J Clin Aesthet Dermatol. 2026;19(5):28–34.
by Mojan DeriSs, PA-C; James Q. Del Rosso, DO; and Natasha Mesinkovska, MD
Ms. Deriss is with Roseman University College of Medicine (RUCOM), Las Vegas, Nevada. Dr. Del Rosso is with JDR Dermatology Research, Las Vegas, Nevada and Touro University Nevada, Henderson, Nevada. Dr. Mesinkovska is with the Department of Dermatology at the University of California, Irvine, Irvine, California.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Del Rosso has served as a clinical research investigator, consultant/advisor, and/or speaker for AbbVie, Almirall, Amgen, Anaptos Bio, Apogee, Arcutis, Bausch, Beiersdorf, Biofrontera, Botanix, Bristol Myers Squibb, Cage Bio, Cara, Cassiopea, Dermata, Dermavant (Organon), Ferndale, Galderma, Incyte, Johnson and Johnson, La Roche Posay, LEO Pharma, Lilly, L’Oréal, MC2, Moonlake, Nektar, Novan, Nutrafol, Pfizer, Ralexar, RBC Consultants, Regeneron, Sanofi, Sente, Sun Pharma, Takeda, UCB, Verrica, Vyne. Dr. Mesinkovska reports being an advisory board member for AbbVie, Arcutis, Lilly, L’Oreal, Merck, Nektar, Pfizer, Sun Pharma, and Veradermics; a speaker for Lilly, Pfizer, and Sun Pharma; and a principal investigator for AbbVie, Arcutis, Lilly, Pfizer, Regeneron, Sanofi, and Sun Pharma. Ms. Deriss has no relevant conflicts of interest to disclose.
ABSTRACT: Atopic dermatitis (AD) and alopecia areata (AA) are chronic immune-mediated disorders that frequently coexist in the same patient, an observation noted in both pediatric and adult populations. The clinical implications of a patient experiencing both disease states simultaneously underscores the need for a deeper understanding of pathophysiology of both disease states, how to optimally utilize specific therapeutic options to achieve synergistic outcomes, suggest simplified rational approaches to treatment selection, how to integrate care with other specialists when needed, and hopefully reach the ultimate goal of clinicians incorporating more personalized treatment selection through immunophenotype-guided therapy. This article , based primarily on emerging case reports and off-label data, reviews all the above implications when encountering a patient with both AD and AA. Continued research and real-world clinical evidence are needed in this important area that affects many individuals that clinicians encounter in their practice, including within the pediatric population. Keywords: Atopic dermatitis, alopecia areata, immunophenotype-guided therapy, real world evidence
Introduction
Atopic dermatitis (AD) and alopecia areata (AA) are chronic immune-mediated disorders that frequently coexist in the same patient, an observation noted in both pediatric and adult populations. Recent epidemiologic studies confirm that patients with moderate to severe or very severe AA have a significantly higher prevalence of comorbid AD, ranging from 3.2% to 17.4%, compared to the general population estimates in some subgroups.1,2 These findings highlight a population at greater risk for compounded disease burdens and adverse psychosocial effects. The compounded implications of a patient experiencing both disease states simultaneously underscore the importance of integrated care and the need for a deeper understanding of how to optimally utilize specific therapeutic options with the hopeful goal of achieving synergistic outcomes.
Emerging case reports and data on off-label, targeted use of biologics (such as dupilumab and tralokinumab) and Janus kinase (JAK) inhibitors (including abrocitinib and upadacitinib) have demonstrated cross-therapeutic benefits for AD and AA, prompting a re-evaluation of sequential and combination treatment strategies.3–26 Nonetheless, optimal patient selection and personalization of therapeutic choices to comprehensively address both disease states warrant the integration of immunophenotyping into clinical practice with the development of precision-guided approaches to maximize therapeutic outcomes. Unlike prior reviews that primarily summarize efficacy of individual agents, this article integrates immunophenotyping data across AD and AA to propose a practical, endotype-based approach for systemic treatment selection and sequencing in comorbid disease. The core premise of this article is to promote understanding of how seemingly divergent and shared immune circuits correlate directly with clinical endotypes that clinicians can efficiently identify when encountering patients with both AD and AA. This premise is essential for advancing the accuracy of individualized therapeutic selection.
What Has Changed Over The Past Decade That DIRECTLY Impacts How We May Change As Clinicians?
Both AD and AA share complex immunopathologic mechanisms, with AD described as being driven primarily by helper T cell (TH) 2/TH22-skewed responses and AA by TH1/interferon (IFN) γ cytokine signatures. Over time, it is also apparent that significant immunologic overlap does exist between these pathways, which provides at least part of the rationale for why patients with the same fundamental disease state exhibit variable responses to different established therapies.27 Until recently, therapeutic options were limited to a traditional stepwise approach (stated under the euphemistic guise called an “algorithm”) because our understanding of the disease mechanism was incomplete; we knew from direct observations in clinical practice over years that disease states such as AD or AA were heterogeneous in their clinical manifestations, but we never fully understood the underlying reasons why. Before the recent “explosion” in mapping cytokines, pathways, cell types, and signaling mediators, we had very limited understanding of which immunologic circuits mattered most for the specific patient with AD/AA and what would work for them. Prior to the past decade with both AD and AA independently, escalation from topical or intralesional therapy (with an occasional “burst” of a systemic corticosteroid to rapidly reduce marked flares) to a conventional systemic immunosuppressant (methotrexate, cyclosporine) was a selective decision reserved for severe, refractory cases. Even greater caution was considered in the pediatric population (where both AD and AA are relatively common), especially with few to no US Food and Drug Administration (FDA)-approved options. Since the FDA approval of dupilumab approximately a decade ago, the addition of other biologics for AD and the subsequent addition of certain JAK inhibitors for AD and/or AA, along with published data in such cases, the landscape of options is steadily changing and evolving with more detailed data on efficacy and safety.
As newer therapies evolve quickly and explanations of pathophysiology accrue at a fast pace, there is an inevitable pitfall in how and when specific advances become accepted enough to be consistently integrated into everyday practice across the country. The cadence of changes in patient care does not occur at a steady and predictable rate among clinicians who are treating these patients. The rate of adoption of newer approaches by clinicians is affected by the level of understanding of the underlying pathophysiology or modes of action, confidence in the data presented, true differentiation of therapies, believability of the presenters of data, comfort level with proper monitoring and safety information, concerns regarding practice efficiency, third-party coverage, cost to patients, and ease of authorizations. Clinicians often continue suboptimal therapies with poor long-term efficacy, minimal impact on underlying inflammation, and safety concerns, overall limiting adoption of newer, more effective options. This becomes an important consideration in disease states when response to an advanced treatment is adversely affected by the duration of the disease, such as with AA. At present, there is a limited ability to predict which patients with AD and AA may respond to specific therapies that are established as both efficient and cost effective. Collectively, this highlights the need for development of endotype-driven, targeted approaches that become established within the mainstream of dermatology and can dramatically improve treatment selection for all clinicians and patients.
Pathophysiology and Immunophenotyping
AD is classically characterized primarily as a type 2 inflammatory dermatosis, with TH2 cytokines such as interleukin (IL) 4, IL-13, and IL-31 driving immunoglobulin (Ig) E production, eosinophil recruitment, and epidermal barrier dysfunction.28 Exogenous (high-IgE) AD exhibits marked TH2 skewing and filaggrin (FLG) loss, whereas endogenous (normal-IgE) AD shows relatively lower TH2 signatures and more prominent TH1/IFN-γ activity, especially in chronic lesions. With disease chronicity, there is a shift from a purely TH2-dominant profile toward mixed TH2/TH1 and TH22 signatures, including increased IFN-γ and IL‑22, which further disrupt keratinocyte homeostasis, amplify pruritus, and perpetuate inflammation.1
AA, by contrast, has traditionally been framed as a TH1- and IFN‑γ–mediated autoimmune attack on the hair follicle immune privilege, with CD8+ T cells, natural killer cells, and cytokines such as IFN‑γ and tumor necrosis factor (TNF) α central to follicular damage and hair cycle arrest.29 However, Cheng et al1 demonstrated that AA also exhibits TH2 and TH17/TH22 involvement, with elevated IL‑4, IL‑13, IL‑17, IL‑22, IgE, and eosinophils in subsets of patients, particularly in those with concomitant atopic disease or high serum IgE. Perifollicular TH17 cell infiltrates and increased IL‑17/IL‑22 correlate with AA severity and early onset, suggesting that AA exists along a continuum of TH1–TH2–TH17/TH22 activation rather than a purely TH1-polarized entity.1
Across both conditions, the JAK–signal transducers and activators of transcription (STAT) pathway serves as a central intracellular hub integrating these cytokine signals.1,29 In AD, IL‑4 and IL‑13 signaling via JAK1/JAK3 and STAT6 downregulates FLG and other barrier proteins, promotes class-switching to IgE, and amplifies type 2 inflammation and pruritus. In AA, genome-wide association studies have identified JAK–STAT components (eg, JAK1, JAK3, STAT3, STAT5) as susceptibility loci, and aberrant JAK–STAT activation in hair follicles is associated with failure to re-enter anagen, supporting the clinical efficacy of JAK inhibition in restoring hair growth. These shared JAK–STAT dependencies provide a mechanistic rationale for overlapping responsiveness of AD and AA to targeted small-molecule inhibitors and selected biologics.1,29
TH17/TH22 axes further link AD and AA. In AD, IL‑17 and IL‑22 contribute to epidermal hyperplasia, barrier dysfunction, and nonhistaminergic itch. In AA, elevated IL‑17 and IL‑22 are associated with severe and early-onset disease and may reflect stress-induced skewing toward TH17 dominance, while TH22 products can perpetuate chronic inflammation in both skin and hair follicles. The OX40/OX40L costimulatory pathway, overexpressed in AD lesional skin and upregulated in AA around follicles, promotes expansion of TH1, TH2, TH17, and TH22 populations and impairs regulatory T-cell function, further eroding follicular immune privilege. This shared T-cell costimulation axis represents an additional convergent therapeutic target in overlapping AD/AA.1
Genetically, AD and AA share susceptibility loci that reinforce their mechanistic overlap.1,30 FLG loss-of-function variants, the strongest known genetic risk factor for AD, are also associated with increased AA risk and more severe AA in patients with a history of AD, likely by compromising barrier integrity and facilitating antigen penetration into the follicular unit.1 Polymorphisms in IL-4, IL-13, and immune regulatory loci such as CLEC16A/KIAA0350 contribute to both AD and AA and to broader atopic or autoimmune diatheses, supporting the concept of shared immunogenetic architecture.1,30 Building on these mechanistic endotypes, JAK–STAT activation and barrier-genetic background exist across the AD/AA spectrum.
Current Evidence: Biologics and JAK Inhibitor Use in AD and AA
Biologics such as dupilumab, tralokinumab, and lebrikizumab have transformed AD control, and the impact of these biologic agents on AA may be synergistic. Randomized controlled trial extension data and meta-analyses of the dupilumab AD program confirm durable skin clearance, corticosteroid-sparing effects, and a largely acceptable long-term safety profile.31 Furthermore, some patients with concurrent AD and AA experience substantial hair regrowth on dupilumab, particularly in TH2‑high, high-IgE phenotypes; however, new-onset or worsening AA has been observed in some cases, suggesting that pure TH2 blockade can unmask or shift disease toward a TH1/IFN‑γ‑dominant pattern.32 Similar patterns are now emerging for tralokinumab, with observational cohorts exhibiting marked AD improvement yet isolated cases suggest varied effects on AA. This indicates that IL‑13–selective inhibition alone may potentially be insufficient to reliably modulate follicular disease; more data are needed.1,11–13 Thus, AD biologics appear to be reliable anchors for TH2‑high AD, but their effects on AA are nuanced, heterogeneous, and endotype‑dependent.
JAK inhibitors offer a distinct therapeutic profile, with consistent evidence of dual benefit in AD and AA, particularly for abrocitinib and upadacitinib, the oral agents that predominantly inhibit JAK1.9,10,15–26 In AD, network meta-analyses and head-to-head comparisons show that these JAK inhibitors provide rapid, progressive improvements in both marked itch reduction and increased clearance of eczematous dermatitis, with results that exceed or at least match the therapeutic outcomes with biologics; however, they carry more potential safety signals and a need for baseline and periodic laboratory monitoring.33,34 For AA, systematic reviews find that JAK inhibitors significantly increase complete or near-complete scalp, eyebrow, and eyelash regrowth across all disease severities.35,36 JAK1-selective regimens can also improve both AD and AA in patients with overlapping immunophenotypes. Case reports of upadacitinib and abrocitinib in severe AD with long-standing AA describe rapid eczema clearance with progressive, sometimes dramatic, hair regrowth, reinforcing the concept that convergent JAK–STAT blockade can simultaneously address TH2‑ and IFN‑γ–driven pathophysiologic cascades.9,10,15–26,35,36 JAK inhibitors provide the advantage of achieving the most consistent dual control of AD and AA, accompanied by the need to perform baseline and periodic clinical and laboratory monitoring due to potential adverse effects.
Immunophenotype-Guided Stratification
Given this complexity, immunophenotyping and endotyping have become important advances in guiding targeted therapy. Biomarker panels, including total and specific IgE, blood eosinophil counts, cytokine and chemokine signatures (eg, IL‑4/IL‑13, IFN‑γ, IL‑17, IL‑22, CXCL9/10), and genetic markers such as FLG and IL-4/IL-13 polymorphisms, can help differentiate TH2‑high vs TH1/IFN‑γ‑high profiles in individual patients.34 In AD with comorbid or emerging AA, TH2‑high, high-IgE, eosinophilic phenotypes are likely to respond to IL‑4/IL‑13–targeting biologics, whereas TH1/IFN‑γ–high or mixed TH1/TH17 signatures are likely to favor JAK inhibition or combination strategies. Similarly, in patients with AA who have strong atopic backgrounds, IgE elevation and TH2/TH22 activation could identify a subgroup in whom AD biologics influence hair disease course, whereas TH1‑dominant, low-IgE AA may be at risk of paradoxical worsening with pure TH2 blockade.19 Incorporating such endotyping into therapeutic decision-making is valuable in improving the precision of treatment stratification.
Emerging evidence supports this stratified approach in both AD and AA. In AD, serum IgE, eosinophils, and type 2-skewed transcriptomic signatures (IL‑4/IL‑13 axes, periostin, TARC/CCL17) identify TH2‑high endotypes that show the most robust and durable responses to biologics such as dupilumab, tralokinumab, and lebrikizumab, while patients with higher IFN-γ expression, stronger IFN‑γ–secreting T‑cell capacity, or mixed TH1/TH17 profiles display relatively attenuated responses and may benefit more from JAK inhibition.1,34,37 In AA, immunophenotyping studies indicate that non-atopic, low-IgE AA is enriched for type 1 cytotoxic T cells/TH1 and IFN‑γ signatures, whereas atopic AA shows increased TH2/TH17 skewing and B‑cell activation, suggesting that JAK inhibitors may be preferable in the former while the latter overlaps more closely with TH2‑high AD biology and may be modulated by AD biologics in selected cases. This continuum helps explain clinical observations of AA improvement in highly atopic patients treated with dupilumab or tralokinumab, in contrast to paradoxical AA onset or worsening in TH1‑dominant, low-IgE phenotypes.1
These data are translated into practical archetypes to assist clinicians with optimizing therapeutic selection in patients with AD and AA:
– TH2‑high AD/AA archetype
– TH1/IFN‑γ‑high AA‑dominant archetype
– Mixed or ‘switching’ archetype
A TH2‑high AD/AA archetype includes patients with long-standing AD, early onset, personal or family atopy, markedly elevated total IgE, eosinophilia, and elevated TH2/TH22 biomarkers. By contrast, a TH1/IFN‑γ–high AA‑dominant archetype comprises individuals with severe, often non-atopic AA, low or normal IgE, minimal eosinophilia, and strong IFN‑γ/TH1 or mixed TH1/TH17 signatures. A third mixed or “switching” archetype includes patients whose AD has transitioned from a TH2-dominated acute phase to a TH1-dominant chronic phase, with declining IgE/eosinophils, increased IFN-γ signatures, and lichenified skin, or whose AA arises in the context of chronic AD, reflecting a dynamic shift along the TH2–TH1/TH17 continuum.38 For example, an adult with early‑onset and long-standing AD, very high IgE, and new patchy AA would be categorized as TH2‑high AD/AA, whereas a non-atopic young adult with alopecia universalis, low IgE, and minimal eczema would fit the TH1/IFN‑γ‑high AA‑dominant archetype.
Proposed Therapeutic Selection Approach
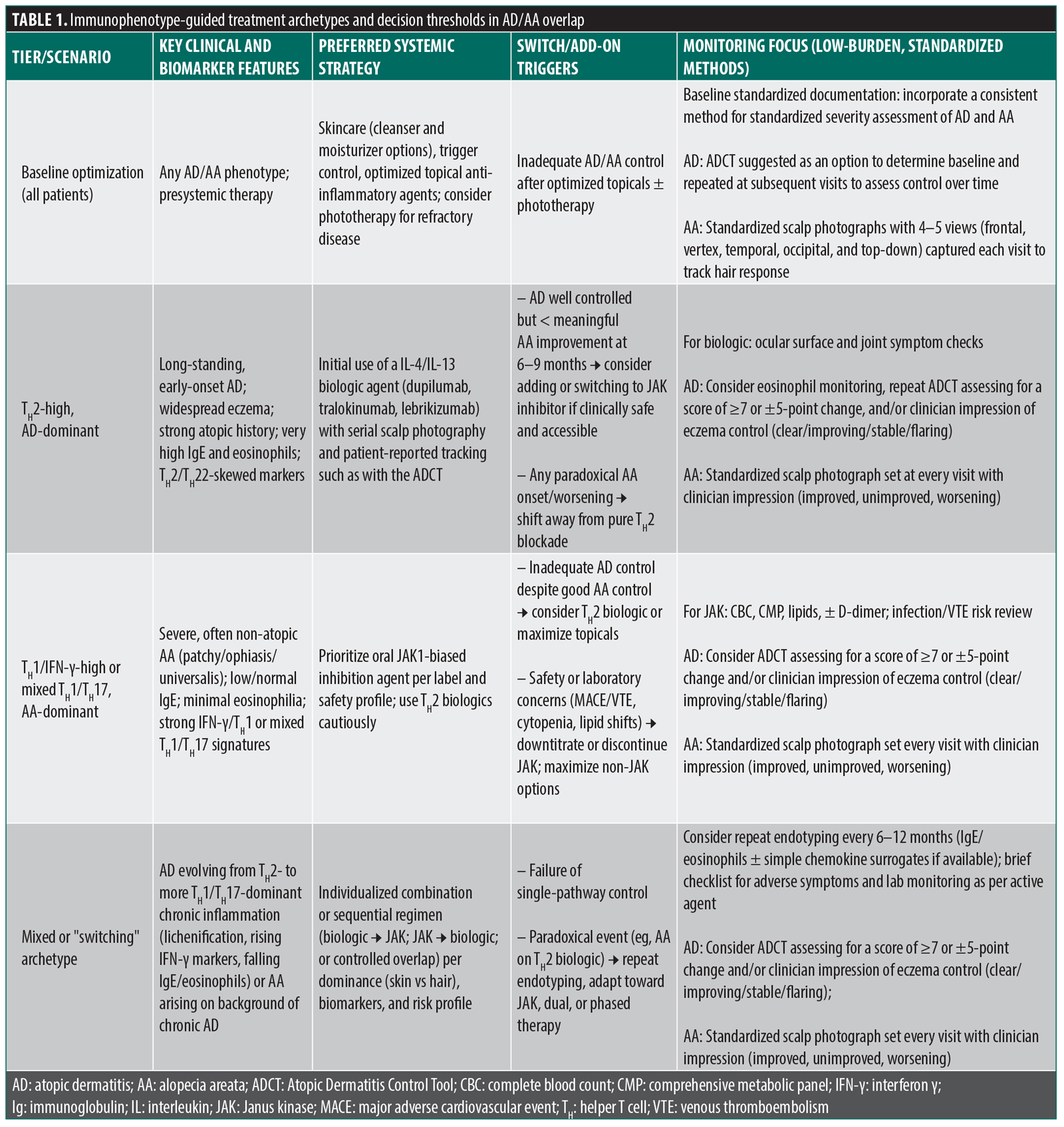
A practical therapeutic selection approach (often called an “algorithm”) for patients with AD and comorbid or emerging AA effectively integrates standard AD severity-based pathways with immunophenotype-guided and directed assistance. (Table 1) Existing AD algorithms and/or consensus guidelines rarely address coexisting AA or paradoxical hair outcomes; this approach recommended here extends the frameworks of therapeutic management by coupling systematic AA assessments and immunophenotype‑guided options.
Building on this, a proposed treatment selection approach for AD/AA overlap could proceed in 3 tiers: baseline optimization, immunophenotype-guided initial systemic choice, and structured switching/combination recommendations. After confirming diagnosis and optimizing skincare, trigger control, and topical therapy, patients with moderate to severe AD and any degree of AA would undergo a focused stratification: clinical pattern (AD‑dominant vs AA‑dominant vs balanced), atopic history (personal or family asthma, rhinitis, food allergy), and a minimal biomarker panel (total IgE, eosinophils, with basic cytokine/chemokine surrogates if available). In TH2‑high, AD‑dominant patients (early-onset AD, widespread eczema, high IgE, eosinophilia, strong atopic diathesis), initial systemic therapy would usually favor IL‑4/IL‑13–targeting biologics (dupilumab, tralokinumab, lebrikizumab) with a consistent methodology for baseline and serial documentation of hair status, reserving JAK inhibitors for inadequate AA response or emergent TH1/TH17 features. In TH1/IFN‑γ‑high or mixed TH1/TH17, AA‑dominant phenotypes (non-atopic AA, low or normal IgE, minimal eosinophilia, severe patchy, ophiasis, or universalis patterns), oral JAK inhibition (eg, abrocitinib, upadacitinib, deuruxolitinib, or another systemic JAK1‑biased agent chosen per label, risk profile, and/or other specific factors) with high-quality published data would be prioritized, with TH2 biologics used cautiously, if at all. Factors such as younger patient age groups below adolescence will warrant consideration, as this requires a more detailed review of experience, literature review, and pediatric dermatology expertise and consultation.
Within this framework, this treatment selection approach defines explicit “switch” and “add‑on” thresholds based on both skin and hair outcomes. For example, in a patient with TH2‑high AD/AA started on dupilumab, failure to achieve meaningful AA improvement despite good AD control would trigger either addition of a JAK inhibitor (where safety and coverage allow) or a switch to JAK inhibitor monotherapy to address both AA and AD, whereas any clear paradoxical AA worsening would prompt earlier transition away from pure TH2 blockade. Conversely, in a JAK‑first, AA‑dominant patient, marked hair regrowth with persistent, inadequately controlled AD can justify adding a TH2 biologic, particularly in individuals with elevated cardiometabolic or thromboembolic risk, with the goal of potentially de-escalating JAK inhibitor exposure (such as lowering the daily dose exposure, every-other day dosing, etc) while maintaining dual disease control.
Across all branches, this approach emphasizes practical, consistent monitoring methods rather than mandated scoring systems. Baseline and periodic laboratory testing (complete blood count, comprehensive metabolic panel, lipid panel, with or without D‑dimer) and infection or venous thromboembolism risk evaluation remain essential for JAK inhibitors, while monitoring for ocular surface disease (OSD) symptoms, eosinophil trends, and joint complaints is recommended for biologics. Evaluation of OSD at baseline and periodic intervals is optimally conducted by an ophthalmologist, as slit‑lamp examination may be required for accurate assessment.
Streamlined objective assessment strategies for AD such as the Atopic Dermatitis Control Tool (ADCT) completed by patients in the waiting room and standardized scalp photography for AA (4–5 consistent views—frontal, vertex, temporal, occipital, top-down—taken by staff at each visit) provide reproducible documentation of treatment progress to guide management decisions over time. The ADCT represents 1 validated, low-burden option for practices seeking efficient, longitudinal monitoring of AD control.39,40 This brief, 6-item patient-reported survey, validated for adult and pediatric patients (aged 12 years or older, with modified 6-month or older versions available), enables rapid, consistent assessment of disease control at each visit and is completed independently in the waiting room, thereby optimizing clinical discussions.41 The ADCT assists with treatment decisions and addresses core pertinent clinical questions such as “Over the last week, how many nights did you have trouble falling asleep or staying asleep because of your eczema?” followed by 5 answer choices and qualitative values assigned to each. The ADCT total score (range: 0–24) is calculated by summing responses (0–4 points each) to its 6 questions, with scores ≥7 indicating poor current control and ±5-point changes signifying clinically meaningful improvement or worsening over time.42 Readers are referred to the cited references for free access to the ADCT instrument, validation data, implementation guidance, and evidence of its impact on practice quality and efficiency.39–42
Above all, consistency in the clinician’s chosen tracking method is most important to ensure longitudinal reliability for both skin and hair outcomes. Such a stepwise, clinical, and biomarker-based flow supports a more-informed decision-making process at each point of evaluation that the clinician can use in discussing with the patient when deciding together on next steps.
Real-World Factors and Effects on Prescribing Patterns
Real-world use of biologics and JAK inhibitors for AD and AA is frequently constrained by access, with off-label prescribing falling into third-party coverage barriers. Patients with moderate to severe AD may already face hurdles for prescribing of on-label biologics. Survey data from the National Eczema Association show nearly half of respondents experienced at least 1 insurance-related delay or denial in the previous year, and over 40% of biologic prescriptions encountered a third-party coverage issue, often requiring prior authorization or step therapy leading to weeks-long treatment delays.43,44 Prior authorizations for AD biologics cause the majority of delays (55%), often requiring multiple weeks of back-and-forth communications to resolve the delay; such coverage barriers impose a substantial increase in administrative costs, with biologics and other systemic agents facing more coverage barriers and longer wait times than topical therapies.43 These barriers can delay effective care for the patient and prolong the suffering they experience from their disease without access to a potential option used in the study. These same barriers can create frustrations for clinicians and distract from their primary obligation to provide care to their patients. The increased workload and time constraints caused by these third-party coverage barriers can sometimes deter clinicians and/or their staff from pursuing advanced treatments even when medically appropriate.
When therapeutic agents are prescribed off-label, such as using AD biologics or JAK inhibitors to treat AA or to leverage cross-efficacy in comorbid AD/AA, these coverage barriers are often magnified. A published series in AA documented very high initial denial rates for JAK inhibitor coverage in off‑label use, with many requests denied on first submission and “cosmetic” classification or lack of on‑label status cited as common reasons.44 Patients who do obtain access frequently do so only after multiple appeals, letters of medical necessity, or external review processes that are time-intensive for dermatology practices and not uniformly successful, especially in community settings with fewer administrative resources. When coverage is denied or only partially approved, individuals can face high monthly out-of-pocket expenses for oral JAK inhibitors or injectable biologics, leading to treatment abandonment, dose stretching, or cycling back to less effective but reimbursed therapies. This pattern has commonly been characterized as “financial toxicity” in dermatologic care.45 In this context, the practical implementation of immunophenotype-guided, off-label strategies for AD and AA is limited not by scientific rationale but by payor policies, variable benefit design, and patients’ capacity to absorb substantial direct costs, all of which must be explicitly considered when proposing real-world clinical treatment plans.
Future Directions and Research Gaps
Future directions and research gaps in managing overlapping AD and AA center on moving from empiric, label-driven prescribing toward genuinely stratified and data-informed care. Current evidence is predominantly dominated by short- to medium-term randomized controlled trials designed for the primary purpose of FDA evaluation and drug approval. Published case series that are often heterogeneous in nature regarding patient inclusion may add a fraction of additional perspective on management of patients with both AD and AA. There are relatively few prospective studies specifically designed around comorbid AD and AA or cross-disease effects of AD-targeted agents in AA. There is a pressing need for pragmatic, real-world trials that embed simple immunophenotyping (eg, IgE, eosinophils, basic cytokine/chemokine panels) and follow patients longitudinally as they cycle through biologics and JAK inhibitors, capturing coprimary endpoints that evaluate both eczematous dermatitis/pruritus and hair growth outcomes. Large, multicenter registries that systematically record clinical phenotype, biomarker data, treatment sequences, and patient-reported outcomes in AD/AA overlap would help define durable response patterns, clarify predictors of paradoxical events, and contribute to informed reimbursement and policy decisions.1,29
Opportunities for precision medicine include operationalizing routine immunophenotyping in everyday practice in a time- and cost-efficient manner and integrating these data into optimizing clinical decisions related to treatment selection. Standardized, affordable panels linking IgE/eosinophils, a small set of signature cytokines or chemokines, and key genetic markers (such as FLG and IL-4/IL-13 variants) could be used to assign patients to TH2‑high, TH1/IFN‑γ–high, or mixed endotypes, with treatment plans that adapt over time as those profiles shift. Accurately designed artificial intelligence and machine-learning models that are properly vetted and trained on registry and trial datasets could then synthesize demographics, comorbidities, laboratory profiles, and prior treatment responses to generate individualized recommendations for initial therapy, timing of escalation or switching, and risk of paradoxical AA with TH2 blockade or inadequate AA response with biologic monotherapy. Finally, long-term outcome studies spanning safety, durability of dual disease control, quality of life, and economic impact are essential to validate these precision strategies, refine risk-benefit assessments for combination regimens, and ensure that advances in immunology and data science translate into equitable, sustainable care for patients with concurrent AD and AA.34,37,38
Conclusion
AD and AA exemplify a spectrum of overlapping, dynamically shifting immune endotypes in which shared TH1/TH2/TH17/TH22 and JAK–STAT pathways drive heterogeneous cutaneous and follicular inflammation.38 The current evidence base shows that TH2‑targeting biologics (such as dupilumab, tralokinumab, and lebrikizumab) and JAK inhibitors (including abrocitinib, upadacitinib, and related agents) can each achieve robust control of moderate to severe AD and that JAK inhibitors in particular offer consistent efficacy for AA, with biologics demonstrating more variable and sometimes paradoxical effects on hair disease.3,31,32,35 These findings collectively support moving from a one-size-fits-all treatment model toward an immunophenotype-guided approach that stratifies patients by clinical dominance (AD vs AA), atopic history, and simple biomarkers (IgE, eosinophils, cytokine signatures, and key genetic variants) when selecting and sequencing systemic therapies.1,34,37 It is hoped that future research will further convert this conceptual framework into validated, clinically actionable pathways that will streamline and simplify the accuracy of treatment selection and improve therapeutic outcomes for affected patients.
References
- Cheng J, Jiang Y, Chen Q, Xiao M. Overlapping features of atopic dermatitis and alopecia areata: from pathogenesis to treatment. Front Immunol. 2025;16:1641918.
- Bunick CG, Armstrong AW, Grada A, et al. The epidemiology of atopic dermatitis among adults and adolescents with alopecia areata in the United States. J Investigative Dermatol. 2025;146(2):365–373.e3.
- Dekkers C, Zuithoff N, Bakker D, et al. Tralokinumab treatment in adult atopic dermatitis patients: 28-week evaluation of cinical effectiveness, safety, serum proteins and total IgE levels. Allergy. 2025;80(4):1060–1073.
- Guttman-Yassky E, Renert-Yuval Y, Bares J, et al. Phase 2a randomized clinical trial of dupilumab (anti-IL-4Rα) for alopecia areata patients. Allergy. 2022;77(3):897–906.
- Huang J, Jian J, Li T, et al. Dupilumab therapy for alopecia areata: a case series and review of the literature. J Dermatolog Treat. 2024;35(1):2312245.
- Magdaleno-Tapial J, Valenzuela-Oñate C, García-Legaz-Martínez M, Martínez-Domenech Á, Pérez-Ferriols A. Improvement of alopecia areata with dupilumab in a patient with severe atopic dermatitis and review the literature. Australas J Dermatol. 2020;61(2):e223-e225.
- Cho SK, Craiglow BG. Dupilumab for the treatment of alopecia areata in children with atopic dermatitis. JAAD Case Rep. 2021;16:82–85.
- McKenzie PL, Castelo-Soccio L. Dupilumab therapy for alopecia areata in pediatric patients with concomitant atopic dermatitis. J Am Acad Dermatol. 2021;84(6):1691–1694.
- Liu X, Song B, Jin H. Abrocitinib improved dupilumab-resistant severe atopic dermatitis with comorbid mild alopecia areata in a 12-year-old boy: a case report with 1-year follow-up. J Asthma Allergy. 2024;17:305–311.
- Zhang J, Zuo YG. Successful treatment of alopecia universalis with abrocitinib: a case report. J Dermatolog Treat. 2023;34(1):2242706.
- Kussini J, Pfützner W, Mühlenbein S. A case of unexpected successful treatment of alopecia areata with tralokinumab in a patient with atopic dermatitis. Int J Dermatol. 2025;64(10):1905–1906.
- Tavoletti G, Chiei-Gallo A, Barei F, Marzano AV, Ferrucci SM. Tralokinumab as a therapeutic option for patients with concurrent atopic dermatitis and alopecia areata. Int J Dermatol. 2024;63(3):374–375.
- Greenberg ABW, Shahriari M, Cameron MC, et al. Tralokinumab as a therapeutic alternative for dupilumab-associated arthralgia in atopic dermatitis: a multi-center case series. J Clin Aesthet Dermatol. 2025;18(5):16–19.
- David E, Shokrian N, Del Duca E, et al. Dupilumab induces hair regrowth in pediatric alopecia areata: a real-world, single-center observational study. Arch Dermatol Res. 2024;316(7):487.
- Mitroi GG, Mitroi GF, Ică OM, Anghelina F, Ciolofan MS, Mitroi MR. Off-label uses of abrocitinib: review of emerging therapeutic applications beyond atopic dermatitis. Life (Basel). 2024;14(9):1127.
- Bennett M, Moussa A, Sinclair R. Successful treatment of chronic severe alopecia areata with abrocitinib. Australas J Dermatol. 2022;63(2):274–276.
- Battilotti C, Azzella G, Dattola A, et al. Efficacy of upadacitinib in treating alopecia areata, atopic dermatitis, and Th1 comorbidities in pediatric patients: a comprehensive case series and literature review. J Clin Med. 2025;14(11):3881.
- Asfour L, Getsos Colla T, Moussa A, Sinclair RD. Concurrent chronic alopecia areata and severe atopic dermatitis successfully treated with upadacitinib. Int J Dermatol. 2022;61(11):e416-e417.
- Brănișteanu DE, Huțanu AE, Brănișteanu DC, et al. Dual relief: how atopic dermatitis treatments affect alopecia areata-a small retrospective cohort study. Diagnostics (Basel). 2025;15(5):520.
- Cantelli M, Martora F, Patruno C, Nappa P, Fabbrocini G, Napolitano M. Upadacitinib improved alopecia areata in a patient with atopic dermatitis: a case report. Dermatol Ther. 2022;35(4):e15346.
- Bourkas AN, Sibbald C. Upadacitinib for the treatment of alopecia areata and severe atopic dermatitis in a paediatric patient: a case report. SAGE Open Med Case Rep. 2022;10:2050313X221138452.
- Walls B, Reguiai Z. Dual efficacy of upadacitinib in a patient with concomitant severe atopic dermatitis and alopecia areata. Ann Dermatol Venereol. 2023;150(4):281–283.
- Gao Y, Zhu C, Jin H. Upadacitinib therapy in adolescent severe alopecia areata: a case series and narrative review. Clin Cosmet Investig Dermatol. 2025;18:2141–2148.
- Kołcz K, Żychowska M, Sawińska E, Reich A. Alopecia universalis in an adolescent successfully treated with upadacitinib-a case report and review of the literature on the use of JAK inhibitors in pediatric alopecia areata. Dermatol Ther (Heidelb). 2023;13(3):843–856.
- Gambardella A, Licata G, Calabrese G, De Rosa A, Alfano R, Argenziano G. Dual efficacy of upadacitinib in 2 patients with concomitant severe atopic dermatitis and alopecia areata. Dermatitis. 2021;32(1S):e85–e86.
- Giavina-Bianchi M, Giavina-Bianchi P. Successful treatment of severe atopic dermatitis and alopecia universalis with upadacitinib in a 29-year-old male patient. J Allergy Clin Immunol Glob. 2024;3(3):100269.
- Starace M, Cedirian S, Quadrelli F, et al. Dupilumab and alopecia areata: a possible combined or disturbance therapy? A review of the literature. Dermatol Pract Concept. 2024;14(4):e2024270.
- Gavrilita E, Silion SI, Bitca ML, Tatu AL. Insights into intrinsic atopic dermatitis: immunogenicity, dysbiosis, and imaging (reflectance confocal microscopy, optical coherence tomography). Clin Cosmet Investig Dermatol. 2024;17:1377–1386.
- Passeron T , King B, Seneschal J, et al. Inhibition of T-cell activity in alopecia areata: recent developments and new directions. Front Immunol. 2023;14:1243556.
- Weidinger S, Willis-Owen SA, Kamatani Y, et al. A genome-wide association study of atopic dermatitis identifies loci with overlapping effects on asthma and psoriasis. Hum Mol Genet. 2013;22(23):4841–4856.
- Beck LA, Bissonnette R, Deleuran M, et al. Dupilumab in adults with moderate to severe atopic dermatitis: a 5-year open-label extension study. JAMA Dermatol. 2024;160(8):805–812.
- Sachdeva M, Witol A, Mufti A, Maliyar K, Yeung J. Alopecia areata related paradoxical reactions in patients on dupilumab therapy: a systematic review. J Cutan Med Surg. 2021;25(4):451–452.
- Silverberg JI, Thyssen JP, Fahrbach K, et al. Comparative efficacy and safety of systemic therapies used in moderate-to-severe atopic dermatitis: a systematic literature review and network meta-analysis. J Eur Acad Dermatol Venereol. 2021;35(9):1797–1810.
- Park CO, Kim SM, Lee KH, Bieber T. Biomarkers for phenotype-endotype relationship in atopic dermatitis: a critical review. EBioMedicine. 2024;103:105121.
- Yan D, Fan H, Chen M, et al. The efficacy and safety of JAK inhibitors for alopecia areata: a systematic review and meta-analysis of prospective studies. Front Pharmacol. 2022;13:950450.
- Liu M, Gao Y, Yuan Y, et al. Janus kinase inhibitors for alopecia areata: a systematic review and meta-analysis. JAMA Netw Open. 2023;6(6):e2320351.
- Maintz L, Welchowski T, Herrmann N, et al. IL-13, periostin and dipeptidyl-peptidase-4 reveal endotype-phenotype associations in atopic dermatitis. Allergy. Published online 17 Jan 2023.
- Bai R, Zheng Y, Dai X. Atopic dermatitis: diagnosis, molecular pathogenesis, and therapeutics. Mol Biomed. 2025;6(1):71.
- Staumont-Sallé D, Taieb C, Merhand S, Shourick J. The atopic dermatitis control tool: a high-performance tool for optimal support. Acta Derm Venereol. 2021;101(12):adv00618.
- Simpson E, Eckert L, Gadkari A, et al. Validation of the Atopic Dermatitis Control Tool (ADCT©) using a longitudinal survey of biologic-treated patients with atopic dermatitis. BMC Dermatol. 2019;19(1):15.
- Chuang CC, Pariser DM, Simpson E, et al. The Atopic Dermatitis Control Tool: adaptation and content validation for children and caregivers of children with atopic dermatitis. Dermatol Ther (Heidelb). 2024;14(12):3261–3271.
- Atopic Dermatitis Control Tool. Accessed 5 Jan 2026. https://www.adcontroltool.com
- Loiselle AR, Chovatiya R, Thibau IJ, et al. Evaluating access to prescription medications in the atopic dermatitis patient population in the USA. Dermatol Ther (Heidelb). 2024;14(7):1811–1821.
- Thompson HJ, Vavra T, Jabbari A. Factors associated with insurance coverage of tofacitinib for alopecia areata: a retrospective review from an academic institution. J Am Acad Dermatol. 2020;83(5):1509–1510.
- Burkhart C. Sequelae from financial toxicity of biologics and JAK inhibitors in the United States: health insurance companies may eliminate dermatology from the list of specialists in their physician panels. Open Dermatol J. 2023;17:e187437222304180.