J Clin Aesthet Dermatol. 2026;19(5):17–27.
by Luisa V. Ramirez, MD; Ebony P. Gayle, MD; Ana Ormaza, MD; Sami K. Saikaly, MD; Elisha Singer, MD; Thais Pincelli, MD; Deanna C. Menapace, MD; Courtney L. James-Newsome, MD; Roy S. Rogers III, MD; Alison J. Bruce, MBChB; Leila Tolaymat, MD; and Miguel A. Aristizabal-Torres, MD
Drs. Ramirez and Gayle are with ADEI, Aesthetics & Dermatology Institute, Bogota, Colombia. Dr. Ormaza is with the Department of Dermatology, Eastern Virginia Medical School, Norfolk, Virginia. Dr. Saikaly is with Rising Sun Dermatology & Cosmetics Center, St. Augustine, Florida. Drs. Singer, Pincelli, Bruce, Tolaymat, and Aristizabal-Torres are with the Department of Dermatology, Mayo Clinic, Jacksonville, Florida. Dr. Menapace is with the Department of Otolaryngology, Division of Facial Plastic Surgery, Mayo Clinic, Jacksonville, Florida. Dr. James-Newsome is with the Department of Emergency Medicine, Mayo Clinic, Jacksonville, Florida. Dr. Rogers is with the Department of Dermatology, Mayo Clinic, Scottsdale, Arizona.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no relevant conflicts of interest.
ABSTRACT: Background: Hyperbaric oxygen therapy (HBOT) has been used in ischemic and inflammatory conditions due to its ability to enhance tissue oxygenation and support wound healing. Filler-induced vascular occlusion (FIVO) is a rare but potentially devastating complication of dermal filler injections that may result in skin necrosis or vision loss. HBOT has been increasingly reported as an adjunctive intervention in FIVO, but its reported use has not been systematically summarized.
Objective: To summarize the existing literature on HBOT use in FIVO, describe reported clinical contexts and treatment parameters, and identify gaps in current knowledge.
Methods: This scoping review was conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews guidelines. A comprehensive search of PubMed, MEDLINE, Embase, and Google Scholar identified case reports, case series, experimental studies, and reviews describing FIVO cases managed with HBOT. Articles published in English or Spanish between January 2011 and May 2025 were included. Data were synthesized descriptively.
Results: Twenty-four studies met inclusion criteria, consisting primarily of case reports and small case series. HBOT was most often used as an adjunct to established therapies, including hyaluronidase, antiplatelet agents, vasodilators, and thrombolytic therapy. Reported HBOT protocols varied substantially, with treatment pressures of 2.0 to 3.0 atmospheres absolute and session durations of 60 to 120 minutes. Outcomes were heterogeneous and frequently confounded by multimodal management. CONCLUSION: HBOT has been reported as an adjunctive intervention in selected cases of FIVO, but evidence remains limited to low-level observational data, highlighting the need for standardized protocols and prospective studies.
Keywords: Hyperbaric medicine, hyperbaric oxygen therapy, hyperbaric oxygen treatment, filler-induced vascular occlusion, derma filler, filler complications
Introduction
Dermal fillers are widely used in minimally invasive cosmetic facial rejuvenation and have gained global popularity due to their biocompatibility, accessibility, and ease of use.1,2 According to the 2023 Procedural Statistics Report by the American Society of Plastic Surgeons, approximately 25.4 million minimally invasive procedures were performed.3 Among these, dermal fillers accounted for 3,441,534 procedures, representing a 4% increase from 2022.3 This notable rise in frequency may also be accompanied by a corresponding increase in complications, underscoring the need for clinicians to have a broad and readily accessible vascular occlusion protocol.
Dermal fillers may be composed of various materials, including hyaluronic acid, poly-L-lactic acid, calcium hydroxyapatite, polycaprolactone, and others.4,5 While generally considered safe, dermal fillers can occasionally lead to serious complications, most notably filler-induced vascular occlusion (FIVO).6,7 FIVO refers to the obstruction of blood flow in areas treated with dermal fillers and is estimated to occur in approximately 0.01% to 0.05% of cases.8 Although rare, FIVO can result in significant aesthetic and physiological consequences.9,10 Despite its severity, FIVO remains underreported and insufficiently documented, emphasizing the importance of improved awareness, prevention strategies, and treatment protocols.8,11
Several treatment options for FIVO have been described, including vasodilators, hyaluronidase, antiplatelet agents, and corticosteroids. These therapies have demonstrated varying degrees of efficacy in restoring perfusion to ischemic tissues.12,13 Given the potential limitations of current approaches, further exploration of adjunctive treatments is warranted. One such modality is hyperbaric oxygen therapy (HBOT), which may enhance oxygen delivery and thereby promote tissue viability in affected areas. Owing to its ability to improve tissue oxygenation, reduce ischemia, and stimulate healing, HBOT has been proposed as a potential adjuvant treatment to minimize long-term sequelae in FIVO. However, the current evidence remains limited, primarily derived from case reports and small case series.14–16 Therefore, the objective of this scoping review is to map the existing literature on the use of HBOT in FIVO, describe reported clinical contexts and treatment parameters, and identify knowledge gaps.

Methods
Review design and reporting framework. This scoping review was conducted and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) guidelines.
Eligibility criteria. Eligible sources of evidence included published case reports, case series, experimental studies, and review articles describing cases of FIVO in which HBOT was used as part of the management strategy. Articles published in English or Spanish between January 2011 and May 2025 were considered.
Studies were excluded if HBOT was not part of the treatment regimen for FIVO, if HBOT was used exclusively for non–filler-related dermatologic conditions (eg, chronic ulcers, scars, or graft compromise unrelated to dermal fillers), if insufficient clinical detail regarding HBOT use was provided, or if the report was unrelated to dermal filler complications.
Information sources and search strategy. A comprehensive literature search was performed using PubMed, MEDLINE, Embase, and Google Scholar. Search terms included combinations of keywords and subject headings related to HBOT and dermal filler complications, including “hyperbaric oxygen therapy,” “HBOT,” “dermal fillers,” “filler-induced vascular occlusion,” “vascular occlusion,” “hyaluronic acid,” “skin ischemia,” “skin necrosis,” “calcium hydroxyapatite,” and “filler complications” (Table 1). Reference lists of included articles were manually reviewed to identify additional relevant sources. The literature search was conducted by L.V.R., E.P.G., A.O., and M.A.A.-T.
Study selection. All records identified through database searching were exported to a reference management system, and duplicate records were removed. Titles and abstracts were screened independently by 4 reviewers (L.V.R., E.P.G., A.O., M.A.A.-T.) to identify potentially relevant articles. Full-text articles were subsequently assessed for eligibility by 5 reviewers (L.V.R., E.P.G., A.O., R.S.R, M.A.A.-T.). Discrepancies were resolved through discussion and consensus, with consultation of a senior author (S.K.S., E.S., D.C.M., C.L.J.-N., R.S.R., A.J.B., L.L.T., M.A.A.-T.) when necessary. Reasons for exclusion at the full-text stage were documented.
Data charting process. Data were extracted using a standardized data charting form developed for this review. Extracted variables included author and year of publication, study design, type of dermal filler, clinical manifestations of FIVO (cutaneous and/or ocular), timing of HBOT initiation relative to symptom onset, number of HBOT sessions, chamber type (monoplace or multiplace), atmospheric pressure applied, duration of each session, concomitant therapies, and reported clinical outcomes. Extracted data were reviewed by all authors to ensure accuracy and completeness.
Synthesis of results. Data were synthesized descriptively to map patterns of HBOT use in the management of FIVO. Given the heterogeneity of study designs, interventions, and outcomes, no quantitative synthesis or meta-analysis was performed. Consistent with PRISMA-ScR guidance, no formal risk of bias assessment was undertaken.
Results
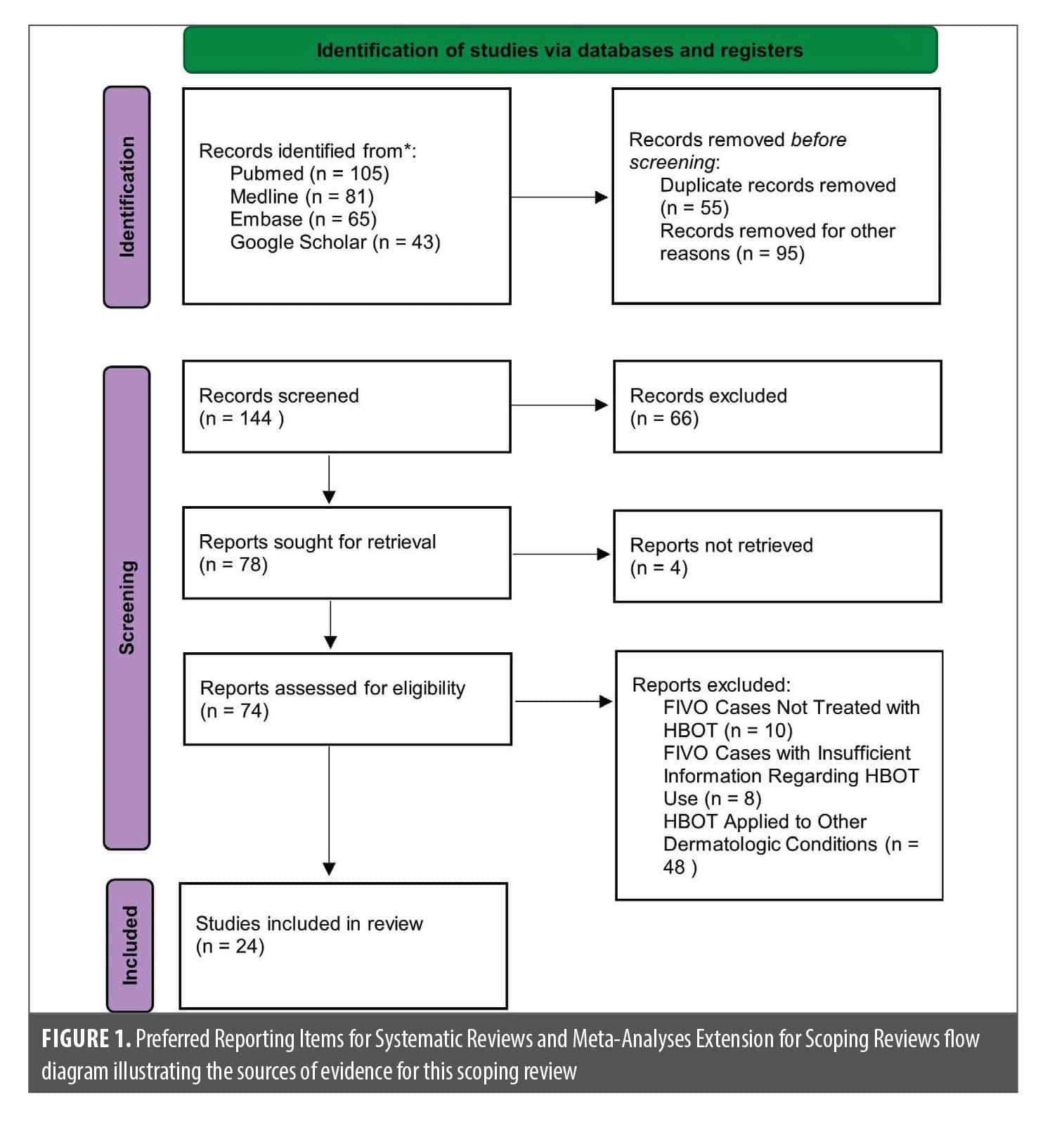
Search results. The database search identified a total of 294 records across PubMed, MEDLINE, Embase, and Google Scholar. After removal of 55 duplicate records, 239 unique articles remained. Following exclusion of studies that addressed nondermatologic indications for HBOT or were unrelated to dermal filler complications, 144 records were screened based on title and abstract.
Seventy-eight full-text articles were assessed for eligibility. Four reports could not be retrieved. Of the remaining articles, 50 were excluded due to lack of relevance to FIVO or insufficient information regarding the use of HBOT. Ultimately, 24 studies met inclusion criteria and were included in this scoping review. The study selection process is summarized in the PRISMA-ScR flow diagram (Figure 1).
Characteristics of included sources. The 24 included studies consisted predominantly of case reports and small case series, with a limited number of narrative reviews and experimental reports. Publication years ranged from 2011 to 2025. Most reports described female patients undergoing cosmetic facial filler procedures, with hyaluronic acid being the most commonly implicated filler material.
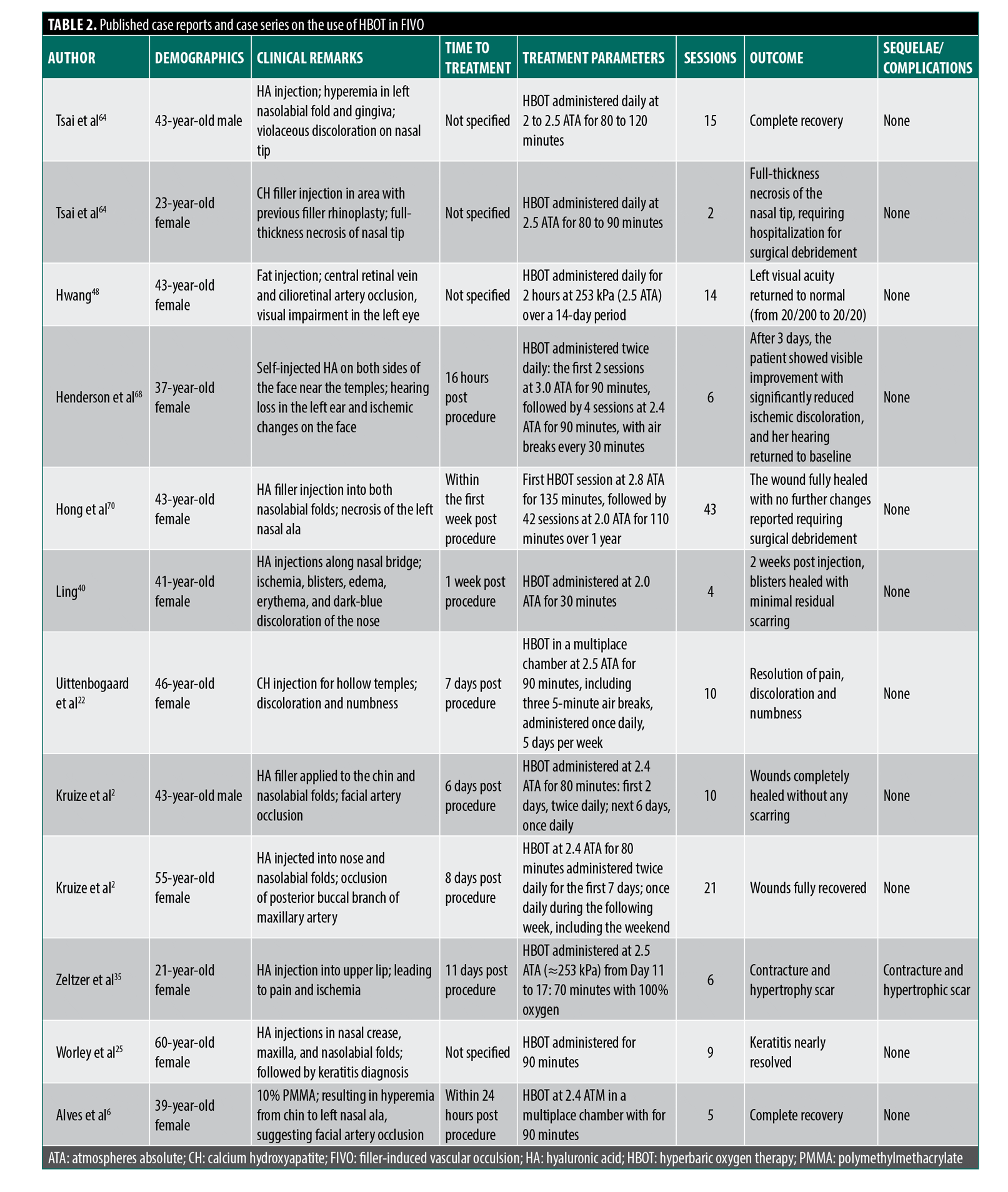
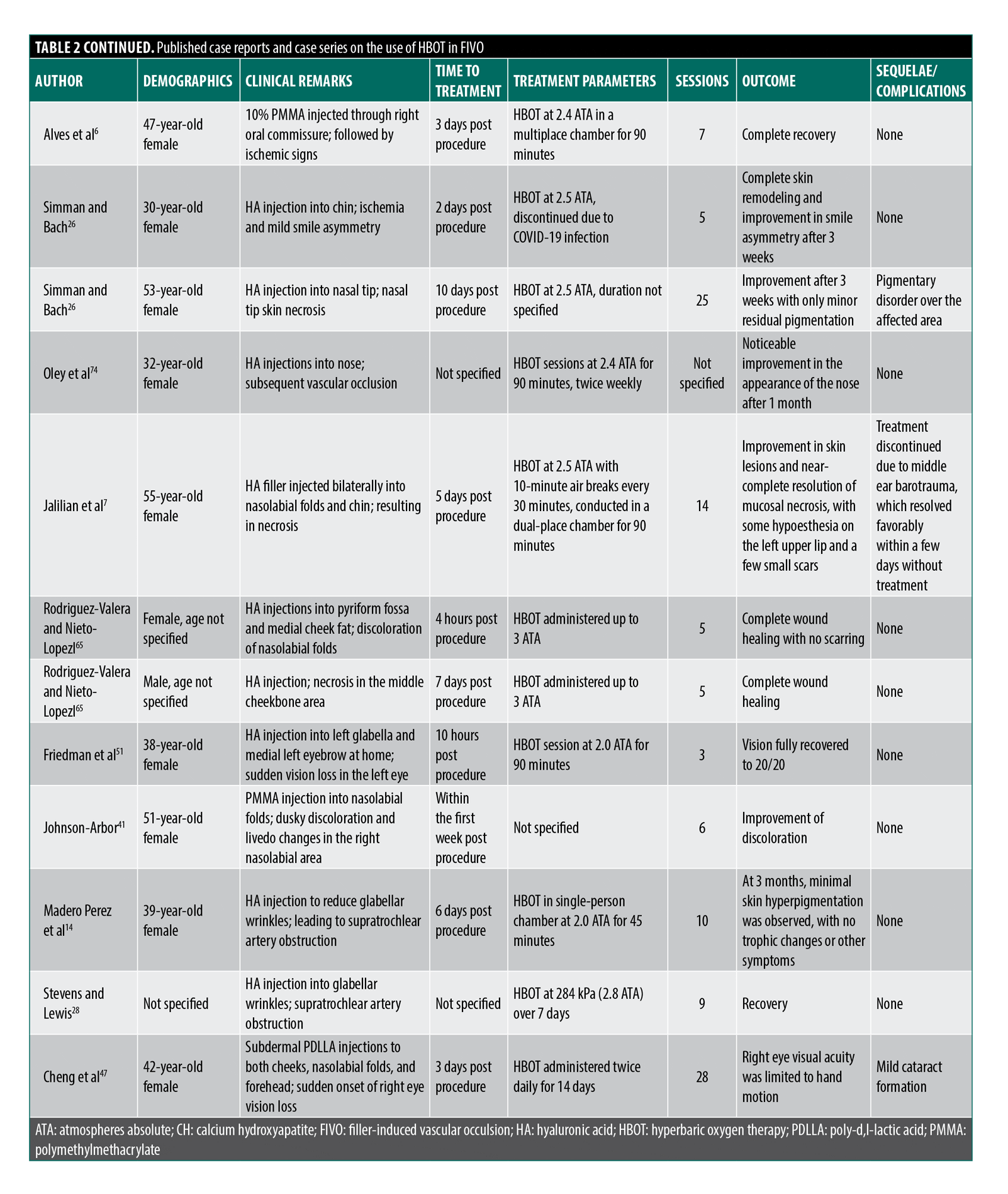
Clinical presentations included cutaneous ischemia, skin necrosis, and, less frequently, ocular complications such as visual impairment or retinal ischemia. HBOT was consistently reported as an adjunctive intervention and was administered alongside established therapies, including hyaluronidase, antiplatelet agents, vasodilators, corticosteroids, and thrombolytic therapy. The extracted data from the included studies are presented in Table 2.
Overview of mapped evidence. Across the included sources, authors consistently described FIVO as resulting from either intravascular injection or external compression, leading to ischemia that may progress to tissue necrosis, ocular ischemia, or neurologic injury depending on the regions involved and the extent of anatomical compromise. The following subsections summarize key concepts and clinical features reported in the literature to contextualize the use of HBOT.
Key concepts and clinical context. Dermal fillers are commonly used to enhance facial volume and to improve the appearance of rhytides.17 Despite their widespread use and generally favorable safety profile, these agents carry a known risk of vascular complications.18,19 Inadvertent intra- or perivascular injection may occur due to several leading factors such as injection technique, anatomical variations, and the physical characteristics of the filler material itself, including its composition, particle size, viscosity, and volume.20,21 When a filler is unintentionally applied into or near a blood vessel, it can lead to mechanical obstruction of blood flow, which may result in tissue congestion or hypoxia and, in more severe cases, necrosis of the overlying skin and soft tissue.6,22
The risk of vascular compromise is heightened by the complex anatomy of the facial arterial system, which features an extensive network of anastomoses and collateral vessels.23 These interconnections allow filler material to propagate unpredictably within the vasculature, even beyond the initial site of injection.24 Once within the arterial system, intravascular filler particles can migrate and occlude smaller vessels, causing ischemia in more distal or even contralateral regions.25,26 Embolization of filler material into critical vascular territories, such as the ophthalmic or cerebral arteries, can result in severe outcomes, including retinal ischemia, cerebral infarction, and stroke.27,28
FIVO has been described as a pathophysiological process comprising 4 progressive stages. The first stage, vasocannulation, involves mechanical penetration of the arterial wall by the needle or cannula. This is followed by vasoinoculation, in which the filler material enters the arterial lumen. Vasodissemination, the third stage, entails the dissemination of filler through anastomotic pathways to more distal vascular territories. Finally, vasoocclusion results in the obstruction of blood flow, culminating in tissue ischemia and necrosis.29,30
In addition to direct intravascular injection, vascular compromise may also occur through external compression. Large boluses of filler can exert extrinsic pressure on adjacent vessels, particularly in areas with limited tissue compliance, leading to perivascular obstruction.31,32 Moreover, fillers may induce vasospasm, activate the coagulation cascade, and stimulate platelet aggregation and thrombosis.20 These vascular responses are often accompanied by an inflammatory reaction mediated by cytokines, notably interleukin (IL) 6 and IL-8, which can further impair perfusion and oxygen delivery at the tissue level.33
Certain anatomical sites are recognized as being at a particularly high risk for vascular events due to their proximity to critical arteries and limited collateral circulation. These include the nasolabial folds, nasal dorsum, glabella, and forehead.34,35 Risk is also influenced by procedural factors, including the volume of filler injected; volumes exceeding 0.1 mL are associated with increased complication rates.17,36 The choice of delivery instrument can also increase risk. Needle-based injections and the use of small-diameter cannulas increase the likelihood of vascular penetration.37,38 Additionally, prior scarring or fibrosis in the treatment area may distort normal anatomy and elevate the risk of vascular injury.21,39
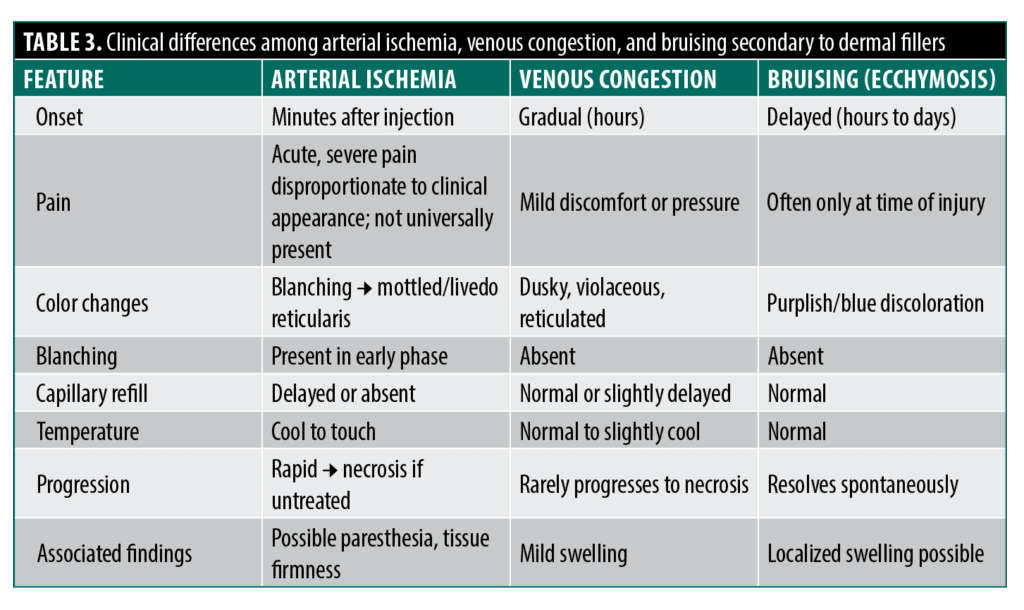
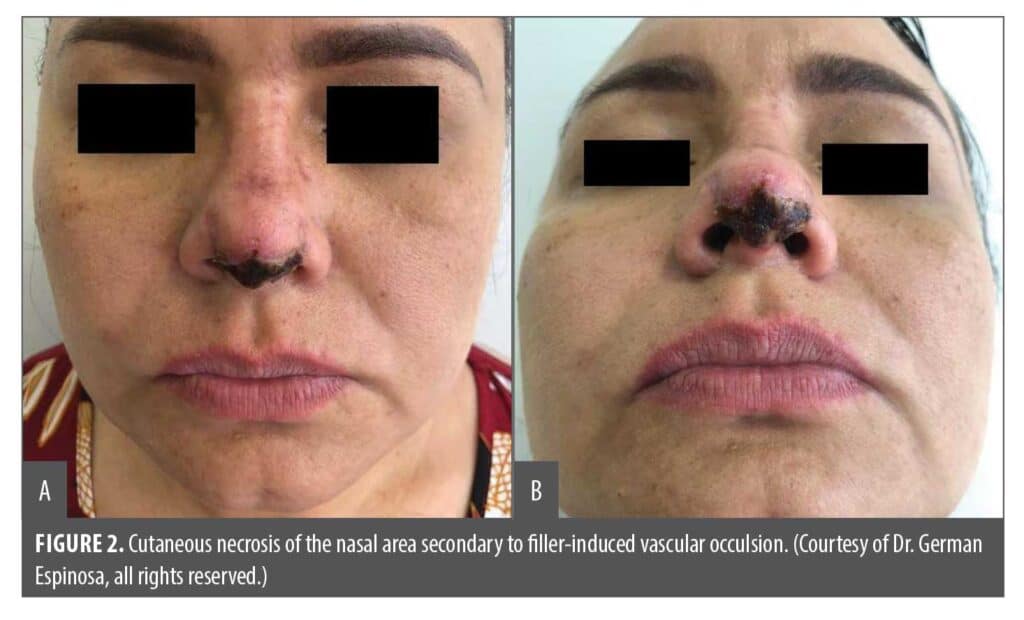
Clinical manifestations of filler-induced vascular complications. Early signs and symptoms of FIVO that may precede ischemic skin necrosis include localized blanching, acute pain, livedo reticularis, and delayed capillary refill.20,22,40,41 In contrast, venous congestion is more often characterized by a gradual onset of dusky, violaceous discoloration with a reticulated pattern, typically accompanied by mild swelling rather than acute pain. Bruising (ecchymosis), which may be mistaken for early vascular compromise, generally presents post procedure with well-demarcated purplish discoloration, is nonblanching, and lacks associated blanching or severe pain (Table 3).42 Distinguishing these entities at the earliest stage is essential, as unrecognized or inadequately treated arterial ischemia may progress to epidermal and dermal disruption, culminating in full-thickness skin necrosis (Figures 2A and 2B).21,28,43
In addition to cutaneous complications, FIVO may result in devastating ophthalmic and neurologic outcomes. A recent comprehensive literature review identified 365 cases of partial or complete vision loss following dermal filler injections.44 These complications were most frequently associated with injections in high-risk anatomical regions, particularly the glabella, forehead, and nasal dorsum, where critical vessels are closely linked to the ophthalmic arterial system.34,45,46 The underlying mechanisms of vision loss included occlusion of the ophthalmic artery, central retinal artery, branch retinal arteries, and posterior ciliary arteries.47,48
Clinical manifestations of filler-associated ocular ischemia included sudden-onset partial or complete vision loss, visual field deficits, afferent pupillary defects, ophthalmoplegia, blepharoptosis, and, in some cases, neurologic symptoms such as ptosis, nausea, vomiting, and headache.27 Rarely, patients exhibited stroke-like symptoms, brain infarction, hemiplegia, or limb weakness due to retrograde embolization of dermal fillers from the injection area into the internal carotid artery and cerebral circulation. This may progress to postinfarction hemorrhage and carries a high risk of severe brain injury or death.44,49 Vision loss typically occurred immediately or within 10 minutes following the injection, highlighting the need for prompt recognition and urgent intervention. Outcomes ranged from irreversible vision loss to partial or, in some cases, complete visual recovery.44,50,51
Management of filler-induced vascular occlusion. Prompt recognition and intervention are critical in cases of FIVO to minimize tissue ischemia and prevent irreversible complications.11 The immediate priority is to stop injection of the filler at the first sign of vascular compromise. Warm compresses should be applied to promote vasodilation and improve local tissue perfusion. In any case of stroke or visual symptoms, urgent referral to a neurologist, ophthalmologist, or retinal specialist is mandatory. Multidisciplinary management, including neurology, ophthalmology, and plastic surgery, may be necessary for optimal outcomes.52 Although the standard therapeutic window for effective ocular reperfusion is generally considered to be up to 90 minutes, some reports suggest that retinal ischemia may become irreversible in as little as 10 to 15 minutes.29,34,44
For FIVO involving hyaluronic acid fillers, prompt administration of hyaluronidase is the cornerstone of treatment.5 Enzymatic degradation of the filler may be achieved through multiple high-dose intradermal or subcutaneous injections in the affected area. In severe cases, including vision loss, hyaluronidase has been administered into the retrobulbar, supratrochlear, or periorbital regions, and in some cases, both hyaluronidase and thrombolysis have been delivered via intra-arterial injection.40,53–56 A report described a 27-year-old woman with complete vision loss following nasal hyaluronic acid injection. Intra-arterial hyaluronidase led to partial recovery, and subsequent thrombolysis with alteplase and hyaluronidase achieved ophthalmic artery recanalization and further improvement.57
Adjunctive therapies may include systemic or topical vasodilators such as nitroglycerin paste or oral tadalafil or sildenafil to enhance microcirculation, although care must be taken to avoid excessive vasodilation, which may inadvertently promote migration of filler material to other vascular territories.31,58,59 Additional pharmacologic interventions include the use of antiplatelet agents such as aspirin, corticosteroids to mitigate inflammation, and anticoagulants such as low-molecular-weight heparin. In select cases, thrombolytic agents like alteplase have been considered.20,21,23 Ultrasound assessment may be performed following emergency intervention to evaluate vascular competence in potentially compromised areas.23
HBOT is an emerging and scientifically supported adjunctive treatment (Table 2). It is particularly beneficial in improving tissue oxygenation in both early and delayed presentations of vascular occlusion.7,15 HBOT may help reduce tissue hypoxia, modulate inflammation, and support neovascularization, especially when used alongside conventional therapies.26
HBOT in the setting of filler-induced vascular occlusion. HBOT represents a valuable adjunctive modality in the treatment of FIVO, particularly when tissue ischemia is prolonged. Under physiological conditions, oxygen is primarily transported in the blood bound to hemoglobin, with only a small fraction dissolved in the plasma.15,60 The therapeutic effect of HBOT can be understood through Henry’s law, which states that the amount of gas dissolved in a liquid is directly proportional to the pressure applied to it.14,16 Accordingly, breathing 100% oxygen under elevated atmospheric pressure increases the concentration of dissolved oxygen in plasma, potentially exceeding the oxygen-carrying capacity of hemoglobin.61–63
At sea level (1 atmosphere absolute [ATA]), nearly all oxygen is bound to hemoglobin, with plasma containing only about 0.32% dissolved oxygen.15,64 Breathing 100% oxygen at 1 ATA increases this value to approximately 2.09%, and, at 3 ATA, the dissolved oxygen concentration can reach 6.8%, over 21 times higher than normobaric conditions.25,62 Arterial oxygen tensions during HBOT may rise to levels as high as 2,000 mm Hg.14 This hyperoxygenation promotes oxygen delivery even in areas with impaired microcirculation, thereby preventing tissue hypoxia and necrosis.41,65
HBOT is delivered in specialized single- or multiperson chambers where patients inhale 100% oxygen in a pressurized environment.66 The minimum therapeutic pressure for HBOT is typically 1.4 ATA, though elective treatments are generally conducted at 2 to 3 ATA for durations of 60 to 120 minutes, depending on clinical indications.62,67,68 In dermatology, plastic surgery, and aesthetic medicine, HBOT has been used successfully for ischemic ulcers, compromised flaps and grafts, posthair transplantation recovery, and keloid scars.2,22 Its primary goal is to improve oxygenation in hypoxic tissues, reduce inflammation, enhance regeneration, and prevent necrosis.61,69
Tissue oxygen pressures of at least 30 mm Hg are considered necessary for adequate wound healing.64 HBOT raises the partial pressure of oxygen in the blood and tissues, supporting oxygen-dependent cellular processes and the recovery of mitochondrial function even when arterial perfusion is inadequate, as in cases of FIVO.33,39,45 Additionally, HBOT exerts effects on erythrocytes by promoting osmotic fluid loss, leading to shrinkage and deformation of red blood cells.27 This may enhance microvascular flow and tissue oxygenation.60,64
Another relevant mechanism is the Robin Hood effect, wherein hyperoxia-induced vasoconstriction occurs in normoxic tissues but not in hypoxic regions.51,65 This differential response leads to a redistribution of blood flow toward ischemic areas. Similarly, HBOT reduces interstitial edema by decreasing capillary hydrostatic pressure, consistent with Starling’s law, while elevating plasma oncotic pressure, thereby promoting reabsorption of fluid from the interstitial space.14,60,66
HBOT also demonstrates bactericidal and immunomodulatory effects. High oxygen concentrations are toxic to anaerobic bacteria and improve leukocyte function by enhancing phagocytosis and oxidative burst capacity.51,70,71 Inflammatory signaling is also modulated through suppression of proinflammatory cytokines, such as IL-1, IL-6, IL-8, and tumor necrosis factor α, while levels of the anti-inflammatory cytokine IL-10 are increased.33,68,72
The upregulation of hypoxia-inducible factor (HIF) 1, a transcription factor critical to wound healing, is also another key mechanism.60 HIF-1 promotes keratinocyte migration and epithelial regeneration and induces the production of several prorepair growth factors, including vascular endothelial growth factor, platelet-derived growth factor, and stromal cell-derived factor.33,40,73 These signaling molecules enhance neovascularization, erythropoiesis, osteogenesis, fibroblast activation, and collagen synthesis, key processes for tissue recovery in hypoxic or ischemic environments.2,14,47,61
HBOT has been successfully used for the salvage of compromised skin flaps and grafts.16,69 The European Underwater and Baromedical Society recommends initiating HBOT immediately for such cases, using pressures of 2 to 2.5 ATA for 60 to 120 minutes per session.17 Initial treatment typically includes 2 to 3 sessions daily, followed by 1 to 2 sessions per day until clinical improvement is observed or necrosis resolves.26,66
In ophthalmology, HBOT has also been applied in the treatment of central retinal artery occlusion (CRAO) related to filler embolization.15,65 The therapy improves oxygenation of the retina and macula, particularly in hypoperfused regions affected by interstitial edema.15,25 In the first 10 minutes of HBOT, vasoconstriction may occur, but this is soon followed by vasodilation mediated by increased nitric oxide production. The choroidal circulation, which supplies approximately 60% of the oxygen delivered to the retina, plays a critical role during CRAO. HBOT can augment oxygen delivery from choroidal and collateral vessels to maintain retinal metabolism until reperfusion occurs.27,34 Adequate oxygenation of the inner retinal layers is critical to preserving vision, and early HBOT may help mitigate ischemic injury.
Although HBOT is considered an adjunctive therapy, its timely initiation is crucial for maximizing benefit in both cutaneous and ocular FIVO.73 Incorporating HBOT into a multidisciplinary management plan may significantly improve clinical outcomes in patients at risk of tissue necrosis or permanent vision loss.68,70
Although generally well tolerated, HBOT carries potential risks due to the elevated pressures and increased oxygen concentrations. The most common adverse effects are related to barotrauma and include ear and sinus discomfort, middle ear injuries such as tympanic membrane rupture, and, less frequently, sinus squeeze or barotitis.67,68 More serious complications include arterial gas embolism, pulmonary barotrauma, and, in rare instances, lung collapse, pulmonary oxygen toxicity, or pulmonary edema.66
One of the key mechanisms underlying HBOT-related toxicity is oxidative stress from excessive production of reactive oxygen species. This can contribute to cellular and tissue damage, including ocular effects such as lens toxicity and cataract development, particularly after prolonged exposure or cumulative treatment sessions exceeding 100.45,47 Although uncommon, central nervous system oxygen toxicity may also occur and can manifest as tonic-clonic (grand mal) seizures during or shortly after treatment.62
To mitigate the risk of adverse events, treatments are generally limited to pressures below 3 ATA and durations of less than 2 hours per session.42 Careful patient selection, pretreatment screening (eg, for existing pulmonary or otolaryngologic conditions, such as pneumothorax and eustachian tube dysfunction, respectively), and close monitoring during therapy are essential for safety.73
Another critical concern is the risk of fire due to the high oxygen concentrations in HBOT chambers. To mitigate this risk, the US National Fire Protection Association mandates strict operational and engineering safety standards in all HBOT facilities.16,65 These guidelines include rigorous protocols for ignition source control, patient attire, chamber construction materials, and staff training.
Contraindications to HBOT. HBOT is contraindicated in cases of untreated pneumothorax due to the risk of gas expansion during decompression, according to Boyle’s law.66 The use of certain chemotherapeutic agents, such as doxorubicin or cisplatin, is also considered an absolute contraindication because of the potential for enhanced oxidative tissue damage.45,65
HBOT can lead to fluid shifts from the third space into the intravascular compartment under elevated pressure, potentially exacerbating congestive heart failure.6 Additionally, the vasoconstrictive effects of hyperoxia may increase afterload, further complicating cardiac function.51 As such, HBOT should be administered with caution in patients with known heart failure.51,62
Other relative contraindications include pregnancy, epilepsy, upper respiratory infection, chronic obstructive pulmonary disease, asthma, pulmonary bullae, optic neuritis, claustrophobic individuals, and recent thoracic surgery.69,72
Guidelines and Future Directions
HBOT has emerged as a valuable adjunct in the management of FIVO and has been incorporated into treatment protocols reported in multiple case reports and case series (Table 2). However, despite encouraging clinical outcomes, further rigorous controlled studies are needed to establish optimal timing, dosing, and treatment parameters that influence efficacy. Available data suggest that HBOT should ideally be initiated within 24 hours of suspected FIVO to maximize therapeutic benefit. In cases involving ocular compromise, particularly CRAO, time is even more critical.55 The retina can typically tolerate no more than 90 to 100 minutes of ischemia before irreversible damage occurs, underscoring the importance of immediate ophthalmologic intervention and the potential use of subsequent HBOT.15,25,45
In the studies reviewed, the majority of reported cases involved female patients, predominantly in the third to fifth decades of life. The timing of intervention varied considerably across cases, ranging from within the first 24 hours to as late as 11 days postprocedure. In several reports, the exact timing of treatment initiation was not specified. Most cases demonstrated complete clinical improvement without residual sequelae. However, some cases reported persistent complications, including scarring, hypoesthesia, hyperpigmentation, or visual acuity impairment, in which treatment was initiated 11, 5, 3, and 6 days after the procedure, respectively. These findings suggest that earlier initiation of therapy may be associated with greater clinical benefit and a reduced risk of long-term sequelae. HBOT was generally administered on a daily basis at pressures ranging between 2.0 and 3.0 ATA, with session durations of 60 to 120 minutes, most commonly 90 minutes. The total number of sessions varied depending on clinical response, disease severity, and patient compliance. Importantly, in most cases, patients did not develop functional or anatomical sequelae; however, treatment was multimodal, with HBOT used as an adjunct to other therapies such as hyaluronidase, antiplatelet agents, vasodilators, and thrombolytic therapy.
Based on the author’s clinical experience, a preliminary protocol of 5 to 15 consecutive sessions of HBOT at 2 to 3 ATA, with 100% oxygen administered for at least 60 minutes per session, combined with daily clinical assessments of the affected area, may offer therapeutic benefit.
Despite these promising mechanistic and clinical findings, HBOT use in FIVO is limited by several challenges, and it is important to acknowledge the presence of several confounding factors when interpreting its role. There is no standardized treatment protocol specific to FIVO, and most current recommendations are extrapolated from protocols for compromised grafts, flaps, or other ischemic injuries. The optimal timing, pressure, session duration, and number of treatments remain undefined, and practice patterns vary widely across institutions. The available evidence is largely limited to case reports, small case series, and retrospective analyses, with a paucity of prospective studies or randomized controlled trials to confirm efficacy, determine cost effectiveness, and define patient selection criteria. Furthermore, most reported cases of FIVO have been managed using a multimodal therapeutic approach, including hyaluronidase, antiplatelet agents, thrombolytic therapy, and HBOT administered concurrently. As a result, clinical improvement cannot be attributed solely to HBOT, making it difficult to determine its isolated therapeutic effect. Consequently, HBOT has been applied as an adjunctive treatment, with reported benefits observed in combination with other interventions rather than as a monotherapy. HBOT should not replace first-line interventions such as prompt hyaluronidase administration and urgent ophthalmologic referral in the case of ocular compromise; its role as an adjunctive therapy warrants further clarification. Additional barriers include limited access in nonhospital or aesthetic practice settings, which can delay initiation, as well as the high cost of HBOT, limited insurance coverage, and the absence of robust data on long-term outcomes.
Future research should prioritize the development of standardized, evidence-based HBOT protocols for FIVO, the conduct of multicenter prospective studies or randomized trials to assess efficacy and safety, and the identification of patient- and procedure-specific factors predictive of treatment response. Such efforts are essential to optimize clinical outcomes and establish the definitive role of HBOT in managing filler-induced vascular complications.
Conclusion
The management of FIVO remains a complex, time-sensitive challenge that demands both technical expertise and rapid clinical decision-making. Successful outcomes depend on a thorough understanding of facial vascular anatomy, recognition of high-risk injection zones, and immediate initiation of appropriate diagnostic and therapeutic measures. Early diagnosis and prompt intervention are critical to minimizing ischemic injury and preserving tissue viability.
As a scoping review, this study does not aim to determine efficacy but rather to summarize how HBOT has been reported and applied in clinical practice. By analyzing reported patient outcomes in cases where HBOT was used as an adjunctive treatment, this review seeks to clarify its potential therapeutic contribution within a multimodal management approach. In doing so, this study aims to synthesize currently fragmented evidence, highlight existing knowledge gaps, and encourage further investigation into HBOT as a supportive intervention. Given the increasing and widespread use of facial fillers in aesthetic practice, along with the rising incidence of associated vascular complications, a clearer understanding of adjunctive therapies is clinically relevant.
Recognizing the limitations of this study, the available evidence is derived exclusively from case reports and case series, and no well-designed prospective or randomized studies currently exist. As such, this review represents a systematic synthesis of the existing literature rather than definitive evidence of efficacy, underscoring the need for future well-designed prospective studies to better define the role of HBOT in FIVO managemen and optimal treatment parameters, assess safety, and clarify the role of HBOT in managing vascular complications related to dermal fillers. Ultimately, this review intends to inform clinical decision-making, promote future research, and support strategies that may help reduce functional and anatomical sequelae associated with FIVO.
While HBOT is not a substitute for first-line measures such as hyaluronidase administration and urgent ophthalmologic referral, preliminary clinical observations support its role as a valuable adjunct in both early and select delayed presentations of FIVO. Its potential to improve oxygen delivery, attenuate inflammation, and support tissue repair warrants consideration in refractory or severe cases. Clinicians should be aware of the location and accessibility of nearby HBOT facilities to avoid delays in initiation.
Until standardized, evidence-based protocols are established, the integration of HBOT into the therapeutic algorithm for FIVO should be guided by multidisciplinary collaboration, individualized treatment planning, and ongoing clinical reassessment.
References
- Lacombe VG. Volumizing fillers for skin: selection strategies. Facial Plast Surg Clin North Am. 2023;31(4):521–524.
- Kruize RGF, Teguh DN, van Hulst RA. Hyperbaric oxygen therapy in hyaluronic acid filler-induced dermal ischemia. Dermatol Surg. 2020;46(12):1755–1757.
- American Society of Plastic Surgeons. 2023 ASPS procedural statics release. Plast Recon Surg. 2024;154(3S):1–41.
- Liu MH, Beynet DP, Gharavi NM. Overview of deep dermal fillers. Facial Plast Surg. 2019;35(3):224–229.
- Kroumpouzos G, Treacy P. Hyaluronidase for dermal filler complications: review of applications and dosage recommendations. JMIR Dermatol. 2024;7:e50403.
- Alves DD, Menezes HS, Chacur R, et al. Vascular complications with injection implants: treatment with hyperbaric chamber. Case Rep Plast Surg Hand Surg. 2021;8(1):81–86.
- Jalilian F, Hetz SP, Bostwick J, Boet S. Hyperbaric oxygen therapy for treatment of a late presenting ischaemic complication from hyaluronic acid cosmetic filler injection. BMJ Case Rep. 2022;15(7):e249190.
- Schelke L, Decates T, Kadouch J, Velthuis P. Incidence of vascular obstruction after filler injections. Aesthet Surg J. 2020;40(8):NP457–NP460.
- Soares DJ, Hynes SD, Yi CH, Shah-Desai S, Irving SC. Cosmetic filler-induced vascular occlusion: a rising threat presenting to emergency departments. Ann Emerg Med. 2024;83(1):59–67.
- Quach B, Clevens RA. Complications of injectables. Atlas Oral Maxillofac Surg Clin. 2024;32(1):57–63.
- van Loghem J, Funt D, Pavicic T, et al. Managing intravascular complications following treatment with calcium hydroxylapatite: an expert consensus. J Cosmet Dermatol. 2020;19(11):2845–2858.
- Aviv U, Haik J, Weiss N, et al. Treatment algorithm for hyaluronic acid-related complication based on a systematic review of case reports, case series, and clinical experience. Craniomaxillofacial Trauma Reconstr. 2020;13(4):313–328.
- Zaccaria G, Dotti A, Benanti E, Vigliarolo C, Vaienti L. A treatment algorithm for hyaluronic acid filler related complications of the face. J Plast Reconstr Aesthet Surg. 2024;91:207–217.
- Madero J, Salvador M, Kadouch J, Muñoz-Gonzalez C, Fakih-Gomez N. Role of hyperbaric oxygen in filler-induced vascular occlusion. Aesthetic Plast Surg. 2024;48(14):2713–2721.
- Celebi ARC. Hyperbaric oxygen therapy for central retinal artery occlusion: patient selection and perspectives. Clin Ophthalmol. 2021;15:3443–3457.
- Kleban SK, Baynosa RC. The effect of hyperbaric oxygen on compromised grafts and flaps. Undersea Hyperb Med. 2020;47(4):635–648.
- Sito G, Manzoni V, Sommariva R. Vascular complications after facial filler injection: a literature review and meta-analysis. J Clin Aesthetic Dermatol. 2019;12(6):E65–E72.
- Vargas-Laguna E, García-Gavín J, Bárcena-Ruiz E. Seguridad en los procedimientos dermatológicos: oclusión vascular por materiales de relleno. Actas Dermosifiliogr. 2021;112(9):794–797.
- Zengarini C, Sapigni C, Benati M, et al; Società Italiana di Dermatologia Chirurgica Oncologica Correttiva Ed Estetica Sidco. An out-of-season cold. Filler-induced vascular occlusion of the columellar artery causing necrosis of the cartilaginous anterior nasal septum: case report and a literature review. Dermatol Reports. 2025;17(1):10061.
- Murray G, Convery C, Walker L, Davies E. Guideline for the management of hyaluronic acid filler-induced vascular occlusion. J Clin Aesthetic Dermatol. 2021;14(5):E61–E69.
- DeLorenzi C. Complications of injectable fillers, part 2: vascular complications. Aesthet Surg J. 2014;34(4):584–600.
- Uittenbogaard D, Lansdorp CA, Bauland CG, Boonstra O. Hyperbaric oxygen therapy for dermal ischemia after dermal filler injection with calcium hydroxylapatite: a case report. Undersea Hyperb Med. 2019;46(2):207–210.
- Mehta P, Kaplan JB, Zhang-Nunes S. Ischemic complications of dermal fillers. Plast Aesthetic Res. 2022;9(10):57.
- Yang H, Zheng Y, Li D, et al. Facial artery branch thrombolysis for nasal vascular embolism induced by hyaluronic acid injection. Ann Plast Surg. 2024;93(6):658–663.
- Worley N, Lupo M, Holcomb K, Kullman G, Elahi E, Elison J. Hyperbaric oxygen treatment of keratitis following facial hyaluronic acid injection. Ochsner J. 2020;20(2):193–196.
- Simman R, Bach K. Role of hyperbaric oxygen therapy in cosmetic and reconstructive surgery in ischemic soft tissue wounds: a case series. Eplasty. 2022;22:e61.
- Nishant P, Morya AK, Sinha S, Sinha RK. Ophthalmic complications of injectable facial fillers. World J Clin Cases. 2024;12(34):6736–6739.
- Stevens G, Lewis I. A case of facial vascular occlusion after hyaluronic acid cosmetic filler injection treated with adjunctive hyperbaric oxygen. Diving Hyperb Med. 2025;55(1):56–58.
- Soares DJ. Bridging a century-old problem: the pathophysiology and molecular mechanisms of HA filler-induced vascular occlusion (FIVO)—implications for therapeutic interventions. Molecules. 2022;27(17):5398.
- Zaresharifi S, Gheisari M, Mohammadzadeh H, Layegh H, Amani M. Case report: management of polycaprolactone‐based dermal filler–induced vascular occlusion. J Cosmet Dermatol. 2025;24(1):e16779.
- Urdiales-Gálvez F, Delgado NE, Figueiredo V, et al. Treatment of soft tissue filler complications: expert consensus recommendations. Aesthetic Plast Surg. 2018;42(2):498–510.
- Balazic E, Nazarian RS, Hawkins K, Kobets K. Vessel infiltration with microcannula during filler injection: a rare but consequential occurrence. J Cosmet Laser Ther. 2023;25(5–8):74–76.
- Růžička J, Dejmek J, Bolek L, Beneš J, Kuncová J. Hyperbaric oxygen influences chronic wound healing – a cellular level review. Physiol Res. 2021;70(S3):S261–S273.
- Ortiz Middleton E. Evidence-based clinical guide for the management of peripheral vascular occlusion with hyaluronic acid, report of 6 successful cases. Adv Oral Maxillofac Surg. 2022;8:100367.
- Zeltzer A, Geeroms M, Antoniazzi E, et al. The “ART” of facial filler injections: avoid, recognize, and treat hyaluronic acid‐induced complications. J Cosmet Dermatol. 2020;19(9):2229–2236.
- Siperstein R. The use of a 27-gauge cannula in aesthetic medicine. Aesthetic Surg J Open Forum. 2022;4:ojac018.
- Siperstein RD, Broadfield DC, Yerke Hansen P, Conger JR, Zhang-Nunes SX. Arterial wall and tissue penetration force with various cannulas and needles. Aesthet Surg J. 2023;43(12):1521–1529.
- Pavicic T, Webb KL, Frank K, Gotkin RH, Tamura B, Cotofana S. Arterial wall penetration forces in needles versus cannulas. Plast Reconstr Surg. 2019;143(3):504e–512e.
- Clark NW, Pan DR, Barrett DM. Facial fillers: relevant anatomy, injection techniques, and complications. World J Otorhinolaryngol Head Neck Surg. 2023;9(3):227–235.
- Ling LIH. Successful management of nose arterial occlusion and impending skin necrosis after filler injection. J Cosmet Med. 2019;3(2):108–113.
- Johnson-Arbor K. Hyperbaric oxygen therapy for treatment of vascular occlusion after permanent dermal filler injection. Undersea Hyperb Med. 2024;51(4):403–406.
- King M, Walker L, Convery C, Davies E. Management of a vascular occlusion associated with cosmetic injections. J Clin Aesthetic Dermatol. 2020;13(1):E53–E58.
- Soares DJ, Bowhay A, Blevins LW, Patel SM, Zuliani GF. Patterns of filler-induced facial skin ischemia: a systematic review of 243 cases and introduction of the FOEM scoring system and grading scale. Plast Reconstr Surg. 2023;151(4):592e–608e.
- Doyon VC, Liu C, Fitzgerald R, et al. Update on blindness from filler: review of prognostic factors, management approaches, and a century of published cases. Aesthet Surg J. 2024;44(10):1091–1104.
- Tamaki A, Silverman DA, Ozer E. The role of hyperbaric oxygen in head and neck reconstruction and facial cosmetic surgery.Facial Plast Surg. 2020;36(6):753–759.
- Wollina U, Goldman A. Facial vascular danger zones for filler injections. Dermatol Ther. 2020;33(6):e14285.
- Chen IH, Cheng KC, Chen KJ. Iatrogenic ocular ischemic syndrome combined with central retinal artery occlusion following periorbital aesthetic poly‐D, L‐lactic acid filler injections – a case report. J Cosmet Laser Ther. 2025;27(3):98–101.
- Hwang K. Hyperbaric oxygen therapy to avoid blindness from filler injection. J Craniofac Surg. 2016;27(8):2154–2155.
- Zhao F, Chen Y, He D, You X, Xu Y. Disastrous cerebral and ocular vascular complications after cosmetic facial filler injections: a retrospective case series study. Sci Rep. 2024;14(1):3495.
- Lee W, Koh I, Oh W, Yang E. Ocular complications of soft tissue filler injections: a review of literature. J Cosmet Dermatol. 2020;19(4):772–781.
- Friedman R, Coombs AV, Stevens S, Lisman RD, Chiu ES. Complete vision recovery after filler-induced blindness using hyperbaric oxygen therapy: case report and literature review. Aesthetic Surg J Open Forum. 2024;6:ojae036.
- Arlette JP, Ashenhurst M, Hill V, Jiang K. Prevention and management of filler induced iatrogenic stroke of the eye. J Cutan Med Surg. 2021;25(5):543–552.
- Fakih-Gomez N, Muñoz-Gonzalez C, Porcar Plana CA, Puzo Bayod M, Madero J. Retrobulbar hyaluronidase in hyaluronic acid-induced ocular vascular occlusion: efficacy, challenges, and implications for clinical practice. Aesthetic Plast Surg. 2025;49(5):1458–1468.
- Navarro-Hernandez E, Pérez-López M. Effectiveness of retrobulbar hyaluronidase in the treatment of visual loss caused by periocular hyaluronic acid injection. A systematic review. Arch Soc Esp Oftalmol Engl Ed (Engl Ed). 2022;97(9):521–538.
- Thanasarnaksorn W, Cotofana S, Rudolph C, Kraisak P, Chanasumon N, Suwanchinda A. Severe vision loss caused by cosmetic filler augmentation: case series with review of cause and therapy. J Cosmet Dermatol. 2018;17(5):712–718.
- Borzabadi-Farahani A, Mosahebi A, Zargaran D. A scoping review of hyaluronidase use in managing the complications of aesthetic interventions. Aesthetic Plast Surg. 2024;48(6):1193–1209.
- Nguyen HH, Tran HTT, Duong QH, Nguyen MD, Dao HX, Le DT. Significant vision recovery from filler-induced complete blindness with combined intra-arterial injection of hyaluron-idase and thrombolytic agents. Aesthetic Plast Surg. 2022;46(2):907–911.
- Kleydman K, Cohen JL, Marmur E. Nitroglycerin: a review of its use in the treatment of vascular occlusion after soft tissue augmentation. Dermatol Surg. 2012;38(12):1889–1897.
- Chuchvara N, Alamgir M, John AM, Rao B. Dermal filler-induced vascular occlusion successfully treated with tadalafil, hyaluronidase, and aspirin. Dermatol Surg. 2021;47(8):1160–1162.
- Cannellotto M, Yasells García A, Landa MS. Hyperoxia: effective mechanism of hyperbaric treatment at mild-pressure. Int J Mol Sci. 2024;25(2):777.
- Gehmert S, Geis S, Lamby P, et al. Evaluation of hyperbaric oxygen therapy for free flaps using planar optical oxygen sensors. Preliminary results. Clin Hemorheol Microcirc. 2011;48(1–3):75–79.
- Kirby JP, Snyder J, Schuerer DJE, Peters JS, Bochicchio GV. Essentials of hyperbaric oxygen therapy: 2019 review. Mo Med. 2019;116(3):176–179.
- Tsai M, Hsia T, Han Y, Wu H, Lin Y. Successful hyperbaric oxygen therapy in complications of fillers rhinoplasty-cases report. Internet J Altern Med. 2014;9(1):6.
- Rodriguez‐Valera A, Nieto‐Lopez F. Hyperbaric oxygenation therapy improve recovery in early or late vascular occlusion generates by tissue fillers. J Cosmet Dermatol. 2023;22(7):1969–1972.
- Parnis J, Magrin AMF, Hassan H. The role, safety, and efficacy of hyperbaric oxygen therapy in aesthetic practice–an evidence‐based review. J Cosmet Dermatol. 2024;23(6):1940–1955.
- Jeter JP, Wong EB. Hyperbaric oxygen therapy in dermatology. Cutis. 2020;105(1):24–27.
- Henderson R, Reilly DA, Cooper JS. Hyperbaric oxygen for ischemia due to injection of cosmetic fillers: case report and issues. Plast Reconstr Surg Glob Open. 2018;6(1):e1618.
- Ríos-Gómez M, Gómez-Ortega V, Cardona C, De la Hoz-Valle JA. Hyperbaric oxygen therapy in plastic, aesthetic, and reconstructive surgery: systematic review. Surg Tech Dev. 2023;12(1):43–52.
- Hong WT, Kim J, Kim SW. Minimizing tissue damage due to filler injection with systemic hyperbaric oxygen therapy. Arch Craniofacial Surg. 2019;20(4):246–250.
- Latusek K, Slotwinska A, Michniak A, Orzechowska-Wyelgala B. Effects of hyperbaric oxygen therapy on periodontal disease: a literature review. Undersea Hyperb Med. 2023;50(1):17–27.
- Memar MY, Yekani M, Alizadeh N, Baghi HB. Hyperbaric oxygen therapy: Antimicrobial mechanisms and clinical application for infections. Biomed Pharmacother. 2019;109:440–447.
- Sen S, Sen S. Therapeutic effects of hyperbaric oxygen: integrated review. Med Gas Res. 2021;11(1):30–33.
- Oley MH, Oley MC, Mawu FO, Aling DMR, Faruk M. Hyperbaric oxygen therapy in managing minimally invasive aesthetic procedure complications: a report of three cases. Clin Cosmet Investig Dermatol. 2022;15:63–68.
