J Clin Aesthet Dermatol. 2026;19(5):35–42.
by Alice B. Gottlieb, MD, PhD, and Joseph F. Merola, MD, MMSc
Drs. Gottlieb and Merola are with the University of Texas Southwestern Medical Center, Dallas, Texas.
FUNDING: Medical writing and editorial support were provided by Jordyn Karliner, PhD, and Nitish Chaudhari, PhD, of Red Nucleus, and funded by Sun Pharma.
DISCLOSURES: Dr. Gottlieb has received research/educational grants all paid to The Icahn School of Medicine at Mount Sinai until May 1, 2025, from Bristol Myers Squibb, Janssen, MoonLake Immunotherapeutics, Sanofi, and UCB; and has received honoraria as an advisory board member, consultant, and/or speaker from AbbVie, Amgen, Boehringer Ingelheim, Bristol Myers Squibb, Janssen, Lilly, Novartis, Oruka Therapeutics, Sanofi-Aventis, Sun Pharma, Takeda, and UCB. Dr. Merola is a consultant and/or investigator for AbbVie, Amgen, Biogen, Boehringer Ingelheim, Bristol Myers Squibb, Dermavant, Incyte, Janssen, LEO Pharma, Lilly, Novartis, Pfizer, Regeneron Pharmaceuticals, Sanofi, Sun Pharma, and UCB.
ABSTRACT: BACKGROUND: Tildrakizumab is an anti-interleukin 23 p19 monoclonal antibody approved for the treatment of adults with moderate-to-severe plaque psoriasis. Globally, patients aged ≥65 years are 9.8% to 12.8% of patients with psoriasis who are treated with biologics, but they are often underrepresented in clinical trials. OBJECTIVE: To summarize the available evidence on the efficacy and safety of tildrakizumab in patients aged ≥65 years with psoriasis for a United States (US)-based healthcare practitioners audience. METHODS: A PubMed search was conducted using the search terms “(tildrakizumab OR MK-3222 OR Ilumya) AND psoriasis AND (elderly OR 65 years)” to identify English-language clinical trials and real-world studies evaluating efficacy and safety of tildrakizumab in elderly patients with psoriasis. RESULTS: Two clinical trials and 5 real-world studies describing the efficacy and safety of tildrakizumab in patients aged ≥65 years were identified. The short- and long-term efficacy of tildrakizumab was comparable between patients aged <65 and ≥65 years. Adverse events were consistent with those observed among the overall clinical trial population, with some expected increases in age-related events. LIMITATIONS: The number of studies with data for patients aged ≥65 years, and sample sizes within some studies, were small, limiting interpretation. CONCLUSION: Based on available data, the established efficacy and safety profiles of tildrakizumab are maintained in patients aged ≥65 years with moderate-to-severe plaque psoriasis. Practical considerations, including insurance coverage, quarterly maintenance dosing, and in-office administration, in the US may increase treatment adherence and make tildrakizumab an appropriate treatment option for elderly patients. Keywords: Biologics, elderly patients, narrative review, psoriasis, tildrakizumab
Introduction
Psoriasis is a chronic, immune-mediated inflammatory skin disease that affects approximately 0.5% to 11.4% of adults globally as of a 2017 systematic review.1,2 The age at psoriasis onset has a bimodal distribution, with peaks between 15 to 25 years and 50 to 60 years of age.3 The prevalence of psoriasis tends to increase with age due to its chronic nature and the relatively normal life expectancy of affected patients compared to the general population.4,5 For example, based on electronic health records in England as of 2019, the prevalence of psoriasis was approximately 2.5% in the overall population and 4.25% in patients aged ≥65 years.6 In the United States (US), 2011 Medicare claims data indicate a prevalence of 0.51% to 1.23% among patients aged ≥65 years.7 Global data from national registries for patients receiving biologic treatment for psoriasis indicate that 9.8% to 12.8% of enrollees are aged ≥65 years,8–10 and this number will likely grow due to increasing life expectancy of the general population.11 Despite the substantial population of elderly patients among those with psoriasis, patients aged ≥65 years are often underrepresented in clinical trials of psoriasis treatments, leading to limited data on efficacy and safety in this population.12,13
Treatments for psoriasis in elderly patients. The management of psoriasis in elderly patients is challenging for many reasons, including the higher prevalence of comorbidities, associated polypharmacy, decreased immune function, reliance on assistance for treatment, and potential cognitive impairments due to dementia.4,14–17 Treatment options include topical therapies, phototherapies, systemic therapies, and biologics; however, most options have drawbacks both in general and specifically for elderly patients.
Topical therapies, including corticosteroids, calcineurin inhibitors, vitamin D analogs, retinoids, and salicylic acid, are associated with poor compliance and the potential for cutaneous adverse events (AEs), including atrophy, purpura, telangiectasia, secondary skin infections, rebound phenomenon, and tachyphylaxis.7,18 Additionally, these therapies are often not sufficiently effective for patients with involvement of extensive body surface area or high-impact areas, including the face, palms, soles, genitalia, scalp, or nails.1,19 Phototherapy options, such as narrowband UV-B and psoralen plus UV-A, are effective but may lead to development of skin cancer, for which elderly patients are already at increased risk due to cumulative lifetime sun exposure.1,14,20–22 Furthermore, many medications commonly used by elderly patients can lead to phototoxic or photoallergic cutaneous reactions.23,24 Additionally, phototherapy requires transportation to and from the hospital and the ability to tolerate long treatment sessions, which may pose logistical challenges for older patients, especially those with mobility limitations.25 In patients of all ages, conventional systemic therapies, including methotrexate, acitretin, and cyclosporine, carry risks of multiple drug interactions, immunosuppression, mucocutaneous ulcerations, hematologic abnormalities, hepatotoxicity, renal impairment, hypertension, and dry mouth; elderly patients are already at increased risk for many of these conditions.4,14,26–30
Biologic therapies, including tumor necrosis factor (TNF) α, interleukin (IL)-12/23, IL-17, and IL-23 inhibitors, offer targeted treatment but are associated with infections, including an increased risk of tuberculosis with TNF-α inhibitors.31–33 Consistent with these findings, the prescribing information for biologics approved for the treatment of psoriasis warns of the potential for infections, serious infections, malignancies, hypersensitivity reactions, inflammatory bowel disease, and noninfectious pneumonia with treatment, along with the need to monitor patients for tuberculosis and avoid live vaccines.34–37 Safety is frequently a concern for elderly patients; in a real-world study using data from the British Association of Dermatologists Biologics and Immunomodulators Register, patients aged ≥65 years were more likely to discontinue treatment with biologics due to AEs relative to those aged 45 to 54 years.38 Consistent with this finding, in a retrospective claims analysis of US Medicare patients who initiated treatment with adalimumab, etanercept, infliximab, or ustekinumab, 38.5% to 53.2% of patients aged ≥65 years discontinued treatment compared to 30.6% to 50.3% of patients aged <65 years.39 Another potential barrier to treatment is that newer oral medications and many biologics may be prohibitively expensive for elderly patients; additionally, self-administered medications are often unaffordable in the US due to the high co-insurance required by Medicare Part D.
The pro-inflammatory cytokine IL-23 is a key mediator of inflammation in psoriasis and thus represents an important target for systemic treatment.40 Three IL-23 inhibitors, guselkumab, risankizumab, and tildrakizumab, are approved in the US for the treatment of adults with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.36,41,42 In our clinical experience, tildrakizumab, a humanized anti–IL-23 p19 monoclonal antibody, may be an underappreciated option for the treatment of elderly patients with psoriasis, as it offers a variety of benefits over other biologics approved to treat psoriasis. For example, while safety warnings include hypersensitivity, infections, avoidance of live vaccines, and screening for tuberculosis, there are no warnings for malignancies, serious infections, inflammatory bowel disease, or noninfectious pneumonia with treatment. Additionally, the efficacy and safety of tildrakizumab are similar in patients with vs without comorbidities. In clinical trials, tildrakizumab efficacy and safety were comparable in patients with vs without metabolic syndrome through 5 years of treatment, and there were no clinically meaningful differences in efficacy between patients with vs without psoriatic arthritis (PsA) after 12 weeks of treatment.43,44 In a real-world study of patients treated with tildrakizumab for 52 weeks, the efficacy of tildrakizumab was comparable in patients with vs without PsA and significantly greater in patients with vs without cardiometabolic comorbidities.45 Additional benefits of tildrakizumab for use in the US include that it is administered by a healthcare practitioner with a 12-week maintenance dosing interval and is therefore the only inhibitor of the IL-23/T helper 17 cell pathway that qualifies for reimbursement under Medicare Part B.34–36,41,42,46–48 Herein, we review the published literature describing the efficacy and safety of tildrakizumab in elderly patients with moderate-to-severe plaque psoriasis for a US healthcare practitioner audience to further explore its utility in this population.
Methods
A PubMed search was conducted on August 6, 2025, using the search terms “(tildrakizumab OR MK-3222 OR Ilumya) AND psoriasis AND (elderly OR 65 years)” to identify English-language clinical trials and real-world studies evaluating the efficacy and safety of tildrakizumab in elderly patients with psoriasis. Two articles reporting clinical trial results and 4 publications describing real-world studies were identified; a fifth real-world study not indexed in PubMed was identified serendipitously and included after review.
Results
Clinical trials of tildrakizumab. The efficacy and safety of tildrakizumab in patients with moderate-to-severe plaque psoriasis were demonstrated in two phase 3, randomized, double-blind clinical trials, reSURFACE 1 (NCT01722331) and reSURFACE 2 (NCT01729754).In both trials, treatment with tildrakizumab 100 mg was significantly more effective than placebo with few specific safety concerns.49 In pooled analyses from the reSURFACE 1 and reSURFACE 2 extension trials, the efficacy and safety profiles of tildrakizumab were sustained for up to 5 years.50 These results represent data from patients aged ≥18 years.49 Given the high burden of comorbidities and the potential impact of immunosenescence in older adults, data from clinical trial populations with a wide age range may not be representative of results in elderly patients4,51; however, subgroup analyses of the trial data provide evidence on the efficacy and safety of tildrakizumab in patients aged ≥65 years.
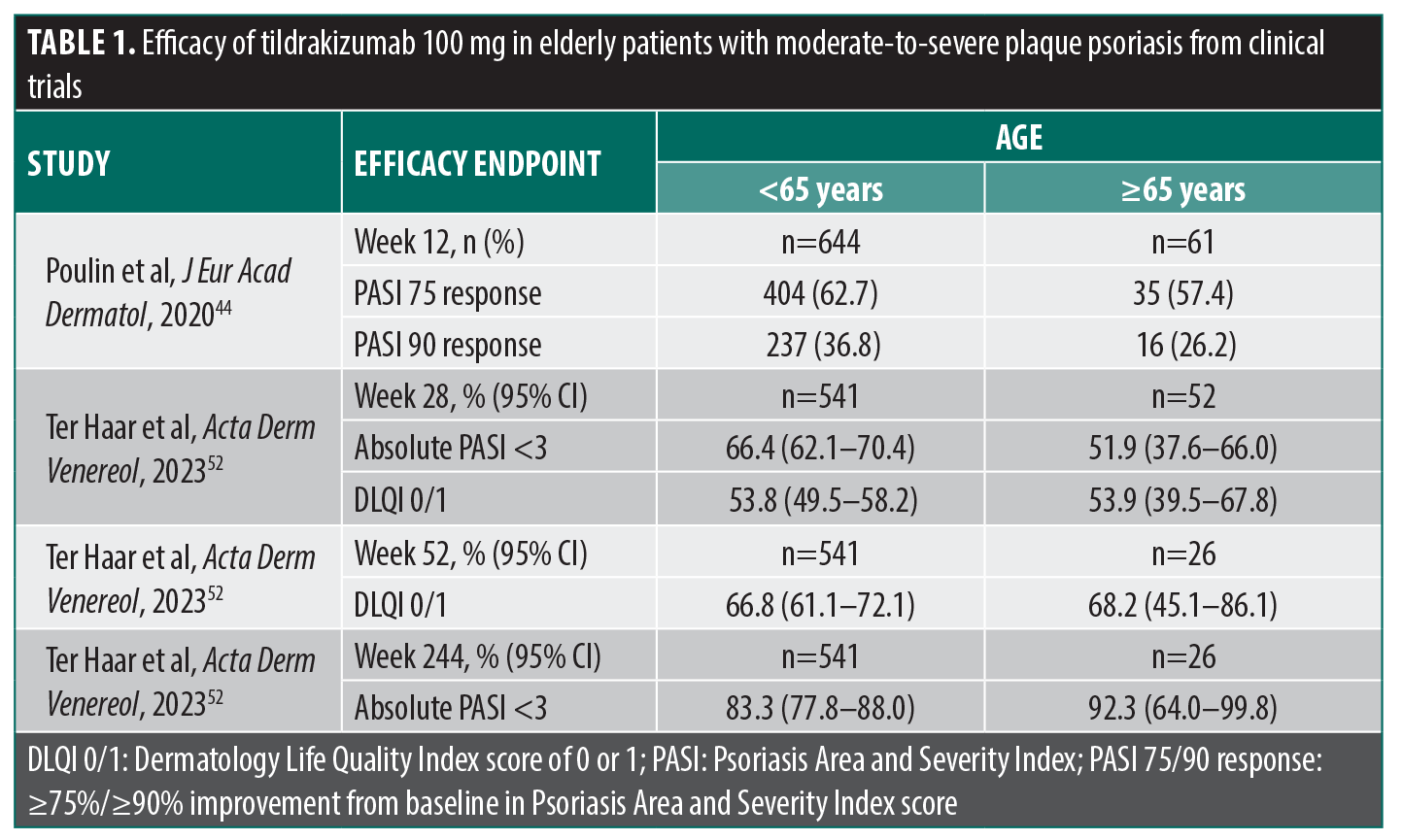
Efficacy of tildrakizumab in elderly patients with psoriasis in clinical trials. Short-term efficacy. A post hoc pooled data analysis from the phase 2b trial and the two phase 3 trials (reSURFACE 1 and reSURFACE 2) evaluated the efficacy of tildrakizumab in patients with moderate-to-severe plaque psoriasis stratified by patient characteristics; data were summarized for the subgroups of patients treated with tildrakizumab 100 mg, which is the dose approved in the US, defined by age (<65 years [n=644] and ≥65 years [n=61]).42,44 The proportions of patients achieving ≥75% and ≥90% improvement from baseline in Psoriasis Area and Severity Index (PASI 75 and PASI 90, respectively) score at Week 12 were evaluated.44 Among patients treated with tildrakizumab 100 mg, 62.7% of patients aged <65 years and 57.4% of patients ≥65 years achieved PASI 75 response at Week 12; 36.8% of patients aged <65 years vs 26.2% of patients ≥65 years achieved PASI 90 response at Week 12 (Table 1).44 While formal statistical analyses were not conducted to evaluate age-related differences, both age categories had higher rates of response to tildrakizumab vs placebo. For PASI 75 response, the point estimate difference for tildrakizumab vs placebo was 57.3% (95% CI: 52.5%–61.7%) for patients aged <65 years and 50.2% (95% CI: 31.5%–65.3%) for those ≥65 years; for PASI 90 response, the point estimate difference was 34.7% (95% CI: 30.2%–38.9%) for patients aged <65 years of age and 24.9% (95% CI: 11.1%–39.6%) for those ≥65 years.44
Long-term efficacy. A post hoc pooled data analysis of reSURFACE 1, reSURFACE 2, and their extension studies assessed the efficacy of tildrakizumab 100 mg in patients aged <65 years and ≥65 years through Week 244 of treatment.52 The percentage of patients achieving absolute PASI <3 was evaluated at Weeks 28, 52, and 244; the percentage of patients achieving a Dermatology Life Quality Index score of 0 or 1 (DLQI 0/1) was assessed at Weeks 28 and 52.52 Week 28 analyses included all patients randomized to tildrakizumab who received at least 1 dose of study medication; due to study design limitations, analyses at Week 52 and Week 244 included only patients who achieved a PASI 75 response at Week 28. At baseline, the mean (SD) PASI score of patients treated with tildrakizumab 100 mg was 20.2 (7.9) for patients <65 years of age and 18.9 (6.2) for patients ≥65 years of age. At Week 28, 66.4% of patients <65 years of age vs 51.9% of patients ≥65 years of age achieved a PASI score <3; long term, 83.3% of patients aged <65 years and 92.3% of patients ≥65 years had a PASI score <3 at Week 244 (Table 1).52 At baseline, mean (SD) DLQI score was 14.4 (7.0) for patients aged <65 years and 12.8 (7.6) for patients ≥65 years; 53.8% of patients aged <65 years and 53.9% of patients ≥65 years achieved DLQI 0/1 at Week 28. At Week 52, 66.8% of patients aged <65 years and 68.2% of patients ≥65 years achieved DLQI 0/1 (Table 1).52 Overall, although the number of patients aged ≥65 years was limited, long-term PASI and DLQI responses were comparable between patients aged <65 and ≥65 years.
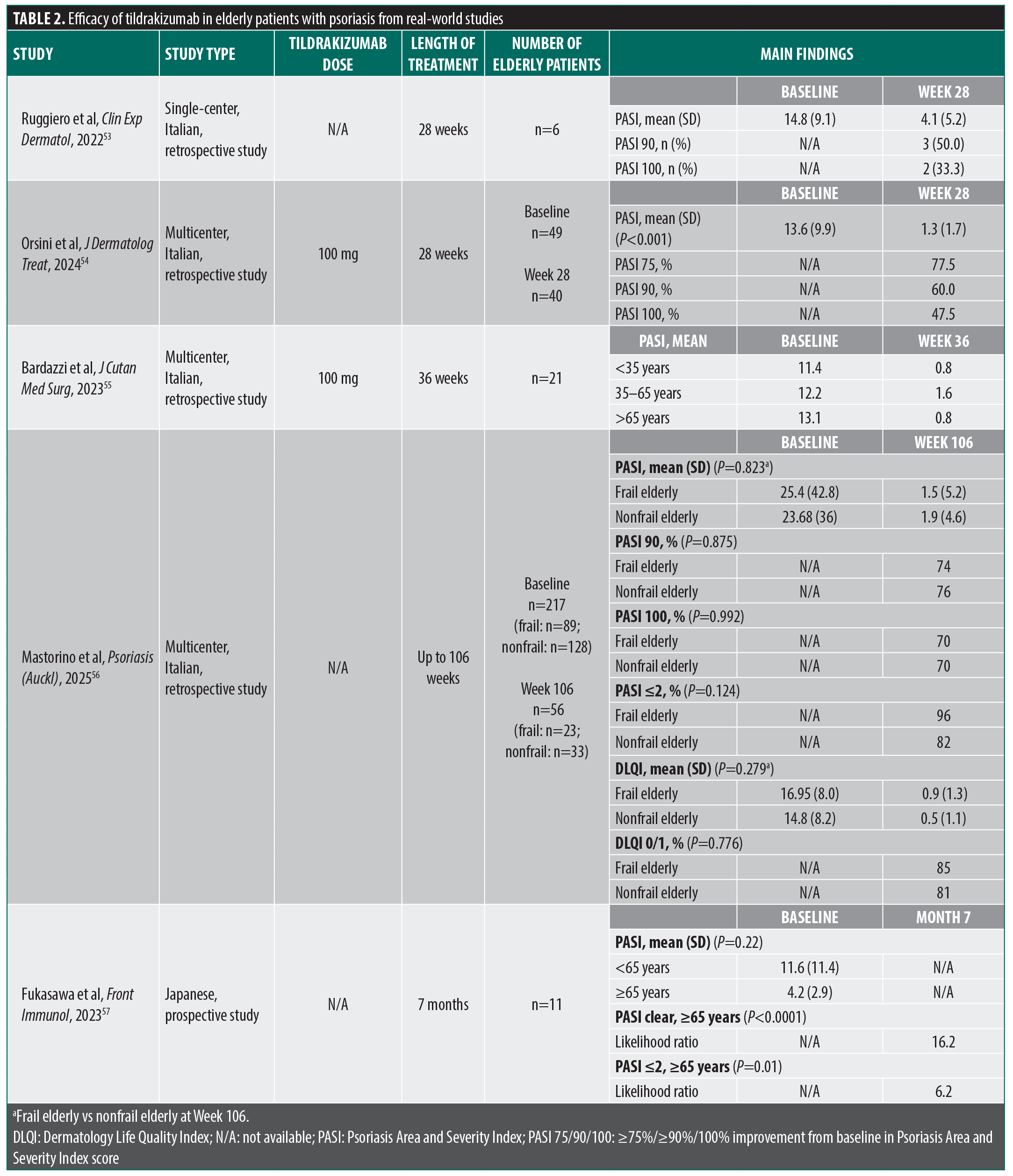
Efficacy in real-world studies. Real-world studies evaluating the efficacy of tildrakizumab in elderly patients with psoriasis were conducted in Italy and Japan (Table 2). A single-center, Italian retrospective study included patients aged ≥65 years with moderate-to-severe plaque psoriasis treated with tildrakizumab, guselkumab, or risankizumab.53 A total of 6 patients treated with tildrakizumab (dose not reported) for 28 weeks were included. The mean (SD) PASI score decreased from 14.8 (9.1) at baseline to 4.1 (5.2) at Week 28 (Table 2).53 At Week 28, 3 of 6 (50.0%) patients achieved PASI 90 response and 2 of 6 (33.3%) achieved 100% improvement from baseline PASI score (PASI 100 response; Table 2).53 No significant difference in effectiveness at Week 28 was observed among patients treated with guselkumab, risankizumab, and tildrakizumab.53
A multicenter Italian retrospective study (ESTER) evaluated 49 patients aged ≥65 years with moderate-to-severe plaque psoriasis treated with tildrakizumab 100 mg for 28 weeks (n=40).54 Mean (SD) PASI score significantly decreased from 13.6 (9.9) at baseline to 1.3 (1.7) at Week 28 (P<0.001; Table 2).54 At Week 28, 77.5% of patients achieved PASI 75 response, 60.0% achieved PASI 90 response, and 47.5% achieved PASI 100 response (Table 2).54
Another multicenter Italian retrospective study evaluated the efficacy and safety of tildrakizumab 100 mg in patients with moderate-to-severe plaque psoriasis treated with tildrakizumab for up to 36 weeks.55 Response to treatment was assessed in patients stratified by age: <35, 35 to 65, and >65 years (n=21).55 The oldest group of patients (>65 years) achieved greater improvement in mean PASI score (from 13.1 at baseline to 0.8 at Week 36) compared to those <35 years (from 11.4 at baseline to 0.8 at Week 36) and 35 to 65 years (from 12.2 at baseline to 1.6 at Week 36; Table 2).55
A third multicenter Italian retrospective study evaluated the efficacy and safety of tildrakizumab (dose not reported) for up to 2 years in patients aged ≥65 years (n=217) stratified into frail elderly (n=89) and nonfrail elderly cohorts (n=128).56 Patients were considered frail if they had 2 major comorbidities or had low income with 1 major comorbidity and/or had 2 of the following: weight loss, weakness, sluggishness, low activity level, and exhaustion. Among all patients aged ≥65 years, mean (SD) PASI score decreased from 24.36 (38.8) at baseline to 4.09 (6.6) at Week 16 (n=187), 2.35 (5.7) at Week 28 (n=179), 2.8 (8.1) at Week 52 (n=165), 4.8 (24.5) at Week 80 (n=86), and 1.7 (4.8) at Week 106 (n=56).56 By Week 106, 75% of patients achieved PASI 90 response, 70% achieved PASI 100 response, and 88% achieved a PASI score ≤2.56 Patients’ mean (SD) DLQI score decreased from 15.7 (8.1) at baseline to 1.5 (2.2) at Week 52 and 0.7 (1.2) at Week 106; 83.3% of patients achieved DLQI 0/1 at Week 106.56 No significant differences in PASI or DLQI scores were observed between frail and nonfrail patients aged ≥65 years (P>0.05). The percentage of patients who achieved PASI 90 response, PASI 100 response, a PASI score ≤2, and DLQI 0/1 did not significantly differ between frail and nonfrail elderly patients (P>0.05; Table 2).56
Finally, a Japanese prospective study evaluated patients with plaque psoriasis without PsA (n=246) who were treated with tildrakizumab (dose not reported; n=20) for 7 months.57 Patients were stratified by age into elderly (aged ≥65 years, n=11) and nonelderly (<65 years, n=9) cohorts.57 At baseline, mean (SD) PASI score was 4.2 (2.9) for patients aged ≥65 years and 11.6 (11.4) for patients <65 years; the difference in PASI scores was not statistically significant (P=0.22; Table 2).57 An age ≥65 years significantly increased the likelihood of achieving PASI scores of clear (P<0.0001) and ≤2 (P=0.01) after 7 months of tildrakizumab treatment (Table 2).57 Treatment with tildrakizumab significantly increased the number and function of regulatory T and T helper 17 cells, potentially contributing to the efficacy of tildrakizumab in elderly patients.57
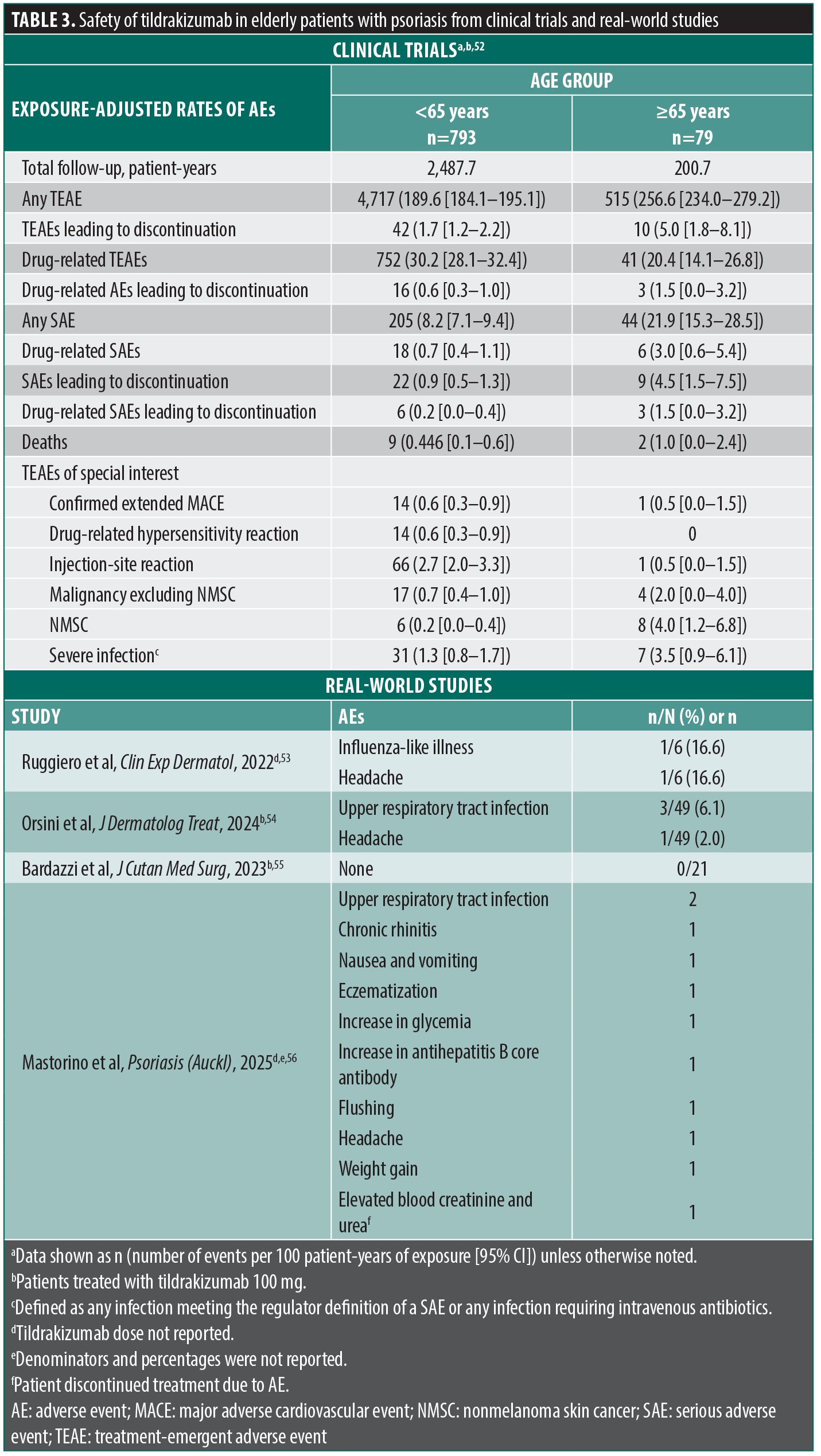
Safety of tildrakizumab in elderly patients with psoriasis. Among the above studies evaluating the efficacy of tildrakizumab in elderly patients with psoriasis, safety was also evaluated in 1 of the clinical trial analyses and 3 of the real-world studies. Overall, analysis of the reSURFACE 1 and reSURFACE 2 trials showed a consistent safety profile between patients aged <65 and ≥65 years treated with tildrakizumab 100 mg (all patients as-treated from baseline; Table 3).52 Total follow-up was 2,487.7 patient-years for patients aged <65 years and 200.7 patient-years for patients ≥65 years. The total number of AEs per 100 patient-years of exposure was 189.6 for patients aged <65 years and 256.6 for patients ≥65 years. The most frequent AE in both age groups was nasopharyngitis (cumulative incidence: <65 years, 621 per 100 patient-years of exposure; ≥65 years, 23 per 100 patient-years of exposure), followed by upper respiratory tract infection (cumulative incidence: <65 years, 168 per 100 patient-years of exposure; ≥65 years, 11 per 100 patient-years of exposure) and influenza (cumulative incidence: <65 years, 76 per 100 patient-years of exposure; ≥65 years, 3 per 100 patient-years of exposure).52 The number of serious AEs (SAEs) per 100 patient-years of exposure was 8.2 for patients aged <65 years and 21.9 for patients ≥65 years, and the number of AEs leading to discontinuation per 100 patient-years of exposure was 1.7 for patients aged <65 years vs 5.0 for patients ≥65 years. The number of deaths per 100 patient-years of exposure was 0.446 among patients aged <65 years and 1.0 among patients ≥65 years. Among AEs of special interest, patients aged ≥65 vs <65 years experienced proportionally greater numbers of serious infections (<65 years, 1.3 events per 100 patient-years of exposure; ≥65 years, 3.5 events per 100 patient-years of exposure), malignancies excluding nonmelanoma skin cancer (NMSC; <65 years, 0.7 events per 100 patient-years of exposure; ≥65 years, 2.0 events per 100 patient-years of exposure), and NMSC (<65 years, 0.2 events per 100 patient-years of exposure; ≥65 years, 4.0 events per 100 patient-years of exposure); these differences were attributed to the aging process rather than to the effects of tildrakizumab.52 Notably, other AEs of special interest occurred at similar or lower rates in elderly vs nonelderly patients, including major adverse cardiovascular events (<65 years, 0.6 events per 100 patient-years of exposure; ≥65 years, 0.5 events per 100 patient-years of exposure), injection-site reactions (<65 years, 2.7 events per 100 patient-years of exposure; ≥65 years, 0.5 events per 100 patient-years of exposure), and drug-related hypersensitivity reactions (<65 years, 0.6 events per 100 patient-years of exposure; ≥65 years, 0 events per 100 patient-years of exposure).52
The 4 Italian real-world studies described above included limited safety data, with few concerns described53–56; the Japanese prospective study did not include safety analyses.57 In 3 of the 4 Italian real-world studies (1 evaluated tildrakizumab 100 mg, 2 did not specify dosing), no patients experienced cardiovascular events or malignancies; the fourth Italian real-world study (tildrakizumab 100 mg) did not report on cardiovascular events or malignancies, though no patient experienced any SAE.53–55 Individual AEs were reported in 3 of the 4 studies (1 evaluated tildrakizumab 100 mg, 2 dosing not specified) and included headache, influenza-like illness, upper respiratory tract infection, chronic rhinitis, nausea and vomiting, eczematization, increase in glycemia, increase in anti-hepatitis B core antibody, flushing, and weight gain.53,54,56 In the study comparing frail elderly to nonfrail elderly patients, 4 of 10 AEs were reported in frail patients and 6 of 10 in nonfrail patients.56 In 2 of 3 studies reporting treatment discontinuations (tildrakizumab 100 mg and dosing not specified), no patients discontinued due to AEs (Table 3).53,55 In the study evaluating frail elderly vs nonfrail elderly patients (tildrakizumab dose not specified), 2 (7.4%) patients discontinued due to treatment-related AEs (1 event of elevated blood creatinine and urea, 1 unknown; Table 3); 3 patients died during the study, and all deaths were considered related to their comorbidities and not to tildrakizumab treatment.56 In the study including patients treated with guselkumab and risankizumab, the safety profile of tildrakizumab (dose not reported) in patients aged ≥65 years with psoriasis was comparable to those of the other IL-23 p19 inhibitors.53 Notably, the AEs identified in patients aged ≥65 years in the above studies were similar to those reported in clinical trials of tildrakizumab 100 mg in all patients aged ≥18 years, suggesting minimal additional safety concerns with tildrakizumab use with increasing age.49
Practical aspects of tildrakizumab for the treatment of psoriasis in elderly patients
Relative to other biologics, tildrakizumab has several characteristics that may contribute to treatment adherence and persistence in elderly patients with psoriasis. First, tildrakizumab has a reassuring safety profile.58 In a short-term (12–16 weeks from baseline) network meta-analysis, tildrakizumab 100 mg was associated with the lowest rates of AEs (46.3%) compared to other biologics, including etanercept (49.1%), certolizumab (52.2%), risankizumab (52.4%), and guselkumab (55.8%).58 Similarly, in the Cochrane Database systematic review of biologics for the treatment of psoriasis, tildrakizumab 100 mg and 200 mg combined had the highest Surface Under the Cumulative Ranking (ie, greatest probability of being the best treatment) in the analysis of all AEs.59 Safety is an especially relevant consideration for elderly patients; as previously noted, patients aged ≥65 years were more likely than those 45 to 54 years to discontinue treatment due to AEs in a real-world study of patients with psoriasis treated with biologics.38 Although clinical trial patients aged ≥65 years treated with tildrakizumab 100 mg experienced more discontinuations due to AEs relative to younger patients (5.0 vs 1.7 per 100 patient-years of exposure), only 3 patients discontinued due to AEs among the 244 with data in the real-world studies (1 evaluated tildrakizumab 100 mg, 2 did not specify dosing).52,53,55 Furthermore, in a 5-year clinical study of patients with psoriasis and concomitant metabolic syndrome, there were no additional safety concerns during treatment with tildrakizumab 100 mg relative to patients without metabolic syndrome, and both populations discontinued tildrakizumab treatment at similar rates.43 This is notable given that significantly more elderly vs nonelderly patients have metabolic syndrome and related comorbidities.4,14,15 Consistent with these findings, frail elderly patients—defined as those with at least 1 major comorbidity and/or other indicators—and nonfrail elderly patients treated with tildrakizumab (dose not reported) experienced AEs at similar frequencies.56
Tildrakizumab efficacy is also maintained in patients with vs without comorbidities that are common in elderly patients. In clinical and real-world studies, efficacy of tildrakizumab 100 mg was comparable in patients with vs without metabolic syndrome or PsA and significantly greater in a real-world study in patients with vs without cardiometabolic comorbidities.43–45 In the real-world study of frail elderly and nonfrail elderly patients, efficacy of tildrakizumab (dose not reported) was comparable between the 2 cohorts.56 Tildrakizumab also has a convenient dosing schedule; maintenance dosing is every 12 weeks after induction doses at Week 0 and Week 4, among the lowest frequencies for biologics used to treat psoriasis.34,36,37,42,46,47,60–62 Furthermore, in the US, tildrakizumab is administered in office by a healthcare practitioner, which may increase treatment adherence and improve clinical outcomes compared to self-administration.39,42,63 While in-office administration can be a barrier to patients who may require a caregiver for transportation, it can also make tildrakizumab especially financially accessible for elderly patients with psoriasis in the US, as tildrakizumab is covered under Medicare Part B for eligible patients.7
Limitations
There are some limitations to the studies discussed herein that may hinder the generalizability of their efficacy and safety findings. First, the studies included a small number of elderly patients, limiting the inclusion and interpretation of statistical analyses. Additionally, due to the design of the pivotal trials, the long-term efficacy analysis included only patients who achieved a PASI 75 response at Week 28 in the Week 52 and 244 results, introducing a potential survivorship bias. Finally, all of the real-world studies of tildrakizumab in elderly patients with psoriasis were conducted in Italy and Japan, limiting the generalizability of the results to patients in the US.
Conclusion
In clinical trials and real-world studies, the established clinical profile of tildrakizumab 100 mg is maintained in elderly patients. Both short-term and long-term efficacy of tildrakizumab in elderly patients with psoriasis are consistent with results in younger patients, and the few special safety concerns observed were consistent with conditions experienced by elderly patients in general and not attributed to tildrakizumab treatment. The infrequent dosing schedule, administration by healthcare practitioners, and coverage under Medicare Part B in the US are valuable benefits that can enhance accessibility and treatment adherence in elderly patients. Therefore, tildrakizumab may be an appropriate treatment option for many elderly patients with psoriasis.
Acknowledgements
Medical writing and editorial support were provided by Jordyn Karliner, PhD, and Nitish Chaudhari, PhD, of Red Nucleus and funded by Sun Pharma.
References
- Menter A, Strober BE, Kaplan DH, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with biologics. J Am Acad Dermatol. 2019;80(4):1029–1072.
- Michalek IM, Loring B, John SM. A systematic review of worldwide epidemiology of psoriasis. J Eur Acad Dermatol Venereol. 2017;31(2):205–212.
- Henseler T, Christophers E. Psoriasis of early and late onset: characterization of two types of psoriasis vulgaris. J Am Acad Dermatol. 1985;13(3):450–456.
- Fernandez-Torres RM, Paradela S, Fonseca E. Psoriasis in patients older than 65 years. A comparative study with younger adult psoriatic patients. J Nutr Health Aging. 2012;16(6):586–591.
- Armstrong AW, Mehta MD, Schupp CW, Gondo GC, Bell SJ, Griffiths CEM. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157(8):940–946.
- Vivekanantham A, Burn E, Moncusi MP, et al. P177 The prevalence and incidence of psoriasis and psoriatic arthritis in England from 2009-2019: an observational study using primary care data. Rheumatology. 2024;63(suppl 1):keae163.216.
- Takeshita J, Gelfand JM, Li P, et al. Psoriasis in the US Medicare population: prevalence, treatment, and factors associated with biologic use. J Invest Dermatol. 2015;135(12):2955–2963.
- Medina C, Carretero G, Ferrandiz C, et al; BIOBADADERM Study Group. Safety of classic and biologic systemic therapies for the treatment of psoriasis in elderly: an observational study from national BIOBADADERM registry. J Eur Acad Dermatol Venereol. 2015;29(5):858–864.
- Ter Haar ELM, Thomas SE, van den Reek J, et al. Drug survival, safety, and effectiveness of biologics in older patients with psoriasis: a comparison with younger patients-a BioCAPTURE registry study. Drugs Aging. 2022;39(9):715–727.
- Trettel A, Spehr C, Körber A, Augustin M. The impact of age on psoriasis health care in Germany. J Eur Acad Dermatol Venereol. 2017;31(5):870–875.
- Oeppen J, Vaupel JW. Demography. Broken limits to life expectancy. Science. 2002;296(5570):1029–1031.
- van Winden MEC, van der Schoot LS, van de L’Isle Arias M, et al. Effectiveness and safety of systemic therapy for psoriasis in older adults: a systematic review. JAMA Dermatol. 2020;156(11):1229–1239.
- Schaap MJ, van Winden MEC, Seyger MMB, de Jong EMG, Lubeek SFK. Representation of older adults in randomized controlled trials on systemic treatment in plaque psoriasis: a systematic review. J Am Acad Dermatol. 2020;83(2):412–424.
- van Winden MEC, Ter Haar ELM, Groenewoud HMM, van de Kerkhof PCM, de Jong EMGJ, Lubeek SFK. Disease and treatment characteristics in geriatric psoriasis: a patient survey comparing age groups. Acta Derm Venereol. 2020;100(14):adv00215.
- Phan C, Sigal ML, Estève E, et al; GEM RESOPSO. Psoriasis in the elderly: epidemiological and clinical aspects, and evaluation of patients with very late onset psoriasis. J Eur Acad Dermatol Venereol. 2016;30(1):78–82.
- Zingel R, Jacob L, Smith L, Konrad M, Kostev K. Association between psoriasis and dementia: a retrospective cohort study. J Alzheimers Dis Rep. 2023;7(1):41–49.
- Liu Y, Li D, Yu X, Li X. Identification of a novel immunosenescence-related genes and integrative analyses in patients with psoriasis. Hum Immunol. 2025;86(1):111218.
- Grozdev IS, Van Voorhees AS, Gottlieb AB, et al; National Psoriasis Foundation. Psoriasis in the elderly: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2011;65(3):537-545.
- Strober B, Ryan C, van de Kerkhof P, et al; International Psoriasis Council Board Members and Councilors. Recategorization of psoriasis severity: Delphi consensus from the International Psoriasis Council. J Am Acad Dermatol. 2020;82(1):117–122.
- Stern RS, Lange R. Non-melanoma skin cancer occurring in patients treated with PUVA five to ten years after first treatment. J Invest Dermatol. 1988;91(2):120–124.
- Lindelöf B, Sigurgeirsson B, Tegner E, et al. PUVA and cancer: a large-scale epidemiological study. Lancet. 1991;338(8759):91–93.
- Dennis LK. Cumulative sun exposure and melanoma in a population-based case-control study: does sun sensitivity matter? Cancers (Basel). 2022;14(4):1008.
- Hoel RW, Giddings Connolly RM, Takahashi PY. Polypharmacy management in older patients. Mayo Clin Proc. 2021;96(1):242–256.
- Monteiro AF, Rato M, Martins C. Drug-induced photosensitivity: photoallergic and phototoxic reactions. Clin Dermatol. 2016;34(5):571–581.
- Matthews SW, Pike K, Chien AJ. Phototherapy: safe and effective for challenging skin conditions in older adults. Cutis. 2021;108(1):E15–E21.
- Singh GK, Sapra D, Bahuguna A, Das P. Methotrexate toxicity in psoriasis-a multicentric retrospective study. Dermatol Ther. 2022;35(10):e15765.
- Bauer B, Chyou PH, Stratman EJ, Green C. Noninvasive testing for nonalcoholic steatohepatitis and hepatic fibrosis in patients with psoriasis receiving long-term methotrexate sodium therapy. JAMA Dermatol. 2017;153(10):977–982.
- Ohtsuki M, Nakagawa H, Sugai J, et al. Long-term continuous versus intermittent cyclosporin: therapy for psoriasis. J Dermatol. 2003;30(4):290–298.
- Chularojanamontri L, Silpa-Archa N, Wongpraparut C, Limphoka P. Long-term safety and drug survival of acitretin in psoriasis: a retrospective observational study. Int J Dermatol. 2019;58(5):593–599.
- Saurat JH, Guérin A, Yu AP, et al. High prevalence of potential drug-drug interactions for psoriasis patients prescribed methotrexate or cyclosporine for psoriasis: associated clinical and economic outcomes in real-world practice. Dermatology. 2010;220(2):128–137.
- Zhang Z, Fan W, Yang G, et al. Risk of tuberculosis in patients treated with TNF-α antagonists: a systematic review and meta-analysis of randomised controlled trials. BMJ Open. 2017;7(3):e012567.
- Hayashi M, Umezawa Y, Fukuchi O, Ito T, Saeki H, Nakagawa H. Efficacy and safety of ustekinumab treatment in elderly patients with psoriasis. J Dermatol. 2014;41(11):974–980.
- Körber A, Papavassilis C, Bhosekar V, Reinhardt M. Efficacy and safety of secukinumab in elderly subjects with moderate to severe plaque psoriasis: a pooled analysis of phase III studies. Drugs Aging. 2018;35(2):135–144.
- TALTZ (ixekizumab). Prescribing information. Eli Lilly and Company; 2024.
- STELARA (ustekinumab). Prescribing information. Janssen Biotech, Inc.; 2012.
- TREMFYA (guselkumab). Prescribing information. Janssen Biotech, Inc.; 2024.
- ENBREL (etanercept). Prescribing information. Amgen; 2024.
- Alabas OA, Mason KJ, Yiu ZZN, et al; BADBIR study group. Age and biologic survival in patients with moderate-to-severe psoriasis: a cohort study from the British Association of Dermatologists Biologics and Immunomodulators Register (BADBIR). Br J Dermatol. 2025;192(5):907–916.
- Doshi JA, Takeshita J, Pinto L, et al. Biologic therapy adherence, discontinuation, switching, and restarting among patients with psoriasis in the US Medicare population. J Am Acad Dermatol. 2016;74(6):1057-1065.e4.
- Gooderham MJ, Papp KA, Lynde CW. Shifting the focus – the primary role of IL-23 in psoriasis and other inflammatory disorders. J Eur Acad Dermatol Venereol. 2018;32(7):1111–1119.
- SKYRIZI (risankizumab-rzaa). Prescribing information. AbbVie Inc.; 2024.
- ILUMYA (tildrakizumab-asmn). Prescribing information. Sun Pharmaceutical Industries, Inc.; 2024.
- Fernandez AP, Dauden E, Gerdes S, et al. Tildrakizumab efficacy and safety in patients with psoriasis and concomitant metabolic syndrome: post hoc analysis of 5-year data from reSURFACE 1 and reSURFACE 2. J Eur Acad Dermatol Venereol. 2022;36(10):1774–1783.
- Poulin Y, Ramon M, Rosoph L, et al. Efficacy of tildrakizumab by patient demographic and disease characteristics across a phase 2b and 2 phase 3 trials in patients with moderate-to-severe chronic plaque psoriasis. J Eur Acad Dermatol Venereol. 2020;34(7):1500–1509.
- Narcisi A, Valenti M, Gargiulo L, et al. Real-life effectiveness of tildrakizumab in chronic plaque psoriasis: a 52-week multicentre retrospective study-IL PSO (Italian landscape psoriasis). J Eur Acad Dermatol Venereol. 2023;37(1):93–103.
- COSENTYX (secukinumab). Prescribing information. Novartis Pharmaceuticals Corporation, Inc.; 2024.
- SILIQ (brodalumab). Prescribing information. Bausch Health US, LLC; 2024.
- Centers for Medicare & Medicaid Services. Part B drugs and biologicals. 2024. Accessed 17 Sep 2025. https://www.cms.gov/cms-guide-medical-technology-companies-and-other-interested-parties/payment/part-b-drugs
- Reich K, Papp KA, Blauvelt A, et al. Tildrakizumab versus placebo or etanercept for chronic plaque psoriasis (reSURFACE 1 and reSURFACE 2): results from two randomised controlled, phase 3 trials. Lancet. 2017;390(10091):276–288.
- Thaci D, Piaserico S, Warren RB, et al. Five-year efficacy and safety of tildrakizumab in patients with moderate-to-severe psoriasis who respond at week 28: pooled analyses of two randomized phase III clinical trials (reSURFACE 1 and reSURFACE 2). Br J Dermatol. 2021;185(2):323–334.
- Šahmatova L, Sügis E, Šunina M, et al. Signs of innate immune activation and premature immunosenescence in psoriasis patients. Sci Rep. 2017;7(1):7553.
- Ter Haar ELM, Van den Reek J, Gaarn Du Jardin K, Barbero-Castillo A, De Jong EMGJ, Lubeek SFK. Efficacy and safety of tildrakizumab in older patients: pooled analyses of two randomized phase III clinical trials (reSURFACE 1 and reSURFACE 2) through 244 weeks. Acta Derm Venereol. 2023;103:adv17752.
- Ruggiero A, Fabbrocini G, Cinelli E, Ocampo Garza SS, Camela E, Megna M. Anti-interleukin-23 for psoriasis in elderly patients: guselkumab, risankizumab and tildrakizumab in real-world practice. Clin Exp Dermatol. 2022;47(3):561–567.
- Orsini D, Caldarola G, Dattola A, et al. Efficacy and safety of tildrakizumab in elderly patients: real-world multicenter study (ESTER – study). J Dermatolog Treat. 2024;35(1):2319304.
- Bardazzi F, Viviani F, Piraccini BM, et al. Tildrakizumab in complex psoriatic patients: an experience in Emilia-Romagna (Italy). J Cutan Med Surg. 2023;27(2):126–132.
- Mastorino L, Dapavo P, Burlando M, et al; Frail-PsO Group. Effectiveness and safety of tildrakizumab in elderly and frail elderly psoriatic patients up to 2 years. Psoriasis (Auckl). 2025;15:339–350.
- Fukasawa T, Yamashita T, Enomoto A, et al. The optimal use of tildrakizumab in the elderly via improvement of Treg function and its preventive effect of psoriatic arthritis. Front Immunol. 2023;14:1286251.
- Shear NH, Betts KA, Soliman AM, et al. Comparative safety and benefit-risk profile of biologics and oral treatment for moderate-to-severe plaque psoriasis: a network meta-analysis of clinical trial data. J Am Acad Dermatol. 2021;85(3):572–581.
- Sbidian E, Chaimani A, Guelimi R, et al. Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis. Cochrane Database Syst Rev. 2023;7(7):CD011535.
- HUMIRA (adalimumab). Prescribing information. AbbVie Inc.; 2024.
- Brunner M, Holyoak K, DiRuggiero D. Healthcare provider administration of biologics for patients with plaque psoriasis: literature review and clinical considerations. J Clin Aesthet Dermatol. 2023;16(12 suppl 2):S20–S25.
- CIMZIA (certolizumab pegol). Prescribing information. UCB, Inc.; 2024.
- Muzumdar S, Waldman R, Wu R, Kelsey A. In-office administration associated with increased drug survival of ustekinumab for chronic plaque psoriasis when compared with self-administration: a single-institution retrospective chart review. J Am Acad Dermatol. 2023;88(5):1111–1113.