J Clin Aesthet Dermatol. 2025;18(12):66–72.
by Glynis Ablon, MD, FAAD; Jyotsna Paturi, PhD; Franck Juchaux, MS; Rebecca Barresi-Thornton, ME; Benedicte Fallou, BS; I-Chien Liao, PhD; Jinyuan Yan, PhD; Ying Chen, PhD; Julien Laboureau, PhD; Carine Ballihaut, PhD; Elena Catalan Martin, MS; Jean-Thierry Simonnet, PhD; Xavier Marat, PhD; and Qian Zheng, MD, PhD
Dr. Ablon is with the Ablon Skin Institute and Research Center in Manhattan Beach, California. Dr. Paturi, Ms. Barresi-Thornton, Dr. Liao, Dr. Yan, Dr. Chen, and Dr. Zheng are with L’Oréal Research and Innovation in Clark, New Jersey. Ms. Fallou is with Episkin in Lyon, France. Mr. Juchaux, Ms. Catalan Martin, Dr. Simonnet, and Dr. Marat are with L’Oréal Research and Innovation in Chevilly-LaRue, France. Dr. Ballihaut and Dr. Labourea are with L’Oréal Research and Innovation in Aulnay-Sous-Bois, France.
FUNDING: This research was funded by L’Oréal Research and Innovation.
DISCLOSURES: Dr. Ablon has research grants with Galderma and L’Oréal. The remaining authors are employees of L’Oréal Research and Innovation.
ABSTRACT: Objective: With advances in aesthetic procedures, best practices for postprocedure care to support immediate healing and augment efficacy are still without consensus. A postprocedure formula containing hydroxypropyl tetrahydropyrantriol (HPT) and niacinamide (NAM) has been developed. This study evaluated the functional benefits and clinical efficacy of pairing HPT/NAM formula with nonablative fractional laser treatment. Methods: The functional benefits of HPT/NAM were explored using 2D cellular models, reconstructed skin equivalents, and ex vivo skin models. The clinical efficacy of a HPT/NAM formula vs. a clinical control (Aquaphor Healing Ointment) was investigated through a randomized, double-blind, split-face, multicenter, multiethnic study with a White and a Northeast Asian panel. The investigative products were randomly assigned to one side of the face following a single full-face fractional nonablative laser procedure, and the products were applied twice daily for 56 days. Results: In vitro testing showed that the HPT/NAM combination decreased the secretion of prostaglandin E2 (PGE2), encouraged the production of glycosaminoglycan, α-smooth muscle actin, and various angiogenic growth factors. In both panels, the HPT/NAM formula demonstrated significant improvement vs. the clinical control for key anti-aging parameters such as fine lines, crow’s feet wrinkles, skin roughness, and overall healthy appearance over the course of 56 days. Limitations: This study only investigated the pairing of HPT/NAM formula with nonablative laser treatment. Conclusion: This study demonstrated comparable healing benefits to the clinical control along with augmented skin resurfacing benefits with HPT/NAM use following a nonablative fractional laser treatment. Keywords: Anti-aging, hydroxypropyl tetrahydropyrantriol, C-xyloside derivative, niacinamide, nonablative laser treatment, aesthetic procedures, minimally invasive procedures
Introduction
Minimally invasive aesthetic procedures, such as microneedling and nonablative laser treatments, are increasingly popular with consumers who are seeking accessible, affordable, and effective options for facial skin correction and rejuvenation.1-3 The impact of aesthetic procedures on changes to tissue morphology throughout the wound healing process, in regard to skin barrier, growth factor secretion, and proinflammatory cytokine release, has been well studied.4-7 Aesthetic procedures can disrupt the skin integrity, highlighting the need for a postprocedure skincare routine that facilitates skin recovery and enhances its rejuvenation effects.8
Among the minimally invasive procedures, nonablative laser resurfacing is one of the most popular.1,3 During nonablative resurfacing treatment, fractional lasers (with wavelength ranging from 1410nm to 1927nm) create microscopic thermal zones (MTZs) in the dermis and epidermis, which act as reservoirs for tissue remodeling and regeneration.9 The ability to control the depth, width, and density of MTZs with fractional nonablative lasers promotes skin rejuvenation postprocedure with markedly reduced downtime compared to ablative lasers.10 To promote safety and efficacy during the healing process, focus must be placed on postprocedure barrier rejuvenation. It is important to note that the healing process can vary, thus resulting in different needs by different consumer groups; for example, patients with a higher Fitzpatrick skin type are more susceptible to the development of postprocedure hyperpigmentation following laser treatments.11 Therefore, it is imperative to identify postprocedure care routines that take into account the potential for hyperpigmentation, erythema, and other indirect postinflammation effects.12,13 We hypothesize that a successful postprocedure formulation should encourage epidermal regeneration, dermal regeneration, anti-inflammation and antihyperpigmentation properties in order to enhance the skin rejuvenation efficacy of aesthetic procedure.
In order to tailor a product for postprocedure skin, hydroxypropyl tetrahydropyrantriol (HPT) was identified as a key active ingredient. HPT is well known for its glycosaminoglycan synthesis, growth factor modulation, and extracellular matrix boosting-benefits to enhance skin repair.14-16 Daily use of HPT as a topical serum has been found to improve facial skin attributes such as ptosis, nasolabial folds, skin radiance, and complexion evenness.17-19 These positive benefits of HPT make it an attractive active ingredient to explore as part of a postprocedure formulation. Niacinamide (NAM) has previously demonstrated benefits in antioxidation, anti-inflammation, antisenescence, and regulation of pigmentation.20,21 Therefore, we hypothesized that the addition of NAM to the postprocedure skincare formula would complement the actions of HPT. Considering the properties of each active ingredient (HPT/NAM), the association ensures an optimized skin care routine by targeting different key areas of need for postprocedure skin. The objective of this study is to both elucidate the complementary functional benefits of the topical regeneration formula containing HPT and NAM using in vitro evaluations and to evaluate the clinical efficacy of the formula following nonablative fractional laser treatment.
Methods
In vitro and ex vivo assays. Keratinocyte filaggrin expression. Normal adult human epidermal keratinocytes (NHEK-Ad) were used for two distinct experiments, with initial cell maintenance in basal growth medium. Cells were treated for 48 hours with fresh growth medium containing varying concentrations of HPT alone (0.005%, 0.017%, 0.05%), NAM alone (0.0012%, 0.004%, 0.012%), or HPT/NAM combination. Fluorescent signals were captured via microscope for quantification. To assess barrier maturation, NHEK-Ad cells were seeded at 5×103 cells per well, equilibrated overnight, and then stained with antifilaggrin primary antibody (ab81468; Abcam Inc.) and goat anti-mouse IgG cross-adsorbed secondary antibody (Alexa Fluor 488, A-11001; ThermoFisher Scientific). Filaggrin expression was quantified based on fluorescent intensity.
Fibroblast inflammation model. Normal adult human fibroblasts (NHDF-Ad) were maintained in fibroblast basal medium (FBM) with FGM-2 supplements. For inflammation experiments, fibroblasts (5×103 cells/well) were stimulated with 0.1ng/mL of interleukin 1α (IL-1α) and treated with HPT (1mM, 3mM) and NAM (0.3mM, 1mM) alone or in combination (HPT/NAM). Prostaglandin E2 (PGE2) secretion was quantified in the supernatant using a PGE2 enzyme-linked immunosorbent assay (ELISA) kit.
Fibroblasts α-SMA expression. Normal adult human fibroblasts (NHDF-Ad) were used to assess the ability of HPT or NAM alone or in combination (HPT/NAM) to induce α-smooth muscle actin (α-SMA) expression, indicating wound contraction potential. Cells were maintained in FBM basal medium with FGM-2 supplements. Fibroblasts (5×103 cells/well) were seeded, equilibrated overnight, and then treated for 48 hours with various concentrations of HPT (0.005%, 0.017%, 0.05%) and NAM (0.0012%, 0.004%, 0.012%), or their combination. TGF-β (0.1ng/mL) served as a positive control for α-SMA induction. Cells were stained with mouse anti-α-SMA primary antibody (ab7817; Abcam Inc.) and goat anti-mouse IgG cross-adsorbed secondary antibody (Alexa Fluor 488, A-11001; ThermoFisher Scientific). Fluorescent signals were captured with a microscope, and α-SMA expression was quantified based on fluorescent intensity.
sGAG release in reconstructed human skin model. Reconstructed full-thickness skin samples (T-Skin) were systemically treated with HPT (3mM), NAM (200µM), or their combination (HPT/NAM) from Day 11 to Day 15 (n=3). Tissues were maintained at 37°C with 95% humidity and 5% carbon dioxide (CO2). On Day 15, the medium supernatant was collected to quantify sulfated glycosaminoglycans (sGAGs) using an assay kit.
Ex vivo microneedling model. Fresh human skin samples (post-abdominoplasty; BioIVT Inc.) were defatted and the tissue was then subjected to 5 passes of microneedling treatment at a needle length setting of 1.5mm using a microneedling pen (36-pin needles, Dr. Pen A6 Cartridges Tips, Dr. Pen Inc.).5 Following treatment, 1.2cm skin biopsies were cultured at the air-liquid interface for 6 days. Untreated and microneedling control samples were cultured in standard Dulbecco’s Modified Eagle’s Medium (DMEM). Treated microneedled (MN) samples (MN+HPT/NAM) received daily topical applications of 10% HPT and 3% NAM. After 6 days, biopsies underwent histological and immunohistochemical (filaggrin, transglutaminase-1) analysis. Media supernatant was collected to quantify growth factors (fibroblast growth factor [FGF], vascular endothelial growth factor [VEGF], Angiopoietin-2 [Ang-2], and hepatocyte growth factor [HGF]) via LEGENDplex (BioLegend), normalized to total protein content.
Clinical evaluation: study design and inclusion/exclusion criteria. This randomized, double-blind, split-face, multicenter, multiethnicity study evaluated the clinical efficacy of HPT/NAM formula in conjunction with full-face nonablative fractional laser treatment. The study was performed in accordance with the Good Clinical Practices and the principles of the Declaration of Helsinki.22 The procedures used in this study were approved by an independent Institutional Review Board (Pro00073859, Advarra IRB). Before the study procedure, informed consent and photoconsent was obtained from all study subjects before enrollment. Eligibility was determined by physical examination and confirmation of all inclusion/exclusion criteria. The study was designed to treat participants at two centers targeting two different populations. Center 1 comprised of a panel of non-Hispanic Caucasian women (n=25; aged 30 to 65 years) with Fitzpatrick skin type II-III, with mild to severe crow’s feet wrinkles 2-5.6 (0-6 L’Oréal Atlas scale23), mild-to-severe dyschromia and roughness 3.5-8 (0-9 modified Griffiths scale.24 For Center 1, 37 non-Hispanic Caucasian female subjects were screened, of which 29 were included and randomized for the treatment protocol. 25 subjects finished the study, as four females withdrew due to consent withdrawal or lost to follow-up. No subjects withdrew due to adverse events. Center 2 comprised of a panel of Northeast Asian (Chinese, Korean, or Japanese) females (n=12, 30-65 years) with Fitzpatrick skin type II-IV, with mild to severe dyschromia and roughness, 3.5-8 (0-9 modified Griffiths scale24), mild-to-severe crow’s feet wrinkles 2.5-6 (L’oreal Atlas scale).
Clinical procedure. The study was conducted separately at both centers between September 2022 and May 2023. Nonablative fractional laser treatment was performed using HALO Laser (Sciton), with laser settings of 1470nm wavelength and 15mm scan line. The laser settings were optimized acknowledging the different ethnicities of the panels. A topical regimen was followed for 56 days postlaser resurfacing treatment, with follow-up visits, including safety and tolerance evaluations, scheduled on Days 0 (baseline), 1, 2, 3, 4, 7, 14, 28, and 56.
The investigational products used were the HPT/NAM formula (10% HPT + 3% NAM) versus the clinical control Aquaphor Healing Ointment (Beiersdorf Inc.). 10% HPT and 3% NAM formula was incorporated in an oil-in-water emulsion to create a rich, nonocclusive topical cream compatible with postprocedure skin. For the duration of the study, subjects applied 2mg/cm2 of the test product to the assigned side of the face twice daily. Auxiliary products, including a marketed gentle facial cleaner (twice a day, morning and night) and a commercialized sunscreen, were distributed to all participants to be used throughout the study.
For both centers on Days 0, 7, 14, 28 and 56, the clinical assessment of anti-aging efficacy included global wrinkles, fine lines, undereye and forehead wrinkles, skin dyschromia, skin tone evenness, skin brightness, nasolabial folds, skin radiance, skin roughness, skin laxity, and pores on the cheeks, separately on each side of the face (left versus right), as defined by the L’Oréal Aging Atlas scale23 and the modified Griffiths scale.24 In addition, postprocedure healing was evaluated by assessment of erythema, edema, peeling, and dryness. Transepidermal water loss measurements were performed using the Tewameter TM 300 (Courage + Khazaka electronic GmbH, Koln, Germany) on the cheeks (left, right). Digital images were captured using a VISIA-CR system (Canfield Scientific) and used to visually assess skin repair, signs of aging, and dyschromia.
Safety assessment. Safety was assessed by recording the incidence of adverse events and subjective tolerance, including cutaneous reactions (local intolerance), papule, erythema, peeling, pruritis, acne flare-up, and dryness, from the informed consent signature date until the end of the study duration.
Statistical analysis. All statistical analysis conducted for in vitro tests were performed using Graphpad (Prism) version 10.2.0. Statistical significance between groups was determined with one-way analysis of variance (ANOVA) followed by Tukey’s test with confidence level of 95%. For each clinical parameter, the change from baseline was estimated using a linear mixed model. The model included fixed effects for the baseline score, treatment, visit, an interaction of treatment group with baseline, an interaction of treatment group with visit, and a random subject effect. Improvement versus baseline and comparisons between treatments were made using contrasts of least square mean estimates calculated by the model. In cases where the model’s assumptions were not met, nonparametric Wilcoxon signed-rank tests were performed. All statistical tests produced two-sided p-values, with the Benjamini-Hochberg method applied to adjust p-values for multiple comparisons. Statistical significance was determined at a level of 0.05.
Results
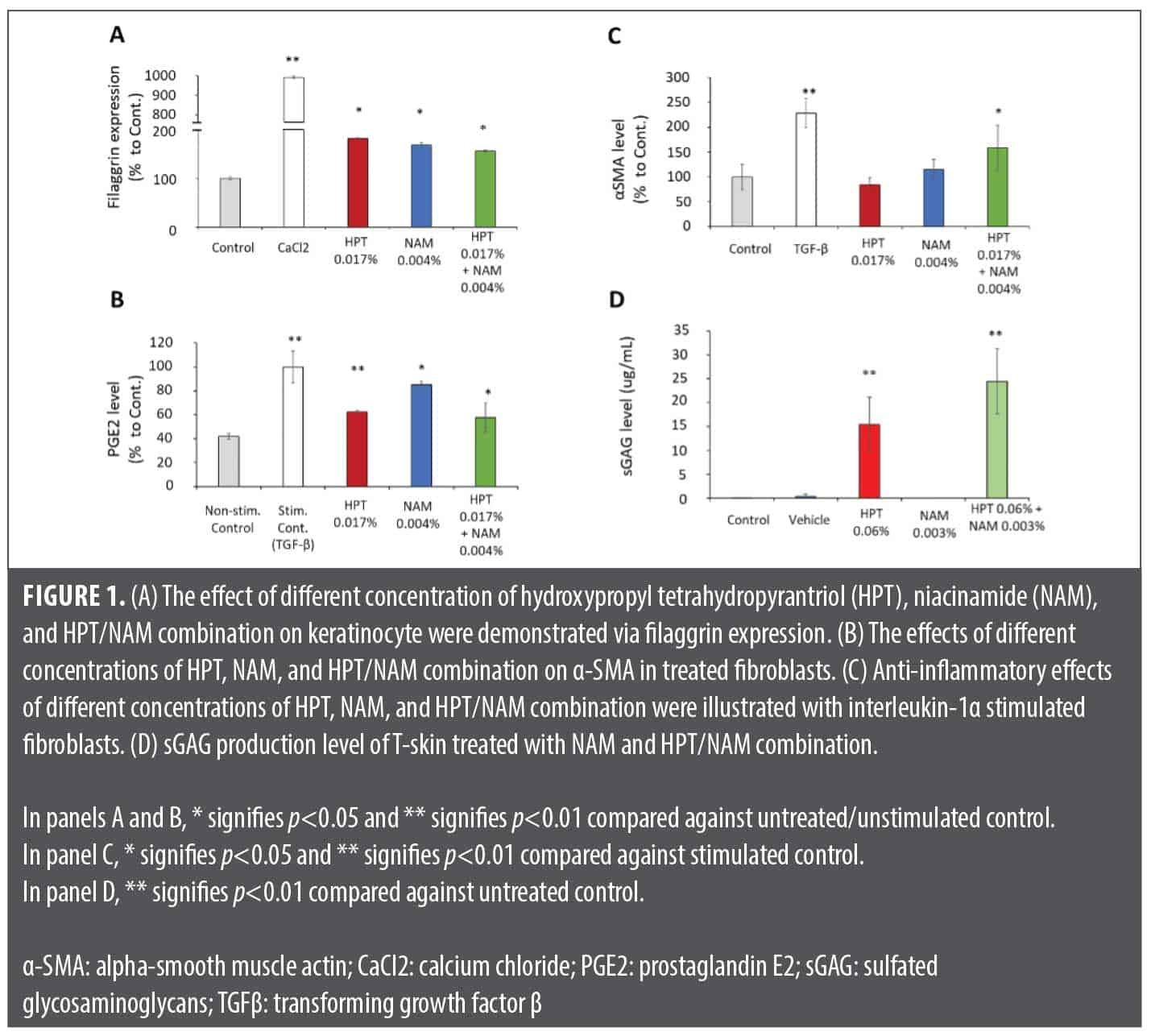
In vitro assays. The in vitro assays evaluated the HPT/NAM combination efficacy on the key post-aesthetic procedure parameters of barrier restoration, anti-inflammation, pigmentation control, wound contraction, and extracellular matrix (ECM) production. Figure 1A shows that HPT, NAM, and HPT/NAM induced elevated filaggrin expression in keratinocytes, promoting barrier renewal. In an IL-1α-stimulated fibroblast model, HPT, NAM, and HPT/NAM inhibited PGE2 production, a key mediator of post-inflammatory hyperpigmentation (Figure 1B). This connects to a PGE2-stimulated melanocyte model simulating inflammation-driven pigmentation.25 HPT and NAM’s postprocedure benefits include stimulating α-SMA expression and sGAG synthesis. In a 48-hour fibroblast experiment (Figure 1C), neither HPT nor NAM alone significantly induced α-SMA, an indicator of beneficial myofibroblast conversion for wound healing.26 However, HPT/NAM significantly stimulated α-SMA expression in treated fibroblasts (Figure 1C). The full-thickness reconstructed skin model showed that NAM and HPT/NAM induced epidermal thickening after one week (data not shown). Additionally, HPT, and consequently HPT/NAM, increased sGAG production in the T-Skin™ supernatant (Figure 1D).
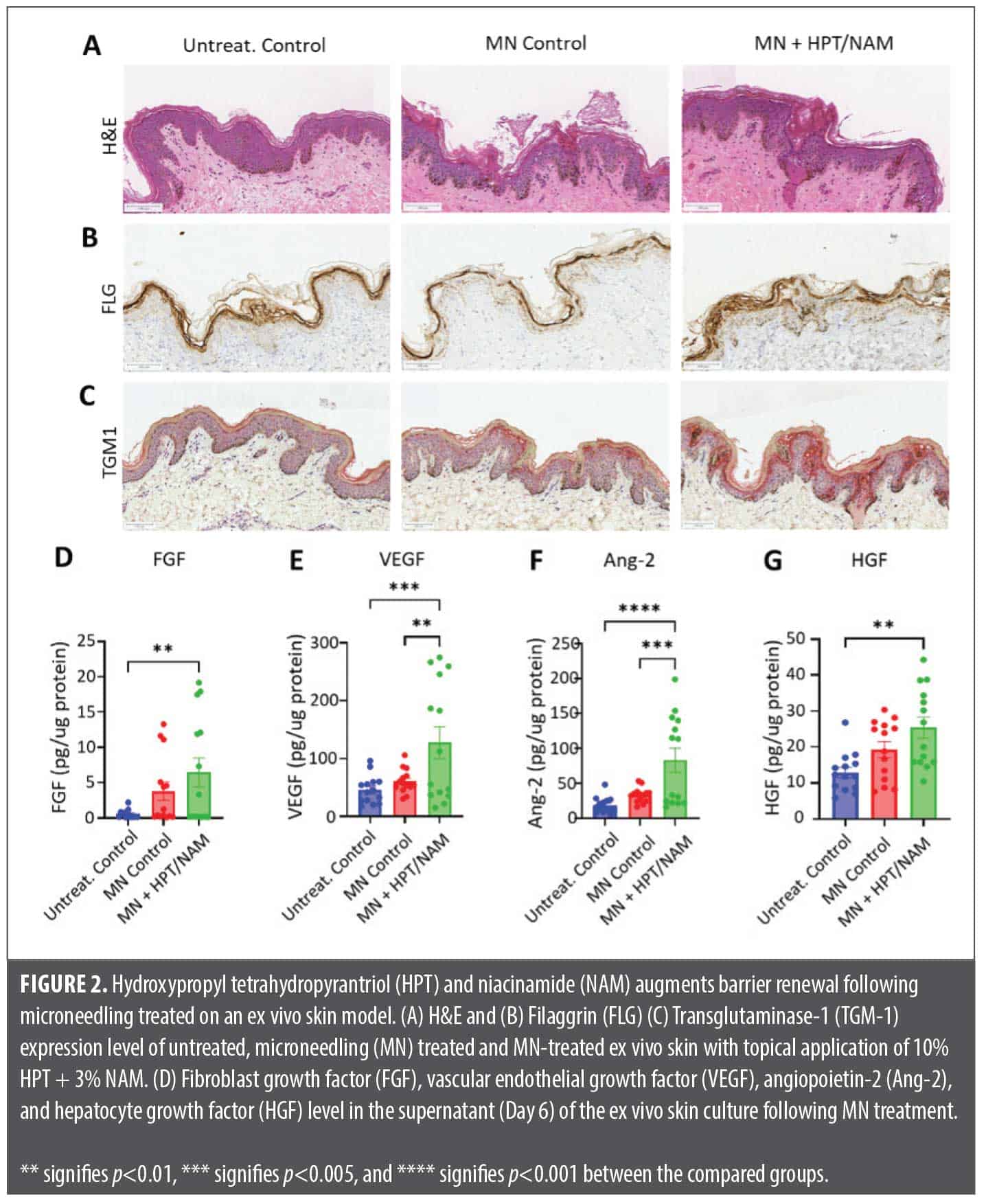
Ex vivo assays. The ex vivo skin microneedling model, simulating post-aesthetic procedure wound healing, assessed the HPT/NAM combination’s impact on barrier restoration and inflammation. While hematoxylin-eosin (H&E) staining (Figure 2A) confirmed epidermal regrowth in both MN and MN+HPT/NAM groups, enhanced filaggrin (FLG) and transglutaminase-1 (TGM1) immunostaining in the MN+HPT/NAM group (Figures 2B and 2C) indicated accelerated epidermal restoration. Furthermore, MN+HPT/NAM treatment significantly elevated key proangiogenic growth factors such as FGF, VEGF, Ang-2, and HGF (Figure 2D-2G) compared to controls.
Clinical study. For both Caucasian and Asian panels, all participants demonstrated no product-related adverse events following laser resurfacing procedure. Moreover, the HPT/NAM combination showed parity with the clinical control for healing parameters such as erythema, edema, and dryness at Days 1, 3, 7, and 14.
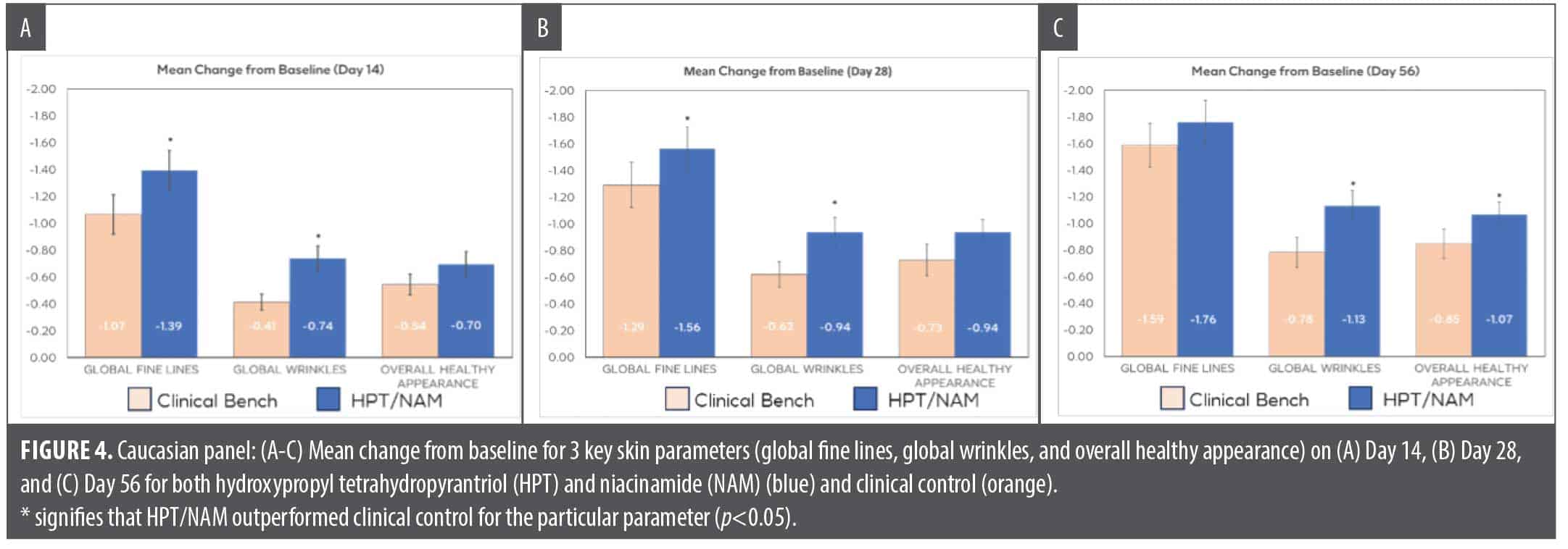
In the Caucasian panel, both the HPT/NAM formula and the control product showed statistically significant improvement over baseline beginning on Day 14, where both formulas showed at least 0.5-grade improvement over baseline for improvement in skin rejuvenation endpoints such as global fine lines, skin brightness, skin roughness, skin dyschromia, and overall healthy appearance. HPT/NAM is significantly superior compared to the control for improvement in global fine lines and global wrinkles at Day 14. The HPT/NAM formula demonstrated continuous improvement in global wrinkles, global fine lines, and skin roughness (not shown) as observed at Day 28 and Day 56 along with significant improvement versus the control for global fine lines at Day 28 and overall healthy appearance for Day 56 (Figure 3 and Figure 4).
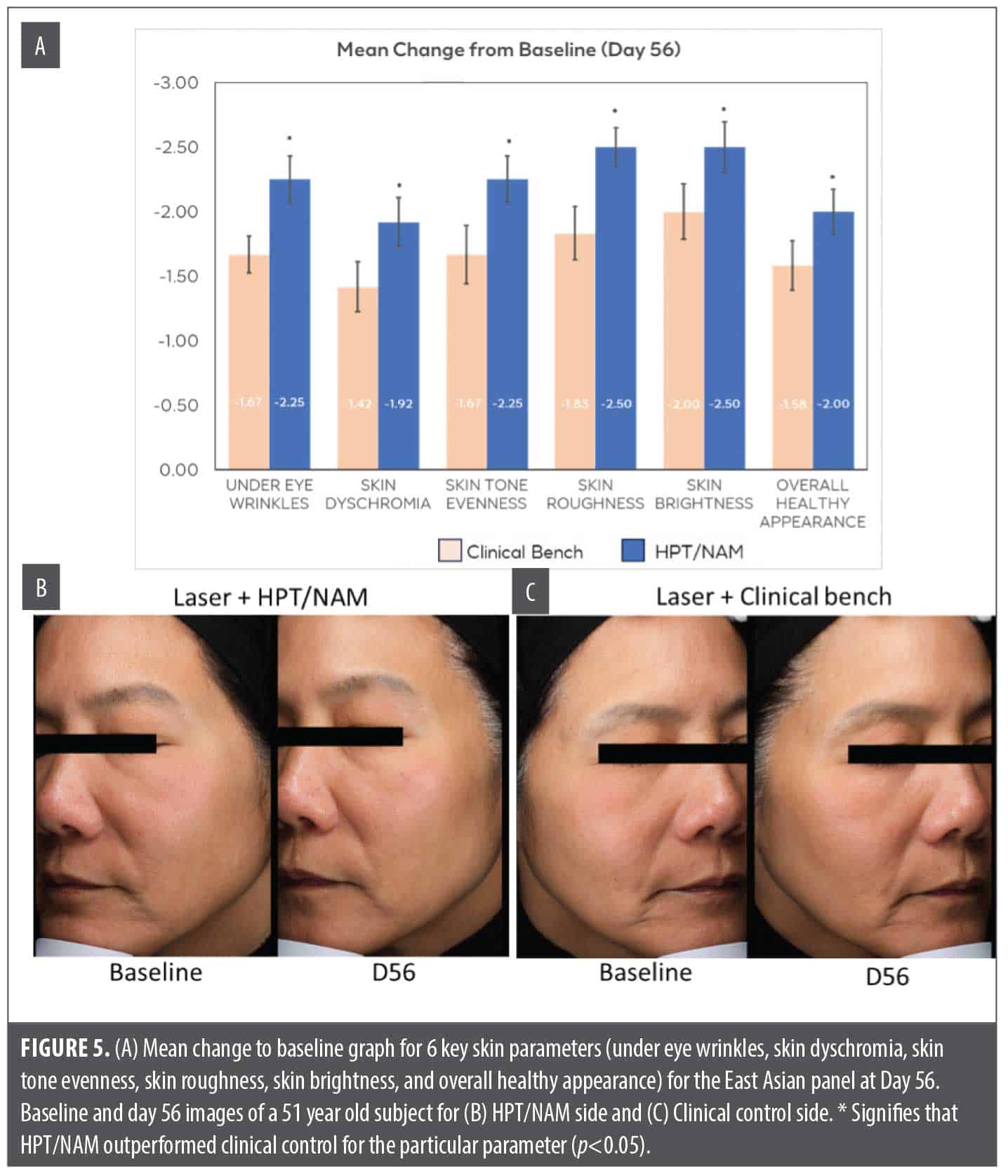
The Northeast Asian panel was tested in the same study design and assessed for the same anti-aging parameters (including undereye wrinkles, pore appearance, skin brightness, skin tone evenness, overall skin dyschromia, and overall healthy appearance). However, the laser settings were modified in this panel to account for skin sensitivity and higher risk of postinflammatory hyperpigmentation (PIH). Similar to the result observed in the Caucasian population, HPT/NAM formula application demonstrated significant improvement over baseline in crow’s feet wrinkles, global fine lines, skin tone evenness, skin brightness, radiance, overall healthy appearance, and undereye wrinkles at Days 14, 28, and 56. In this panel, the benefits of HPT/NAM formula were not statistically significantly different vs. control at Days 14 and 28 for anti-aging parameters evaluated (data not shown). However, at Day 56, HPT/NAM significantly outperformed the control for under eye wrinkles, skin dyschromia, skin tone evenness, skin roughness, skin brightness, and overall health appearance (Figure 5).
Discussion
The objective of this study was to evaluate the functional benefits of the HPT/NAM formula using in vitro evaluation methods and the clinical procedure augmentation efficacy when paired with a nonablative fractional laser procedure. HPT, also known as C-xylopyranoside, has been previously demonstrated to stimulate the synthesis of sGAG and growth factors and the production of key dermal-epidermal junction proteins (such as laminin 5, collagen IV and collagen VII) and it has been suggested to have skin repair functions.14-16,19 The use of HPT, as part of a daily topical formulation, has been established to improve skin hydration and skin quality as well as to reduce visible signs of aging over the course of 2 to 3 months.18 Despite the known anti-aging benefits of HPT, its use in compromised skin and postprocedure settings was not well understood. In previous publications, postprocedure skin benefited from a treatment routine that managed skin inflammation and hyperpigmentation while encouraging dermal and epidermal regeneration.27 Thus, this study combined HPT with NAM to create an active combination with complementary mechanisms of action to support postprocedure skin. The selection of NAM is based on its ability to stimulate cellular mitochondria energy and modulate inflammation and oxidative stress.20
In this study, NAM alone and HPT/NAM were able to stimulate keratinocyte proliferation and reduce the level of PGE2 production in IL-1 α-stimulated fibroblasts (Figure 1). PGE2 is an important mediator of inflammation, as it plays an important role to trigger inflammation and melanin production and has been suggested to be a contributing factor to the induction of post inflammatory hyperpigmentation.28 Therefore, the inclusion of NAM is critically important to manage the level of skin redness and reduce the risk of hyperpigmentation that can potentially be triggered by the procedure treatments. Interestingly, HPT/NAM also stimulates α-SMA in fibroblasts as well as the induction of several key angiogenic growth factors such as FGF, VEGF, and Ang-2 (Figure 1 and Figure 2). Upregulation of α-SMA and proangiogenic growth factors encourages the closure of the procedure induced wounds,29,30 and the stimulation of FGF is also a key anti-aging strategy to encourage cell regeneration, skin repair, and ECM production.31 The functionality to induce skin repair is evaluated in an ex vivo microneedling model because it can recreate the wound healing process after an aesthetic procedure.5 As highlighted in Figure 2, the application of HPT/NAM can enhance the expression of transglutaminase and filaggrin after the ex vivo skin underwent microneedling treatment. Transglutaminase, a key enzyme essential for the assembly of cornified cell envelope, is critically important for the restoration of a mature barrier.32 The assembly of the cornified cell envelope is also driven by filaggrin, a protein that cross-links to form the key components of the uppermost skin barrier.33 Thus, the stimulation of these key biomarkers suggests that HPT/NAM can accelerate epidermal barrier repair kinetics after the skin is compromised by procedure treatments.
In this study, the HPT/NAM formula was compared to the clinical control (Aquaphor Healing Ointment) as a skin care application following nonablative laser procedure for a Caucasian and an Northeast Asian panel. Evaluating the formula for postprocedure enhancement in a diverse ethnic panel is key to demonstrating the versatility of the active combination. The rise in aesthetic procedures in North America and Asia indicates the relevance of including the two ethnicities to demonstrate clinical benefits of HPT/NAM.34 This HPT/NAM formula, being a rich, nonocclusive oil-in-water emulsion topical cream, was designed for a pleasant sensorial experience compared to occlusive, petrolatum-based formulas for post procedure skin. The shift toward including nonocclusive skincare formulas with newer small molecules to accelerate the three key stages of postprocedure repair—inflammation, proliferation, and ECM remodeling—emphasizes the benefit of HPT/NAM’s parity with traditional occlusive formulas in addition to the augmented clinical benefits manifested as early as Day 14.35 Thus, it is encouraging to see the HPT/NAM formula reaching parity with the clinical control for the evaluated healing parameters (erythema, edema, and dryness) at Days 1, 3, 7, and 14. In double-blind, randomized, split-face study, HPT/NAM outperformed the clinical control for crow’s feet wrinkles and fine lines at Day 14, fine lines and wrinkles at Day 28, and wrinkles and overall healthy appearance at Day 56 in the Caucasian panel (Figure 3 and Figure 4). In the Northeast Asian panel, the HPT/NAM formula outperformed the clinical control for 6 of the 8 clinical parameters (undereye wrinkles, skin dyschromia, skin tone unevenness, skin roughness, skin brightness, and overall skin health) at Day 56 (Figure 5). The use of a split-face evaluation attests to the robustness of the study design, therefore strengthening the clinical efficacy of HPT/NAM for postprocedure healing and superiority vs. control for key anti-aging endpoints. Furthermore, this study demonstrated augmentation benefits to anti-aging performance of nonablative laser treatments in both Caucasian and Northeast Asian panels, which suggests the performance feasibility across a broader spectrum of skin types. As illustrated in Figure 3 and Figure 4, the HPT/NAM formula brings strong augmentation benefits at Day 14 and extends benefits upwards to the conclusion of the study at Day 56.
Designing an optimal routine for postprocedure skin is complex due to diverse patient needs and a wide portfolio of different aesthetic procedure. Thus, it is critically important that any investigation of a postprocedure treatment routine have a rigorous, well-controlled study design. In this study, the pairing benefits of HPT/NAM with nonablative laser treatments have been demonstrated in Caucasian and Northeast Asian panels. However, this study did not investigate the potential augmentation effects if the patients were subjected to repeated procedures. Furthermore, the pairing benefits of HPT/NAM with other types of procedures, such as ablative fractional laser, microneedling, and radiofrequency (RF) microneedling, have yet to be investigated. Although this study reported the pairing benefits in Caucasian and Northeast Asian panels, patients with higher Fitzpatrick skin types were not included. These highlighted opportunities are exciting angles for future investigation, and it is logical to believe that a postprocedure formula designed to deliver balanced skin benefits including dermal and epidermal regeneration, skin inflammation, and hyperpigmentation can be a promising postprocedure routine.
Conclusion
In this study, the potential benefits of the HPT/NAM formula as a postprocedure skincare routine were investigated using in vitro evaluation methods and in a clinical study with nonablative fractional laser. The pairing of HPT/NAM encourages barrier renewal, stimulates sGAG production, and manages the level of PGE2 production while promoting α-SMA and growth factor production. Clinically, these biological benefits translated to the augmentation of anti-aging benefits provided by nonablative laser in both the Caucasian and Asian panels. In conclusion, this study introduces the HPT/NAM formula as a promising new technology that contributes towards the development of postprocedure skincare routine.
References
- Krueger N, Luebberding S, Sattler G, et. al. The history of aesthetic medicine and surgery. J Drugs Dermatol. 2013;12(7):737-742.
- Mobayed N, Nguyen JK, Jagdeo J. Minimally invasive facial cosmetic procedures for the millennial aesthetic patient. J Drugs Dermatol. 2020;19(1):100-103.
- Angelo-Khattar M. Nonablative lasers for photorejuvenation. In: Almida Issa MC, Tamura B, eds. Lasers, Lights, and Other Technologies: Springer; 2018. p. 89-103.
- He C, Zhang W, Tu Y, et al. Characterization of an ablative fractional CO2 laser-induced wound-healing model based on in vitro 3D reconstructed skin. J Cosmet Dermatol. 2023; 22(5):1495-1506..
- Liu X, Barresi R, Kaminer M, et al. Utilization of ex vivo tissue model to study skin regeneration following microneedle stimuli. Sci Rep. 2022;12(1):18115.
- Schmitt L, Marquardt Y, Amann P, et al. Comprehensive molecular characterization of microneedling therapy in a human three-dimensional skin model. PLoS One. 2018;13(9):e0204318.
- Marquardt Y, Amann PM, Heise R, et al. Characterization of a novel standardized human three-dimensional skin wound healing model using non-sequential fractional ultrapulsed CO2 laser treatments. Lasers Surg Med. 2015;47(3):257-265.
- Gold MH, Andriessen A, Goldberg DJ, et al. Pre-/postprocedure measures for minimally invasive, nonenergy aesthetic treatments: a survey. J Cosmet Dermatol. 2020;19(7):1587-1592.
- Preissig J, Hamilton K, Markus R. Current laser resurfacing technologies: a review that delves beneath the surface. Semin Plast Surg. 2012;26(3):109-116.
- Pozner JN, DiBernardo BE. Laser resurfacing: full field and fractional. Clin Plast Surg. 2016;43(3):515-525.
- Kaushik SB, Alexis AF. Nonablative fractional laser resurfacing in skin of color: evidence-based review. J Clin Aesthet Dermatol. 2017;10(6):51-67.
- Alexis AF. Lasers and light-based therapies in ethnic skin: treatment options and recommendations for Fitzpatrick skin types V and VI. Br J Dermatol. 2013;169 Suppl 3:91-97.
- Woolery-Lloyd H, Viera MH, Valins W. Laser therapy in black skin. Facial Plast Surg Clin North Am. 2011;19(2):405-416.
- Sok J, Pineau N, Dalko-Csiba M, et al. Improvement of the dermal epidermal junction in human reconstructed skin by a new c-xylopyranoside derivative. Eur J Dermatol. 2008;18(3):297-302.
- Pineau N, Carrino DA, Caplan AI, Breton L. Biological evaluation of a new C-xylopyranoside derivative (C-Xyloside) and its role in glycosaminoglycan biosynthesis. Eur J Dermatol. 2011;21(3):359-370.
- Vassal-Stermann E, Duranton A, Black AF, et al. A new C-Xyloside induces modifications of GAG expression, structure and functional properties. PLoS One. 2012;7(10):e47933.
- Raab S, Yatskayer M, Lynch S, et al. Clinical evaluation of a multimodal facial serum that addresses hyaluronic acid levels in skin. J Drugs Dermatol. 2017;16(9):884-890.
- Bouloc A, Roo E, Moga A, et al. A compensating skin care complex containing pro-xylane in menopausal women: results from a multicentre, evaluator-blinded, randomized study. Acta Derm Venereol. 2017;97(4):541-542.
- Wang ST, Neo BH, Betts RJ. Glycosaminoglycans: sweet as sugar targets for topical skin anti-aging. Clin Cosmet Investig Dermatol. 2021;14:1227-1246.
- Boo YC. Mechanistic basis and clinical evidence for the applications of nicotinamide (niacinamide) to control skin aging and pigmentation. Antioxidants (Basel). 2021;10(8):1315.
- Wohlrab J, Kreft D. Niacinamide – mechanisms of action and its topical use in dermatology. Skin Pharmacol Physiol. 2014;27(6):311-315.
- World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191-2194.
- Bazin R. Skin Aging Atlas. Volume 1, Caucasian Type: MED COM; 2007.
- Griffiths CE, Wang TS, Hamilton TA, et al. A photonumeric scale for the assessment of cutaneous photodamage. Arch Dermatol. 1992;128(3):347-351.
- Desai S, Ayres E, Bak H, et al. Effect of a Tranexamic Acid, Kojic Acid, and Niacinamide Containing Serum on Facial Dyschromia: A Clinical Evaluation. J Drugs Dermatol. 2019;18(5):454-459.
- Grinnell F. Fibroblasts, myofibroblasts, and wound contraction. J Cell Biol. 1994;124(4):401-404.
- Liao IC, He C, Liu XL, Barresi-Thornton R, et al. The application of a multi-component reaction peptide as a model regenerative active to enhance skin wound healing post-laser procedure in a double-blinded placebo-controlled clinical trial. J Cosmet Dermatol. 2024;23(3):898-910.
- Tomita Y, Iwamoto M, Masuda T, Tagami H. Stimulatory effect of prostaglandin E2 on the configuration of normal human melanocytes in vitro. J Invest Dermatol. 1987;89(3):299-301.
- Darby I, Skalli O, Gabbiani G. Alpha-smooth muscle actin is transiently expressed by myofibroblasts during experimental wound healing. Lab Invest. 1990;63(1):21-29.
- Meyer M, Muller AK, Yang J, et al. FGF receptors 1 and 2 are key regulators of keratinocyte migration in vitro and in wounded skin. J Cell Sci. 2012;125(Pt 23):5690-5701.
- de Araujo R, Lobo M, Trindade K, et al. Fibroblast Growth Factors: A Controlling Mechanism of Skin Aging. Skin Pharmacol Physiol. 2019;32(5):275-282.
- Hitomi K. Transglutaminases in skin epidermis. Eur J Dermatol. 2005;15(5):313-319.
- Kezic S, Jakasa I. Filaggrin and skin barrier function. Curr Probl Dermatol. 2016;49:1-7.
- Kwon SH, Lao WWK, Lee CH, et al. Experiences and attitudes toward aesthetic procedures in East Asia: a cross-sectional survey of five geographical regions. Arch Plast Surg. 2021;48(6):660-669.
- Angra K, Lipp MB, Sekhon S, et al. Review of post-laser-resurfacing topical agents for improved healing and cosmesis. J Clin Aesthet Dermatol. 2021;14(8):24-32.