J Clin Aesthet Dermatol. 2025;18(12):50–65.
by Courtney A. Chau, BS; Hilary E. Baldwin, MD; James Q. Del Rosso, DO; Julie C. Harper, MD; Edward Lain, MD, MBA; Todd Schlesinger, MD; Ali Shahbaz, MD; Heather Woolery-Lloyd, MD; Jennifer C. Jaworski, MS; Melissa S. Seal, PhD; Diane Hanna, DNP, DCNP; and Peter Lio, MD
Ms. Chau is with the Icahn School of Medicine at Mount Sinai in New York, New York. Dr. Baldwin is with the Department of Dermatology at Rutgers University Robert Wood Johnson Medical Center in New Brunswick, New Jersey. Dr. Del Rosso is with JDR Dermatology Research Center, LLC in Las Vegas, Nevada and Advanced Dermatology and Cosmetic Surgery in Maitland, Florida. Dr. Harper is with the Dermatology and Skin Care Center of Birmingham in Birmingham, Alabama. Dr. Lain is with Sanova Dermatology in Austin, Texas. Dr. Schlesinger is with Clinical Research Center of the Carolinas in Charleston, South Carolina. Dr. Shahbaz is with Texas A&M School of Medicine in Bryan, Texas and Westlake Dermatology and Cosmetic Surgery in Austin, Texas. Dr. Woolery-Lloyd is with the Dr. Philip Frost Department of Dermatology and Cutaneous Surgery at the University of Miami Miller School of Medicine in Miami, Florida. Ms. Jaworski, Dr. Seal, and Dr. Hanna are with Arcutis Biotherapeutics, Inc. in Westlake Village, California. Dr. Lio is with the Departments of Dermatology & Pediatrics at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
FUNDING: Editorial assistance was provided by Ashfield MedComms, an Inizio Company, and was supported by Arcutis Biotherapeutics, Inc.
DISCLOSURES: Courtney A. Chau has no conflicts of interest to disclose. Hilary E. Baldwin has served as investigator, speaker, and/or advisor for Almirall, Arcutis, Beiersdorf, Botanix, Galderma, Journey Medical Corporation, L’Oréal, La Roche-Posay, Ortho Dermatologics, Phyla Skincare, and Sun Pharma. James Q. Del Rosso reports receiving investigator fees, speaker fees, clinical research grants, and/or honoraria from Aclaris, Almirall, Amgen, AnaptysBio, Arcutis, Athēnix, Biofrontera, BioPharmX, Biorasi, BlueCreek Dermatologics, BMS, Botanix, Brickell Biotech, Cara Therapeutics, Cassiopea SpA, Dermata, Dermavant, Dermira, Encore Dermatology, Evommune, Ferndale Laboratories, Foamix, Galderma, Genentech, Incyte, JEM Health, La Roche-Posay/L’Oréal, LEO Pharma, Lilly, Mayne Pharma, Menlo Therapeutics, Novan, Ortho Dermatologics, Pfizer, Promius Pharma, Ralexar, Regeneron/Sanofi Genzyme, Sebacia, Senté, Sol-Gel Technologies, Sonoma Pharmaceuticals, Sun Pharmaceuticals, Trevi Therapeutics, UCB, Unilever Home & Personal Care, Verrica, and Viamet, and receiving salary as an employee of Regeneron Pharmaceuticals. Julie C. Harper reports consulting fees and/or honoraria from Almirall, Arcutis, Bioderma, Beiersdorf, Bubble Skincare, Cutera, Galderma, Journey Medical Corporation, L’Oréal, Nutrafol, Ortho Dermatologics, and Sun Pharma. Edward Lain is an investigator for and has received grants/research funding and honoraria from Arcutis. Todd Schlesinger is an investigator, speaker, and/or consultant for AbbVie, Allergan, Almirall, Apogee Therapeutics, Arcutis, ASLAN Pharmaceuticals, Beiersdorf, BENEV, Biofrontera, BMS, Boehringer Ingelheim, Cara Therapeutics, Castle Biosciences, ChemoCentryx, Coherus, Concentrics, Concert Pharmaceuticals (acquired by SUN Pharma), Crown Aesthetics, Cutanea, Dermavant, Dermsquared, ExoCoBio, Flint Clinical, Galderma, Genentech, Highlightll, HTL Biotechnology, Incyte, Janssen, L’Oréal, LEO Pharma, Lilly, MJH Life Sciences, Nimbus Therapeutics, Novartis, Pfizer, Processa, Prolacta, RBC Consultants, Regeneron, Sanofi, Senté, Sun Pharma, Takeda, Trevi Pharmaceuticals, UCB, and Verrica; is a shareholder of BMS, Lilly, and Remedly; and received a salary from Avant-Health. Ali Shahbaz is an investigator and/or consultant for AbbVie/Allergan, Apogee Therapeutics, Arcutis, Beiersdorf, BMS, Boehringer Ingelheim, Crown Aesthetics, Dermavant, Galderma, Janssen, LEO Pharma, Lilly, Novartis, Sun Pharma, and UCB. Heather Woolery-Lloyd has served as a consultant, speaker, and/or investigator for Allergan, Arcutis, Eirion, Galderma, Incyte, L’Oréal, Lilly, Ortho Dermatologics, Pfizer, Regeneron, and Vyne. Jennifer C. Jaworski, Melissa S. Seal, and Diane Hanna are employees and shareholders of Arcutis Biotherapeutics, Inc. Peter Lio reports honoraria for participation in speakers bureaus for AbbVie, Arcutis, Galderma, Hyphens Pharma, Incyte, La Roche-Posay/L’Oréal, Lilly, Pfizer, Pierre Fabre, Regeneron/Sanofi Genzyme, and Verrica; personal fees for consulting/participation in advisory boards for AbbVie, Almirall, Alphyn Biologics, Amyris, Arcutis, ASLAN Pharmaceuticals, Astria Therapeutics, BMS, Boston Skin Science, Burt’s Bees, Castle Biosciences, Codex Labs, Concerto Biosciences, Dermavant, Galderma, Kenue, L’Oréal, LEO Pharma, Lilly, Lipidor, Merck, Micreos, MyOr Diagnostics, Pelthos Therapeutics, Regeneron/Sanofi Genzyme, Sibel Health, Skinfix, Soteri Skin, Stratum Biosciences, Sun Pharma, Theraplex, Thimble Health, UCB, Unilever, Verdant Scientific, Verrica, and Yobee Care; and stock options with Alphyn Biologics, Codex Labs, Concerto Biosciences, Soteri Skin, Stratum Biosciences, Thimble Health, Yobee Care, and Verdant Scientific. In addition, Dr. Lio has a patent pending for a Theraplex product with royalties paid and is a Board Member and Scientific Advisory Committee Member of the National Eczema Association.
ABSTRACT: Many inflammatory skin conditions share mechanistic pathways involving multiple receptors and cytokines. Perhaps there is no better endorsement of this idea than the foundational concept of “steroid-responsive dermatoses.” Similarly, targeting the phosphodiesterase 4 (PDE4) enzyme, which exists at the crux of several pathways that contribute to inflammatory skin diseases, offers an avenue by which the pathogenesis of multiple conditions may be interrupted. This suggests that PDE4 inhibitors may assume a similar complementary framework for treating “PDE4 inhibitor-responsive dermatoses.” Certain formulations of PDE4 inhibitors (including apremilast, crisaborole, and roflumilast) have undergone rigorous investigation in clinical trials and demonstrated robust efficacy and safety in many dermatologic conditions, achieving United States Food and Drug Administration approval for atopic dermatitis, psoriasis, and seborrheic dermatitis. Herein, we summarize the clinical evidence supporting the emerging concept of “PDE4 inhibitor-responsive dermatoses” and propose that PDE4 inhibition offers an additional pathway to achieving desired clinical outcomes. Keywords: Steroid-responsive dermatoses, PDE4 inhibitor-responsive dermatoses, crisaborole, apremilast, roflumilast, phosphodiesterase 4 inhibition
Introduction
The era of biologics has brought increasingly targeted therapies to dermatology, unlocking the ability to target specific receptors and even single cytokines. Yet, there is no doubt that many inflammatory conditions share mechanistic pathways involving multiple receptors and cytokines. Perhaps there is no better endorsement of this idea than the foundational concept of “steroid-responsive dermatoses,” a term that arose soon after the introduction of steroids to treat skin diseases in the 1950s. The indication of “inflammatory and pruritic manifestations for corticosteroid-responsive dermatoses,” which has been shortened to “steroid-responsive dermatoses,” encompasses the many conditions that exhibit a therapeutic response to corticosteroids. As the number of indications for corticosteroids grew, the term grew more useful.1 The United States Food and Drug Administration (FDA) has since adopted this term primarily to communicate drug indications; however, this approval pathway now requires specific clinical trials and regulatory endpoints in each disease-specific indication.2-4 An encompassing term for a wide range of indications, “steroid-responsive dermatoses” demonstrates the interwoven pathogenesis of the dermatoses encountered daily in the practice of dermatology.
Patients, however, increasingly voice concerns regarding continuous long-term use of corticosteroids, a phenomenon that has been characterized in the literature and on media platforms as “steroid phobia.” Estimates of the prevalence of steroid phobia have ranged from 21.0% to as high as 95.7%.5,6 Concerns related to adverse events include cutaneous events (eg, skin thinning, skin aging, dyspigmentation, striae), systemic events (eg, hypothalamic-pituitary-adrenal axis suppression, increased fracture risk, osteoporosis, stunted growth and development, rebound flare, topical steroid withdrawal [TSW] syndrome, ophthalmic effects), and other unknown long-term effects.5-10 Meanwhile, nonsteroidal treatment options, which have recently been termed advanced targeted topical (ATT) treatments, for a variety of dermatologic conditions have continued to expand.11 Consensus statements from an expert panel regarding the safety and efficacy of topical corticosteroids (TCS) and newer targeted therapies for inflammatory skin conditions provide a call to action for clinicians to consider newer ATT treatments in patients with chronic inflammatory skin disease.10 The key components of the consensus statements include that practitioners should exercise caution when prescribing topical or systemic corticosteroids, with use only in acute settings due to the risk of cutaneous and systemic adverse events. Additionally, use of multiple TCS to treat various affected areas of the body can burden the healthcare system and patients. Finally, ATT treatments (including roflumilast, tapinarof, ruxolitinib, and crisaborole) should be considered as alternatives to TCS and can be a safe and effective option.
Analogous to “steroid-responsive dermatoses,” there is an emerging concept involving the nonsteroidal pathway of phosphodiesterase 4 (PDE4) inhibition. In this narrative review, we summarize the existing literature, which demonstrates the potential for many conditions to respond to inhibition of the PDE4 inflammatory pathway, and suggest that it may be time to consider framing a larger group of conditions as being responsive to PDE4 inhibition.
History
Since the introduction of hydrocortisone in the 1950s, TCS have become a mainstay in the field of dermatology for their extensive applications in the treatment of various dermatoses.12 The anti-inflammatory, vasoconstrictive, antimitotic, and immunosuppressive effects of TCS cast a wide net, interfering with the many pathological mechanisms of “steroid-responsive dermatoses.” This large group of “steroid-responsive dermatoses” includes eczematous, papulosquamous, autoimmune, blistering, vascular, inflammatory, and granulomatous skin conditions, among others.1
Adverse events associated with TCS became apparent soon after their use expanded in the latter half of the 20th century. Effects such as systemic absorption, adrenal suppression, telangiectasis, purpura, skin atrophy, and striae were already reported by the mid-1970s.13 Unsurprisingly, concerns about TCS use have persisted. By 2000, research examined topical steroid phobia among patients with eczema, with one dermatology outpatient center demonstrating that 145 of 200 patients (72.5%) had concerns about using TCS on their own or their child’s skin, leading to nonadherence in 33.1% of these patients.14 The most prominent concerns among patients were skin thinning and nonspecific long-term adverse events—concerns that remain today, accompanied by concerns of growth stunting, skin aging, dyspigmentation, and the specter of TSW syndrome.5-9,14
In recent years, the discourse surrounding TSW syndrome has soared. From 2016 to 2020, use of the hashtag “#topicalsteroidwithdrawal” increased 274% on Instagram, Twitter, Facebook, Reddit, LinkedIn, YouTube, and Pinterest.15 Descriptions of TSW symptoms included edema, widespread erythema, burning pain, skin sensitivity, oozing, skin exfoliation, acneiform papules, and intense itch upon cessation of long-term mid-to-high-potency TCS use.16,17 A recent study included the first mechanistic evaluation of TSW through metabolomic and transcriptomic evaluation, both in vitro and in patients with atopic dermatitis (AD) reporting TSW symptoms following glucocorticoid exposure, and identified TSW as an iatrogenic dermatopathy distinct from AD in which patients had elevated levels of oxidized nicotinamide adenine dinucleotide (NAD+) in their blood serum and skin as compared to patients without TSW symptoms.18 Yet, TSW remains a debated and controversial topic in the field of dermatology, and the limited existing research on TSW is of variable quality.19 Importantly, however, the government of the United Kingdom (through the Medicines and Healthcare Products Regulatory Agency), the National Eczema Society, the British Dermatological Nursing Group, and the British Association of Dermatologists have recognized TSW.20,21 Additionally, in July 2022, the government of Canada released a safety brief regarding the risk of TSW reactions.22 Although TSW is yet to be officially recognized as an entity in the United States, the National Eczema Association has issued educational information on TSW and highlighted the risks associated with TCS use.23,24 Concerns regarding corticosteroid use nonetheless support the exploration and expansion of ATT treatment options.
PDE4 inhibition for the treatment of dermatological diseases was born out of the work of Jon Hanifin, starting in the late 1980s. In 1988, Hanifin characterized cyclic adenosine monophosphate (cAMP)-PDE forms in monocytes and lymphocytes, exploring the potential role of abnormal PDE elevation in inflammatory response in AD.25 Playing an important role in degradation of cAMP, PDE4 acts to regulate inflammatory activity in many immune cells, such as T cells, neutrophils, dendritic cells, monocytes, macrophages, and eosinophils (Figure 1).26-30 As such, PDE4 inhibition attenuates the production and release of cytokines and chemokines, influencing inflammation and chemotaxis by interfering with a variety of downstream pathways.26,31 Inhibition has also been shown to directly reduce the activation of dermal sensory neurons that mediate the itch sensation as well as normalization of keratinocyte activation and differentiation that causes epidermal barrier dysfunction.30,32-38 Additionally, PDE4 inhibition with roflumilast has been shown to promote melanocyte proliferation, increase melanogenic gene and protein expression in cultured melanocytes, and protect melanocytes from apoptosis.39,40 Thus, targeting this enzyme that exists at the crux of several mechanistic pathways offers an avenue by which the pathogenesis of multiple conditions may be interrupted.
Hanifin’s exploration continued throughout the 1990s with investigations of increased cAMP-PDE activity in leukocytes of patients with AD, as well as of the in vivo and in vitro anti-inflammatory efficacy of PDE4 inhibitors for AD.41,42 This was demonstrated with twice-daily (BID) application of a topical PDE4 inhibitor (CP 80,633) in patients with AD for 28 days, resulting in significantly decreased inflammation within 3 days and overall reductions in erythema, induration, and excoriations.42 Certain formulations of PDE4 inhibitors have since undergone rigorous investigation in clinical trials and demonstrated robust efficacy and safety, achieving FDA approval for apremilast, crisaborole, and roflumilast. The structural properties of these PDE4 inhibitors inform how they bind to the active site in PDE4 and impacts potency, function, and clinical efficacy.43
PDE4 Inhibitors with approved indications in dermatology
Apremilast. Apremilast, a small-molecule oral PDE4 inhibitor, was first approved by the FDA in 2014 for adults with active psoriatic arthritis. Its indications now include adults and children (aged 6 years or older and weighing at least 20kg) with plaque psoriasis who are candidates for phototherapy or systemic therapy and adults with oral ulcers associated with Behçet’s disease at doses ranging from 10 to 30mg.44,45
In randomized control trials, apremilast has demonstrated efficacy in achieving at least 75% improvement in Psoriasis Area and Severity Index (PASI-75) after 16 weeks in 29% to 41% of participants with psoriasis at a dosage of 30mg twice per day (BID) and 29% and 11% at dosages of 20mg and 10mg BID, respectively.46-48 For psoriatic arthritis, 16 weeks of treatment has demonstrated ≥20% improvement in American College of Rheumatology response criteria (ACR20) among 28% to 37% of patients receiving a dose of 20mg BID, and 32% to 41% of patients at 30mg BID.49,50 After 52 weeks, a sustained response to treatment has been observed, with 75% of participants in the ESTEEM 1 trial achieving PASI-70 and over 50% achieving ACR20 in the PALACE 2 trial.47,49,50
While apremilast has demonstrated marked efficacy in clinical trials, several factors modulate patient outcomes in the clinical setting. Continued use of apremilast in patient populations is complicated by its dosing and adverse events. Upon treatment initiation, the dose can be gradually titrated over 5 days to reach the desired daily dosing schedule. Additionally, dosage is maintained at BID (with the exception of patients with impaired renal function).45 The complicated and frequent nature of dosing may reduce patient adherence or result in treatment switching. Additionally, the most commonly reported adverse events include weight loss and gastrointestinal symptoms (nausea, vomiting, and diarrhea). To a lesser degree, patients may experience headache and upper respiratory tract infection.45,47-51 Much less frequently, depression and suicidal ideation have also been reported.45,49,51 Although most adverse events are seemingly mild, they may contribute to discontinuation of treatment. In a retrospective review of 2841 patients receiving apremilast, adherence to apremilast over a 24-month period was estimated at 22.8%.52 Treatment persistence studies have estimated low drug persistence, with 6-month and 1-year drug survival rates ranging from 50.3% to 69.5% and 31.3% to 34.7%, respectively. The 2 primary reasons for discontinuation were lack or loss of efficacy and adverse events.51,53 Less frequently, discontinuation was related to pregnancy plans, as pregnancy and breastfeeding are contraindications to apremilast treatment.45,53
Regardless, apremilast is efficacious and relatively safe for its indications, demonstrating mostly mild-to-moderate adverse events for patients as young as 6 years of age. Additionally, it may be a more accessible option than biologics for some patients.52
Crisaborole. Crisaborole is a topical treatment that was approved by the FDA in 2016. Crisaborole is indicated for mild-to-moderate AD in adult and pediatric patients aged 3 months and older.54 In a pooled analysis of 8 randomized control trials, patients undergoing 4-week treatment with crisaborole BID demonstrated significant improvements in the Pruritus Numerical Rating Scale (NRS), Patient-Oriented Eczema Measure (POEM), Eczema Area and Severity Index (EASI), Investigator Global Assessment (IGA), Dermatology Life Quality Index (DLQI), and Dermatitis Family Impact Questionnaire (DFI) compared with 4 weeks of vehicle treatment. Additionally, patients treated with crisaborole achieved ≥75% improvement in EASI (EASI-75) at a higher rate (relative risk of 1.71) than vehicle. Importantly, there was no difference in the incidence of adverse events between crisaborole and vehicle-treated groups.55 With regard to long-term outcomes, studies of up to 52 weeks have demonstrated continued efficacy.56-58 At 52 weeks, patients treated with crisaborole BID had a longer median flare-free interval, more flare-free days (on average), and fewer flares (on average) than patients treated with vehicle.56
Adverse events associated with crisaborole include AD, application site pain, application site infection, gastrointestinal disorders, upper respiratory tract infection, and nasopharyngitis; although, studies of these adverse events considered them unrelated to treatment, and there is little evidence of serious adverse events with crisaborole use.57-59 Additionally, systemic exposure of crisaborole is minimal, as it is rapidly metabolized to inactive products; however, systemic exposure increases with increased application area.58 Crisaborole provides a nonsteroidal treatment option for patients with AD as young as 3 months of age.
Limitations of crisaborole use include adherence, hypersensitivity, and local tolerability. As a topical ointment, crisaborole is subject to the well-known phenomenon of nonadherence, which is often a result of inconvenience.60,61 Additionally, hypersensitivity reactions to crisaborole are contraindications to its use, although such reactions occur in a minority of patients and are typically mild or moderate.54,57-59 Thirdly, stinging and burning at the application site, while reported at relatively low levels in clinical trials, have been observed to be more common in the postmarketing phase, affecting upwards of 30% of patients in some reports.62
Roflumilast. Roflumilast is a PDE4 inhibitor originally approved by the FDA in 2011 as an oral formulation for reducing the risk of chronic obstructive pulmonary disease (COPD) exacerbations.63 In recent years, topical roflumilast has been approved for several dermatologic indications. The cream formulation is approved for psoriasis in patients aged 6 years and older at 0.3% and for AD in patients aged 6 years and older and 2-5 years at 0.15% and 0.05%, respectively.64,65 An additional trial of roflumilast cream 0.05% is underway in patients with AD aged 3 to 24 months.66 The foam preparation (0.3%) is approved for patients with seborrheic dermatitis aged 9 years and older and for patients with psoriasis of the scalp and body aged 12 years and older.67 Once-daily (QD) topical roflumilast has demonstrated substantial efficacy in a recent meta-analysis of randomized controlled trials on roflumilast cream 0.3% for psoriasis. Patients saw significant achievement of IGA success (ie, clear or almost clear with ≥2-grade improvement from baseline; relative risk of 5.07 compared to vehicle) and PASI-75 (relative risk of 4.48 compared to vehicle) at 8 weeks.68 Patients with seborrheic dermatitis treated with roflumilast foam 0.3% QD demonstrated up to 43.0%, 73.1%, and 73.8% achievement of IGA success at 2, 4, and 8 weeks, respectively, in Phase II and III trials.69,70 Similarly, the INTEGUMENT trials of roflumilast cream 0.15% QD in patients aged ≥6 years with AD demonstrated up to 43.2% achieved EASI-75 by 4 weeks of treatment.71 Topical roflumilast was also well tolerated and demonstrated safety in long-term trials of up to 52 weeks.72,73 The most common adverse events reported were mild or moderate, and included diarrhea, nausea, headache, upper respiratory tract infections, and nasopharyngitis.69-74
The QD dosing regimen offers more convenience than other topical PDE4 inhibitors and is well tolerated, with patient-reported rates of stinging and burning of <2%. Additionally, the cream roflumilast formulation is approved and efficacious in intertriginous areas, providing a well-tolerated treatment option for patients with intertriginous involvement.72
An oral formulation of roflumilast has been explored for the treatment of psoriasis, demonstrating PASI-75 achievement in 44% of patients at 24 weeks, although with more adverse events, specifically gastrointestinal adverse events, than the topical formulation.75
PDE4 Inhibitors in other dermatologic conditions
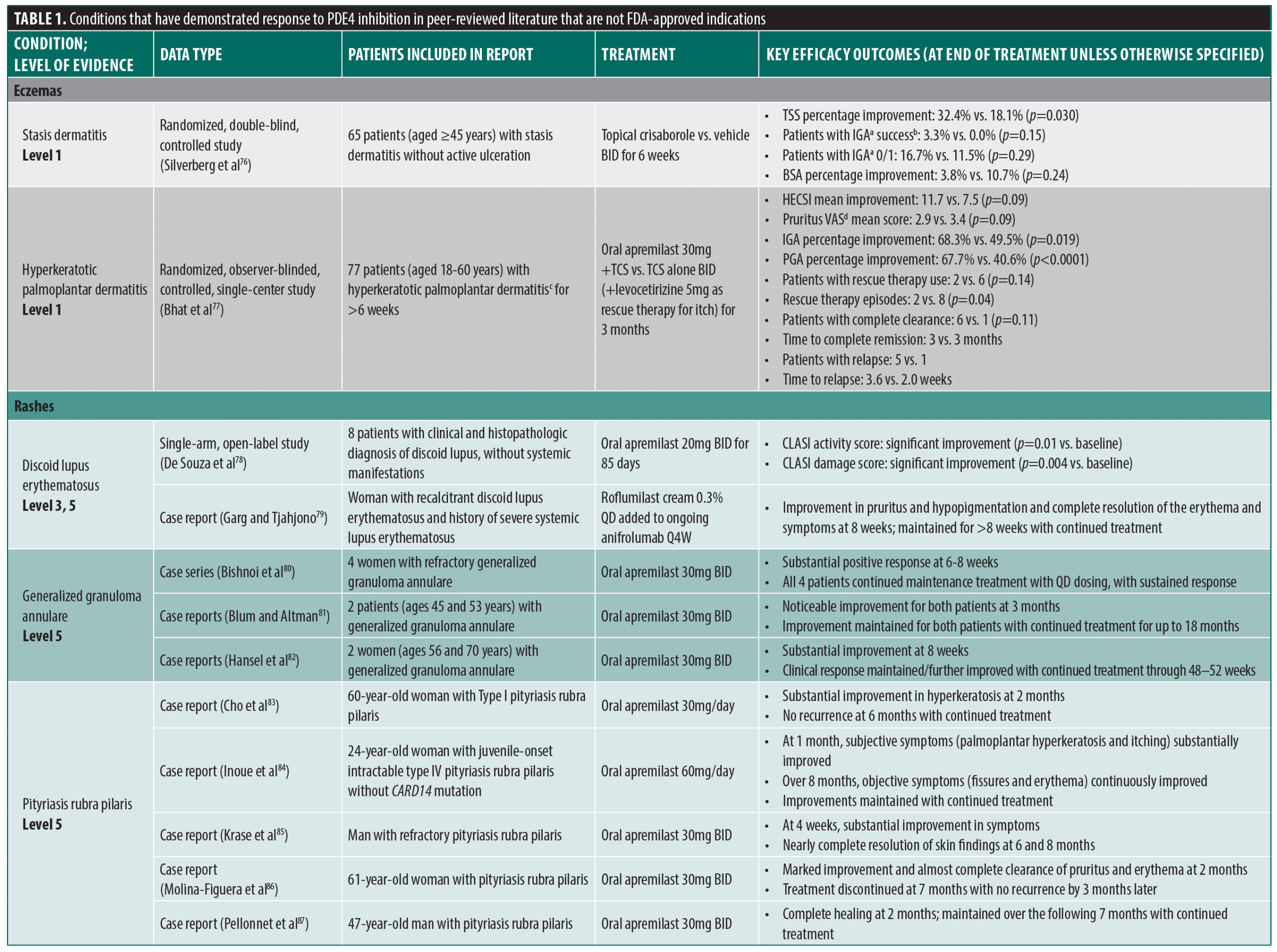
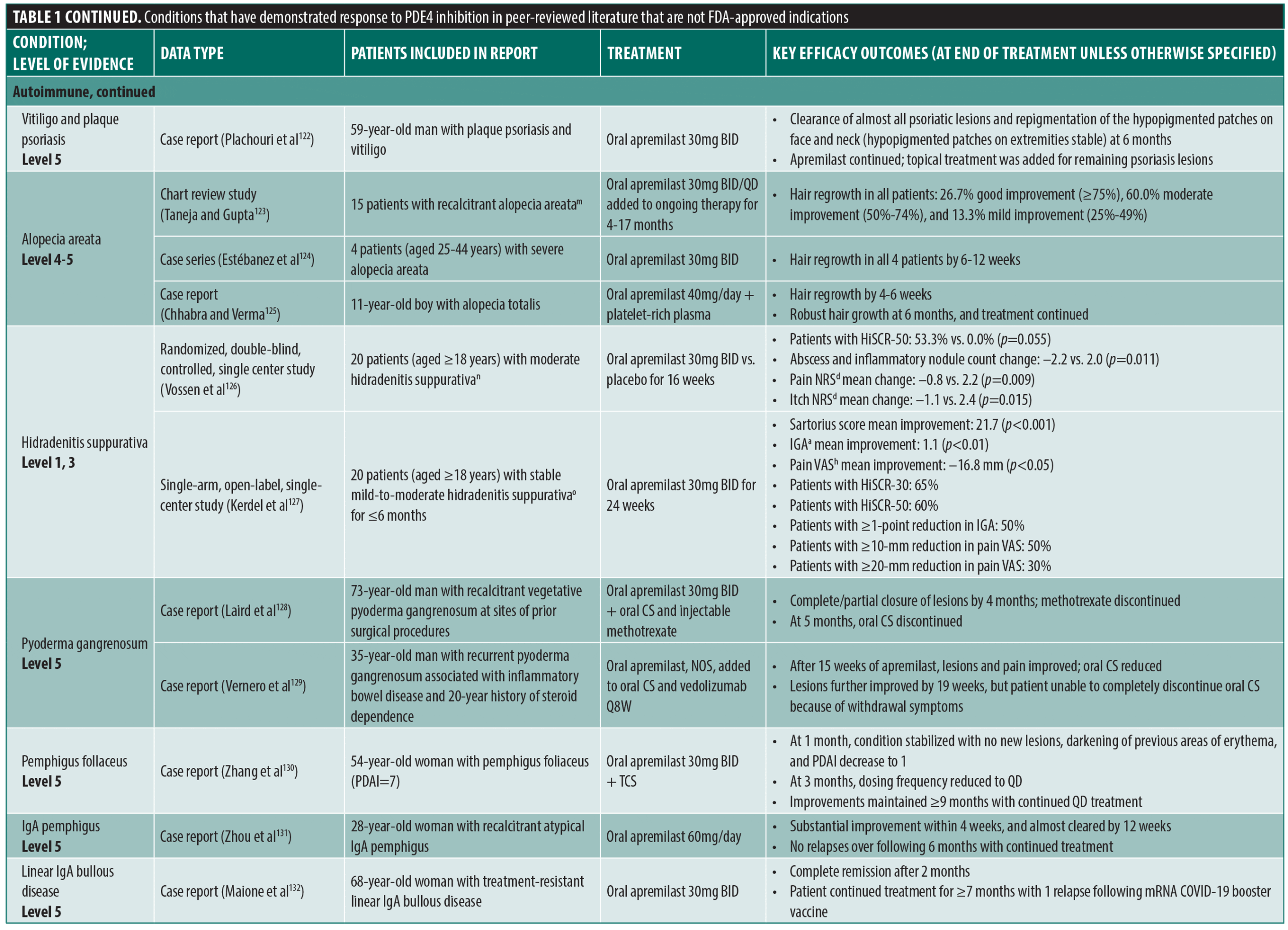
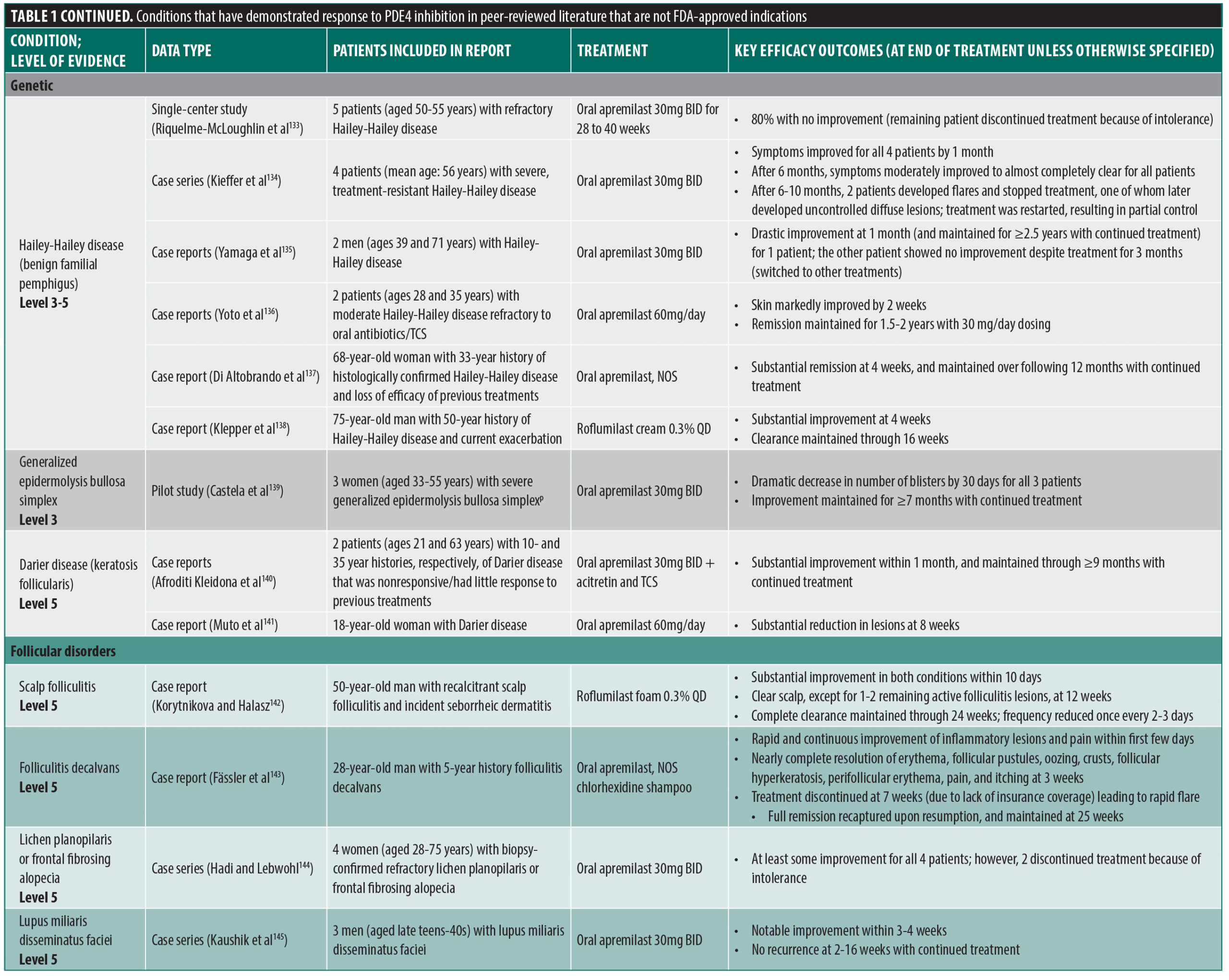
PDE4 inhibitors continue to be explored for their potential applications in a wide range of inflammatory conditions. Table 1 summarizes several reports of other dermatologic conditions that have responded to PDE4 inhibition, identified through a nonsystematic search of PubMed using terms including PDE4 inhibition, case reports, clinical trial, and dermatologic conditions, conducted on February 28, 2025. Reports published up to the date of the search were selected based on novelty, relevance to the field, and the relative unmet medical need for a particular indication, with priority given to reports containing higher levels of evidence. The level of evidence varies from single case reports to randomized controlled trials, and the strength of the conclusions drawn from these reports is commensurate with these differences.
An Emerging Concept: “PDE4-Responsive Dermatoses”
Given the role of PDE4 in the pathogenesis of so many inflammatory skin conditions and the continued exploration into the growing number of potential indications for PDE4 inhibitors, the concept of “PDE4 inhibitor-responsive dermatoses” may be a useful, modern way of thinking about many of the common inflammatory skin conditions that are seen in dermatology each day.
Moreover, PDE4 inhibitors are generally well tolerated and have demonstrated remarkable safety among adult and pediatric patients. Not only are they efficacious, but they are also useful in body areas that may be challenging for TCS, such as facial, eyelid, genital, and flexural areas, provided they are appropriately formulated for these areas. Therefore, PDE4 inhibitors complement the existing concept of “steroid-responsive dermatoses,” offering an additional tool to potentially reach desired clinical outcomes.
Conclusion
The concept of “steroid-responsive dermatoses” has persisted because of its utility in treating a wide range of inflammatory conditions, despite recent advances in the availability of ATT treatments. The field may benefit from welcoming the nonsteroidal complementary framework of “PDE4 inhibitor-responsive dermatoses.” In clinical practice, for cases of overlapping conditions, unclear diagnoses, inadequate responses to targeted agents, and patients for whom targeted agents may not be suitable, PDE4 inhibitors pose an ATT option that can play a key role in the broader treatment of inflammation. TCS have a place in dermatology to address acute self-limiting presentations; however, it seems that PDE4 inhibitors can safely and effectively assume a role analogous to that of TCS in treating a wide variety of inflammatory conditions encountered in dermatology daily. We look forward to the volume and quality of the evidence base for this concept grow and evolve.
References
- Thomas J, Parimalam K. Dermatological indications and usage of topical corticosteroid. A Treatise on Topical Corticosteroids in Dermatology: Use, Misuse and Abuse. Springer Singapore. 2018;23:chap 40.
- Clobetasol propionate cream USP, 0.05%. Prescribing information. Lupin Pharmaceuticals, Inc.; 2024. Accessed March 18, 2025. https://www.lupin.com/US/wp-content/uploads/2024/11/pi-clobetasol-propionate-cream-usp-0.05-11-2024.pdf
- Hydrocortisone cream USP, 2.5%. Prescribing information. Padagis Israel Pharmaceuticals Ltd; 2023. Accessed March 18, 2025. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=7ba59aa6-b828-4048-bf86-90c90d9894a0&type=display
- Betamethasone valerate cream USP, 0.1%. Prescribing information. E. Fougera & Co.; 2021. Accessed March 18, 2025. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=0b386ccc-a2d4-4d18-a2d7-e46ff894c674&type=display
- Contento M, Cline A, Russo M. Steroid phobia: a review of prevalence, risk factors, and interventions. Am J Clin Dermatol. 2021;22(6):837-851.
- Li AW, Yin ES, Antaya RJ. Topical corticosteroid phobia in atopic dermatitis: a systematic review. JAMA Dermatol. 2017;153(10):1036-1042.
- Fitzmaurice W, Silverberg NB. Systematic review of steroid phobia in atopic dermatitis. Dermatitis. 2024;35(6):664-668.
- Sheary B. Steroid withdrawal effects following long-term topical corticosteroid use. Dermatitis. 2018;29(4):213-218.
- Tan S, Phan P, Law JY, Choi E, Chandran NS. Qualitative analysis of topical corticosteroid concerns, topical steroid addiction and withdrawal in dermatological patients. BMJ Open. 2022;12(3):e060867.
- Burshtein J, Chovatiya R, Golant A, et al. Risks of topical corticosteroid therapy and role for advanced targeted topical treatments for inflammatory skin diseases: an expert consensus panel. Dermatol Online J. 2025;31(1):10.5070/D331164978.
- DiRuggiero D, Agopian J, Baker A, et al. Recommendations for topical corticosteroid use and the role of alternative, advanced targeted topical treatments in the treatment of chronic inflammatory skin diseases. J Clin Aesthet Dermatol. 2025;18(5-6 Suppl 1):S10-S13.
- Mehta AB, Nadkarni NJ, Patil SP, Godse KV, Gautam M, Agarwal S. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82(4):371-378.
- Lee SS, Rapp Y. The modern topical steroid. Int J Dermatol. 1975;14(6):412-421.
- Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142(5):931-936.
- Bowe S, Masterson S, Murray G, Haugh I. Topical steroid withdrawal through the lens of social media. Clin Exp Dermatol. 2022;47(8):1554-1557.
- Brookes TS, Barlow R, Mohandas P, Bewley A. Topical steroid withdrawal: an emerging clinical problem. Clin Exp Dermatol. 2023;48(9):1007-1011.
- Tan SY, Chandran NS, Choi EC. Steroid phobia: is there a basis? A review of topical steroid safety, addiction and withdrawal. Clin Drug Investig. 2021;41(10):835-842.
- Shobnam N, Ratley G, Saksena S, et al. Topical steroid withdrawal is a targetable excess of mitochondrial NAD. J Invest Dermatol. 2025;145(8):1953-1968.e14.
- Hwang J, Lio PA. Topical corticosteroid withdrawal (‘steroid addiction’): an update of a systematic review. J Dermatolog Treat. 2022;33(3):1293-1298.
- Medicines and Healthcare Products Regulatory Agency. MHRA Public Assessment Report: Topical Steroid Withdrawal Reactions: A Review of the Evidence. September 15, 2021. Accessed March 18, 2025. https://www.gov.uk/government/publications/topical-steroid-withdrawal-reactions-a-review-of-the-evidence/topical-steroid-withdrawal-reactions-a-review-of-the-evidence
- National Eczema Society, British Dermatological Nursing Group, British Association of Dermatologists. Topical Steroid Withdrawal: A Joint Statement by National Eczema Society, the British Dermatological Nursing Group, and the British Association of Dermatologists. February 2024. Accessed March 18, 2025. https://cdn.bad.org.uk/uploads/2024/02/22095550/Topical-Steroid-Withdrawal-Joint-Statement.pdf
- Health Canada. Safety Brief: Topical Corticosteroids and the Risk of Topical Withdrawal Reactions. Her Majesty the Queen in Right of Canada. July 2022. Accessed March 18, 2025. https://www.canada.ca/en/health-canada/services/drugs-health-products/medeffect-canada/health-product-infowatch/july-2022.html
- National Eczema Association. Education Announcement: Use of Topical Steroids for eczema. July 17, 2021. Accessed March 18, 2025. https://nationaleczema.org/blog/warnings-for-topical-steroids-eczema/
- Hajar T, Leshem YA, Hanifin JM, et al. A systematic review of topical corticosteroid withdrawal (“steroid addiction”) in patients with atopic dermatitis and other dermatoses. J Am Acad Dermatol. 2015;72(3):541-549.e2.
- Hanifin JM, Chan SC. Characterization of cAMP‐phosphodiesterase as a possible laboratory marker of atopic dermatitis. Drug Dev Res. 1988;13(2-3):123-136.
- Chiricozzi A, Caposiena D, Garofalo V, Cannizzaro MV, Chimenti S, Saraceno R. A new therapeutic for the treatment of moderate-to-severe plaque psoriasis: apremilast. Expert Rev Clin Immunol. 2016;12(3):237-249.
- Crocetti L, Floresta G, Cilibrizzi A, Giovannoni MP. An overview of PDE4 inhibitors in clinical trials: 2010 to early 2022. Molecules. 2022;27(15):4964.
- Li H, Zuo J, Tang W. Phosphodiesterase-4 inhibitors for the treatment of inflammatory diseases. Front Pharmacol. 2018;9:1048.
- Milakovic M, Gooderham MJ. Phosphodiesterase-4 inhibition in psoriasis. Psoriasis (Auckl). 2021;11(0):21-29.
- Zebda R, Paller AS. Phosphodiesterase 4 inhibitors. J Am Acad Dermatol. 2018;78(3 Suppl 1):S43-S52.
- Schafer PH, Parton A, Gandhi AK, et al. Apremilast, a cAMP phosphodiesterase-4 inhibitor, demonstrates anti-inflammatory activity in vitro and in a model of psoriasis. Br J Pharmacol. 2010;159(4):842-855.
- Dong C, Virtucio C, Zemska O, et al. Treatment of skin inflammation with benzoxaborole phosphodiesterase inhibitors: selectivity, cellular activity, and effect on cytokines associated with skin inflammation and skin architecture changes. J Pharmacol Exp Ther. 2016;358(3):413-422.
- Bros M, Montermann E, Cholaszczynska A, Reske-Kunz AB. The phosphodiesterase 4 inhibitor roflumilast augments the Th17-promoting capability of dendritic cells by enhancing IL-23 production, and impairs their T cell stimulatory activity due to elevated IL-10. Int Immunopharmacol. 2016;35:174-184.
- Guttman-Yassky E, Hanifin JM, Boguniewicz M, et al. The role of phosphodiesterase 4 in the pathophysiology of atopic dermatitis and the perspective for its inhibition. Exp Dermatol. 2019;28(1):3-10.
- Annunziato F, Romagnani C, Romagnani S. The 3 major types of innate and adaptive cell-mediated effector immunity. J Allergy Clin Immunol. 2015;135(3):626-635.
- Wakita H, Ohkuro M, Ishii N, Hishinuma I, Shirato M. A putative antipruritic mechanism of the phosphodiesterase-4 inhibitor E6005 by attenuating capsaicin-induced depolarization of C-fibre nerves. Exp Dermatol. 2015;24(3):215-216.
- Das P, Mounika P, Yellurkar ML, et al. Keratinocytes: an enigmatic factor in atopic dermatitis. Cells. 2022;11(10):1683.
- Xu R, Feng S, Ao Z, et al. Long-acting β2 adrenergic receptor agonist ameliorates imiquimod-induced psoriasis-like skin lesion by regulating keratinocyte proliferation and apoptosis. Front Pharmacol. 2022;13:865715.
- Chen Z, Li Y, Xie Y, Nie S, Chen B, Wu Z. Roflumilast enhances the melanogenesis and attenuates oxidative stress-triggered damage in melanocytes. J Dermatol Sci. 2023;110(2):44-52.
- Goldstein NB, Berk ZBK, Tomb LC, et al. Phosphodiesterase-4 inhibitors increase pigment cell proliferation and melanization in cultured melanocytes and within a 3-dimensional skin equivalent model. J Invest Dermatol. 2024.
- Hanifin JM, Lloyd R, Okubo K, Guerin LL, Fancher L, Chan SC. Relationship between increased cyclic AMP-phosphodiesterase activity and abnormal adenylyl cyclase regulation in leukocytes from patients with atopic dermatitis. J Invest Dermatol. 1992;98(6 Suppl):100S-105S.
- Hanifin JM, Chan SC, Cheng JB, et al. Type 4 phosphodiesterase inhibitors have clinical and in vitro anti-inflammatory effects in atopic dermatitis. J Invest Dermatol. 1996;107(1):51-56.
- Wang Y, Coyne K, Sofen H, et al. Qualitative analysis and reproducibility assessment of the Scalp Itch Numeric Rating Scale among patients with moderate to severe plaque psoriasis of the scalp. J Dermatolog Treat. 2019;30(8):775-783.
- Otezla. Prescribing information. Amgen Inc.; 2024. Accessed February 19, 2025. https://www.pi.amgen.com/-/media/Project/Amgen/Repository/pi-amgen-com/Otezla/otezla_pi_english.pdf
- Padda IS, Bhatt R, Parmar M. Apremilast. StatPearls [Internet]. StatPearls Publishing; 2025. Accessed March 19, 2025. https://www.ncbi.nlm.nih.gov/books/NBK572078/
- Papp K, Cather JC, Rosoph L, et al. Efficacy of apremilast in the treatment of moderate to severe psoriasis: a randomised controlled trial. Lancet. 2012;380(9843):738-746.
- Papp K, Reich K, Leonardi CL, et al. Apremilast, an oral phosphodiesterase 4 (PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: results of a phase III, randomized, controlled trial (efficacy and safety trial evaluating the effects of apremilast in psoriasis [ESTEEM] 1). J Am Acad Dermatol. 2015;73(1):37-49.
- Paul C, Cather J, Gooderham M, et al. Efficacy and safety of apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate-to-severe plaque psoriasis over 52 weeks: a phase III, randomized controlled trial (ESTEEM 2). Br J Dermatol. 2015;173(6):1387-1399.
- Cutolo M, Myerson GE, Fleischmann RM, et al. A phase III, randomized, controlled trial of apremilast in patients with psoriatic arthritis: results of the PALACE 2 trial. J Rheumatol. 2016;43(9):1724-1734.
- Edwards CJ, Blanco FJ, Crowley J, et al. Apremilast, an oral phosphodiesterase 4 inhibitor, in patients with psoriatic arthritis and current skin involvement: a phase III, randomised, controlled trial (PALACE 3). Ann Rheum Dis. 2016;75(6):1065-1073.
- Distel J, Cazzaniga S, Seyed Jafari SM, et al. Long-term effectiveness and drug survival of apremilast in treating psoriasis: a real-world experience. Dermatology. 2022;238(2):267-275.
- Feldman SR, Zhang J, Martinez DJ, et al. Real-world biologic and apremilast treatment patterns and healthcare costs in moderate-to-severe plaque psoriasis. Dermatol Online J. 2021;27(1).
- Haddad A, Stein N, Lavi I, et al. Treatment persistence of apremilast among patients with psoriatic arthritis. Biologics. 2023;17(0):129-136.
- Eucrisa. Prescribing information. Pfizer; 2023. https://labeling.pfizer.com/showlabeling.aspx?id=5331
- He Y, Liu J, Wang Y, et al. Topical administration of crisaborole in mild to moderate atopic dermatitis: a systematic review and meta‐analysis. Dermatol. Ther. 2023;2023(1):1869934.
- Eichenfield LF, Gower RG, Xu J, et al. Once-daily crisaborole ointment, 2%, as a long-term maintenance treatment in patients aged ≥ 3 months with mild-to-moderate atopic dermatitis: a 52-week clinical study. Am J Clin Dermatol. 2023;24(4):623-635.
- Eichenfield LF, Call RS, Forsha DW, et al. Long-term safety of crisaborole ointment 2% in children and adults with mild to moderate atopic dermatitis. J Am Acad Dermatol. 2017;77(4):641-649.e5.
- Hoy SM. Crisaborole ointment 2%: a review in mild to moderate atopic dermatitis. Am J Clin Dermatol. 2017;18(6):837-843.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75(3):494-503.e6.
- Lee IA, Maibach HI. Pharmionics in dermatology: a review of topical medication adherence. Am J Clin Dermatol. 2006;7(4):231-236.
- Ramachandran V, Cline A, Feldman SR, Strowd LC. Evaluating crisaborole as a treatment option for atopic dermatitis. Expert Opin Pharmacother. 2019;20(9):1057-1063.
- Pao-Ling Lin C, Gordon S, Her MJ, Rosmarin D. A retrospective study: application site pain with the use of crisaborole, a topical phosphodiesterase 4 inhibitor. J Am Acad Dermatol. 2019;80(5):1451-1453.
- Daliresp. Prescribing information. AstraZeneca Pharmaceuticals LP; 2020. https://den8dhaj6zs0e.cloudfront.net/50fd68b9-106b-4550-b5d0-12b045f8b184/704932ce-e104-4c6f-840a-575170971344/704932ce-e104-4c6f-840a-575170971344_viewable_rendition__v.pdf
- Zoryve® (roflumilast) cream 0.3% and 0.15%. Prescribing information. Arcutis Biotherapeutics, Inc.; 2025. Accessed November 17, 2025. https://www.arcutis.com/wp-content/uploads/USPI-roflumilast-cream.pdf
- Eichenfield LF, Serrao R, Prajapati VH, et al. Efficacy and safety of once-daily roflumilast cream 0.05% in pediatric patients aged 2-5 years with mild-to-moderate atopic dermatitis (INTEGUMENT-PED): a phase 3 randomized controlled trial. Pediatr Dermatol. 2025;42(2):296-304.
- Safety Study of Roflumilast Cream 0.05% in Infants Aged 3 Months to Less Than 2 Years With Atopic Dermatitis. ClinicalTrials.gov identifier NCT06998056. Updated June 17, 2025. Accessed November 12, 2025. https://clinicaltrials.gov/study/NCT06998056
- Zoryve® (roflumilast) foam 0.3%. Prescribing information. Arcutis Biotherapeutics, Inc.; 2025. Accessed June 18, 2025. https://www.arcutis.com/wp-content/uploads/zoryve-foam-pi-hcp.pdf
- de Moraes-Souza R, Chahine Chater R, Pera Calvi I, et al. Efficacy and safety of topical roflumilast for the treatment of psoriasis: a systematic review and meta-analysis of randomized controlled trials. Clin Drug Investig. 2024;44(9):655-665.
- Blauvelt A, Draelos ZD, Stein Gold L, et al. Roflumilast foam 0.3% for adolescent and adult patients with seborrheic dermatitis: a randomized, double-blinded, vehicle-controlled, phase 3 trial. J Am Acad Dermatol. 2024;90(5):986-993.
- Zirwas MJ, Draelos ZD, DuBois J, et al. Efficacy of roflumilast foam, 0.3%, in patients with seborrheic dermatitis: a double-blind, vehicle-controlled phase 2a randomized clinical trial. JAMA Dermatol. 2023;159(6):613-620.
- Simpson EL, Eichenfield LF, Alonso-Llamazares J, et al. Roflumilast cream, 0.15%, for atopic dermatitis in adults and children: INTEGUMENT-1 and INTEGUMENT-2 randomized clinical trials. JAMA Dermatol. 2024;160(11):1161-1170.
- Drakos A, Vender R, Torres T. Topical roflumilast for the treatment of psoriasis. Expert Rev Clin Immunol. 2023;19(9):1053-1062.
- Stein Gold L, Adam DN, Albrecht L, et al. Long-term safety and effectiveness of roflumilast cream 0.3% in adults with chronic plaque psoriasis: a 52-week, phase 2, open-label trial. J Am Acad Dermatol. 2024;91(2):273-280.
- Lebwohl MG, Kircik LH, Moore AY, et al. Effect of roflumilast cream vs. vehicle cream on chronic plaque psoriasis: the DERMIS-1 and DERMIS-2 randomized clinical trials. JAMA. 2022;328(11):1073-1084.
- Gyldenløve M, Meteran H, Sorensen JA, et al. Efficacy and safety of oral roflumilast for moderate-to-severe psoriasis-a randomized controlled trial (PSORRO). Lancet Reg Health Eur. 2023;30:100639.
- Silverberg JI, Kirsner RS, Margolis DJ, et al. Efficacy and safety of crisaborole ointment, 2%, in participants aged ≥45 years with stasis dermatitis: results from a fully decentralized, randomized, proof-of-concept phase 2a study. J Am Acad Dermatol. 2024;90(5):945-952.
- Bhat K, Patra S, Bhardwaj A, et al. Efficacy of apremilast in hyperkeratotic hand and foot dermatitis: results from a randomized observer-blinded comparative study. Int J Dermatol. 2024;63(11):1535-1540.
- De Souza A, Strober BE, Merola JF, Oliver S, Franks AG, Jr. Apremilast for discoid lupus erythematosus: results of a phase 2, open-label, single-arm, pilot study. J Drugs Dermatol. 2012;11(10):1224-1226.
- Garg KS, Tjahjono LA. Treatment of recalcitrant discoid lupus erythematosus with roflumilast 0.3% cream following suboptimal response to anifrolumab and improvement of hypopigmentation. JAAD Case Rep. 2024;54:50-52.
- Bishnoi A, Raj D, Vinay K, Dogra S. Refractory generalized granuloma annulare treated with oral apremilast. JAMA Dermatol. 2019;155(11):1318-1320.
- Blum S, Altman D. Treatment of generalized granuloma annulare with apremilast: a report of 2 cases. JAAD Case Rep. 2019;5(11):976-978.
- Hansel K, Biondi F, Bianchi L, Tramontana M, Marietti R, Stingeni L. Generalized granuloma annulare successfully treated with apremilast: report of two cases and literature review. Clin Exp Dermatol. 2021;46(8):1603-1606.
- Cho M, Honda T, Ueshima C, Kataoka T, Otsuka A, Kabashima K. A case of pityriasis rubra pilaris treated successfully with the phosphodiesterase-4 inhibitor apremilast. Acta Derm Venereol. 2018;98(10):975-976.
- Inoue E, Arase N, Hanaoka Y, Tanemura A, Fujimoto M. The beneficial effect of a PDE4 inhibitor in a patient with juvenile-onset intractable pityriasis rubra pilaris without CARD14 mutation. Dermatol Ther. 2021;34(1):e14714.
- Krase IZ, Cavanaugh K, Curiel-Lewandrowski C. Treatment of refractory pityriasis rubra pilaris with novel phosphodiesterase 4 (PDE4) inhibitor apremilast. JAMA Dermatol. 2016;152(3):348-350.
- Molina-Figuera E, Gonzalez-Cantero A, Martinez-Lorenzo E, et al. Successful treatment of refractory type 1 pityriasis rubra pilaris with apremilast. J Cutan Med Surg. 2018;22(1):104-105.
- Pellonnet L, Beltzung F, Franck F, Rouanet J, D’Incan M. A case of severe pityriasis rubra pilaris with a dramatic response to apremilast. Eur J Dermatol. 2018;28(1):128-129.
- Eto A, Nakao M, Furue M. Three cases of palmoplantar pustulosis successfully treated with apremilast. J Dermatol. 2019;46(1):e29-e30.
- Haebich G, Kalavala M. Successful treatment of refractory palmoplantar pustulosis with apremilast. Clin Exp Dermatol. 2017;42(4):471-473.
- Altman DA. Palmoplantar pustulosis successfully treated with topical roflumilast 0.3%. J Dermatol Physician Assist. 2024;18(3):34-35.
- Ravichandran S, Kheterpal MK. Apremilast for the off-label treatment of lichenoid and interface dermatoses. J Am Acad Dermatol. 2020;83(5):1489-1491.
- Chen T, Levitt J, Geller L. Apremilast for treatment of recurrent erythema multiforme. Dermatol Online J. 2017;23(1).
- Kaushik A, Narang T, Handa S. Successful use of apremilast as a steroid-sparing agent in chronic actinic dermatitis. Dermatol Ther. 2020;33(6):e13809.
- Thompson BJ, Furniss M, Zhao W, Chakraborty B, Mackay-Wiggan J. An oral phosphodiesterase inhibitor (apremilast) for inflammatory rosacea in adults: a pilot study. JAMA Dermatol. 2014;150(9):1013-1014.
- Yin X, Huang C, Feng Y, Xia J, Zhang M, Yin Z. Apremilast for the treatment of papuloerythroderma of Ofuji. Clin Exp Dermatol. 2023;48(11):1282-1284.
- Liu X, Liu Q, Guo L, Jiang X. 2% crisaborole ointment for the treatment of chronic cheilitis caused by lip tattoos. J Inflamm Res. 2024;17(0):3419-3422.
- Nakajima K, Nakajima R, Morisaka H, Nakajima H, Sano S. Beneficial impact of apremilast on palmoplantar keratodermas. Acta Derm Venereol. 2021;101(4):adv00437.
- Kaushik A, Raj D, Chatterjee D, Vinay K. Apremilast in orofacial granulomatosis-a report of five cases. Dermatol Ther. 2020;33(6):e14345.
- Waki Y, Kamiya K, Komine M, et al. A case of anti-laminin γ1 (p200) pemphigoid with psoriasis vulgaris successfully treated with apremilast. Eur J Dermatol. 2018;28(3):413-414.
- Ruiz Dasilva D. Palmoplantar psoriasis treatment with topical roflumilast 0.3%. SKIN. 2024;8(6):1981-1983.
- Hanna E, Benchikh El Fegoun A. Anti-TNF alpha-induced paradoxical psoriasis treated with topical roflumilast 0.3%. JAAD Case Rep. 2024;48:115-117.
- Garg KS, Kircik L, Tjahjono L. Polymorphous light eruptions treated with roflumilast 0.3% cream: a case report. J Drugs Dermatol. 2024;23(8):686-687.
- Rich P, Gooderham M, Bachelez H, et al. Apremilast, an oral phosphodiesterase 4 inhibitor, in patients with difficult-to-treat nail and scalp psoriasis: results of 2 phase III randomized, controlled trials (ESTEEM 1 and ESTEEM 2). J Am Acad Dermatol. 2016;74(1):134-142.
- Strober B, Bagel J, Lebwohl M, et al. Efficacy and safety of apremilast in patients with moderate plaque psoriasis with lower BSA: week 16 results from the UNVEIL study. J Drugs Dermatol. 2017;16(8):801-808.
- Johnston LA, Poelman SM. Successful treatment of nail psoriasis with topical roflumilast: a case report. SAGE Open Med Case Rep. 2024;12:2050313X241289594.
- Jeon C, Nakamura M, Sekhon S, et al. Generalized pustular psoriasis treated with apremilast in a patient with multiple medical comorbidities. JAAD Case Rep. 2017;3(6):495-497.
- Kurihara Y, Nakano K, Eto A, Furue M. Successful treatment of acrodermatitis continua of Hallopeau with apremilast. J Dermatol. 2019;46(10):e370-e371.
- Lanna C, Cesaroni GM, Mazzilli S, et al. Nails as immune-privileged sites: a case of disabling acrodermatitis continua of Hallopeau successfully treated with apremilast. Dermatol Ther. 2019;32(4):e12946.
- Georgakopoulos JR, Ighani A, Yeung J. Short- and long-term management of an acute pustular psoriasis flare: a case report. J Cutan Med Surg. 2017;21(5):452-456.
- Viswanath V, Joshi P, Dhakne M, Dhoot D, Mahadkar N, Barkate H. Evaluation of the efficacy and safety of apremilast in the management of lichen planus. Clin Cosmet Investig Dermatol. 2022;15(0):2593-2600.
- Baker A. The treatment of lichen planus with topical roflumilast 0.3%. J Dermatol Physician Assist. 2025.
- Bettencourt M. Oral lichen planus treated with apremilast. J Drugs Dermatol. 2016;15(8):1026-1028.
- Fage S, Johansen C. Severe and therapeutic challenging oral erosive lichen planus treated with oral roflumilast. Clin Exp Dermatol. 2023;48(5):556-557.
- Kim-Lim P, Thomas C. Crushed apremilast for the treatment of oral lichen planus. JAAD Case Rep. 2023;37:114-115.
- Domingues E. The treatment of lichen sclerosus with topical roflumilast 0.3% case study. SKIN. 2024;8(6):1997-2000.
- Kim HJ, Singer GK, Del Duca E, et al. Combination of apremilast and narrowband ultraviolet B light in the treatment of generalized vitiligo in skin phototypes IV to VI: a randomized split-body pilot study. J Am Acad Dermatol. 2021;85(6):1657-1660.
- Kim HJ, Del Duca E, Pavel AB, et al. Apremilast and narrowband ultraviolet B combination therapy suppresses Th17 axis and promotes melanogenesis in vitiligo skin: a randomized, split-body, pilot study in skin types IV-VI. Arch Dermatol Res. 2023;315(2):215-221.
- Sharma S, Bhardwaj A, Dwivedi P, et al. Apremilast add-on benefits over conventional drugs (ABCD) in unstable non-segmental vitiligo: a 12-week single-center randomized controlled trial. Cureus. 2023;15(4):e37180.
- Majid I, Imran S, Batool S. Apremilast is effective in controlling the progression of adult vitiligo: a case series. Dermatol Ther. 2019;32(4):e12923.
- Warren KJ, Sanchez S. Recalcitrant pediatric facial vitiligo successfully treated with roflumilast cream 0.3% once daily. J Clin Aesthet Dermatol. 2025;18(1):52-54.
- Huff SB, Gottwald LD. Repigmentation of tenacious vitiligo on apremilast. Case Rep Dermatol Med. 2017;2017(1):2386234.
- Plachouri KM, Kyriakou G, Chourdakis V, Georgiou S, Grafanaki K. One stone, two birds: improvement of early-onset vitiligo under apremilast in a patient with plaque psoriasis. Dermatol Ther. 2019;32(5):e13064.
- Taneja N, Gupta S. Apremilast is efficacious in refractory alopecia areata. J Dermatolog Treat. 2020;31(7):727-729.
- Estébanez A, Estebanez N, Martin JM, Montesinos E. Apremilast in refractory alopecia areata. Int J Trichology. 2019;11(5):213-215.
- Chhabra G, Verma P. Steroid-resistant alopecia totalis in a child successfully treated with oral apremilast and platelet-rich plasma therapy. Dermatol Ther. 2019;32(6):e13082.
- Vossen A, van Doorn MBA, van der Zee HH, Prens EP. Apremilast for moderate hidradenitis suppurativa: results of a randomized controlled trial. J Am Acad Dermatol. 2019;80(1):80-88.
- Kerdel FR, Azevedo FA, Kerdel Don C, Don FA, Fabbrocini G, Kerdel FA. Apremilast for the treatment of mild-to-moderate hidradenitis suppurativa in a prospective, open-label, phase 2 study. J Drugs Dermatol. 2019;18(2):170-176.
- Laird ME, Tong LX, Lo Sicco KI, Kim RH, Meehan SA, Franks AG, Jr. Novel use of apremilast for adjunctive treatment of recalcitrant pyoderma gangrenosum. JAAD Case Rep. 2017;3(3):228-229.
- Vernero M, Ribaldone DG, Cariti C, et al. Dual-targeted therapy with apremilast and vedolizumab in pyoderma gangrenosum associated with Crohn’s disease. J Dermatol. 2020;47(6):e216-e217.
- Zhang Q, Yu L, Wan L, Chen L, Chen J. Case report: managing pemphigus foliaceus using apremilast without systemic glucocorticosteroids or immunosuppressive agents. Front Immunol. 2024;15:1408116.
- Zhou Y, Xiao Y, Wang Y, Li W. Refractory atypical IgA pemphigus successfully treated with apremilast. J Dermatol. 2024;51(3):e86-e87.
- Maione V, Bettolini L, Venturuzzo A, Tonon F, Battocchio S, Calzavara-Pinton P. Efficacy of apremilast in a case of resistant linear IgA bullous disease. Int J Dermatol. 2023;62(5):e300-e302.
- Riquelme-Mc Loughlin C, Iranzo P, Mascaro JM, Jr. Apremilast in benign chronic pemphigus (Hailey-Hailey disease). Clin Exp Dermatol. 2020;45(6):737-739.
- Kieffer J, Le Duff F, Montaudie H, Chiaverini C, Lacour JP, Passeron T. Treatment of severe Hailey-Hailey disease with apremilast. JAMA Dermatol. 2018;154(12):1453-1456.
- Yamaga M, Miyauchi T, Peh JT, et al. Case report: difference in outcomes between two cases of Hailey-Hailey disease treated with apremilast. Front Genet. 2022;13:884359.
- Yoto A, Makino T, Mizawa M, et al. Two cases of Hailey-Hailey disease effectively treated with apremilast and a review of reported cases. J Dermatol. 2021;48(12):1945-1948.
- Di Altobrando A, Sacchelli L, Patrizi A, Bardazzi F. Successful treatment of refractory Hailey-Hailey disease with apremilast. Clin Exp Dermatol. 2020;45(5):604-605.
- Klepper EM. Case report of Hailey-Hailey disease successfully treated with topical roflumilast 0.3% cream. J Dermatol Res. 2024:1-4.
- Castela E, Tulic MK, Rozieres A, et al. Epidermolysis bullosa simplex generalized severe induces a T helper 17 response and is improved by apremilast treatment. Br J Dermatol. 2019;180(2):357-364.
- Afroditi Kleidona I, Agiasofitou E, Tsiogka A, et al. Darier disease responding to apremilast: report of two cases. Indian J Dermatol Venereol Leprol. 2024;90:1-3.
- Muto Y, Kinjyo K, Asano Y. Successful treatment of Darier’s disease with apremilast and review of reported cases. J Dermatol. 2025;52(1):138-141.
- Korytnikova E, Halasz CL. Chronic nonscarring scalp folliculitis responding to topical phosphodiesterase-4 inhibitor roflumilast. JAAD Case Rep. 2025;55:94-96.
- Fässler M, Radonjic-Hoesli S, Feldmeyer L, et al. Successful treatment of refractory folliculitis decalvans with apremilast. JAAD Case Rep. 2020;6(10):1079-1081.
- Hadi A, Lebwohl M. Apremilast for lichen planopilaris and frontal fibrosing alopecia: a case series. SKIN. 2017;1(1):32-36.
- Kaushik A, Kumaran MS, Chatterjee D, De D. The search for a uniformly effective treatment in patients with lupus miliaris disseminatus faciei. JAMA Dermatol. 2020;156(10):841-842.
- Sánchez-Velázquez A, Falkenhain-Lopez D, Arroyo-Andres J, Montero-Menarguez J, Garcia-Donoso C, Postigo-Llorente C. Apremilast: a novel adjuvant treatment for refractory isotretinoin-induced acne fulminans. Dermatol Ther. 2022;35(8):e15637.
- Koschitzky M, Khattri S. Apremilast as a treatment for morphea: a case series. JAAD Case Rep. 2022;19:58-63.
- Baughman RP, Judson MA, Ingledue R, Craft NL, Lower EE. Efficacy and safety of apremilast in chronic cutaneous sarcoidosis. Arch Dermatol. 2012;148(2):262-264.
- Narang T, Ashraf R, Kaushik A, Dogra S. Apremilast in multibacillary leprosy patients with chronic and recurrent erythema nodosum leprosum: a prospective single-centre pilot study. J Eur Acad Dermatol Venereol. 2021;35(12):e917-e919.
- Bitar C, Ninh T, Brag K, et al. Apremilast in recalcitrant cutaneous dermatomyositis: a nonrandomized controlled trial. JAMA Dermatol. 2022;158(12):1357-1366.
- Qiblawi SH, Fivenson DP. Apremilast as an adjuvant therapy for calcinosis cutis. JAAD Case Rep. 2019;5(10):874-876.
- Calderon P, Ajmal H, Brady M, Kartono F. Refractory erythema annulare centrifugum treated with roflumilast. JAAD Case Rep. 2024;47:17-19.
- Adamo S, Nilsson J, Krebs A, et al. Successful treatment of SAPHO syndrome with apremilast. Br J Dermatol. 2018;179(4):959-962.
- Magdaleno-Tapial J, Valenzuela-Onate C, Alonso-Carpio M, et al. Improvement of recalcitrant Sneddon-Wilkinson disease with apremilast. Australas J Dermatol. 2020;61(2):185-186.
- Yamauchi PS. Management of scrotal pruritus with topical roflumilast 0.3% once daily treatment. JAAD Case Rep. 2024;47:4-5.
- Hanna E, El Moussawi N. Neurodermatitis of the scalp associated with trichotillomania treated with roflumilast cream 0.3%. JAAD Case Rep. 2025;56:57-59.
- McMichael J, Stoff BK. Treatment of a perforating dermatosis with apremilast. JAAD Case Rep. 2021;16:155-157.