J Clin Aesthet Dermatol. 2026;19(5):9–16.
by Saeid Amini-Nik, MD, PhD; Robert M. Law, MD; Vinay Bhardwaj, PhD; Stephen Lynch, PhD; and Diane B. Nelson, RN, MPH
Dr. Amini-Nik is with Société Générale de Surveillance (SGS) North America, Inc., Clinical Research, Union, New Jersey. Dr. Law is with the Dermatopathology Division, Sonic Healthcare USA, Dallas, Texas. Dr. Bhardwaj is with Société Générale de Surveillance (SGS) North America, Inc., Clinical Research, Cosmetic and Personal Care, Richardson, Texas. Dr. Lynch is with L’Oréal USA Research & Innovation, Clark, New Jersey. Ms. Nelson was with skinbetter science, a Dermatological Beauty Brand of L’Oréal USA, Inc.
FUNDING: This study was sponsored by skinbetter science, a Dermatological Beauty Brand of L’Oréal USA Inc.
DISCLOSURES: Drs. Amini-Nik, Law, and Bhardwaj served as study investigators, and were employed full-time or part-time by SGS which received funding from the sponsor at the outset of the study. Upon study completion and submission of the final report, the authors conducted a post-hoc analysis and data evaluation independently and were not compensated, directly or indirectly, for this analysis or the drafting of the manuscript. Dr. Lynch works for L’Oréal USA Research & Innovation, and Ms. Nelson was an employee of skinbetter science, a Dermatological Beauty Brand of L’Oréal USA, Inc.
ABSTRACT: Objective. To evaluate the protective effects of 2 antioxidant products against UV light by assessing UV-induced erythema and skin damage biomarkers.
Methods: A randomized, controlled trial enrolled 15 healthy women aged 35 to 60 years. Participants’ lower backs were marked with 4 test sites (5 cm × 6 cm). Test products (4 mg/cm2) were applied daily for 4 days. Untreated/irradiated and naïve sites served as controls. After determining each participant’s minimal erythema dose (MED), skin was exposed to UV irradiation at 1× and 2× MED. Erythema, imaging, and skin damage were evaluated. Analysis of markers was determined from biopsies using histochemical imaging and scoring by a dermatopathologist (2× MED site).
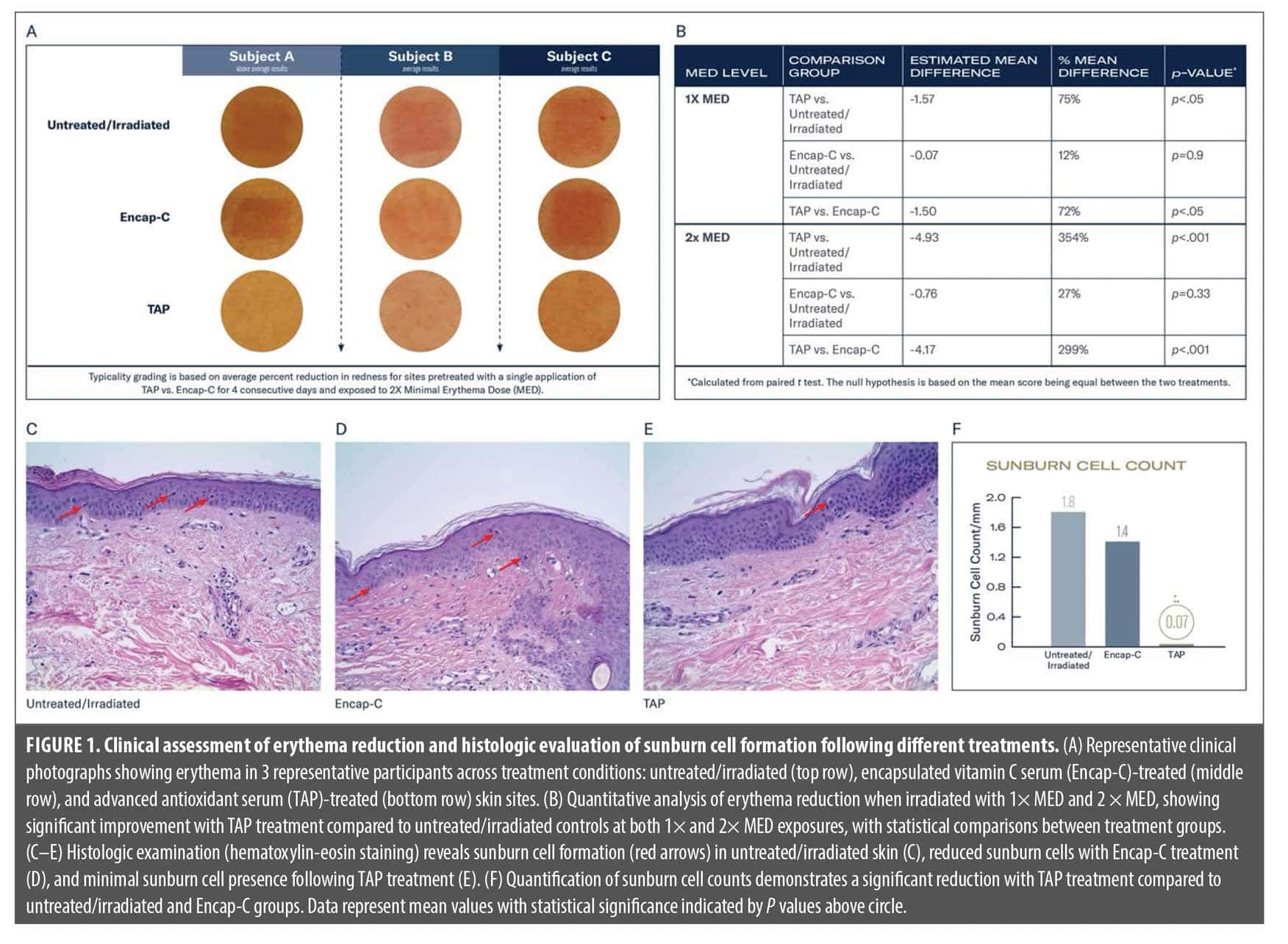
Results: Pretreatment with the advanced antioxidant serum (TAP) demonstrated 72% (1× MED, P=0.02) and 300% less erythema (2× MED, P<0.001) vs an encapsulated vitamin C serum (Encap-C) and 75% (1× MED, P=0.01) and 354% (2× MED, P<0.001) vs control. Pretreatment with TAP demonstrated significant protective effects vs Encap-C and control at 1× and 2× MED for matrix metalloproteinase 9 (P=0.004 and P=0.005, respectively), thymine dimers (both P<0.001), sunburn cell count (P=0.008 and P<0.001, respectively), p53 (P<0.001, both), CD68 (P=0.001 and P<0.001, respectively), and total lymphocyte count (both P<0.001). Encap-C did not demonstrate significant differences in erythema or biomarker levels vs control.
Limitations: This study was conducted in healthy participants (mean age: 49 years), with limited representation across Fitzpatrick skin types. Future studies should include a more diverse study population.
Conclusion: Pretreatment with TAP demonstrated significantly less UV-induced erythema and provided significantly greater cellular protection vs Encap-C serum and untreated/irradiated skin.
Keywords: Oxidative stress, topical antioxidants, skin biomarkers, MED, UV-induced erythema, photoprotection
Introduction
Skin aging occurs as a consequence of the cumulative generation of free radicals both intrinsically, through normal metabolic processes, and extrinsically, through exposure to UV radiation, pollution, ozone, cigarette smoke, and other environmental insults.1–6 Mitochondria are a primary source of the intrinsic production of free radicals. Aging gradually reduces the efficiency of mitochondria and accelerates mitochondrial damage and dysfunction, enabling increased generation of free radicals and the accumulation of cellular debris.7,8 The ability of skin to naturally defend itself against free radicals and reactive oxygen species (ROS) through its endogenous antioxidant defense system diminishes with age, leading to oxidative stress.1,4–6,9 This accelerates the aging process, impeding the ability of skin to function efficiently and causing structural, cellular, and molecular changes.10,11 Additionally, a reduction in the proliferation of basal cells results in epidermal thinning and decreased lipid levels in the stratum corneum.12,13 As a consequence, clinical manifestations of skin aging present as lines and wrinkles, dyschromia, sallowness, rough skin texture, skin laxity, and dehydration.1,5,14,15
Approximately 80% of free radical damage is attributed to UV-A and UV-B exposure.1–3 Given that inflammatory cells are often the first responders to skin insults such as UV radiation, enhancing their ability to respond early and fortifying cellular resilience has become a focal point in research aimed at mitigating age-related skin and systemic decline.16–18 Recent insights suggest that therapies targeting barrier improvement by modulating inflammatory signaling via topical products can attenuate cutaneous inflammation linked to aging. Sunscreens are critical for preventing photodamage and act by scattering, blocking, or absorbing UV radiation prior to the formation of free radicals. Topical antioxidants act in a complementary manner, augmenting the skin’s defenses by stabilizing and neutralizing free radicals and replenishing depleted antioxidant levels.9,11,19 Understanding how to fine-tune the inflammatory response and reinforce barrier function is critical to slowing inflammaging.
The American Academy of Dermatology notes that approximately 9,500 people are diagnosed with skin cancer every day in the United States.20 Despite the relative effectiveness of high-SPF sunscreens, their protective effects are reliant on proper and adequate use and only act on the top layers of skin. Topical antioxidants afford complementary benefits to sunscreens, bolstering defense against UV radiation by neutralizing free radicals and replenishing depleted antioxidant levels. Antioxidants each possess unique properties and capabilities, combining multiple types of antioxidants provides broad-based protection against a variety of ROS. Furthermore, distinct types of antioxidants work synergistically with one another to comprehensively support skin barrier against both intrinsic aging and extrinsic insults.1,21–24
Antioxidants are derived from both enzymatic and nonenzymatic sources, and each possesses unique properties to counteract free radical damage via prevention, interception, or repair.11,25 Enzymatic antioxidants, including superoxide dismutase (SOD), catalase, and ubiquinone, help to supplement skin’s natural defenses.11 Hydrophilic antioxidants, such as vitamin C and glutathione, protect the water-containing portions of cells, interior cell structures, and interstitial fluid.11 α-tocopherol and other lipophilic antioxidants aid in protection of the lipid-rich cellular membrane.22 Combining different types of antioxidants facilitates synergistic interaction and provides broad-based protection from the various types of ROS at all cellular levels of skin.3,9,22,24,26
A topical antioxidant serum comprised of a balanced ratio of 19 water-soluble, enzymatic, and lipid-soluble antioxidants (WEL) was purposefully constructed to provide broad cellular protection of the skin against a variety of ROS.27 Prior studies conducted with a serum containing WEL technology demonstrated its capacity to neutralize ROS (hydrogen peroxide) and protect against the damaging effects of UV-A and UV-B, ozone, blue light, and tobacco-induced oxidative stress and to visibly improve the appearance of photoaged skin.26–28
Targeting both intrinsic and extrinsic aging, an advanced antioxidant serum (TAP) combines WEL technology with topical allyl pyrroloquinoline quinone (PQQ), an efficient scavenger of intrinsically-generated ROS.29,30 PQQ is a potent micronutrient and free radical scavenger of superoxide and hydroxyl radicals, supporting cellular growth and differentiation, DNA repair, and mitochondrial biogenesis.29,30 Found in a variety of foods including spinach, green pepper, kiwi, carrots, and fava beans,31 PQQ has historically been delivered as a dietary supplement due to challenges encountered with topical formulation and subsequent instability and irritation. The addition of an allyl group enables topical delivery,7 supporting more efficient creation of mitochondrial energy and skin renewal and repair.
TAP combines water-soluble (ergothioneine, carnosine, and Buddleja officinalis flower extract), enzymatic (SOD and Arabidopsis thaliana extract) and lipid-soluble (coenzyme Q10, vitamins C and E, and licorice root extract) antioxidants in combination with topical allyl PQQ to comprehensively address both intrinsic and extrinsic skin aging. Previous research conducted with TAP in ex vivo skin tissue exposed to UV demonstrated significant expression of key markers associated with epidermal homeostasis and repair, recycling and removal, and antioxidative response vs untreated/irradiated skin.7 Significant reduction in enzymes associated with collagen degradation vs control occurred in tissues treated with TAP. A multicenter study evaluated twice-daily application of TAP in 44 patients, demonstrating improvements in the appearance of photoaged skin from baseline at 12 weeks.7
Herein, we describe a study evaluating TAP and its effects on UV-induced erythema and biomarkers associated with skin damage compared to a serum containing an encapsulated form of vitamin C (Encap-C).
Methods
A single-center, double-blinded, randomized controlled trial evaluated the protective effects of 2 antioxidant serums against UV-induced erythema and biomarkers associated with oxidative stress and photodamage. The trial was conducted after seeking protocol approval from an independent review board (Advarra Institutional Review Board, Columbia, MD) and according to Good Clinical Practice guidelines. The study enrolled 16 healthy female participants with Fitzpatrick skin type (FST) I to III who were aged 35 to 60 years and in good general health.
Participants who met criteria were eligible to enroll in the study if they were nonsmokers, their lower backs were free of markings or tattoos and uniform in texture and color, and they were able to provide informed consent. Participants were excluded from study participation if they had known allergies to skincare products or any of the ingredients in the study products; known sensitivity to sunscreen, lidocaine, or other anesthetics; a medical history of allergy or hypersensitivity or any serious reaction to a local antibiotic, antiseptic, or anesthetic; or were undergoing any treatment that could affect blood coagulation and hemostasis, such as the use of anticoagulant medications.
Participants were also excluded from participation if they had a medical need to avoid sunlight; had a history of photosensitivity disorders, phototoxicity, or a photo-allergy; or regularly used any medication with photosensitizing potential (including antihypertensives, acne medications, certain psychiatric medications, tricyclic antidepressants, antibiotics, antihistamines, aminolevulinic acid HCl, or psoralen). Participants who were breastfeeding, pregnant, or planning to become pregnant were excluded, as were participants with a personal or family history of skin cancer or any diagnosis of dysplastic nevi, who had developed an abnormal pigmentation response from a medical procedure, or had a history of dermatologic healing defects (such as hypertrophy or keloid scarring). Participants who had a history of any medical comorbidities, immunosuppressive and/or immune deficiency disorders, or chronic illnesses or current use of immunosuppressive or biologic medications were also excluded from participating in the study. Lastly, participants were excluded if they had any health condition and/or pre-existing or dormant dermatologic disease on the back or test area (including psoriatic arthritis, moderate to severe acne, acne conglobata, nodules, cysts, eczema, seborrheic dermatitis, severe excoriations) that the investigator deemed inappropriate for participation or had observable sunburn, suntan, scars, nevi, or previous UV radiation mark or other dermal conditions on the test area of the back that might influence the test results.
On Day 1, participants who met eligibility requirements had 4 test sites (5 cm × 6 cm) marked on their lower back, all at least 1 cm from the adjacent test site. Using predetermined randomization, each test site was designated as: (1) Pretreatment/Encap-C, (2) Pretreatment/TAP, (3) Untreated/Irradiated Control, and (4) Untreated/Unirradiated control (Naïve).
Over the course of 4 consecutive days, 4 mg/cm2 of the 2 test products were applied by clinic personnel to the assigned test site and rubbed into skin. On Day 2, a designated minimal erythema dose (MED) determination site on untreated skin (MEDu) was exposed to a series of UV bursts using a multiport solar simulator (Model Multiport 601-300-v2.5, Solar UV Simulator, Solar Light Col). Measurements were obtained on each participant’s MEDu test area using the Chroma Meter CR-400 (Konic Minolta) set to D65. Illuminant was used to determine the individual typology angle (ITA) characterizing skin clarity and pigmentation to calculate each participant’s estimated or anticipated MEDu. On Day 3, each participant returned to the clinic approximately 24 hours after MEDu irradiation to have their MEDu test area assessed for the level of erythema. The lowest dose of UV radiation producing an erythema score of 1 was selected as the initial MED for the participant. An erythema value (a*) of 1 indicated perceptible unambiguous erythema with defined borders filling >50% of the exposure site.
On Day 4, pretreated test sites and an untreated control site were irradiated at 1× and 2× of each participant’s individual MED, as previously determined. Each irradiation subsite measured approximately 1 cm2 and was exposed to UV-B/UV-A using a multiport solar simulator that complies with International Organization for Standardization and FDA guidelines. The simulator met regulatory requirements for spectral match, including relative cumulative erythemal effectiveness (RCEE) and adequate UV-A output, ensuring standardized and reproducible exposure across all test sites. The designated naïve site was not treated or irradiated. On Day 5, digital images were taken of each participant’s test sites and analyzed for a* using the handheld Canfield Twinflash system (Canfield Imaging Systems) and Canon EOS Rebel T7i camera with a 60-mm EF-S lens (Canon Corporation) under a cross-polarized filter. A 4-mm punch biopsy was performed to obtain full-thickness skin samples, including the epidermis and dermis. The biopsy was conducted under sterile conditions following local anesthesia. The circular tissue core was excised using a sterile punch tool and immediately collected for downstream histologic analysis. To promote proper healing and minimize scarring, the biopsy site was sutured using nonabsorbable stitches as appropriate. Postprocedural wound care instructions were provided, and suture removal was scheduled based on standard clinical timelines. The samples were fixed in formalin and embedded in paraffin before undergoing immunohistochemical analysis, quantified by a dermatopathologist blinded to the study, to evaluate the following: (1) thymine dimers, (2) p53, (3) sunburn cells, (4) matrix metalloproteinase 9 (MMP-9), (5) CD68 (a marker of inflammation), and (6) total lymphocyte count. Participants returned to the clinic for suture removal at least 10 days after biopsy collection. In addition, participants were questioned regarding any changes to their health, and all adverse events (AEs) were recorded.
Images of each test site were analyzed for a* using the macro version “MED_20130420” developed by SGS using Image Pro Plus v7 software (Media Cybernetics, Inc.). A circular area of interest was selected for each subsite within the test sites for the analysis. The a* component of the CIELAB color space (L*a*b*) was extracted from the images and used for quantitative analysis with higher values indicating more redness at the site. To account for baseline skin tone variation and potential treatment-related changes in skin color, 2 background correction methods were implemented:
Naïve-site adjustment: This approach involved subtracting the a* value of the untreated (naïve) site from each of the 3 test sites to correct for anatomical differences in baseline skin color.
Subsite adjustment: Since each test site contained only 2 small irradiated areas, a localized correction was also applied. The a* value from the center of each test site (a nonirradiated area) was subtracted from the corresponding irradiated subsites within that site. This method helps isolate localized treatment effects on skin color.
The background-corrected measurements were used in the statistical analysis.
An independent data committee performed data review and analyses. Statistical analyses of the study data and data management were conducted in accordance with the approved study protocol.
Results
Sixteen participants were enrolled and 15 completed the study, with a mean age of 49 years. One participant experienced an unrelated injury requiring medication and was excluded from the study for missing the final visit. Of the study population, 20% had FST I, 20% FST II, and 60% FST III.
Erythema response (a* values) following UV radiation. As illustrated in Figure 1A, representative images from 3 participants exposed to 2× MED, TAP-treated sites consistently exhibited markedly reduced redness, quantified in Panel 2. Pretreatment with TAP induced significantly lower levels of erythema vs sites pretreated with Encap-C: 72% (1× MED; P=0.05) and 299% (2× MED; P<0.001). Additionally, significantly lower levels of UV-induced erythema occurred on sites pretreated with TAP vs the untreated/irradiated control: 75% (1× MED; P=0.05) and 354% (2× MED; P<.001; Figure 1B). No significant reductions in UV-induced erythema were demonstrated on sites pretreated with Encap-C vs untreated/irradiated control.
Histologic evaluation: damaged cells and sunburn cell formation. Hematoxylin-eosin (H&E)-stained skin biopsies (Figure 1C–E) corroborated the erythema observations at the cellular level. Numerous sunburn cells (identified by pyknotic nuclei and eosinophilic cytoplasm) were present in both the untreated, irradiated skin (Figure 1C, red arrows) and the Encap-C–treated sites (Figure 1D). In contrast, TAP-treated skin (Figure 1E) exhibited only occasional sunburn cells and maintained normal epidermal architecture. Total sunburn cell counts were highest in the untreated/irradiated (1.8 cells/mm) and Encap-C (1.4 cells/mm) groups. The TAP-treated group exhibited significantly fewer total sunburn cells vs both comparators (0.07 cells/mm; P<0.05; Figure 1F).
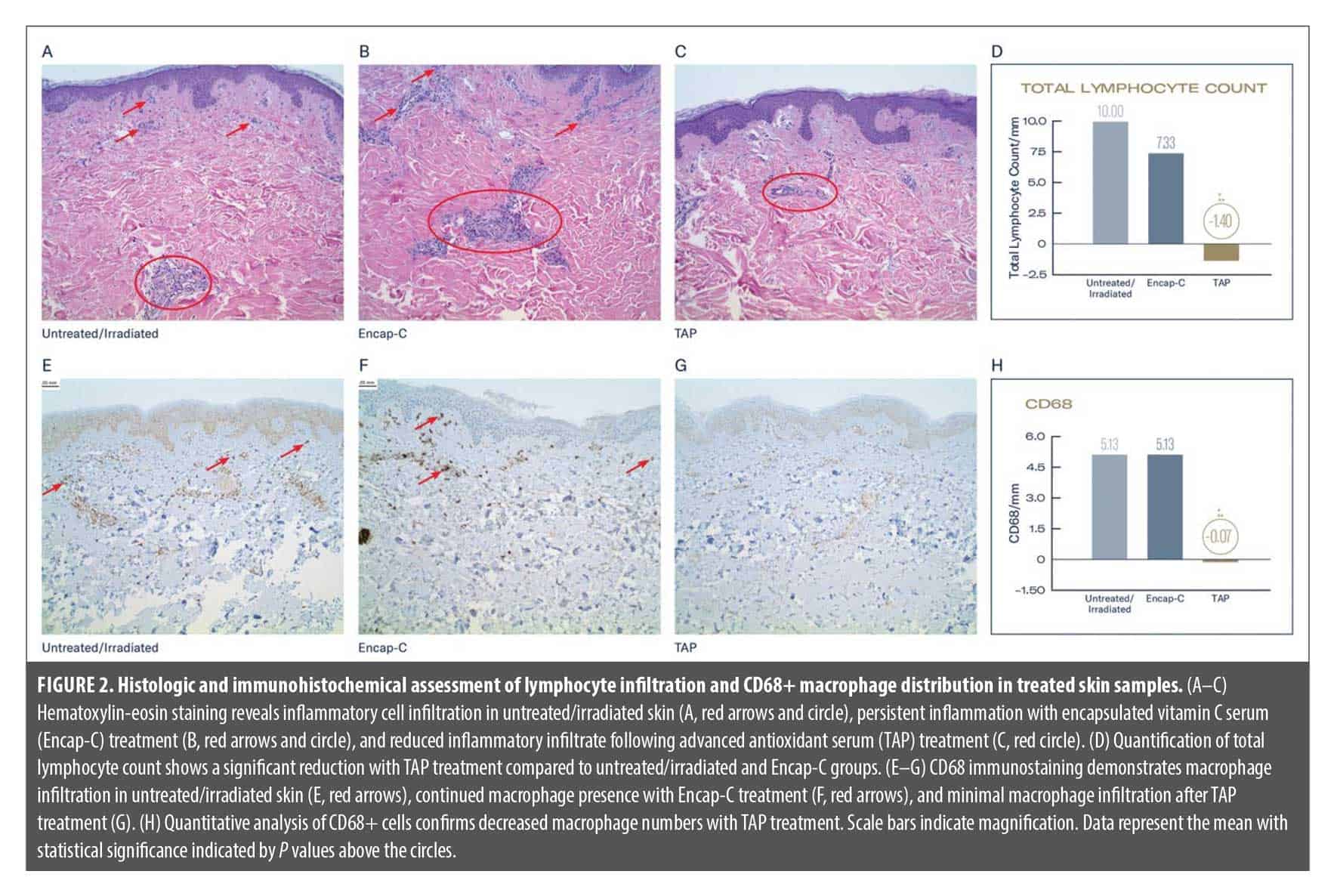
TAP preserves dermal immune homeostasis following UV irradiation. Illustrated in Figure 2, H&E staining revealed marked infiltration of lymphocytes in both untreated/irradiated skin (Figure 2A) and Encap-C–treated skin (Figure 2B), as indicated by perivascular and interstitial lymphocyte clusters and associated epidermal disruption. In contrast, TAP-treated skin (Figure 2C) exhibited substantially reduced lymphocytic presence, with preserved epidermal integrity and minimal inflammatory cell clustering. Quantitative analysis (Figure 2D) confirmed these histologic observations: total lymphocyte counts were highest in the untreated/irradiated group (10.00 cells/mm2), modestly reduced in the Encap-C group (7.33 cells/mm2), and significantly decreased in the TAP-treated group (1.40 cells/mm²; P<0.05 vs both controls).
To evaluate the skin’s innate immune response to UV-induced damage, the presence of CD68+ cell were evaluated. Immunohistochemical staining for CD68, a macrophage marker, further demonstrated the immunomodulatory effect of TAP. In the untreated/irradiated and Encap-C–treated groups (Figure 2E, F), CD68+ cells were frequently observed (arrows), particularly along the dermal-epidermal junction and within the upper dermis, indicating an active inflammatory response. In contrast, TAP-treated samples (Figure 2G) showed a dramatic reduction in CD68+ macrophage density. This observation was supported by quantification (Figure 2H), with both untreated and Encap-C groups showing CD68+ counts of 5.13 cells/mm2, while TAP-treated skin displayed a significantly lower macrophage presence (0.07 cells/mm2; P<0.05).
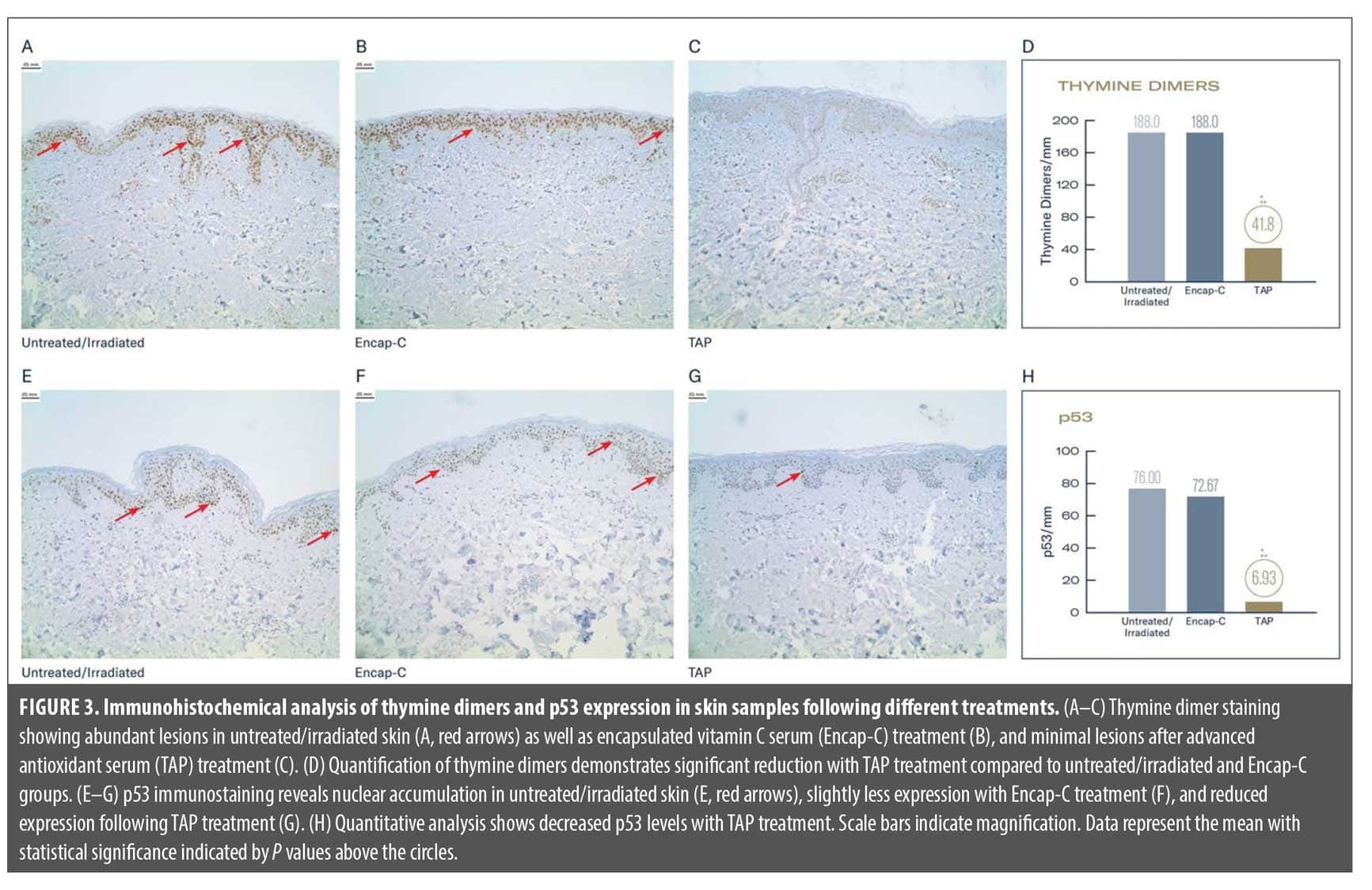
TAP reduces UV-induced DNA damage and p53 activation. Immunohistochemical analysis revealed pronounced differences in thymine dimer formation across treatment groups following UV exposure (Figure 3A–D). In the untreated/irradiated control group, keratinocyte nuclei in the epidermis showed widespread and intense brown nuclear staining, indicating extensive thymine dimer formation (Figure 3A, red arrows). Quantitative analysis confirmed high levels of damage in this group. Treatment with Encap-C resulted in minimal protection (Figure 3B), with thymine dimer staining and quantified levels closely resembling the untreated control.
Skin pretreated with TAP demonstrated a marked reduction in thymine dimer formation (Figure 3C). Immunostaining revealed only faint nuclear staining across the epidermis, indicating significantly less DNA photodamage. Quantification confirmed this finding, with a statistically robust decrease in thymine dimer levels compared to both untreated and Encap-C–treated sites (P<0.001; Figure 3D).
Parallel analysis of p53 showed a similar trend (Figure 3E–H). In untreated/irradiated skin, intense nuclear p53 staining was observed in basal and suprabasal keratinocytes (Figure 3E), reflecting activation of the DNA damage response pathway. Pretreatment with Encap-C offered only modest attenuation of p53 activation (Figure 3F), with levels still elevated. In contrast, TAP-treated skin exhibited a striking reduction in p53-positive nuclei (Figure 3G), indicating a blunted activation of the DNA damage signaling cascade. Quantitative data showed an average of 6.93 p53+ nuclei per μm in TAP-treated samples, representing a significant reduction compared to controls (P<0.001; Figure 3H).
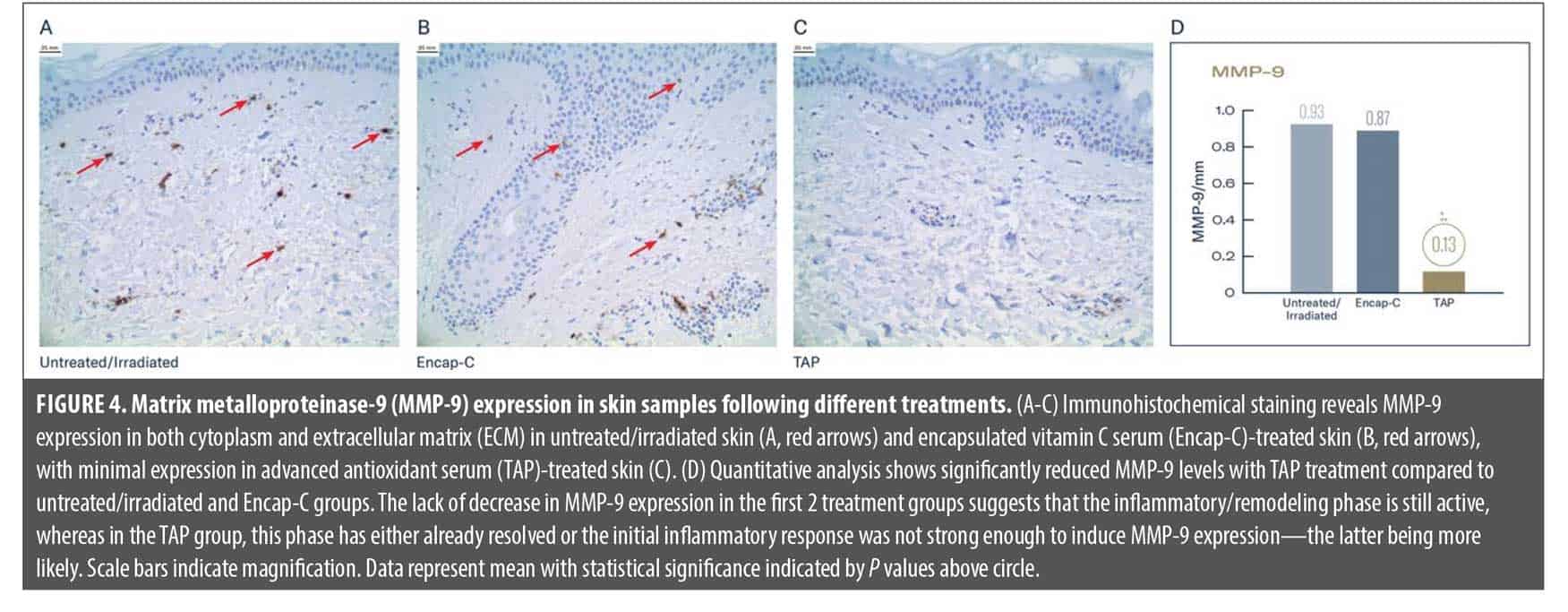
TAP attenuates UV-induced MMP-9 expression. MMP-9 immunohistochemical analysis revealed striking differences between treatment groups following UV irradiation (Figure 4A–D). In the untreated/irradiated control group, robust MMP-9 expression was observed throughout the dermis, evident in dermal fibroblasts and inflammatory cells (Figure 4A; red arrows). The widespread distribution of MMP-9–positive cells indicated an active inflammatory and tissue remodeling response to UV-induced damage, confirmed by quantification as conducted by blinded grader. Treatment with Encap-C demonstrated minimal protective effect against UV-induced MMP-9 upregulation (Figure 4B). The immunohistochemical staining pattern remained prominent, with numerous MMP-9-positive cells visible throughout the dermal compartment (Figure 4B, red arrows). Quantitative measurements showed only marginal reduction compared to untreated controls. In contrast, pretreatment with TAP provided substantial protection against UV-induced MMP-9 upregulation (Figure 4C). The immunohistochemical analysis revealed dramatically reduced MMP-9 staining throughout the dermis, with notably fewer positive cells and lighter overall staining intensity. The dermal architecture appeared better preserved, with less evidence of inflammatory cell infiltration and tissue disruption. Quantitative analysis confirmed this protective effect, showing MMP-9 expression reduced significantly compared to untreated controls (P<0.001; Figure 4D).
Discussion
The present study demonstrates that pretreatment with an advanced antioxidant serum (TAP) provided superior protection compared to a serum containing encapsulated vitamin C (Encap-C), offering comprehensive defense against UV-induced skin damage through multiple complementary mechanisms. The findings reveal that the multitargeted approach of TAP effectively prevents the cascade of molecular and cellular events that characterize acute UV-induced damage and contribute to chronic photoaging. Specifically, quantitative and visual assessments of erythema demonstrated that pretreatment with TAP significantly reduced UV-induced skin redness compared to both Encap-C and the untreated/irradiated control sites. These results suggest that TAP delivers superior protection against UV-induced erythema at both exposure levels.
Time and environmental exposures weaken the skin’s natural antioxidant defenses. Topical antioxidants work by neutralizing free radicals and counteracting cellular damage caused by intrinsic and extrinsic factors and aid in supplementing skin’s antioxidant defense mechanisms.11,25 As various enzymatic and nonenzymatic antioxidants exert unique benefits in counteracting free radical damage, topical formulations that combine enzymatic, lipophilic, and hydrophilic antioxidants offer a multipronged mechanism of action and broad protection against both intrinsic and extrinsic sources of free radicals.
In this study, pretreatment with TAP led to significantly less UV-induced erythema vs pretreatment with Encap-C and the untreated/irradiated control at 1× and 2× MED exposures in healthy participants. In addition, pretreatment with TAP afforded significantly greater cellular protection vs Encap-C and the untreated/irradiated control against UV-induced markers associated with skin damage at 2× MED, including thymine dimers, p53, sunburn cells, MMP-9, CD68, and total lymphocyte count.
Histologic and immunohistochemical analysis of skin biopsies demonstrated that pretreatment with TAP effectively mitigated UV-induced immune cell infiltration and inflammation in the superficial dermis. CD68+ cells are pivotal in both acute inflammation through clearance of apoptotic keratinocytes and cytokine amplification and in chronic photoaging via sustained low-grade inflammation, extracellular matrix degradation, and impaired repair.
Thymine dimers and p53 are formed and activated as a result of UV exposure and can lead to DNA mutations.32–35 Thymine dimers are a well-established marker of UV-induced DNA damage and are directly implicated in sunburn, premature skin aging, and photocarcinogenesis.33,36,37 Thymine dimers result from chemical changes in DNA owing to UV light exposure and can block DNA replication and transcription, impeding the ability of the cell to replicate its DNA. The marked reduction in thymine dimers suggests TAP can prevent DNA damage at the earliest stages of UV-induced injury.38,39 The parallel reduction in both thymine dimer formation and activation of p53, a key transcription factor activated in response to DNA damage,40 supports the conclusion that TAP prevents the initiation of UV-induced genotoxic events rather than merely modulating downstream signaling. This early intervention may limit chronic outcomes such as inflammation, apoptosis, and senescence that contribute to photoaging.41,42
Collectively, these results demonstrate that TAP offers superior protection compared to Encap-C by maintaining genomic integrity under UV stress. These findings underscore the potential of next-generation antioxidant systems for effective protection.
UV irradiation also causes mutations in p53, a tumor suppressor, impairing the cell cycle for DNA and, with substantial enough damage, inducing cellular apoptosis.25,43 Sunburn cells are keratinocytes impacted by UV-B radiation, appearing in the epidermis following acute UV damage. Sufficient exposure of sunburn cells to UV-B leads to apoptosis.44 The lower levels of sunburn cells demonstrated on sites pretreated with TAP suggest a protective effect following UV exposure.
MMP-9 is a type IV collagenase that is upregulated following UV exposure, leading to the breakdown of collagen and elastin.45 MMP-9 plays a dual role in skin homeostasis, serving essential functions in normal tissue remodeling and wound healing while becoming pathologically elevated following UV exposure, where it contributes to extracellular matrix degradation, inflammation, and the characteristic erythema and tissue damage associated with sunburn. The dramatic reduction in MMP-9 expression following TAP treatment suggests multiple protective mechanisms. First, the decreased MMP-9 levels indicate reduced extracellular matrix degradation, which helps maintain dermal structural integrity and prevents the breakdown of collagen and elastin fibers that contribute to photoaging. Second, lower MMP-9 expression reflects attenuated inflammatory responses, as this enzyme is both a mediator and marker of inflammation in UV-damaged skin.
The preservation of dermal architecture observed in TAP-treated samples, evidenced by the organized tissue structure and reduced inflammatory cell infiltration, supports the conclusion that pretreatment with TAP effectively prevents the cascade of molecular events that lead to tissue damage following UV exposure. This protection extends beyond immediate sunburn prevention to include preservation of the dermal matrix components essential for maintaining skin elasticity, strength, and youthful appearance. Together, these findings suggest that TAP provides substantial cellular protection against UV-induced erythema and oxidative damage and was superior to Encap-C.
Study Limitations and Future Directions
While this study provides compelling evidence for the UV-induced protective benefits of TAP, several limitations should be acknowledged. The study was conducted in healthy participants, with a mean age of 49 years and limited representation across all FST. Future studies should include more diverse populations, including individuals with different skin types, ages, and baseline skin conditions. Additionally, longer-term studies are needed to evaluate the cumulative benefits of regular antioxidant use on photoaging prevention and skin health maintenance.
The mechanistic insights provided by this study open several avenues for future research, including investigation of the specific molecular pathways through which different antioxidant combinations exert their protective effects, optimal formulation strategies for maximizing synergistic interactions, and the development of personalized antioxidant approaches based on individual skin characteristics and environmental exposure patterns.
Conclusion
Pretreatment with TAP demonstrated significant reductions in UV-induced erythema and biomarkers associated with oxidative skin damage vs Encap-C and untreated/irradiated skin. No significant reductions occurred in UV-induced erythema or biomarkers associated with oxidative damage following pretreatment with Encap-C. The comprehensive protective effects observed, including significant reductions in erythema, DNA damage, inflammatory cell infiltration, and matrix degradation, highlight the importance of multitargeted antioxidant protection strategies.
These results underscore the potential of next-generation antioxidant systems as effective photoprotective agents that complement traditional sunscreen use. As our understanding of skin aging mechanisms continues to evolve, particularly in the context of inflammaging and barrier dysfunction, the development of sophisticated antioxidant interventions offers promising approaches for maintaining skin health in an increasingly challenging environmental context.
References
- Allemann IB, Baumann L. Antioxidants used in skin care formulations. Skin Therapy Lett. 2008;13(7):5–9.
- Chen L, Hu JY, Wang SQ. The role of antioxidants in photoprotection: a critical review. J Am Acad Dermatol. 2012;67(5):1013–1024.
- Grether-Beck S, Marini A, Jaenicke T, Krutmann J. Effective photoprotection of human skin against infrared A radiation by topically applied antioxidants: results from a vehicle controlled, double-blind, randomized study. Photochem Photobiol. 2015;91(1):248–250.
- Liebel F, Kaur S, Ruvolo E, Kollias N, Southall MD. Irradiation of skin with visible light induces reactive oxygen species and matrix-degrading enzymes. J Invest Dermatol. 2012;132(7):1901–1907.
- Poljšak B, Dahmane R. Free radicals and extrinsic skin aging. Dermatol Res Pract. 2012;2012:135206.
- Stojilijkovic D, Pavlovic D, Arsic I. Oxidative stress, skin aging and antioxidant therapy. Scient J Fac Med Niš. 2014;31(4):207–217.
- Draelos ZD, McDaniel DH, Yeolin S, Pot S, Sotir O, Nelson DB. Evaluation of a new, advanced antioxidant containing topical allyl pyrrolyquinoline quinone: analysis of antioxidant properties and visible effects in subjects with facial photodamage. J Clin Aesthet Dermatol. 2023;16(4):53–59.
- Sarniak A, Lipińska J, Tytman K, Lipińska S. Endogenous mechanisms of reactive oxygen species (ROS) generation. Postepy Hig Med Dosw (Online). 2016;70(0):1150–1165.
- Papaccio F, D’Arino A, Caputo S, Bellei B. Focus on the contribution of oxidative stress in skin aging. Antioxidants (Basel). 2022;11(6):1121.
- Franco AC, Aveleira C, Cavadas C. Skin senescence: mechanisms and impact on whole-body aging. Trends Mol Med. 2022;28(2):97–109.
- Michalak M. Plant-derived antioxidants: significance in skin health and the ageing process. Int J Mol Sci. 2022;23(2):585.
- Farage MA, Miller KW, Elsner P, Maibach HI. Intrinsic and extrinsic factors in skin ageing: a review. Int J Cosmet Sci. 2008;30(2):87–95.
- Zouboulis CC, Makrantonaki E, Mikolakis G. When the skin is in the center of interest: an aging issue. Clin Dermatol. 2019;37(4):296–305.
- Hozier AM, Athar M, Elmets CA. The other end of the rainbow: infrared and skin. J Invest Dermatol. 2010;130(6):1496–1499.
- Sander CS, Chang H, Salzmann S, et al. Photoaging is associated with protein oxidation in human skin in vivo. J Investig Dermatol. 2002;118(4):618–625.
- Amini-Nik S, Abdullahi A, Vinaik R, et al. Aging impairs the cellular interplay between myeloid cells and mesenchymal cells during skin healing in mice. Aging Dis. 2022;13(2):540–551.
- Franceschi RT, Wang D, Krebsbach PH, Rutherford RB. Gene therapy for bone formation: in vitro and in vivo osteogenic activity of an adenovirus expressing BMP7. J Cell Biochem. 2000;78(3):476–486.
- Weyand CM, Goronzy JJ. Aging of the immune system. Mechanisms and therapeutic targets. Ann Am Thorac Soc. 2016;13 Suppl 5(Suppl 5):S422–S428.
- Burke KE. Protection from environmental skin damage with topical antioxidants. Clin Pharmacol Ther. 2019;105(1):36–38.
- American Academy of Dermatology. Skin cancer. Accessed 22 Jan 2025. https://www.aad.org/media/stats-skin-cancer
- Abla MJ, Banga AK. Quantification of skin penetration of antioxidants of varying lipophilicity. Int J Cosmet Sci. 2013;35(1):19–26.
- Alonso C, Rubio L, Touriño S, et al. Antioxidative effects and percutaneous absorption of five polyphenols. Free Radic Biol Med. 2014;75:149–155.
- Pandel R, Poljšak B, Godic A, Dahmane R. Skin photoaging and the role of antioxidants in its prevention. ISRN Dermatology. 2013;2013:930164.
- Rahman K. Studies on free radicals, antioxidants, and co-factors. Clin Interv Aging. 2007;2(2):219–236.
- Rinnerthaler M, Bischof J, Streubel MK, Trost A, Richter K. Oxidative stress in aging human skin. Biomolecules. 2015;5(2):545–589.
- Wortzman M, Nelson D. A comprehensive topical antioxidant inhibits oxidative stress induced by blue light exposure and cigarette smoke in human skin tissue. J Cosmet Dermatol. 2021;20(4):1160–1165.
- McDaniel DH, Waugh JM, Jiang LI, et al. Evaluation of the antioxidant capacity and protective effects of a comprehensive topical antioxidant containing water-soluble, enzymatic, and lipid-soluble antioxidants. J Clin Aesthet Dermatol. 2019;12(4):46–53.
- Pecorelli A, McDaniel DH, Wortzman M, Nelson DB. Protective effects of a comprehensive topical antioxidant against ozone-induced damage in a reconstructed human skin model. Arch Dermatol Res. 2021;313(3):139–146.
- Jonscher KR, Chowanadisai W, Rucker RB. Pyrroloquinoline-quinone is more than an antioxidant: a vitamin-like accessory factor important in health and disease prevention. Biomolecules. 2021;11(10):1441.
- Misra H-S, Rajpurohit YS, Khairnar NP. Pyrroloquinoline-quinone and its versatile role in biological processes. J Biosci. 2012;37(2):313–325.
- Chowanadisai W, Bauerly KA, Tchaparian E, Wong A, Cortopassi GA, Rucker RB. Pyrroloquinoline quinone stimulates mitochondrial biogenesis through cAMP response element-binding protein phosphorylation and increased PGC-1 α expression. J Biol Chem. 2010;285(1):142–152.
- Kappes UP, Luo D, Potter M, Schulmeister K, Rünger TM. Short- and long-wave UV light (UVB and UVA) induce similar mutations in human skin cells. J Invest Dermatol. 2006;126(3):667–675.
- Mouret S, Baudouin C, Charveron M, Favier A, Cadet J, Douki T. Cyclobutane pyrimidine dimers are predominant DNA lesions in whole human skin exposed to UVA radiation. Proc Natl Acad Sci U S A. 2006;103(37):13765–13770.
- Chao C, Saito S, Anderson CW, Appella E, Xu Y. Phosphorylation of murine p53 at ser-18 regulates the p53 responses to DNA damage. Proc Natl Acad Sci U S A. 2000;97(22):11936–11941.
- Méplan C, Richard MJ, Hainaut P. Redox signaling and transition metals in the control of the p53 pathway. Biochem Pharmacol. 2000;59(1):25–33
- Brash DE. UV signature mutations. Photochem Photobiol. 2015;91(1):15–26.
- Pfeifer GP, Besaratinia A. UV wavelength-dependent DNA damage and human non-melanoma and melanoma skin cancer. Photochem Photobiol Sci. 2012;11(1):90–97.
- D’Orazio J, Jarrett S, Amaro-Ortiz A, Scott T. UV radiation and the skin. Int J Mol Sci. 2013;14(6):12222–12248.
- Gonzalez Maglio DH, Paz ML, Ferrari A, et al. Alterations in skin immune response throughout chronic UVB irradiation– skin cancer development and prevention by naproxen. Photochem Photobiol. 2010;86(1):146–152.
- Levine AJ. p53, the cellular gatekeeper for growth and division. Cell. 1997;88(3):323–331.
- Krutmann J, Schroeder P. Role of mitochondria in photoaging of human skin: the defective powerhouse model. J Investig Dermatol Symp Proc. 2009;14(1):44–49.
- Campisi J. The role of cellular senescence in skin aging. J Investig Dermatol Symp Proc. 1998;3(1):1–5.
- Rivlin N, Brosh R, Oren M, Rotter V. Mutations in the p53 tumor suppressor gene. Genes Cancer. 2011;2(4):466–474.
- Van Laethem A, Claerhout S, Garmyn M, Agostinis P. The sunburn cell: regulation of death and survival of the keratinocyte. Internat J Biochem Cell Biol. 2005;37(8):1547–1553.
- Van Doren SR. Matrix metalloproteinase interactions with collagen and elastin. Matrix Biol. 2015;44-46:224–2311.
