J Clin Aesthet Dermatol. 2026;19(5):43–49.
by Lauren S. Jones, DO; Emily McEldrew, DO; and Cynthia L. Bartus, MD
Dr. Jones is with the Dermatology Residency Program at Lehigh Valley Health Network, Allentown, Pennsylvania. Dr. McEldrew is with Advanced Dermatology Associates, Allentown, Pennsylvania. Dr. Bartus is with Macaione and Papa Dermatology, Voorhees Township, New Jersey.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no relevant conflicts of interest.
ABSTRACT: OBJECTIVE: To investigate the association between skin bleaching agents and the development of cutaneous malignancy. METHODS: A scoping literature search was conducted in the PubMed and Google Scholar databases to identify studies and reports that included both skin depigmentation practices and development of cutaneous malignancy. Articles published in English and French were considered for review. RESULTS: To date, reports are limited to a relatively small number of case reports in Africa. The most common skin bleaching agents used were topical clobetasol and hydroquinone with a duration of at least 15 years. Most patients had a reported skin phenotype of Fitzpatrick VI, and the most common skin cancer reported in patients was squamous cell carcinoma (SCC). The majority of these lesions were large, invasive, and/or ulcerated lesions in chronically sun-exposed areas. The most common site of metastasis was the lungs, and the mortality rate was high. LIMITATIONS: This is a literature review; information included is limited to what is available in the literature. CONCLUSION: Skin bleaching agents have a powerful presence among a broad population, yet the consequences of their use in skin of color are poorly understood and potentially dangerous. Based on the available literature, there is potential for skin bleaching practices to be implicated as the causative agent for cutaneous SCC, especially in darker skin phenotypes. Therefore, there is a growing need for dermatologists to address this topic as a major public health concern with high morbidity and potential mortality to prompt further investigation. Keywords: Skin bleaching, corticosteroid, hydroquinone, exogenous ochronosis, squamous cell carcinoma
Introduction
Skin lightening is practiced globally, with the highest rates found in sub-Saharan Africa, South Asia, the Middle East, and the Americas.1–3 There are a variety of skin lightening modalities including topical, oral, and/or systemic agents. The most common active ingredients in topical products include corticosteroids, inorganic mercury, and hydroquinone.1,2,4,5 Skin bleaching agents have a variety of methods to reduce pigmentation but primarily function by disrupting tyrosinase and thereby inhibiting melanogenesis.6–9 In most cases, these agents must be applied consistently to achieve the desired lightening effects, as pigmentation returns after cessation of use.10 Depending on the lightening agent used, chronic application may lead to local and systemic adverse effects including striae, atrophy, infections (viral, bacterial, fungal), exogenous ochronosis, hypercortisolism, nephropathy, cataracts, dermatitis, diabetes, hypertension, impaired wound healing, and trimethylaminuria (“fish odor syndrome”).3–5,9 Given that some agents function by inhibiting melanogenesis and act as immunosuppressants, there is growing concern among dermatologists and public health experts that long-term use of bleaching agents may lead to the development of cutaneous malignancies, specifically squamous cell carcinoma (SCC).11–17 Typically, skin cancers arise more frequently in patients who are older, have a history of extensive UV exposure, are classified as lighter Fitzpatrick skin types (I-II), and/or are immunosuppressed.18,19 The proposed mechanism of developing SCC in the setting of skin depigmentation has yet to be elucidated, but there are 4 main factors suggested: UV radiation, the lack of photoprotection resulting from inhibition of melanogenesis, immunosuppression due to corticosteroids, and the carcinogenic nature of hydroquinone.11–17 This literature review addresses the mechanism of skin depigmentation and investigates the suggested association of skin depigmentation agents with the development of cutaneous malignancy currently reported in the literature. A scoping literature search was conducted to identify studies and reports that included both skin depigmentation practices and development of cutaneous malignancy. Articles published in English and French were considered for review.
Overview Of Skin Depigmentation
Skin bleaching is also known as skin depigmentation, skin lightening, voluntary depigmentation, and skin whitening.1,3 It is the practice of changing a natural skin tone to a lighter one through the application or ingestion of exogenous substances.3 Skin bleaching is an incredibly lucrative industry with a global market. A 2022 world market report estimated the market value of the skin bleaching industry to be $8.8 billion, with the expectation that it will reach $12 billion by 2026.20 Its practice predates modern dermatology. Historically, ancient methods of skin depigmentation and skin lightening were employed to change a complexion in the hopes of gaining economic and social benefits.21–23 Ancient skin lightening methods often employed toxic and caustic substances such as white lead, mercury, hydroquinone, and arsenic wafers to lighten the skin.22 These practices resulted in numerous adverse effects including cancer, disfigurement, ochronosis, poisoning, and death. Despite many serious adverse effects, many of these agents are still in use today, especially in Asian and African countries.3,22 In the United States (US), skin bleaching products containing mercury and topical corticosteroids have been found to be readily available in pharmacies and beauty stores in major cities.24 Dermatologists across the globe encounter patients who apply bleaching creams for various conditions such as focal hyperpigmentation and melasma.3–5,24 Therefore, it is imperative that we recognize the implications of chronic and widespread skin bleaching.
The practice of skin depigmentation is most widespread in Asian and African countries and has a female predominance ranging from 25% to 75%.1,2,5,25,26 A meta-analysis of the global prevalence and correlates of skin bleaching suggests the global lifetime prevalence of skin bleaching is nearly 30%, with 43.8% female prevalence and 35.2% male prevalence.2 Skin bleaching is not limited to 1 geographic, social, or ethnic group; its practice spans all socioeconomic categories and education levels.1,2 However, multiple studies have shown certain factors are associated with an increased lifetime likelihood of skin bleaching, including female sex, younger age (<30 years), urban/semi-urban residence, depressive symptoms, Fitzpatrick skin phenotypes IV-VI, and lower-to middle-income levels.1,2,25 Additionally, studies have shown that despite awareness of the numerous dangers associated with skin bleaching, individuals will begin or continue use of bleaching products for social benefits associated with lighter skin tones.1,25,26 This illustrates the complexity of skin bleaching and the need to address the societal pressures that may influence individuals to initiate this potentially dangerous practice.
Overview of Skin Bleaching Agents
Skin lightening agents are available to consumers in numerous topical, oral, and intravenous formulations. These products are often marketed as “skin brighteners” or “skin lighteners’’ as opposed to “skin bleaching” creams to avoid scrutiny from consumers and regulators.1–3,24 There are several mechanisms used to bleach the skin, including inhibition of melanogenesis, melanocyte destruction, inhibition of the conversion of tyrosine to dihydroxyphenylalanine (DOPA), and inhibition of tyrosinase.10,22,27 Topical bleaching agents include zinc oxide, inorganic mercury, corticosteroids, azelaic acid, hydroquinone, iron oxide, niacinamide, and topical vitamin A derivatives.3,10,27 The most frequently used agents are topical corticosteroids, inorganic mercury (also known as mercury salts), and hydroquinone.2,5,28 Combination therapies of topical bleaching agents, including hydroquinone, topical corticosteroids, and inorganic mercury, have gained popularity because dual or triple therapy agents, as opposed to monotherapy agents, are believed to reduce toxicity and improve effectiveness.28 Common combination therapies include triple therapy (hydroquinone 4%, tretinoin 0.05%, and fluocinolone acetonide 0.01%), which is approved by the US Food and Drug Administration for the treatment of melasma, and dual therapy options such as fluocinolone and retinoic acid, hydroquinone and retinoic acid, and hydroquinone and fluocinolone acetonide.28 Studies in patients with melasma and other pigmentary disorders have shown that triple or dual therapies can have improved efficacy, safety, and patient satisfaction.29–31
Topical corticosteroids. The mechanism of skin lightening effect of topical corticosteroids is poorly understood. It is theorized to lighten the skin via local vasoconstriction and by decreasing the number of melanocytes and production of pro-opiomelanocortin, which is the precursor to α-melanocyte-stimulating hormone.9,32 Superpotent topical corticosteroids are most commonly used in bleaching agents and include clobetasol propionate, fluocinonide, and betamethasone dipropionate.4,9 Adverse effects of corticosteroids include delayed wound healing and systemic absorption leading to adrenal insufficiency, hypertension, and diabetes.4,5,9,32 Additional cutaneous adverse effects related to topical corticosteroids include cutaneous infections (cellulitis, dermatophytosis), acne, striae, telangiectasia, and skin atrophy, with the latter 3 being irreversible.5,9,32
Mercury. Mercury is one of the most dangerous and toxic agents used to bleach the skin. The mechanism of action of mercury to lighten the skin is unclear, but there are 2 proposed mechanisms reported in the literature. The first suggests that mercury effectively replaces the copper ion required for tyrosinase, thereby inhibiting melanin production.7 The second postulates that mercury chloride inhibits tyrosinase activity in an irreversible noncompetitive manner by binding to histidine residues in the catalytic center of tyrosinase, inducing a conformational change.33
Adverse effects of inorganic mercury are numerous and may be acute or chronic. They include gingivitis, gastrointestinal pain, neurologic disturbance, cognitive impairment, muscle weakness, nephrotoxicity, and psychiatric disorders.9,26,34 Dermatologic adverse effects include contact dermatitis, gray-blue discoloration, erythroderma, purpura, hyperpigmentation, and nail dyspigmentation.24,26,28,34 Due to its well-established toxicity, mercury is banned by many countries. However, due to its widespread availability and low cost, it is still found at dangerous levels in readily available products, particularly those imported from abroad, at beauty-supply stores and pharmacies in the US and across the world.3,24,26,34,35
Hydroquinone. Hydroquinone is a ubiquitous bleaching agent due to its effectiveness and widespread availability. One study found that US-based participants most commonly used hydroquinone creams (59.1%), followed by a triple combination cream containing fluocinolone acetonide, hydroquinone, and tretinoin (16.3%).36 A meta-analysis and meta-regression analysis of the global (Africa, Asia, and Middle East) prevalence of skin bleaching practices revealed that hydroquinone was the second most common agent used (47.5%), behind topical corticosteroids (51.8%).2
Hydroquinone can be used topically or ingested orally to produce skin depigmentation.7,28 Its use began in the early 20th century as a skin protective agent and was used topically as a sunscreen in the 1950s.8,28 Hydroquinone lightens the skin by blocking the conversion of tyrosine to DOPA, thereby inhibiting tyrosinase and melanogenesis.8,37 Hydroquinone has also been found to be directly toxic to melanocytes through generation of free radicals and subsequent damage to melanosomes.37 As with other skin bleaching agents, the adverse effects of hydroquinone can be classified as acute or chronic. Adverse effects are especially common with products containing greater than 2% hydroquinone.4,9 Acute adverse effects include irritant contact dermatitis, allergic contact dermatitis, postinflammatory hyperpigmentation, and hypopigmentation.38–40 The most common chronic adverse effects of hydroquinone include exogenous ochronosis (EO), trimethylaminuria, fetal growth restriction, peripheral neuropathy, decreased skin elasticity, ophthalmic damage, patchy depigmentation, and nail discoloration.4,5,9,26
EO, the most common chronic adverse effect, is challenging to treat and has been observed after as little as 3 months of topical hydroquinone use.9,41,42 It is characterized clinically by areas of erythema; asymptomatic hyperpigmentation; gray-blue colloid milia and pigmented nodules and papules; and poikiloderma on sun-exposed areas of the skin, especially the face, neck, shoulders, and upper back.9,41,43 The prevailing theory of the role of hydroquinone in EO is its ability to inhibit homogentisic acid oxidase, resulting in buildup of homogentisic acid.44 This accumulation of homogentisic acid leads to polymerization and formation of ochre pigments in the papillary dermis.44 It has also been suggested that hydroquinone directly damages elastic fibers in addition to UV exposure, leading to abnormal fiber aggregates and creation of pigmented foci in the papillary dermis.42
In summary, hydroquinone is associated with numerous acute and chronic adverse effects, which are difficult to treat and may result in cutaneous hyperpigmentation and disfigurement.
Systemic therapies. Systemic therapies are becoming more common for skin lightening purposes due to ease of use and fewer adverse effects.10,45,46 Their investigation and use are more established in Asian countries. Most studies have been performed in the context of treating melasma and hyperpigmentation in Asian women and skin phototypes III to V.10 Systemic agents that are currently used, especially to treat melasma, include oral tranexamic acid (TXA), Polypodium leucotomos extract (PLE), carotenoids, melatonin, glutathione, and procyanidin.45 TXA is the most well-established treatment for melasma and postinflammatory hyperpigmentation.10,45 It reduces pigmentation by competitively inhibiting plasminogen activator at its lysine-binding sites, subsequently suppressing melanin synthesis.45,47 In general, systemic skin lightening treatments are considered safe and appear to have less reported adverse effects compared to the topical agents.10,45,46 To date, there are no cases of SCC associated with the aforementioned systemic skin lightening agents reported in the literature. While it remains possible that systemic agents are a safer alternative for skin lightening, more research is needed to understand their safety, efficacy, and long-term effects.
The reported association between skin-lightening agents and cutaneous malignancy in the literature
Cutaneous malignancies, especially nonmelanoma skin cancers (NMSC), are generally less common in skin of color as compared to people with lighter skin phenotypes.18,19 In the US, the annual age-adjusted incidence rate for NMSC in White persons has been reported to be 232.6 per 100,000 population, in contrast to 3.4 per 100,000 population in Black persons, suggesting that Black individuals are nearly 70 times less likely to develop NMSC than White individuals.18,19 Despite the lower incidence of NMSC in persons of color, there is an increased morbidity and mortality as compared to the White population.48,49 This discrepancy may be attributed to decreased preventative measures and later detection.18,49,50 In contrast to White patients, numerous studies have shown that SCC in Black patients tends to develop in less sun-exposed areas such as the anogenital region, lower legs, and the feet, suggesting that UV damage may be less important for the development of SCC in Black people.18,50 In the current literature, most Black patients who have developed SCC are middle-aged and male (slight predominance) and have SCC lesions that are associated with inherited and/or acquired preneoplastic dermatoses including albinism, xeroderma pigmentosum, chronic discoid lupus erythematosus, tropical ulcer, sites of human papillomavirus infection, and scarring burns.18,50,51 These findings suggest that the development of SCC in darker skin types may be less related to UV damage and more likely related to disturbances to skin structure and function such as viral infection, inflammation, and/or scarring from chemical and thermal burns.
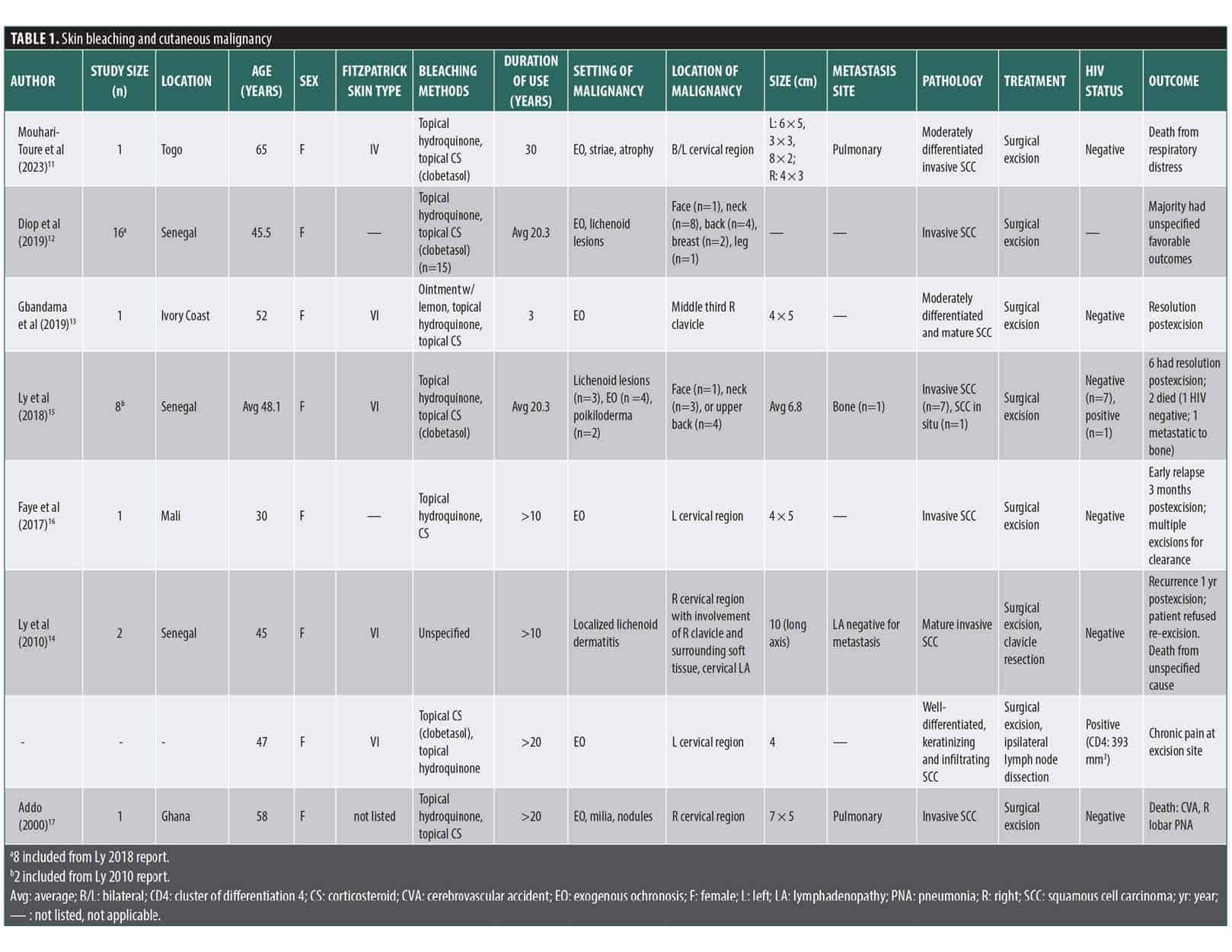
Reports of skin bleaching agents as a cause for cutaneous malignancy are limited to a small number of case reports and exclusively involve patients from African countries including the Ivory Coast, Ghana, Mali, Togo, and Senegal (Table 1). SCC and SCC in situ were the only malignancies reported in association with use of skin bleaching agents. Regarding the demographics of reported patients, the majority were younger than 50 years, female, and HIV-negative and had skin phenotype VI and a duration of skin lightening product use >10 years prior to SCC development. Nearly all the reported cases involved the use of agents containing topical corticosteroids and hydroquinone, with no use of systemic agents reported. The most common topical corticosteroid used was clobetasol. All reported cases of SCC developed in UV-exposed areas, and most cases were found within regions of EO and/or atrophic skin. Areas with lichenoid dermatitis were a less common site for SCC development. The most common location for skin lesion development was the neck/cervical region and upper back. The lesions were typically large, exophytic, and ulcerative, with sizes ranging from 3 to 12 cm on the long axis. The prevailing treatment modality was surgical excision. If the cancer was widely invasive, resection of involved surrounding soft tissue or bone was employed as well as repeat excision for recurrent disease. Notably, multiple cases involved local recurrence and/or metastasis. There was 1 case of bony metastasis and 2 cases of pulmonary metastasis. Of the 20 unique cases reported, 3 patients died due to complications associated with invasive and metastatic SCC, equating to a mortality rate of nearly 15%. For nearly all cases, there were no other precancerous lesions outside of the regions where bleaching products were used.
Despite the widespread practice of skin bleaching globally, the reported association of skin cancer is limited to a small number of case reports in Africa.
Discussion
Although SCC is rare in darker skin types when compared to lighter skin phenotypes, it is the most common cutaneous malignancy in Black and Indian people.18,52 Of the reported cases, the patients who developed SCC are younger, lack preneoplastic dermatoses, and have darker skin phenotypes than conventional patients who develop SCC without the use of skin bleaching agents. This could suggest a possible association between skin bleaching agents and SCC development. The development of SCC in the context of skin bleaching appears to be secondary to a complex interaction between UV exposure, melanocyte inhibition and/or destruction, increased susceptibility to UV damage, irritant contact dermatitis, EO, and skin atrophy. These factors may precipitate skin irritation and inflammation, resulting in malignant transformation due to increased UV susceptibility and damage in otherwise low-risk individuals.
Both UV-A and UV-B cause DNA photodamage; however, UV-B has been shown to be more cytotoxic and damaging to the skin than UV-A due to its smaller wavelength.53,54 On average, 5 times as much UV light reaches the upper dermis of White skin as compared to Black skin.55 The main location of UV filtration in White skin is the stratum corneum compared to the Malpighian layers in Black skin, the deepest layers of the epidermis.55 Additionally, Black skin has been shown to remove nearly 2 times as much UV-B than White skin, and the predicted UV protection factor of Black skin is 4-fold that of White skin.55 In addition to the deeper location of UV filtration, the greater photoprotection in Black skin can partially be explained by increased melanocyte size, activity, and melanin production.55–58 However, the previously proposed role of melanin as the sole protective agent from skin cancer in darker-phenotype individuals has been challenged, and the pathophysiology of photoprotection and cancer development may be more complex than previously thought.59,60 Other factors that may influence the risk of UV-related damage include the production of reactive oxidative species, UV-induced immunosuppression, and DNA damage.60 In darker skin types, UV-induced DNA damage is more superficial, in the upper dermis, as compared to lighter skin types, where the damage is throughout the dermis.60 Regardless of the exact photoprotective mechanism, there is a clear association between reduced skin pigment and higher rates of cutaneous malignancy.18,19 Therefore, skin bleaching agents may increase the risk of SCC due to reduction in endogenous photoprotection and an increased susceptibility to UV damage, in addition to other factors such as immunosuppression from corticosteroids and chemical irritation.
Corticosteroids are one of the most common skin lightening agents used across the globe. Superpotent corticosteroids are becoming increasingly prevalent in bleaching products and causing numerous local and systemic adverse effects.4,9 Topical clobetasol was the most common corticosteroid used in the reported cases. It was typically used continuously for extended durations of time, as long as 2 to 3 decades in a majority of the cases. In addition to decreasing tyrosinase and melanocyte activity, corticosteroids also act as immunosuppressants.9,61 It is well established that patients who use systemic corticosteroids and other immunosuppressants are at a higher risk for cutaneous malignancy than age-matched controls.62 Specifically, the risk for SCC is increased 65 to 200 times for organ transplant recipients.62 Chronic systemic immunosuppression reduces the ability of the adaptive immune system to address the consequences of DNA photodamage such as atypical cell growth, and this leads to a higher incidence of skin cancers and aggressive lesions.63 Despite the association between systemic immunosuppressants such as oral corticosteroids use and skin cancer, the association with topical corticosteroids has not been shown. A systematic review published in 2018 found no evidence to suggest an association with skin cancer in patients with long-term use of topical corticosteroids.64 However, as seen with the reported cases, topical corticosteroids are rarely used alone in bleaching products. This suggests that corticosteroids alone may not be carcinogenic but may increase the risk of SCC when used in conjunction with other cutaneous depigmentation agents.
The safety profile and possible carcinogenic effects of hydroquinone remain unclear. Multiple studies have shown it to be mutagenic in high concentrations in cell and animal models, but evidence is lacking in human models.65 Hydroquinone is a major metabolite of the well-known carcinogen benzene, and this association may indicate carcinogenic potential.65 However, there is inadequate evidence to determine if hydroquinone is carcinogenic.38,65
In the context of skin bleaching, EO almost always develops with the use of topical hydroquinone, especially long-term use. SCC arose in the setting of EO in nearly all cases of SCC reported in patients with a history of skin bleaching.11–17 The pathophysiology of SCC development in foci of EO is unclear and has not been clearly elucidated in the literature. There is uncertainty on whether EO is a precursor for malignancy or if hydroquinone has a more direct role, especially in combination with UV damage, topical corticosteroids, and caustic agents such as inorganic mercury.4,9,14,15 There are no studies that have established EO as a preneoplastic dermatosis or a precursor to malignancy, and this needs to be investigated further. In relation to cutaneous complications of bleaching agents, use of hydroquinone has been associated with lupus-like reactions.4 The ability of hydroquinone to potentiate a preneoplastic-like dermatitis and the association of EO with cutaneous malignancy illustrates a possible association between hydroquinone use and SCC that warrants further investigation.
The mortality rate of cutaneous SCC has been estimated to be 2.8%.66 The mortality rate of SCC in patients who perform skin bleaching is approximately 15%. This suggests a nearly 5-fold increase in mortality. Two of the 3 patients reportedly died due to complications of metastatic SCC, most commonly respiratory failure in the context of pulmonary metastasis. Reasons for increased mortality in this patient population are unclear, but most of these patients waited several years to seek care and access to care was limited in certain settings. Additionally, some of the cases were reported to have tried natural remedies (eg, poultices, herbal treatments) before seeking specialized care.
While millions of people use skin bleaching products across the globe, the reported association of bleaching practices and SCC is quite rare and limited to a small number of case reports. Given the many systemic and cutaneous adverse effects of skin bleaching agents, there may be an association between the agents themselves and the development of SCC, but more research is needed to understand the significance of this relationship. Continued investigation and monitoring for the risk of developing other cutaneous malignancies, including cutaneous lymphomas, melanomas, and other NMSCs, in the users of skin-bleaching agents is imperative. Dermatologists and other physicians are urged to educate and closely follow patients with a history of skin bleaching.
Conclusion
Skin lightening is a widely adopted practice across the globe and is an incredibly lucrative industry. Africa and Asia represent the largest consumers of skin bleaching products. These products commonly contain corticosteroids, hydroquinone, and inorganic mercury, all of which have adverse cutaneous and systemic effects.
Skin depigmentation is mostly commonly practiced in populations with darker skin phenotypes, which are known to have a much lower incidence of NMSC when compared to lighter skin phenotypes. While the incidence is rare, the most common NMSC seen in Black skin is SCC, and its development seems to be impacted more by chronic inflammation and irritation than by chronic UV damage. There are limited reports of SCC in patients with a history of chronic skin bleaching. These reports are characterized by long duration of skin bleaching, darker skin phenotypes, and larger, invasive SCC lesions with higher mortality rates relative to the general population. While millions of people regularly use bleaching products, the overall known incidence of SCC is extremely rare due to the small number of cases reported. The relationship between skin bleaching practices and SCC remains unclear. Further research is necessary to determine the full malignant potential of bleaching agents.
References
- Peltzer K, Pengpid S, James C. The globalization of whitening: prevalence of skin lighteners (or bleachers) use and its social correlates among university students in 26 countries. Int J Dermatol. 2016;55(2):165–172.
- Sagoe D, Pallesen S, Dlova NC, Lartey M, Ezzedine K, Dadzie O. The global prevalence and correlates of skin bleaching: a meta-analysis and meta-regression analysis. Int J Dermatol. 2019;58(1):24–44.
- Sommerlad M. Skin lightening: causes and complications. Clin Exp Dermatol. 2022;47(2):264–270.
- Mahé A, Ly F, Aymard G, Dangou JM. Skin diseases associated with the cosmetic use of bleaching products in women from Dakar, Senegal. Br J Dermatol. 2003;148(3):493–500.
- Petit A, Cohen-Ludmann C, Clevenbergh P, Bergmann JF, Dubertret L. Skin lightening and its complications among African people living in Paris. J Am Acad Dermatol. 2006;55(5):873–878.
- Sardana K, Ghunawat S. Rationale of using hypopigmentating drugs and their clinical application in melisma. Expert Rev Clin Pharmacol. 2015;8(1):123–134.
- Denton CR. Skin protective agents. Med Bull (Ann Arbor). 1952;18(5):143–152.
- Denton CR, Lerner AB, Fitzpatrick TB. Inhibition of melanin formation by chemical agents. J Invest Dermatol. 1952;18(2):119–135.
- Olumide YM, Akinkugbe AO, Altraide D, et al. Complications of chronic use of skin lightening cosmetics. Int J Dermatol. 2008;47(4):344–353.
- Juhasz MLW, Levin MK. The role of systemic treatments for skin lightening. J Cosmet Dermatol. 2018;17(6):1144–1157.
- Mouhari-Toure A, Kassang P, Foma W, et al. Multiple squamous cell carcinoma in a patient using skin bleaching products in Togo. Case Rep Dermatol Med. 2023;2023:8002896.
- Diop K, Ly F, Diop A, et al. Squamous cell carcinomas and voluntary cosmetic depigmentation: more new cases in Senegal, Advocacy for Effective Prevention. Health Sci Dis. 2019;20:6S1.
- Gbandama KKP, Diabaté A, Kouassi KA, Kouassi YI, Allou AS, Kaloga M. Squamous cell carcinoma associated with cosmetic use of bleaching agents: about a case in Ivory Coast. Case Rep Dermatol. 2019;11(3):322–326.
- Ly F, Kane A, Déme A, et al. Premiers cas de carcinomes épidermoïdes sur terrain de dépigmentation artificielle. First cases of squamous cell carcinoma associated with cosmetic use of bleaching compounds. Ann Dermatol Venereol. 2010;137(2):128–131.
- Ly F, Diousse P, Ndiaye C, et al. Cutaneous squamous cell carcinomas (SCC) associated with cosmetic skin whitening: 8 cases reported in Senegal. Ann Dermatol Venereol. 2018;145(2):83–88.
- Faye O, Dicko AA, Berthé S, et al. Carcinome épidermoïde associé à une dépigmentation volontaire. Squamous cell carcinoma associated with use of skin-lightening cream. Ann Dermatol Venereol. 2018;145(2):100–103.
- Addo HA. Squamous cell carcinoma associated with prolonged bleaching. Ghana Med J. 2000;34(3):144–146.
- Gloster HM Jr, Neal K. Skin cancer in skin of color. J Am Acad Dermatol. 2006;55(5):741–764.
- Scotto J, Fears TR, Fraumeni JF Jr. Incidence of Nonmelanoma Skin Cancer in the United States. National Cancer Institute; 1981. DHHS Report No. NIH 82-2433.
- StrategyR. Skin Lighteners: World Market Report. Feb 2022. Accessed 1 April 2022. https://www.strategyr.com/market-report-skin-lighteners-forecasts-global-industry-analysts-inc.asp
- Jablonski NG. Living Color: The Biological and Social Meaning of Skin Colour. University of California Press; 2012.
- Iftekhar N, Zhitny VP. Overview of skin bleaching history and origins. Dermatology. 2021;237(2):306–308.
- Hsieh S, Maranda EL, Salih T, Nguyen A, Jimenez J. The quest for the ultimate skin-lightening agent. JAMA Dermatol. 2016;152(4):372.
- Engler DE. Mercury “bleaching” creams. J Am Acad Dermatol. 2005;52(6):1113–1114.
- Kourouma S, Gbery IP, Kaloga M, et al. Dépigmentation cutanée cosmétique des femmes noires: résultats d’une enquête CAP à Abidjan (Côte d’Ivoire). Cutaneous depigmentation in black female population for cosmetic purposes: results of a KAP survey conducted in Abidjan (Ivory Coast). Pan Afr Med J. 2016;24:159.
- Pollock S, Taylor S, Oyerinde O, et al. The dark side of skin lightening: an international collaboration and review of a public health issue affecting dermatology. Int J Womens Dermatol 2020;7(2):158–164.
- Austin E, Nguyen JK, Jagdeo J. Topical treatments for melasma: a systematic review of randomized controlled trials. J Drugs Dermatol. 2019;18(11):S1545961619P1156X.
- Ladizinski B, Mistry N, Kundu RV. Widespread use of toxic skin lightening compounds: medical and psychosocial aspects. Dermatol Clin. 2011;29(1):111–123.
- Chan R, Park KC, Lee MH, et al. A randomized controlled trial of the efficacy and safety of a fixed triple combination (fluocinolone acetonide 0.01%, hydroquinone 4%, tretinoin 0.05%) compared with hydroquinone 4% cream in Asian patients with moderate to severe melasma. Br J Dermatol 2008;159(3):697–703.
- Cestari TF, Hexsel D, Viegas ML, et. al. Validation of a melasma quality of life questionnaire for Brazilian Portuguese language: the MelasQoL-BP study and improvement of QoL of melasma patients after triple combination therapy. Br J Dermatol. 2006;156(Suppl 1):13–20.
- Torok H, Taylor S, Baumann L, et al. A large 12-month extension study of an 8-week trial to evaluate the safety and efficacy of triple combination (TC) cream in melasma patients previously treated with TC cream or one of its dyads. J Drugs Dermatol 2005;4(5):592–597.
- Coondoo A, Phiske M, Verma S, Lahiri K. Side-effects of topical steroids: a long overdue revisit. Indian Dermatol Online J. 2014;5(4):416–425.
- Chen J, Ye Y, Ran M, Li Q, Ruan Z, Jin N. Inhibition of tyrosinase by mercury chloride: spectroscopic and docking studies. Front Pharmacol. 2020;11:81.
- Graeme KA, Pollack CV Jr. Heavy metal toxicity, part I: arsenic and mercury. J Emerg Med. 1998;16(1):45–56.
- Al-Saleh I, Al-Doush I. Mercury content in skin-lightening creams and potential hazards to the health of Saudi women. J Toxicol Envir Health. 1997;51(2):123–130.
- Saade DS, Maymone MBC, Secemsky EA, Kennedy KF, Vashi NA. Patterns of over-the-counter lightening agent use among patients with hyperpigmentation disorders: a United States-based cohort study. J Clin Aesthet Dermatol. 2018;11(7):26–30.
- Jimbow K, Obata H, Pathak MA, Fitzpatrick TB. Mechanism of depigmentation by hydroquinone. J Invest Dermatol. 1974;62(4):436–449.
- Nordlund JJ, Grimes PE, Ortonne JP. The safety of hydroquinone. J Eur Acad Dermatol Venereol 2006;20(7):781–787.
- Dorsey CS. Dermatitic and pigmentary reactions to monobenzyl ether of hydroquinone. AMA Arch Dermat. 1960;81:245–248.
- Arndt KA, Fitzpatrick TB. Topical use of hydroquinone as a depigmenting agent. JAMA. 1965;194(9):965–967.
- Findlay GH, Morrison JG, Simson IW. Exogenous ochronosis and pigmented colloid milium from hydroquinone bleaching creams. Br J Dermatol. 1975;93(6):613–622.
- Hoshaw RA, Zimmerman KG, Menter A. Ochronosis-like pigmentation from hydroquinone bleaching creams in American Blacks. Arch Dermatol. 1985;121(1):105–108.
- Dogliotte M, Leibowitz M. Granulomatous ochronosis—a cosmetic-induced skin disorder in blacks. S Afr Med J. 1979;56(19):757–760.
- Penneys NS. Ochronosis like pigmentation from hydroquinone bleaching creams. Arch Dermatol. 1985;121(10):1239–1240.
- Zhou LL, Baibergenova A. Melasma: systemic review of the systemic treatments. Int J Dermatol. 2017;56(9):902–908.
- Malathi M, Thappa DM. Systemic skin whitening/lightening agents: what is the evidence? Indian J Dermatol Venereol Leprol. 2013;79(6):842–846.
- Karn D, Kc S, Amatya A, Razouria EA, Timalsina M. Oral tranexamic acid for the treatment of melasma. Kathmandu Univ Med J. 2012;10(40): 40–43.
- Weinstock MA. Nonmelanoma skin cancer mortality in the United States, 1969 through 1988. Arch Dermatol. 1993;129(10):1286–1290.
- Gupta AK, Bharadwaj M, Mehrotra R. Skin cancer concerns in people of color: risk factors and prevention. Asian Pac J Cancer Prev. 2016;17(12):5257–5264.
- Cordel N, Bonnecarrère L, Tressières B. Squamous cell carcinoma in the Afro-Caribbean community: an 11-year retrospective study. J Eur Acad Dermatol Venereol. 2017;31(9):1462–1467.
- Dieng MT, Diop NN, Déme A, Sy TN, Niang SO, Ndiayte B. Carcinome épidermoïde sur peau noire: 80 cas. Squamous cell carcinoma in Black patients: 80 cases. Ann Dermatol Venereol. 2004;131(12):1055–1057.
- Halder RM, Bang KM. Skin cancer in Blacks in the United States. Dermatol Clin. 1988;6(3):397–405.
- Gilchrest BA, Eller MS, Geller AC, Yaar M. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med. 1999;340(17):1341–1348.
- Young AR, Chadwick CA, Harrison GI, Nikaido O, Ramsden J, Potten CS. The similarity of action spectra for thymine dimers in human epidermis and erythema suggests that DNA is the chromophore for erythema. J Invest Dermatol. 1998;111(6):982–988.
- Kaidbey KH, Agin PP, Sayre RM, Kligman AM. Photoprotection by melanin–a comparison of Black and Caucasian skin. J Am Acad Dermatol. 1979;1(3):249–260.
- Jimbow K, Quevedo WC, Fitzpatrick TB, Szabo G. Some aspects of melanin biology: 1950-1975. J Invest Dermatol. 1976;67(1):72-89.
- Ito S, Wakamatsu K. Quantitative analysis of eumelanin and pheomelanin in humans, mice, and other animals: a comparative review. Pigment Cell Res. 2003;16(5):523–531.
- Szabó G, Gerald AB, Pathak MA, Fitzpatrick TB. Racial differences in the fate of melanosomes in human epidermis. Nature. 1969;222(5198):1081–1082.
- Agbai ON, Buster K, Sanchez M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70(4):748–762.
- Taylor SC, Alexis AF, Armstrong AW, et al. Misconceptions of photoprotection in skin of color. J Am Acad Dermatol. 2022;86(3S):S9–S17.
- Williams DM. Clinical pharmacology of corticosteroids. Respir Care. 2018;63(6):655–670.
- Krynitz B, Edgren G, Lindelöf B, et al. Risk of skin cancer and other malignancies in kidney, liver, heart and lung transplant recipients 1970 to 2008–a Swedish population-based study. Int J Cancer. 2013;132(6):1429–1438.
- Griffith CF. Skin cancer in immunosuppressed patients. JAAPA. 2022; 35(2):19–27.
- Ratib S, Burden-Teh E, Leonardi-Bee J, Harwood C, Bath-Hextall F. Long-term topical corticosteroid use and risk of skin cancer: a systematic review. JBI Database System Rev Implement Rep. 2018;16(6):1387–1397.
- McGregor D. Hydroquinone: an evaluation of the human risks from its carcinogenic and mutagenic properties. Crit Rev Toxicol. 2007;37(10):887–914.
- Eigentler TK, Leiter U, Häfner HM, Garbe C, Röcken M, Breuninger H. Survival of patients with cutaneous squamous cell carcinoma: results of a prospective cohort study. J Invest Dermatol. 2017;137(11):2309–2315.