J Clin Aesthet Dermatol. 2026;19(5):63–65.
by Sergio Santos-Alarcón, MD; Javier Mataix Diaz, MD; Luca Schneller-Pavelescu, MD; Isabel Belinchon, MD; Ines Poveda-Montoyo, MD; Víctor González-Delgado, MD; Santiago Guillen-Climent, MD; Francisco Javier Melgosa Ramos, MD; Almudena Mateu, MD
Drs. Santos-Alarcón and Guillen-Climent are with Hospital Verge dels Lliris, Alcoi, Spain. Dr. Mataix Diazis is with Hospital Marina Baixa, Villajoyosa, Spain. Dr. Schneller-Pavelescu is with Hospital Vega Baja, Orihuela, Spain. Drs. Belinchon and Poveda-Montoyo are with General University Hospital of Alicante, Alicante (Alacant), Spain. Drs. González-Delgado is with Hospital Clinic Universitari, València, Spain. Dr. Melgosa Ramos is with Hospital General d’Ontinyent, Ontinyent, Spain. Dr. Mateu is with Doctor Peset University Hospital, València, Spain.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Santos Alarcón has served as a speaker, consultant, or investigator for Abbvie, Adium Pharma, Amgen, Bristol Myers Squibb, Celgene, Isdin, Janssen-Cilag, LEO Pharma, Lilly, Novartis, Pfizer, Pierre Fabre, Sanofi, and UCB. The remaining authors have no conflicts of interest to declare.
ABSTRACT: OBJECTIVE: To evaluate the effectiveness and safety of switching to tildrakizumab, a selective interleukin (IL) 23p19 inhibitor, in patients with moderate-to-severe plaque psoriasis who had an inadequate response to ustekinumab (IL-12/23 inhibitor) in a real-world setting. METHODS: A multicenter retrospective observational study was conducted and included 14 patients who switched to tildrakizumab following ustekinumab failure. Effectiveness was assessed using Psoriasis Area and Severity Index (PASI), Physician Global Assessment (PGA), and Dermatology Life Quality Index (DLQI) at Weeks 0, 12, 28, 36, and 52. RESULTS: The cohort (mean age: 45.92 years; 85.7% prior systemic therapy) showed rapid improvement. Mean PASI decreased from 9.36 at baseline to 2.60 at Week 12, maintaining low disease activity through Week 52 with 100% of patients achieving an absolute PASI score <3. Similar trends were observed in PGA and DLQI scores. No new safety signals were identified. LIMITATIONS: This study included a small sample size (n=14) and retrospective design. CONCLUSION: Tildrakizumab showed consistent clinical improvement as a rescue therapy for patients after ustekinumab failure, supporting the clinical benefit of switching from p40 to specific p19 inhibition. KEYWORDS: Psoriasis, tildrakizumab, ustekinumab, biologic switching, IL-23 inhibitor, real-world evidence
Introduction
Psoriasis is a chronic immune-mediated inflammatory disease that significantly impacts patient quality of life. The introduction of biologic therapies targeting specific cytokines has revolutionized the management of moderate-to-severe plaque psoriasis. However, a subset of “biologic-experienced” patients remains difficult to treat due to primary failure, secondary loss of response, or adverse events.1
Ustekinumab, a monoclonal antibody targeting the p40 subunit shared by both interleukin (IL) 12 and IL-23, is a well-established therapy. Despite its efficacy, some patients experience suboptimal response or loss of efficacy over time. Recent advances in immunology have highlighted the distinct and critical role of the IL-23 and helper T cell (TH) 17 axis in the pathogenesis of psoriasis, distinguishing it from the IL-12/TH1 pathway.2 The specific inhibition of the IL-23 p19 subunit has emerged as a targeted strategy that may offer superior efficacy compared to dual IL-12/23 inhibition, potentially due to the preservation of the IL-12-dependent TH1 response.
Tildrakizumab is a high-affinity, humanized immunoglobulin G1k monoclonal antibody that specifically targets the p19 subunit of IL-23. Clinical trials and long-term extension studies have demonstrated its sustained efficacy and safety.3 However, real-world evidence specifically addressing the “intraclass” switch from ustekinumab (anti-p40) to tildrakizumab (anti-p19) remains limited but crucial for clinical decision-making.
The objective of this study was to assess the clinical response in patients with moderate-to-severe plaque psoriasis who were unresponsive (defined as suboptimal response or inadequate response) to ustekinumab following a switch to tildrakizumab.
Methods
Study design and population. We performed a multicenter retrospective observational study involving patients from 7 hospital centers in the Valencian community, Spain. The study population consisted of adult patients (aged ≥18 years) with moderate-to-severe plaque psoriasis.
Inclusion criteria required patients to have demonstrated an inadequate or suboptimal response to prior treatment with ustekinumab, defined as failure to achieve a Physician Global Assessment (PGA) score of 0 or 1 after standard induction or a secondary loss of efficacy characterized by a relapse of plaques (Psoriasis Area and Severity Index [PASI] score >3) despite continuous treatment. These patients were subsequently switched to tildrakizumab according to standard clinical practice and the Summary of Product Characteristics.
Assessments. Effectiveness was assessed using 3 primary metrics: PASI, PGA, and Dermatology Life Quality Index (DLQI). Data were collected at baseline (Week 0) and at follow-up visits at Weeks 12, 28, 36, and 52 (±1 week). Safety was assessed by monitoring adverse events throughout the study period.
Results
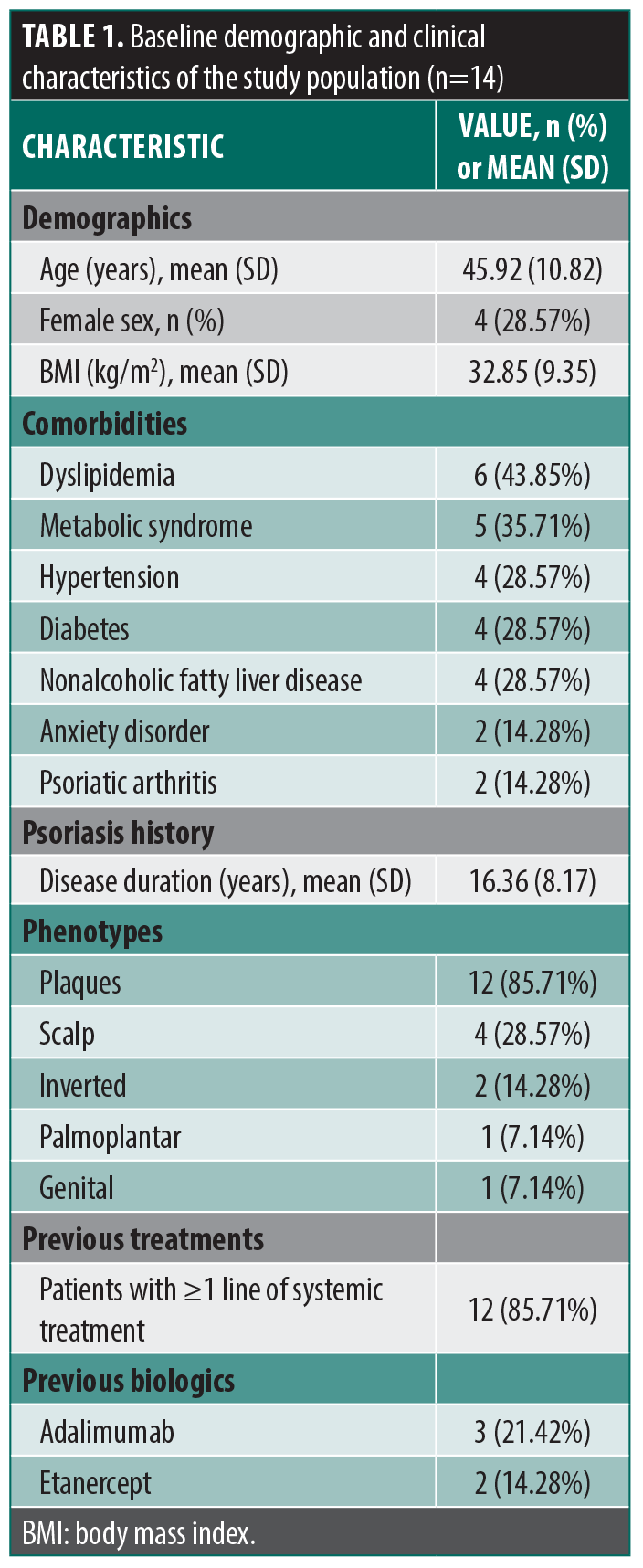
Baseline characteristics. A total of 14 patients were included in the study. (Table 1) The demographic analysis revealed a mean age of 45.92 years (SD: 10.82) and a mean body mass index of 32.85 kg/m² (SD: 9.35), indicating a high prevalence of obesity/overweight in the cohort. Women represented 28.57% (n=4) of the study group.
Comorbidities were common: dyslipidemia was the most frequent (43.85%, n=6), followed by metabolic syndrome (35.71%, n=5), hypertension (28.57%, n=4), and diabetes (28.57%, n=4). Regarding psoriasis history, the mean disease duration was 16.36 years. The predominant phenotype was plaque psoriasis (85.71%), followed by scalp involvement (28.57%). Notably, 85.71% of patients had received at least 1 prior line of systemic treatment. Prior biologic treatments included adalimumab (21.42%) and etanercept (14.28%).
Effectiveness outcomes. Following the switch to tildrakizumab, rapid and sustained improvements were observed across all measured indices:
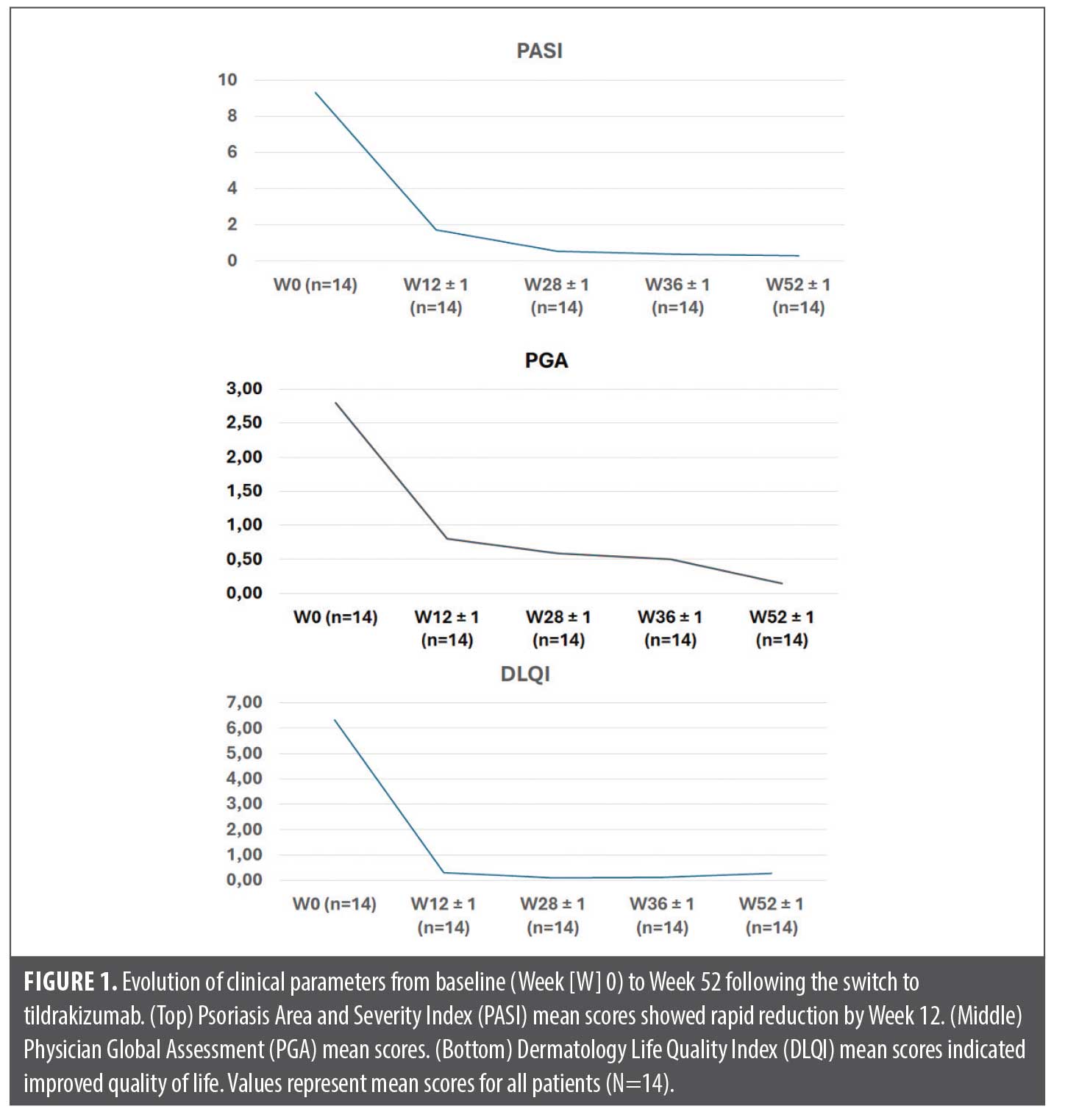
PASI: At baseline (Week 0), patients exhibited high disease activity. Following treatment initiation, PASI scores decreased significantly by Week 12. This improvement was maintained through Weeks 28, 36, and 52, as demonstrated in Figure 1. Notably, at Week 52, 100% of patients (n=14) achieved an absolute PASI score of <3, indicating robust disease control.
PGA: The PGA followed a similar trajectory. Baseline scores indicated moderate-to-severe disease (mean approx. 2.80), dropping sharply by Week 12 (mean <1.00) and trending toward clearance (mean approx. 0.14) by Week 52.
DLQI: The impact on quality of life was significantly reduced. Baseline DLQI scores (mean >6.00) dropped to negligible levels (mean approx. 0.50) by Week 52, reflecting a substantial improvement in patient wellbeing.
Safety. The adverse event rate observed in this cohort was consistent with the known safety profile of tildrakizumab. No severe adverse events or discontinuations due to safety were reported. The safety profile was consistent with clinical trials.
Discussion
This study provides real-world evidence supporting the effectiveness and safety of switching to tildrakizumab in patients with plaque psoriasis after ustekinumab failure. The results suggest that an intraclass switch from an IL-12/23 inhibitor to a selective IL-23p19 inhibitor allows for the recapture of disease control.
Our findings align with recent data from the POSITIVE study,4 which demonstrated high effectiveness of tildrakizumab in both biologic-naïve and biologic-experienced patients in a real-world setting. Similarly, a multicenter Italian study by Di Brizzi et al5 reported effective outcomes after switching to tildrakizumab regardless of the previous biologic agent.
The mechanism underlying this successful switch likely involves the precise targeting of the p19 subunit. While ustekinumab blocks p40 (inhibiting both IL-12 and IL-23), tildrakizumab selectively targets IL-23. This selective inhibition is crucial as IL-23 is the primary driver of the pathogenic TH17 axis in psoriasis. Furthermore, real-world registry data (IL-PSO) has indicated high drug survival rates for IL-23 inhibitors compared to other biologic classes,6 supporting the durability of response observed in our 52-week follow-up.
This study contributes valuable evidence on the utility of intraclass switching of interleukin inhibitors, expanding therapeutic options for this challenging patient population.
The mean baseline PASI in our cohort (9.36) reflects a real-world switching scenario where patients retain some benefit from the previous biologic but fail to achieve clear skin, contrasting with the higher baseline scores typically seen in washout studies.
Limitations. This study has limitations inherent to its design, including the retrospective nature and the small sample size (n=14). Additionally, the lack of a comparator group limits the ability to draw definitive causal conclusions regarding superiority over other therapeutic options.
Conclusion
In conclusion, our findings suggest that tildrakizumab may offer an effective and safe option for patients with moderate-to-severe plaque psoriasis unresponsive (suboptimal or inadequate response) to ustekinumab. Prospective studies are needed to confirm these findings.
References
- Maurelli M, Gisondi P, Girolomoni G. Tailored biological treatment for patients with moderate-to-severe psoriasis. Expert Rev Clin Immunol. 2023;19(1):37–43.
- Krueger JG, Eyerich K, Kuchroo VK, et al. IL-23 past, present, and future: a roadmap to advancing IL-23 science and therapy. Front Immunol. 2024;15:1331217.
- Thaci D, Piaserico S, Warren RB, et al. Five-year efficacy and safety of tildrakizumab in patients with moderate-to-severe psoriasis who respond at week 28: pooled analyses of two randomized phase III clinical trials (reSURFACE 1 and reSURFACE 2). Br J Dermatol. 2021;185(2):242–243.
- Mrowietz U, Sommer R, Reguiai Z, et al. High effectiveness of tildrakizumab in bio-naïve and bio-experienced patients with moderate-to-severe psoriasis: 52-week results from the POSITIVE study. Poster presented at: 33rd EADV Congress; 25–28 Sep 2024; Amsterdam, Netherlands. Poster P3341.
- Di Brizzi EV, Buononato D, Benvenuto P, et al. Effectiveness and safety after a switch to tildrakizumab: a real world multicenter Italian study in psoriasis. Dermatol Pract Concept. 2023;13(4):e2023215.
- Gargiulo L, Ibba L, Malvaso D, et al. Drug survival of IL-12/23, IL-17 and IL-23 inhibitors for moderate-to-severe plaque psoriasis: a retrospective multicenter real-world experience on 5932 treatment courses – IL PSO (Italian landscape psoriasis). Front Immunol. 2024;14:1341708.