J Clin Aesthet Dermatol. 2025;18(12):21–29.
by Angela L. Rosenberg, DO; Milaan Shah, MD; James Q. Del Rosso, DO; Hilary Baldwin, MD; Julie Harper, MD; Adelaide A. Hebert, MD; Jonette Keri, MD, PhD; Pearl Kwong, MD, PhD; Evan Rieder, MD; Linda Stein Gold, MD; Brooke Bartley, MD; Joshua Burshtein, MD; Lauren DeBusk, MD; and Danny Zakria, MD, MBA
Drs. Rosenberg, Bartley, and DeBusk are with the Department of Dermatology at UT Southwestern Medical Center in Dallas, Texas. Dr. Shah is with the Department of Dermatology at the Medical University of South Carolina in Charleston, South Carolina. Dr. Del Rosso is with JDR Dermatology Research in Las Vegas, Nevada and Section of Dermatology at Touro University Nevada in Henderson, Nevada. Dr. Baldwin is with The Acne Treatment and Research Center in Brooklyn, New York and Robert Wood Johnson University Hospital in New Brunswick, New Jersey. Dr. Harper is with the Dermatology and Skin Care Center of Birmingham in Birmingham, Alabama. Dr. Hebert is with the Departments of Dermatology and Pediatrics at UT Health McGovern Medical School and Children’s Memorial Hermann Hospital in Houston, Texas. Dr. Keri is with University of Miami, Miller School of Medicine and the Miami VA Medical Center in Miami, Florida. Dr. Kwong is with PCK Derm LLC in Jacksonville, Florida and Suncoast Skin Solutions and Apex Clinical Trials in Tampa, Florida. Dr. Rieder is in private practice in New York, New York. Dr. Stein Gold is with the Department of Dermatology at Henry Ford Hospital in Detroit, Michigan. Dr. Burshtein is with the Department of Dermatology at the University of Illinois Chicago in Chicago, Illinois. Dr. Zakria is with the Department of Dermatology at the Icahn School of Medicine at Mount Sinai in New York, New York.
FUNDING: This research was supported by an unrestricted educational grant from Almirall.
DISCLOSURES: Dr. Del Rosso is an investigator, consultant, and/or speaker for Almirall, Bausch (Ortho Dermatologics), Galderma, Journey, LEO Pharma, and Sun Pharmaceuticals. Dr. Harper is a speaker for Galderma, Journey, Nutrafol, Ortho Dermatologics, and Sun Pharmaceuticals, and an advisor for and received honorarium from Almirall, Arcutis, Bioderma, Beiersdorf, Bubble, Cutera, Galderma, Journey, L’Oreal, Nutrafol, Ortho Dermatologics, Pelthos, Sagimet, Sanofi, and Sun Pharmaceuticals. Dr. Hebert has received research grants paid to the medical school from AbbVie, Amgen, Arcutis, Biogen, Dermavant, Oneness, Pfizer, and Takeda, and has received honoraria from Arcutis, Dermavant, Incyte, Ortho Dermatologics, Pfizer, and Sun Pharmaceuticals, and is on the Data and Safety Monitoring Board for Ortho Dermatologics, GSK, and Sanofi Regeneron. Dr. Keri is an author, and or on the advisory board for Ortho Dermatologics, Almirall, L’Oreal, and Sanofi; she is an advisor for National Dairy Council. Dr. Kwong is a consultant, principal investigator, and/or speaker for AbbVie, Almirall, Alphyn Biologics, Amgen, Arcutis, Botanix, Galderma, Incyte, LEO Pharma, Lilly, Organon Dermavant, Ortho Dermatologics, Pelthos, Pfizer, Regeneron, Sanofi, Sun Pharmaceuticals, and Verrica. Dr. Rieder is a consultant for AbbVie, Almirall, L’Oreal, Merz, UCB Pharmaceuticals, Unilever. Dr. Stein Gold is an investigator, advisor, and/or speaker for Almirall, Galderma, Journey, Ortho Dermatologics, and Sun Pharmaceuticals. The remaining authors have no conflicts of interest relevant to the contents of this article.
ABSTRACT: Background: The purpose of this meeting was for an expert panel to analyze current literature and provide updated consensus statements regarding the optimal management, safety, and efficacy of oral antibiotics in acne vulgaris (AV). Methods: A thorough literature search was conducted across PubMed, Scopus, and Google Scholar to identify English-language original research articles evaluating the use of oral antibiotics for AV. An expert panel of 8 dermatologists, specializing in the management of AV, convened to assess the findings and develop statements regarding oral antibiotics for AV. Each statement was approved through a modified Delphi process, and a strength of recommendation was assigned based on the Strength of Recommendation Taxonomy (SORT) criteria. Results: A total of 544 articles regarding the safety, efficacy, and management of oral antibiotics for AV were identified through the literature search. Following a rigorous screening process, 17 articles were deemed relevant to the research questions and provided to the panelists for review prior to the roundtable discussion. The panel reached unanimous agreement on 10 consensus statements and recommendations, assigning seven a strength of A, one a strength of B, and two a strength of C. Conclusion: The expert panel concluded that oral antibiotics can be used in the treatment of moderate-to-severe AV, oral antibiotics should be combined with topical treatments, and duration of use should be determined based on individualized patient care. Tetracyclines are recognized options for AV therapy, with sarecycline considered a preferred option for AV due to its targeted mode of action, unique ribosomal binding properties, narrow spectrum of antibiotic activity, lower bacterial resistance risk, and overall better tolerability. Additionally, tetracyclines should be avoided in pregnancy and in young children. Keywords: Acne vulgaris, oral antibiotics, tetracyclines, sarecycline, minocycline, doxycycline
Introduction
Acne vulgaris (AV) is a common chronic inflammatory skin disease marked by follicular obstruction, inflammation, alterations in sebum, and Cutibacterium acnes (C. acnes) proliferation. Recent Global Burden of Disease (GBD) data from 1990 to 2021 show an increase in age-adjusted prevalence from approximately 8563 to 9790 per 100,000 among individuals aged 10 to 24 years, with a 0.43% annual increase and a nearly 25% higher burden in young women than men, although both genders are often adversely affected (10912 vs. 8728 per 100,000).¹ Affected male patients tend to have greater severity of AV and scarring than female patients. Overall, AV affects an estimated 9.4% of the global population.²
Beyond its high prevalence, AV imposes substantial psychosocial and physical burdens, including embarrassment, anxiety, reduced self-esteem, and long-term sequelae such as scarring and postinflammatory dyspigmentation.3,4 The pathogenesis of AV is multifactorial, involving follicular hyperkeratinization, sebum overproduction, C. acnes proliferation, and inflammation.5
Oral antibiotics are commonly used in the treatment of moderate-to-severe AV, valued for their anti-inflammatory effects and bacteriostatic activity against C. acnes.6 However, the emergence of resistant C. acnes strains has been linked to treatment failure in AV, underscoring the need for judicious antibiotic use to preserve their long-term therapeutic efficacy.7 Rising concerns about microbial resistance in the general population, microbiome disruption, and adverse effects have prompted guideline-directed stewardship. Current American Academy of Dermatology (AAD) recommendations advise using oral antibiotics in combination with topical agents and limiting duration to the shortest time needed for control, typically suggesting a duration of 3 to 4 months.8
Among available agents, extended-release (ER) minocycline and sarecycline are the only oral antibiotics approved by the United States (US) Food and Drug Administration (FDA) for moderate-to-severe AV following modern regulatory review.9 ER minocycline, a once-daily formulation for the treatment of inflammatory lesions of non-nodular moderate-to-severe AV in patients aged 12 years or older, received FDA approval in May 2006.9 Sarecycline was approved in October 2018 for patients aged ≥9 years with non-nodular moderate-to-severe AV, making it the first acne-specific oral antibiotic to receive FDA approval.10 Doxycycline (immediate release [IR]), minocycline (IR), and tetracycline continue to be prescribed under a grandfathered designation in the approved prescribing information but have never received FDA approval through a formally submitted drug application process.11
Considering emerging resistance concerns, expanding antibiotic options, and evolving clinical evidence, an expert panel convened to develop consensus-based statements on the optimal use of oral antibiotics for AV. A modified Delphi process was used to assess the literature and develop statements reflecting clinical efficacy, safety, antimicrobial spectrum, regulatory status, and stewardship principles.
Methods
Literature search and study selection. On April 30, 2025, a comprehensive literature search was conducted using PubMed, Scopus, and Google Scholar with the keywords acne vulgaris, oral antibiotics, and management, combined using the Boolean operator AND. The search was limited to English-language original research articles, clinical trials, meta-analyses, randomized controlled trials, observational studies, and reviews. Institutional review board approval was not required. Articles were reviewed for relevance to the safety, efficacy, and clinical management of oral antibiotics in the treatment of acne vulgaris.
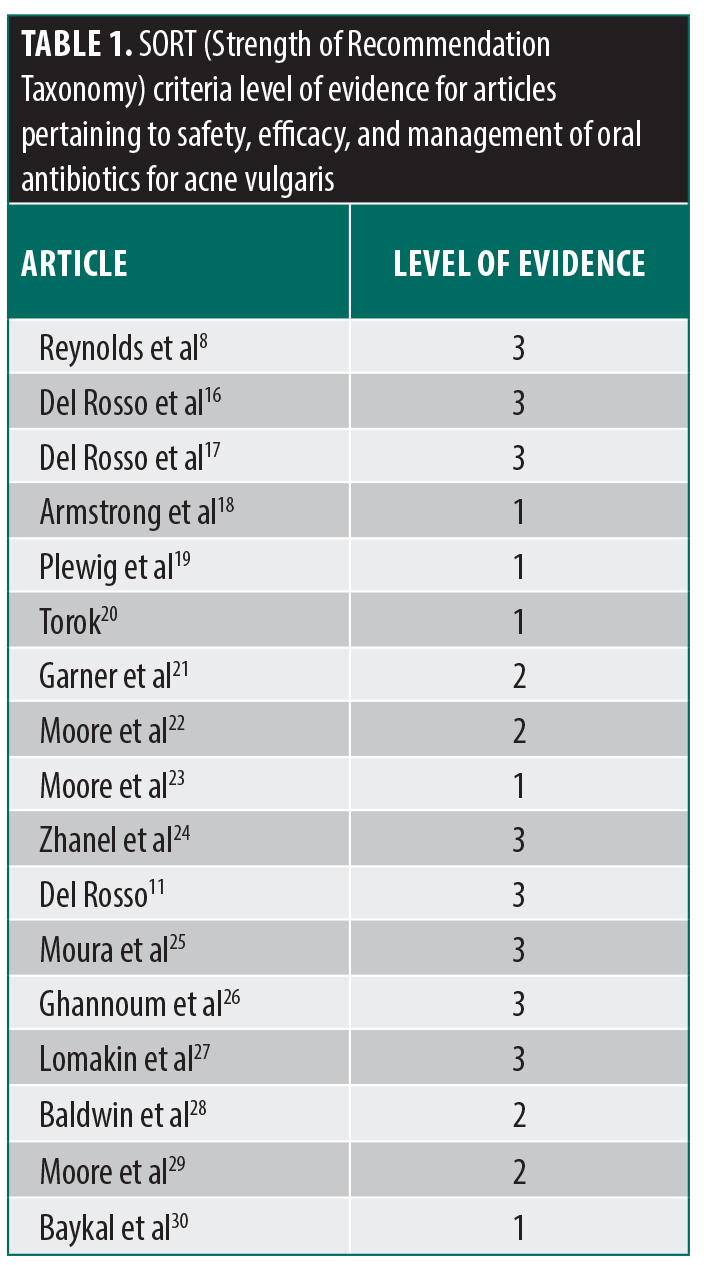
Eligible articles were distributed to the expert panel, who conducted independent reviews and categorized the strength of evidence using the Strength of Recommendation Taxonomy (SORT).12 The SORT system classifies evidence into three levels: Level 1 for high-quality, patient-centered research; Level 2 for moderate-quality, patient-oriented data; and Level 3 for evidence drawn from expert consensuses, clinical practice, or in vitro studies focused on disease processes.12 Notably, in vitro analyses and basic science investigations appropriately assigned to Levels 2 or 3 do not lack validity or imply inadequacy.
Development of consensus statements. The panel comprised eight dermatologists with specialized expertise in the management of acne vulgaris (AV): Hilary Baldwin, MD; James Del Rosso, DO; Julie Harper, MD; Adelaide Hebert, MD; Jonette Keri, MD, PhD; Pearl Kwong, MD, PhD; Evan Rieder, MD; and Linda Stein Gold, MD.
On May 30, 2025, the expert panel gathered to critically evaluate the available literature and formulate consensus statements regarding the optimal use of oral antibiotics in the treatment of AV. A modified Delphi methodology was used to facilitate consensus-building.12 This structured process, frequently employed in dermatology to generate expert guidance, involves multiple iterative rounds of real-time voting and requires a supermajority to establish agreement on any given recommendation.13-15
All panelists, who also serve as coauthors of this manuscript, were fully informed of the study’s aims and collectively endorse its submission to a peer-reviewed and indexed journal.
Results
Literature search and study selection. The literature search initially yielded 544 articles that met the predefined inclusion criteria. Following a thorough screening process, 17 articles were identified as pertinent to the research questions and were distributed to the panelists for review before the roundtable discussion. Of these 17 studies, the panel classified five as Level 1 evidence, four as Level 2 evidence, and eight as Level 3 evidence, in accordance with established grading criteria (Table 1).
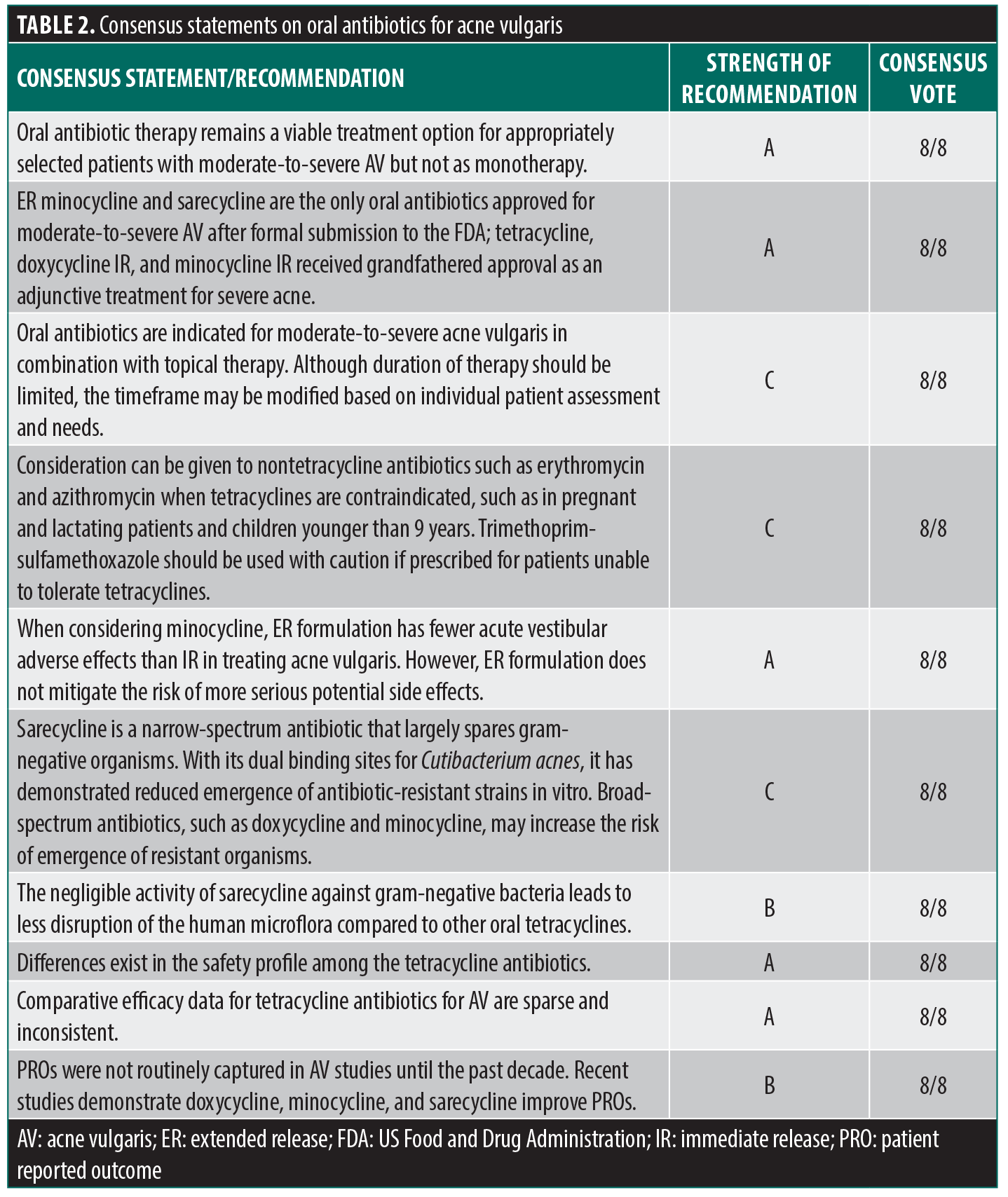
Consensus statements. The panel created 10 consensus statements on the efficacy and safety of oral antibiotics for treating AV as well as guiding management principles. All 10 statements were unanimously approved by all eight panelists. The strength of each recommendation was then rated using the SORT system (Table 2).
Statement 1: Oral antibiotic therapy remains a viable treatment option for appropriately selected patients with moderate-to-severe AV but not as monotherapy. (SORT Level A). Oral antibiotics continue to be a widely used and effective treatment for AV. In an analysis of US outpatient data from 2018 to 2019, oral antibiotics were the second most prescribed systemic therapy for AV in female patients, used in 17.4% of visits, and surpassed only by isotretinoin (31.1% of visits).31 According to 2024 acne management guidelines, the decision to use topical and/or oral antibiotics should be guided by the individual clinical presentation and made in collaboration with the patient.8
Acne severity is classified based on lesion type and extent. Mild AV typically involves comedones and a limited number of inflammatory lesions (papules and pustules).32 Moderate AV is characterized by more numerous inflammatory lesions, occasional nodules, and mild scarring. Severe acne can present as widespread inflammatory lesions, nodules, and/or significant scarring, cases that are resistant to six months of treatment, or any AV severity associated with marked psychological distress.
Randomized controlled trials primarily support that mild-to-moderate AV is effectively managed with a combination of topical retinoids, benzoyl peroxide, and either topical or oral antibiotics; topical clascoterone may be incorporated to address the pathophysiological effects of androgens and sebum.33 Although mild AV can lead to scarring, there is no clinical evidence supporting the use of oral antibiotics for this severity level. Oral antibiotics play a critical role in treating moderate-to-severe inflammatory AV, particularly when topical therapies alone are insufficient or when AV affects the trunk or multiple areas.34,35 However, there is still a lack of standardized, head-to-head trials to determine the optimal dosing and duration for each oral antibiotic used in AV treatment, and no specific antibiotic type or class has demonstrated clear superiority in efficacy.36 When systemic antibiotics are prescribed, it is considered best practice to also use topical agents, primarily benzoyl peroxide, to reduce antibiotic resistance and minimize treatment duration.8
The expert panel agreed that oral antibiotics can be an effective treatment option for patients in whom the above criteria have been considered and re-emphasized the importance of prescribing oral antibiotics in combination with topical therapy, primarily benzoyl peroxide.
Statement 2: ER minocycline and sarecycline are the only oral antibiotics approved for moderate-to-severe AV after formal submission to the FDA; tetracycline, doxycycline IR and minocycline IR received grandfathered approval as an adjunctive treatment for severe acne. (SORT Level A). Tetracyclines are the most recommended oral antibiotics for AV, with doxycycline, minocycline, sarecycline, and tetracycline historically used most frequently.8,18 Of these, doxycycline, minocycline, and tetracycline are broad-spectrum antibiotics with activity against both gram-positive and gram-negative bacteria. Sarecycline is a narrow-spectrum agent that primarily targets C. acnes and other gram-positive organisms, and is supported in its FDA-approved product information as less likely to induce antibiotic-resistant bacterial strains.37 ER minocycline was the first oral antibiotic formally approved by the FDA in 2006 for moderate-to-severe AV, supported by large Phase III trials evaluating its efficacy, safety, and dose response; it is not approved for the treatment of infection.9,20 Sarecycline hydrochloride was later approved in 2018 for the treatment of moderate-to-severe non-nodular inflammatory AV in patients aged 9 years or older, and it is also not approved for the treatment of infection.10 In contrast, tetracycline (approved in 1953) and IR formulations of doxycycline (1967) and minocycline (1971) remain approved by the FDA primarily for infections, with use for AV retained through historical grandfathered status rather than through New Drug Application (NDA) submission and approval.11 Subantibiotic-dose doxycycline was later approved for treatment of rosacea in 2006, but it was not evaluated by the FDA for use in AV.9,17 ER minocycline and sarecycline remain the only oral antibiotics to have undergone full regulatory review with formal NDA submission and receive formal FDA approval for AV.
Statement 3: Oral antibiotics are indicated for moderate-to-severe acne vulgaris in combination with topical therapy. Although duration of therapy should be limited, the timeframe may be modified based on individual patient assessment and needs. (SORT Level C). Given the large population of patients affected by AV and the frequent use of long-term antibiotic therapy, AV treatment contributes significantly to overall antibiotic exposure and emergence of antibiotic-resistant bacterial strains.6,7,17 Oral antibiotic stewardship emphasizes minimizing resistance by using antibiotics judiciously. This includes prescribing them for the shortest effective duration, combining them with non-antibiotic topical treatments such as benzoyl peroxide and topical retinoids, and considering alternative systemic options and/or physical modalities when appropriate.38 While antibiotic stewardship is a universally important term, the recommended time frame of oral antibiotics for AV is variable and dependent on individual patient characteristics.
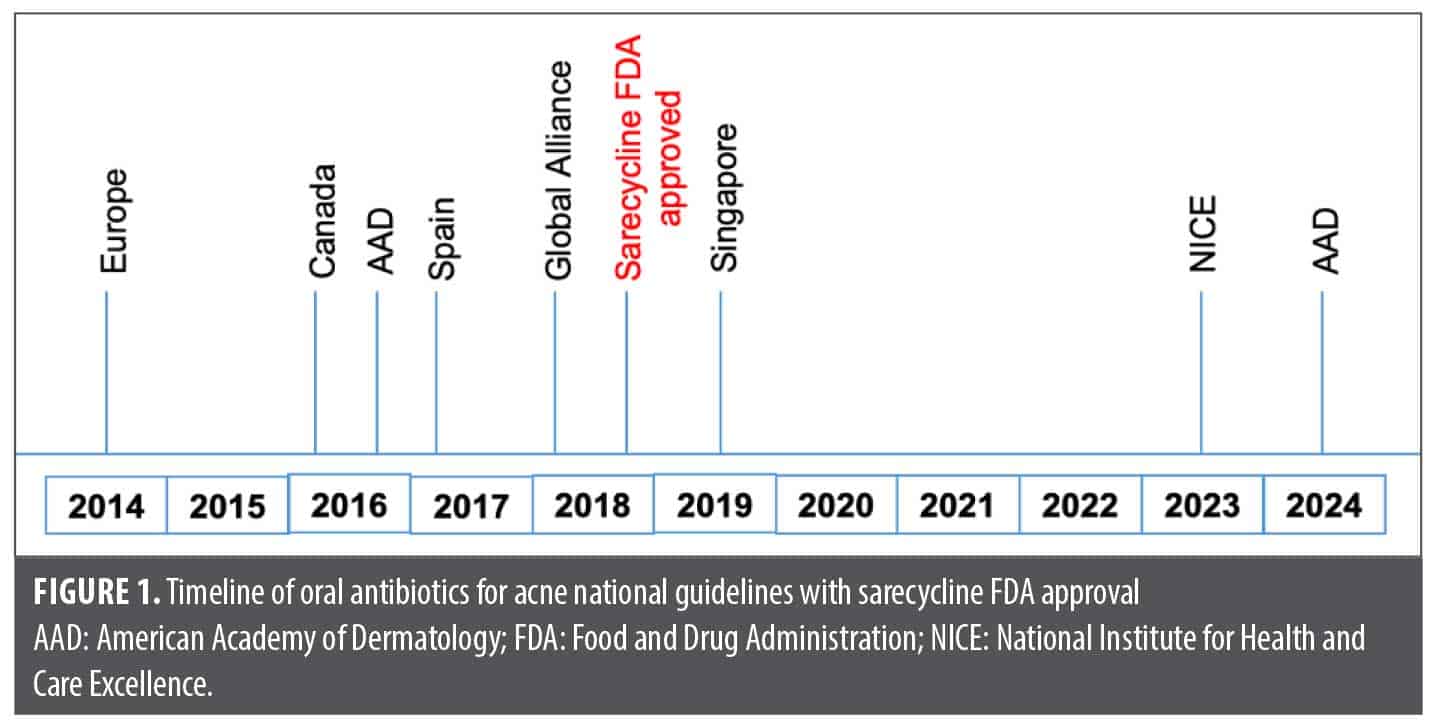
In 2014, European experts recommended restricting systemic antibiotics use for moderate-to-severe acne to 3 to 4 months and advised that topical antibiotics always be combined with benzoyl peroxide to reduce resistance risk.6 The 2016 Canadian guidelines acknowledged uncertainty regarding optimal antibiotic duration, while the AAD that same year endorsed a 3 to 4-month limit but allowed for longer courses in cases where alternatives are not suitable.39,40 Spain’s 2017 consensus adopted an even more conservative approach, recommending a maximum duration of 6 to 12 weeks.41
In 2018, the Global Alliance to Improve Outcomes in Acne recommended reassessing antibiotic therapy at 6 to 8 weeks, limiting treatment to under 3 to 4 months, and transitioning to maintenance therapy with a topical retinoid and either benzoyl peroxide or azelaic acid.35 The 2019 Singapore guidelines echoed this, advising treatment not exceed 3 to 4 months, with at least six weeks required to see clinical improvement.42 The United Kingdom’s National Institute for Health and Care Excellence (NICE) guideline, updated in December 2023, recommends an initial three-month course of oral antibiotics, with reassessment at that point. If improvement is seen but AV is not fully resolved, treatment may continue for another three months, but use beyond six months was strongly discouraged and reserved for exceptional cases.43
Most recently, the 2024 AAD guidelines reinforced these principles, emphasizing that oral antibiotics be used for the shortest effective duration—ideally no longer than 3 to 4 months.8 Despite the FDA approval of sarecycline in 2018 for AV, a 3 to 4-month duration of treatment has remained a commonly stated recommendation over at least the past decade, primarily due to concerns related to antibiotic stewardship (Figure 1). The expert panel notes that treatment length of oral antibiotic therapy for AV frequently needs to be individualized based on clinical response and available therapeutic options, which in some cases may warrant treatment for longer than 4 months.
Statement 4: Consideration can be given to non-tetracycline antibiotics including erythromycin and azithromycin when tetracyclines are contraindicated, such as in pregnant and lactating patients and children younger than 9 years. Trimethoprim-sulfamethoxazole should be used with caution if prescribed for patients unable to tolerate tetracyclines. (SORT Level C). Despite their widespread use in AV treatment, tetracyclines are not recommended, and are sometimes contraindicated, in specific subpopulations. Tetracyclines should be avoided during pregnancy and in children younger than 9 years of age due potential risks, such as maternal hepatotoxicity, impaired fetal bone growth, and/or permanent tooth discoloration in children.44 Although tetracyclines are excreted in breast milk, their bioavailability to the infant is limited by calcium binding, making the antibiotic generally safe but still not recommended during breastfeeding.44 For patients in whom tetracyclines are unsuitable, alternatives such as erythromycin, azithromycin, or trimethoprim-sulfamethoxazole (TMP-SMX) may be considered; however, the current evidence is insufficient to support formal treatment recommendations for these agents in AV.8 A systematic review of randomized controlled trials and meta-analyses found that tetracyclines, macrolides, and TMP-SMX are effective and generally safe for treating moderate-to-severe inflammatory AV.36 The expert panel reiterates that no antibiotic class has demonstrated definitive superiority, so selection is typically guided by individual patient characteristics, clinical experience, and safety profiles.36 The expert panel stresses that caution is particularly important with sulfonamide antimicrobials, such as TMP-SMX, which can cause rare but severe cutaneous adverse reactions, including Stevens-Johnson syndrome, toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS).45 Trimethoprim is commonly combined with sulfamethoxazole to enhance efficacy through complementary and synergistic mechanisms, though it may also be used as monotherapy to avoid sulfonamide reactions.46
Statement 5: When considering minocycline, the ER formulation has fewer acute vestibular side effects than IR in treating acne vulgaris. However, the ER formulation does not mitigate the risk of more serious potential side effects. (SORT Level A). The expert panel notes that differences in formulation account for variation in safety profiles. The lipophilic properties of minocycline enhance its ability to cross the blood-brain barrier, which may contribute to the occurrence of acute vestibular adverse events (AVAEs), including dizziness, vertigo, tinnitus, vomiting, diplopia, and ataxia.47 In the treatment of AV, ER formulations and weight-based dosing of minocycline have been shown to reduce the incidence of AVAEs compared to IR formulations, with a pooled analysis of Phase II and Phase III trials showing minocycline ER (dosed by weight) had vestibular event rates comparable with placebo.20 However, the ER formulation of minocycline has not been shown to definitively reduce the potential risk of serious adverse events, which remain a concern and include autoimmune hepatitis, drug-induced lupus-like syndrome, DRESS, pneumonitis, and vasculitis.48
Minocycline, unlike other tetracyclines, has been associated with a small but measurable risk of drug-induced lupus-like syndrome, estimated at 8.8 cases per 100,000 person-years, with the risk increasing with longer treatment durations.21 In a review of 57 cases of minocycline-induced systemic lupus-like syndrome, nearly all patients experienced polyarthralgia or polyarthritis, often accompanied by liver enzyme abnormalities.49 Additional symptoms included dermatologic manifestations (12 cases), pleuropulmonary involvement (8 cases), and hematologic abnormalities (5 cases), all of which typically resolved upon discontinuation of minocycline.49 Hepatotoxicity associated with minocycline is often an autoimmune response, and typically presents with delayed onset, hepatocellular injury patterns, and responsiveness to corticosteroids along with drug discontinuation.50 The panel concludes that while these findings highlight that ER minocycline may reduce AVAEs, clinicians must remain vigilant about its potential for rare but potentially serious systemic adverse reactions, particularly with prolonged use. In minocycline-associated DRESS syndrome, onset of hepatotoxicity with liver enzyme elevation usually occurs within the first few months after starting minocycline.48
Statement 6: Sarecycline is a narrow-spectrum antibiotic that largely spares gram-negative organisms. With its dual binding sites for C. acnes, it has demonstrated reduced emergence of antibiotic-resistant strains in vitro. Broad-spectrum antibiotics, such as doxycycline and minocycline, may increase the risk of emergence of resistant organisms. (SORT Level C). In the context of antibiotic stewardship, the long-term impact of oral antibiotics on resistance patterns has not been fully defined; however, prolonged changes in the microbiome may persist after oral antibiotic therapy has been stopped.16 Much of the microbiologic data collected derived from in vitro assessments and antibiotic sensitivity profiles rather than from longitudinal clinical studies. In the United States, C. acnes resistance is most prevalent with erythromycin, followed by clindamycin and tetracycline, as reflected by both the frequency of resistant isolates and elevated minimum inhibitory concentrations (MICs).16 Prior to the development of sarecycline, evaluations have shown that resistance is comparatively lower with doxycycline and lowest with minocycline; however, resistance mechanisms such as ribosomal protection and efflux pumps are increasingly common.16,51
Sarecycline was structurally engineered with modifications at the C7 and C9 positions of the tetracycline core to overcome many bacterial resistance mechanisms.11 C. acnes demonstrated a low propensity for resistance to sarecycline, with spontaneous mutation frequencies as low as 10–10 at 4 to 8 times MIC, comparable with minocycline and vancomycin.24 Recent cryogenic electron microscopy studies reveal that sarecycline uniquely binds to two functional sites on the C. acnes 70S ribosome: the A-site of the 30S subunit, inhibiting translation initiation, and the nascent peptide exit tunnel (NPET) of the 50S subunit, impeding early peptide elongation.27 This is in contrast to doxycycline and minocycline, that exert their antibacterial effects by binding to only the A-site of the 30S ribosomal subunit.27 The dual ribosomal binding pattern of sarecycline further reduces the likelihood of the emergence of resistant bacterial organisms, as this would require simultaneous mutations in both ribosomal subunits, which is far less likely to occur.27
The expert panel states that the narrow-spectrum antibacterial profile of sarecycline offers a strategic advantage when selecting an oral antibiotic for AV by delivering targeted efficacy against C. acnes while reducing the bacterial resistance risk commonly associated with broad-spectrum antibiotics. This unique formulation maintains potent activity against gram-positive organisms, including multiple C. acnes strains, while exhibiting significantly reduced activity against aerobic gram-negative bacilli.11,24,52 Importantly, this latter factor is inherent to sarecycline, reduces antibiotic selection pressure within the gastrointestinal (GI) tract microbiome, thus mitigating the emergence of resistant bacterial strains.11,24-27,52,53 Such attributes position sarecycline as a more targeted antibiotic therapy, which emphasizes that broad-spectrum tetracycline agents, in addition to macrolides, cephalosporins, and TMP-SMX, may not be advantageous when choosing an oral antibiotic in the management of AV.11,44,27,52
Statement 7: The negligible activity of sarecycline against gram-negative bacteria leads to less disruption of the human microflora compared to other oral tetracyclines. (SORT Level B). An expanding body of evidence highlights the microbiome-sparing advantages of sarecycline over broader-spectrum tetracyclines in the treatment of AV. In an in vitro human colon model, doxycycline and minocycline significantly disrupted gut microbial diversity, with poor recovery following treatment cessation, whereas sarecycline caused a mild, transient decline in gut flora that fully returned to baseline after discontinuation.25 These findings are supported by MIC and time-kill assays, which demonstrated that sarecycline exhibited significantly less antimicrobial activity than minocycline against key gut commensals, including Escherichia coli, Lactobacillus paracasei, Bifidobacterium adolescentis, and Candida tropicalis.26 Sarecycline also demonstrated higher MICs across several major bacterial phyla present within the GI tract, including Bacteroidota, Actinomycetota, Firmicutes, as well as Propionibacterium freudenreichii.26 Additionally, in studies of normal flora organisms, sarecycline showed markedly lower activity against Enterobacter cloacae (MIC₅₀=32µg/mL) compared to other tetracyclines (MIC₅₀=1-2µg/mL), making it 16 to 32 times less potent than minocycline and doxycycline against aerobic gram-negative bacilli, and 4 to 8 times less active than doxycycline against anaerobic intestinal microbes.24
Sarecycline is also well tolerated. In a randomized, double-blind, placebo-controlled Phase II trial, GI adverse events occurred in only 2.9% of patients treated with sarecycline vs. 6.1% in the placebo group.53 In longer-term Phase III trials (SC1401 and SC1402), GI adverse events (nausea, vomiting) similarly occurred at comparable or lower rates in the sarecycline group relative to placebo.23 A systematic review of Phase II and III trials confirmed this favorable safety profile, reporting low rates of nausea (2.2%) and vomiting (1.0%) with sarecycline, only marginally higher than placebo (1.2% and 0.7%, respectively); rates of vulvovaginal candidiasis were also low (1.1%).11,22,23,29 A notable observation on review of clinical trials with sarecycline is that the reported rates of multiple recognized adverse effects known to occur with specific oral tetracycline antibiotics are low, including GI upset, vulvovaginal candidiasis, and photosensitivity.11,22,23,29,48 The panel concludes that these findings support sarecycline as a narrow-spectrum alternative to broad-spectrum tetracyclines for treatment of AV. Sarecycline may be used as the initial oral antibiotic for treatment of AV as it has established efficacy, minimizes disruption to the gut microbiome, and reduces the potential burden of GI and other tetracycline-associated adverse effects.
Statement 8: Differences exist in the safety profile among the tetracycline antibiotics. (SORT Level A). Oral tetracyclines are commonly associated with adverse effects including gastrointestinal disturbances, vaginal yeast infections, and photosensitivity.11 A review of 13 large interventional and observational studies evaluated the safety of oral tetracyclines—tetracycline, doxycycline, minocycline, and sarecycline—for acne treatment.18 Tetracycline showed general adverse event (AE) rates ranging from 10.3% to 22%, including GI issues such as diarrhea (2.5%), elevated total bilirubin (2.5%), and epigastric pain (1.1%). Additionally, there was an association with an increased risk of new-onset inflammatory bowel disease (IBD).18 Vaginal candidiasis (1.2%) and pseudotumor cerebri (1.2%) were also reported.18
In the same review, doxycycline demonstrated high rates of photosensitivity (15%-30.5%), GI side effects (10%-25%), and vulvovaginal fungal infections at higher doses (2.0%-2.8%) and was linked to an increased risk of IBD.18 In an earlier, smaller, double-blind crossover study, the doxycycline 100mg/day group reported no diarrhea, nausea, or phototoxic reactions.19 Hepatotoxicity associated with oral doxycycline is uncommon, usually presenting as elevation in liver enzymes with short latency (within 60 days) that can sometimes be prolonged.50
Minocycline has demonstrated overall AE rates between 6% to 21%, with marked central nervous system (CNS) effects (17%), AVAEs (~10%), and pigmentation changes, especially at higher doses and with prolonged administration (ie, 200mg/day).18 This drug also carries a 2 to 8 times greater risk of drug-induced lupus-like syndrome (usually with prolonged duration of use) and an increased risk of IBD.18 Among the tetracycline antibiotics, minocycline is most commonly associated with hepatotoxicity, usually as part of an autoimmune or hypersensitivity reaction.50 Minocycline hepatotoxicity generally presents with a long latency, hepatocellular enzyme elevations, and prominent autoimmune features, and is typically responsive to corticosteroid therapy and drug discontinuation.50 However, minocycline hepatotoxicity may also occur early, accompanied by signs of drug hypersensitivity, such as fever, nonspecific diffuse erythema, facial swelling, and eosinophilia (DRESS syndrome).18,50
Sarecycline has demonstrated a favorable safety profile as noted above, with low rates of diarrhea (1.0%-1.2%), nausea (1.9%-4.6%), headache (2.7%-2.9%), photosensitivity (0.2%), and vaginal yeast infections (0.3%-1.1%), thus it appears to have no reported association with IBD or autoimmune conditions to date.18 In two identically designed Phase III trials (SC1401 and SC1402), patients aged 9 to 45 years received either sarecycline once daily with dosing based on weight (1.5mg/kg/day) or placebo once daily for 12 weeks.23 Treatment-emergent adverse events were generally mild and infrequent, with nausea, nasopharyngitis, headache, and vomiting occurring at rates similar to or slightly higher than placebo.23 Additionally, a separate study evaluating sarecycline for cutaneous staphylococcal infections, eyelid styes, and mucous membrane pemphigoid reported significant clinical improvement after short treatment durations as brief as nine days, without any observed adverse events.54 Importantly, oral sarecycline is not approved for the treatment of cutaneous infections. The expert panel notes that based on reported rates and various categories of AEs associated with different oral tetracycline antibiotics, sarecycline offers an overall safe and better tolerated alternative to other oral tetracyclines used to treat AV. The expert panel recognizes that sarecycline has been available for treatment of AV in the US since 2019 and that continued pharmacovigilance is warranted.
Statement 9: Comparative efficacy data for tetracycline antibiotics for AV are sparse and inconsistent. (SORT Level A). Although direct head-to-head studies comparing the efficacy of individual oral tetracyclines for AV are limited, systematic reviews and meta-analyses of randomized controlled trials have found that the oral tetracyclines used to treat AV are effective and generally well tolerated for the treatment of moderate-to-severe inflammatory AV, with no single agent demonstrating clear superiority, including non-tetracycline oral antibiotics.36,55 In a double-blind controlled trial, tetracycline and clindamycin showed comparable efficacy as adjunctive therapies for moderate-to-severe AV, leading to significant reductions in AV lesions and clinical improvement in the majority of patients.56 In another randomized clinical study of 50 patients with AV, participants received either 50mg of doxycycline once daily or 50mg of minocycline twice daily for 12 weeks.57 Clinical improvement or complete clearance was achieved in 78% of patients treated with doxycycline and 82% of those treated with minocycline, while unsatisfactory outcomes were reported in 22% and 18% of patients, respectively. The difference in efficacy between the two regimens was not statistically significant.57
In a double-blind crossover study of 62 patients with predominantly inflammatory AV, oral doxycycline 100mg/day for four weeks led to a good or excellent response in 33% of participants, compared to 22% during the placebo phase.19
Minocycline ER has also demonstrated efficacy in AV management. A review of clinical trials, including two Phase III studies, reported significantly higher treatment success rates at Week 12 with ER-minocycline: 17.3% and 15.9% of patients achieved success compared to 7.9% and 9.5% with placebo (p=0.006 and p=0.018, respectively).20 Additionally, a broader review of 39 RCTs involving 6013 participants confirmed that minocycline is effective for moderate-to-severe AV but found no evidence that it is more effective than other commonly used treatments.21
Sarecycline has been shown to be efficacious for treating AV. In two Phase III trials (SC1401 and SC1402), sarecycline achieved significantly higher Investigator Global Assessment (IGA) facial AV success rates, defined as at least a 2-grade reduction in IGA score from baseline and a final IGA score of 0 or 1 (clear or almost clear), at Week 12—21.9% and 22.6%—compared to 10.5% and 15.3% with placebo (p<0.0001 and p=0.0038).23 Efficacy for truncal AV (chest and back) was also evaluated as a secondary analysis based on IGA assessments, with statistically significant improvements in IGA scores for the chest (29.6% and 36.6%) and back (32.9% and 33.2%) vs. placebo (chest: 19.6% and 21.6%; back: 17.1% and 25.7%).23
Subgroup analyses further support the efficacy of sarecycline for AV across diverse populations. A pooled post hoc analysis of two Phase III trials showed a 26% reduction in facial inflammatory AV lesions by Week 3 and a 55% reduction by Week 12 in Hispanic patients with moderate-to-severe AV.29 Similarly, in a 12-week study of Chinese patients, sarecycline reduced facial inflammatory AV lesions by 73.2% compared to 46.0% with placebo (p<0.001), with 39.7% achieving clear or almost clear skin vs. 16.2% in the placebo group (p<0.001).30 Taken together, these findings highlight that while the available oral tetracyclines are effective options for treatment of moderate-to-severe AV, newer agents like sarecycline may offer added benefit in terms of a narrower antibiotic spectrum, favorable tolerability and safety, and studies evaluating therapeutic outcomes across diverse patient populations.
Statement 10: Patient-reported outcomes (PROs) were not routinely captured in acne studies until the past decade. Recent studies demonstrate doxycycline, minocycline, and sarecycline improve PROs. (SORT Level B). Acne can significantly affect an individual’s quality of life, body image, and self-esteem, often leading to psychological challenges that impact social interactions, relationships, and overall mental health.58,59 Tools such as questionnaires, scales, and structured interviews used to assess psychosocial effects, including anxiety, depression, and self-esteem, have been administered for patients with other inflammatory dermatoses but have only recently been routinely employed in AV studies.60 One such tool developed in 2013, the Acne Symptom and Impact Scale (ASIS), is a PRO measure developed through literature review, expert input, and patient interviews that captures the physical and emotional burden of facial AV from the patient’s perspective.61 Frequently reported impacts included issues with appearance, mood, embarrassment, confidence, and social withdrawal, forming the basis of the 17-item ASIS following refinement through cognitive interviews.62 Given the relatively recent development of this assessment tool, the expert panel emphasizes that prior oral antibiotic trials lack measurement of baseline parameters and robust analysis of the impact of treatment on patients’ psychosocial wellbeing, making interpretation and comparison difficult. However, an emphasis on outcomes of PRO data with ASIS assessment exist for sarecycline, doxycycline, and minocycline.
In the Patient Reported Outcomes for Sarecycline Effectiveness and Safety (PROSES) study, a single-arm, 12-week clinical trial of patients aged 9 years and older with moderate-to-severe non-nodular AV that placed an emphasis on PRO, treatment with oral sarecycline based on body weight (60mg, 100mg, or 150mg) led to significant improvements in ASIS (mean±SD) scores for signs (–0.8±0.7), impact (–1.0±1.0), emotional impact (–1.2±1.1), and social impact (–0.6±1.1) (p<0.0001), along with an IGA success rate of 58.9% and physician satisfaction of 88.1%.28 Similarly, in a multicenter, double-blind study comparing spironolactone and doxycycline, quality of life measured by the Cardiff Acne Disability Index (CADI) improved more substantially with spironolactone by month 6 (2.56±2.38 vs. 3.44±2.44).63 Furthermore, in a randomized trial involving adults with major depressive disorder, minocycline significantly improved measures of life satisfaction and functioning, despite no significant change in core depression scores, supporting its potential benefit on broader wellbeing (Montgomery-Asberg, p<0.001; Clinical Global Impression-Improvement [CGI-I], p=0.02).64 While doxycycline was found to be less effective than spironolactone in improving quality of life, tetracycline-class antibiotics overall demonstrate meaningful benefits in reducing AV severity and enhancing patients’ emotional wellbeing, and daily functioning.
Strengths and limitations. The Delphi method encompasses both inherent advantages and limitations. A notable constraint is its reliance on expert consensus, which may prioritize clinical opinion over empirical evidence for the formulation of consensus statements. However, the present study addressed this limitation by incorporating high-quality, peer-reviewed clinical research to inform and support the development of its recommendations. Consequently, in this methodological context, the Delphi process functioned as a rigorous and systematic approach for generating evidence-informed clinical guidelines.
Conclusion
Following a thorough review of the literature, these 10 expert consensus statements aim to provide optimal clinical guidance on the use of oral antibiotics for AV through summarizing best practices, highlighting gaps and nuances in research, and promoting antibiotic stewardship. This expert panel concludes that oral antibiotics remain an important therapeutic option used in combination with topical therapy for moderate-to-severe AV management but need to be used judiciously, and for limited durations, to balance efficacy with antibiotic stewardship and safety. Among tetracyclines, both ER minocycline and sarecycline have formal FDA approval. The targeted narrow-spectrum action of sarecycline, along with its microbiome-sparing benefits based on microbiologic data and overall safety profile are uniquely beneficial in the management of AV. While all tetracyclines effectively reduce AV severity and improve patient quality of life, careful patient-specific selection is essential to minimize risks, optimize outcomes, and support long-term physical and psychological wellbeing. The expert panel recommends that sarecycline should be strongly considered for initial (first line) oral antibiotic treatment for AV due to its targeted action, lower bacterial resistance risk, and favorable tolerability and safety.
References:
- Chen H, Zhang TC, Yin XL, Man JY, Yang XR, Lu M. Magnitude and temporal trend of acne vulgaris burden in 204 countries and territories from 1990 to 2019: an analysis from the Global Burden of Disease Study 2019. Br J Dermatol. 2022;186(4):673-683.
- Tan JKL, Bhate K. A global perspective on the epidemiology of acne. Br J Dermatol. 2015;172 (Suppl 1):3-12.
- Dréno B, Thiboutot D, Layton AM, et al. Large-scale international study enhances understanding of an emerging acne population: adult females. J Eur Acad Dermatol Venereol. 2015;29(6):1096-1106.
- Tan JKL, Tang J, Fung K, et al. Development and validation of a comprehensive acne severity scale. J Cutan Med Surg. 2007;11(6):211-216.
- Gollnick HPM. From new findings in acne pathogenesis to new approaches in treatment. J Eur Acad Dermatol Venereol. 2015;29(Suppl 5):1-7.
- Dréno B, Thiboutot D, Gollnick H, et al. Global Alliance to Improve Outcomes in Acne. Antibiotic stewardship in dermatology: limiting antibiotic use in acne. Eur J Dermatol. 2014;24(3):330-334.
- Leyden JJ, Del Rosso JQ, Webster GF. Clinical considerations in the treatment of acne vulgaris and other inflammatory skin disorders: a status report. Dermatol Clin. 2009;27(1):1-15.
- Reynolds RV, Yeung H, Cheng CE, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2024;90(5):1006.e1-1006.e30.
- Del Rosso JQ. Recently approved systemic therapies for acne vulgaris and rosacea. Cutis. 2007;80(2):113-120.
- Kaul G, Saxena D, Dasgupta A, Chopra S. Sarecycline hydrochloride for the treatment of acne vulgaris. Drugs Today (Barc). 2019;55(10):615-625.
- Del Rosso JQ. Sarecycline and the narrow-spectrum tetracycline concept: currently available data and potential clinical relevance in dermatology. J Clin Aesthet Dermatol. 2020;13(10):45-48.
- Hsu CC, Sanford B. The Delphi technique: making sense of consensus. Practical Assess Res Eval. 2007;12(10):1-8.
- Berman B, Ceilley R, Cockerell C, et al. Appropriate use criteria for the integration of diagnostic and prognostic gene expression profile assays into the management of cutaneous malignant melanoma: an expert panel consensus-based modified Delphi process assessment. SKIN J Cutan Med. 2019;3(5):291-306.
- Burshtein J, Zakria D, Shah M, et al. Advances in technology for melanoma diagnosis and prognosis: an expert consensus panel. J Drugs Dermatol. 2024;23(9):774-781.
- Zakria D, Brownstone N, Berman B, et al. Incorporating prognostic gene expression profile assays into the management of cutaneous melanoma: an expert consensus panel. SKIN J Cutan Med. 2023;7(1):556–569.
- Del Rosso JQ, Webster GF, Rosen T, et al. Status report from the Scientific Panel on Antibiotic Use in Dermatology of the American Acne and Rosacea Society: part 1: antibiotic prescribing patterns, sources of antibiotic exposure, antibiotic consumption and emergence of antibiotic resistance, impact of alterations in antibiotic prescribing, and clinical sequelae of antibiotic use. J Clin Aesthet Dermatol. 2016;9(4):18-24.
- Del Rosso JQ, Gallo RL, Thiboutot D, et al. Status report from the Scientific Panel on Antibiotic Use in Dermatology of the American Acne and Rosacea Society, part 2: perspectives on antibiotic use and the microbiome and review of microbiologic effects of selected specific therapeutic agents commonly used by dermatologists. J Clin Aesthet Dermatol. 2016;9(5):11–17.
- Armstrong AW, Hekmatjah J, Kircik LH. Oral tetracyclines and acne: a systematic review for dermatologists. J Drugs Dermatol. 2020;19(11):s6-s13.
- Plewig G, Petrozzi JW, Berendes U. Double-blind study of doxycycline in acne vulgaris. Arch Dermatol. 1970;101(4):435-438.
- Torok HM. Extended-release formulation of minocycline in the treatment of moderate-to-severe acne vulgaris in patients over the age of 12 years. J Clin Aesthet Dermatol. 2013;6(7):19-22.
- Garner SE, Eady A, Bennett C, Newton JN, Thomas K, Popescu CM. Minocycline for acne vulgaris: efficacy and safety. Cochrane Database Syst Rev. 2012;2012(8):CD002086.
- Moore AY, Del Rosso J, Johnson JL, Grada A. Sarecycline: a review of preclinical and clinical evidence. Clin Cosmet Investig Dermatol. 2020;13:553-560.
- Moore A, Green LJ, Bruce S, et al. Once-daily oral sarecycline 1.5 mg/kg/day is effective for moderate-to-severe acne vulgaris: results from two identically designed, phase 3, randomized, double-blind clinical trials. J Drugs Dermatol. 2018;17(9):987-996.
- Zhanel G, Critchley I, Lin LY, Alvandi N. Microbiological profile of sarecycline, a novel targeted spectrum tetracycline for the treatment of acne vulgaris. Antimicrob Agents Chemother. 2018;63(1):e01297-18.
- Moura IB, Grada A, Spittal W, et al. Profiling the effects of systemic antibiotics for acne, including the narrow-spectrum antibiotic sarecycline, on the human gut microbiota. Front Microbiol. 2022;13:901911.
- Ghannoum MA, Long L, Bunick CG, et al. Sarecycline demonstrated reduced activity compared to minocycline against microbial species representing human gastrointestinal microbiota. Antibiotics (Basel). 2022;11(3):324.
- Lomakin IB, Devarkar SC, Patel S, Grada A, Bunick CG. Sarecycline inhibits protein translation in Cutibacterium acnes 70S ribosome using a two-site mechanism. Nucleic Acids Res. 2023;51(6):2915-2930.
- Baldwin HE, Graber E, Harper JC, et al. Sarecycline improves acne severity, symptoms, and psychosocial burden in non-nodular acne vulgaris: PROSES study. J Drugs Dermatol. 2024;23(2):SF405634s12-SF405634s18.
- Moore AY, Hurley K, Moore SA. A post hoc analysis of efficacy data on sarecycline in Hispanics with acne from two phase 3, multicenter, randomized, double-blind, placebo-controlled clinical trials. Antibiotics (Basel). 2023;12(1):89.
- Baykal T, Chen X, Zhang J, et al. Efficacy and safety of sarecycline in Chinese patients with acne vulgaris: a randomized, multicenter, double blind, placebo controlled, phase 3 trial. J Am Acad Dermatol. 2024;91(3 Suppl):AB195.
- Mohammad AP, Bae GH. A cross-sectional study analyzing recent trends in the treatment of acne vulgaris: prescription patterns and demographic data from the 2018-2019 National Ambulatory Medical Care Survey (NAMCS) database. Cureus. 2025;17(2):e79170.
- Purdy S, Deberker D. Acne vulgaris. BMJ Clin Evid. 2008;2008:1714.
- Sanchez C, Keri J. Androgen receptor inhibitors in the treatment of acne vulgaris: efficacy and safety profiles of clascoterone 1% cream. Clin Cosmet Investig Dermatol. 2022;15:1357-1366.
- Leung AK, Barankin B, Lam JM, Leong KF, Hon KL. Dermatology: how to manage acne vulgaris. Drugs Context. 2021;10:2021-8-6.
- Thiboutot DM, Dréno B, Abanmi A, et al. Practical management of acne for clinicians: an international consensus from the Global Alliance to Improve Outcomes in Acne. J Am Acad Dermatol. 2018;78(2 Suppl 1):S1-S23.e1.
- Bienenfeld A, Nagler AR, Orlow SJ. Oral antibacterial therapy for acne vulgaris: an evidence-based review. Am J Clin Dermatol. 2017;18(4):469-490.
- Grossman TH. Tetracycline antibiotics and resistance. Cold Spring Harb Perspect Med. 2016;6(4):a025387.
- Adler BL, Kornmehl H, Armstrong AW. Antibiotic resistance in acne treatment. JAMA Dermatol. 2017;153(8):810-811.
- Asai Y, Baibergenova A, Dutil M, et al. Management of acne: Canadian clinical practice guideline. CMAJ. 2016;188(2):118-126.
- Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris [published correction appears in J Am Acad Dermatol. 2020 Jun;82(6):1576. doi: 10.1016/j.jaad.2020.02.010.]. J Am Acad Dermatol. 2016;74(5):945-73.e33.
- López-Estebaranz JL, Herranz-Pinto P, Dréno B; el grupo de dermatólogos expertos en acné. Consensus-based acne classification system and treatment algorithm for Spain. Consenso español para establecer una clasificación y un algoritmo de tratamiento del acné. Actas Dermosifiliogr. 2017;108(2):120-131.
- Oon HH, Wong SN, Aw DCW, Cheong WK, Goh CL, Tan HH. Acne management guidelines by the Dermatological Society of Singapore. J Clin Aesthet Dermatol. 2019;12(7):34-50.
- Acne vulgaris: management. In: NICE Guideline, No.198. National Institute for Health and Care Excellence (NICE); 2023. https://www.ncbi.nlm.nih.gov/books/NBK573056/
- Shutter MC, Akhondi H. Tetracycline. In: StatPearls. StatPearls Publishing; 2025-. Updated June 3, 2023. https://www.ncbi.nlm.nih.gov/books/NBK549905/
- Khan DA, Knowles SR, Shear NH. Sulfonamide hypersensitivity: fact and fiction. J Allergy Clin Immunol Pract. 2019;7(7):2116-2123.
- Gleckman R, Blagg N, Joubert DW. Trimethoprim: mechanisms of action, antimicrobial activity, bacterial resistance, pharmacokinetics, adverse reactions, and therapeutic indications. Pharmacotherapy. 1981;1(1):14-20.
- Fleischer AB, Dinehart S, Stough D, Plott RT; Solodyn Phase 2 Study Group; Solodyn Phase 3 Study Group. Safety and efficacy of a new extended-release formulation of minocycline. Cutis. 2006;78(4 suppl):21– 31.
- Ochsendorf F. Minocycline in acne vulgaris: benefits and risks. Am J Clin Dermatol. 2010;11(5):327-341.
- Schlienger RG, Bircher AJ, Meier CR. Minocycline-induced lupus. a systematic review. Dermatology. 2000;200(3):223-231.
- Tetracyclines. In: Liver Tox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Updated April 10, 2019. https://www.ncbi.nlm.nih.gov/books/NBK548040/
- Schnappinger D, Hillen W. Tetracyclines: antibiotic action, uptake, and resistance mechanisms. Arch Microbiol. 1996;165(6):359-369.
- Moore AY, Charles JEM, Moore S. Sarecycline: a narrow spectrum tetracycline for the treatment of moderate-to-severe acne vulgaris. Future Microbiol. 2019;14(14):1235-1242.
- Leyden JJ, Sniukiene V, Berk DR, Kaoukhov A. Efficacy and safety of sarecycline, a novel, once-daily, narrow spectrum antibiotic for the treatment of moderate-to-severe facial acne vulgaris: results of a phase 2, dose-ranging study. J Drugs Dermatol. 2018;17(3):333-338.
- Grada A, Ghannoum MA, Bunick CG. Sarecycline demonstrates clinical effectiveness against staphylococcal infections and inflammatory dermatoses: evidence for improving antibiotic stewardship in dermatology. Antibiotics (Basel). 2022;11(6):722.
- Simonart T, Dramaix M, De Maertelaer V. Efficacy of tetracyclines in the treatment of acne vulgaris: a review. Br J Dermatol. 2008;158(2):208-216.
- Panzer JD, Poche W, Meek TJ, Derbes VJ, Atkinson W. Acne treatment: a comparative efficacy trial of clindamycin and tetracycline. Cutis. 1977;19(1):109-111.
- Laux B. Treatment of acne vulgaris. A comparison of doxycycline versus minocycline. [Article in German]. Hautarzt. 1989;40(9):577-581.
- Davern J, O’Donnell AT. Stigma predicts health-related quality of life impairment, psychological distress, and somatic symptoms in acne sufferers. PLoS One. 2018;13(9):e0205009.
- Lasek RJ, Chren MM. Acne vulgaris and the quality of life of adult dermatology patients. Arch Dermatol. 1998;134(4):454-458.
- Stamu-O’Brien C, Jafferany M, Carniciu S, Abdelmaksoud A. Psychodermatology of acne: psychological aspects and effects of acne vulgaris. J Cosmet Dermatol. 2021;20(4):1080-1083.
- Zaraa I, Belghith I, Ben Alaya N, Trojjet S, Mokni M, Ben Osman A. Severity of acne and its impact on quality of life. Skinmed. 2013;11(3):148-153.
- Alexis A, Daniels SR, Johnson N, Pompilus F, Burgess SM, Harper JC. Development of a new patient-reported outcome measure for facial acne: the Acne Symptom and Impact Scale (ASIS). J Drugs Dermatol. 2014;13(3):333-340.
- Dréno B, Nguyen JM, Hainaut E, et al. Efficacy of spironolactone compared with doxycycline in moderate acne in adult females: results of the multicentre, controlled, randomized, double-blind prospective and parallel female acne spironolactone vs doxycycline efficacy (FASCE) study. Acta Derm Venereol. 2024;104:adv26002.
- Dean OM, Kanchanatawan B, Ashton M, et al. Adjunctive minocycline treatment for major depressive disorder: A proof of concept trial. Aust N Z J Psychiatry. 2017;51(8):829-840.