J Clin Aesthet Dermatol. 2025;18(11):44–47.
by Carolina Puyana, MD, MSPH, and Jose R. Montes, MD
Dr. Puyana is with Skin Center Boston in Brookline, Massachusetts. Dr. Montes is with the Department of Ophthalmology at the University of Puerto Rico in San Juan, Puerto Rico and José Raúl Montes Eyes & Facial Rejuvenation in San Juan, Puerto Rico.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Montes is a speaker, trainer, and advisory board member for Allergan-AbbVie, Galderma, and Merz. Dr. Puyana has no conflicts of interest to disclose.
ABSTRACT: Objective: This retrospective study aims to assess the extended efficacy of tear trough hyaluronic acid (TT-HA) filler treatments beyond the commonly reported duration of 6 to 12 months. Methods: A retrospective analysis of 155 patients who received TT-HA filler treatments from 2007 to 2023 was conducted. Patient records and photographs were reviewed, and the severity of infraorbital hollowing was graded using the Merz Infraorbital Hollow Assessment Scale (MIHAS). Multivariate regression models were used to analyze factors influencing treatment longevity. Results: Most patients were female (84%) with a mean age of 48 years. Moderate-to-severe infraorbital hollowing was most prevalent at baseline. On average, 0.45mL of filler was injected into each infraorbital hollow using a 27 gauge x 1-inch cannula. Various HA filler products were used, including Belotero Balance®, Juvederm Vollure® XC, Restylane®, and Juvederm Volbella® XC. Most patients experienced an improvement in MIHAS grade posttreatment, with significant results persisting at 18 months. Multivariate regression analysis revealed sustained efficacy over time, with no significant differences in MIHAS grade changes between 6, 12, and 18-month follow-up periods. Conclusion: This study challenges conventional beliefs by demonstrating the extended efficacy of TT-HA fillers, providing evidence of significant improvement in infraorbital hollowing up to 18 months posttreatment. These findings offer valuable insights for clinicians and patients, guiding expectations, and treatment planning in cosmetic dermatology practice. Further research is warranted to elucidate factors contributing to the prolonged longevity of TT-HA fillers to optimize treatment outcomes. Keywords: Tear trough, hyaluronic acid filler, infraorbital hollowing, periorbital rejuvenation
Introduction
Correction of the tear trough (TT) deformity with hyaluronic acid (HA) filler is a minimally invasive procedure with high patient satisfaction and long-term results. The duration of effect reported in the literature ranges from 8 to 12 months, with an average effect of 10.8 months.1 This retrospective study aims to assess the long-term effects of TT-HA filler treatments. By evaluating before and after photographs of 155 patients who received TT-HA filler, this study seeks to investigate whether the duration of improvement in the infraorbital area exceeds the widely reported timeframe.
Methods
Study design. A comprehensive analysis of patient records and photographic documentation was conducted for 155 individuals who underwent TT-HA filler treatments from 2007 to 2023. The Merz Infraorbital Hollow Assessment Scale (MIHAS)2, ranging from 0 (none to minimal) to 4 (extreme), was used to grade the severity of undereye depressions based on patient photographs taken at baseline and follow-up. Patients’ progress was monitored at a close follow-up visit, and a subset of patients underwent extended follow-up visits as well. All treatments were performed by a single oculoplastics surgeon at a private practice office. Patient rating was performed by a board-certified dermatologist blinded to the timing of such photographs. Patients provided informed consent and photoconsent.
Statistical analysis. Cases were identified, and baseline and follow-up photographs were collected from the patient charts. Patient demographics and treatment characteristics were summarized. Multivariate logistic regression models controlling for product used, amount injected, and patient age were fitted to analyze factors influencing treatment longevity. The significance level was set at p<0.05. Odds ratios (OR), 95% confidence intervals (95% CI), and p-values were calculated. SAS software, version 8.6 (SAS Institute Inc.), was used to perform all data analysis.
Results
The majority of participants identified as female 130 (84%) and had a mean age of 48 years. At baseline, 53 patients had moderate, 50 had severe, 34 had extreme, and 20 had mild infraorbital hollows. On average, 0.45 (standard deviation [SD]: ±0.1) mL of product was injected per infraorbital hollow. Most cases were performed utilizing a 27 gauge × 1-inch cannula (n=102; 82%). Products used included: Belotero Balance® (n=36; 23%), Juvederm Vollure XC® (n=28; 18%), Restylane® (n=26; 17%), Restylane-L® (n=26; 17%), and Juvederm Volbella XC® (n=25; 16%), among others (Table 1). Most patients who had TT-HA filler treatment saw an improvement in infraorbital hollowing of 1 MIHAS grade (n=105; 68%), then 2 MIHAS grades (n=22; 14%) and some saw subjective improvement (n=23; 15%) however not enough to indicate an improvement in grade (Table 2). The extended follow-up photographs demonstrated that the results of TT-HA fillers persisted well beyond the commonly reported duration of 6 to 12 months. Mean follow-up was 785 ± 536 days. Multivariate regression models controlling for filler type, amount used, and patient age demonstrated no significant differences in changes in MIHAS grade at 12 or 18 months compared with 6-month follow-up (p=0.25; p=0.57). Patients evaluated after 18 months were less likely to demonstrate a change in MIHAS grade compared with those at the 6-month follow-up (p=0.008).
Discussion
The natural anatomical features of the lid-cheek junction undergo accentuation with age due to alterations in the skin and periorbital supporting structures. Over time, a deepening hollow emerges from the inner corner of the eye, running parallel and just below the lower bony ridge of the eye socket. In some patients, lack of volume and fat loss in the area may lead to a sunken appearance beneath the eyes. In others, herniated orbital fat leads to a swollen or puffy appearance under the lower lid. Additionally, pigmentation and skin texture abnormalities in the area may contribute to an overall aged and tired appearance.1 Managing TT deformities poses a significant challenge for aesthetic providers, as correcting these features often requires a multifactorial approach.3 TT-HA fillers are an effective treatment to replenish volume and restore support in the area.4 A recent meta-analysis examining the effectiveness and safety of TT rejuvenation with HA filler demonstrated a low rate of complications and a high level of patient satisfaction with this procedure.5
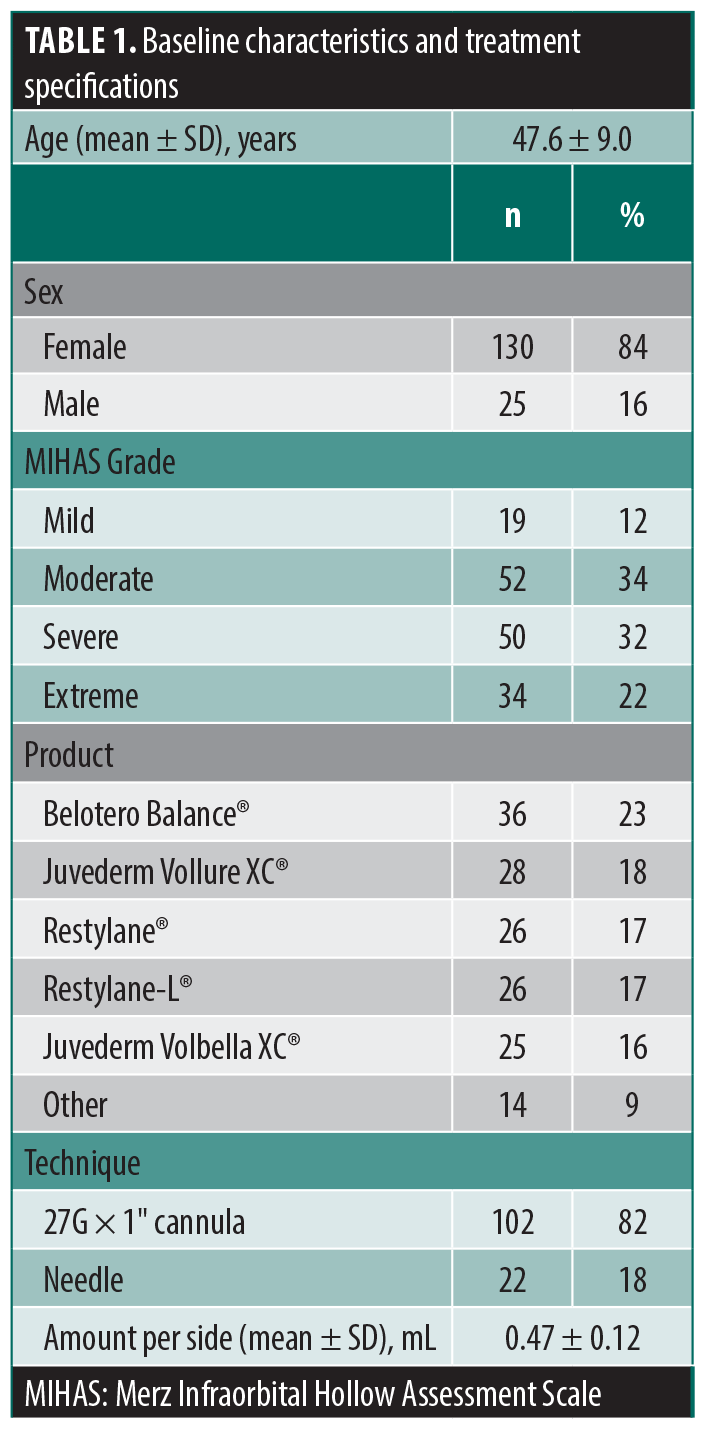
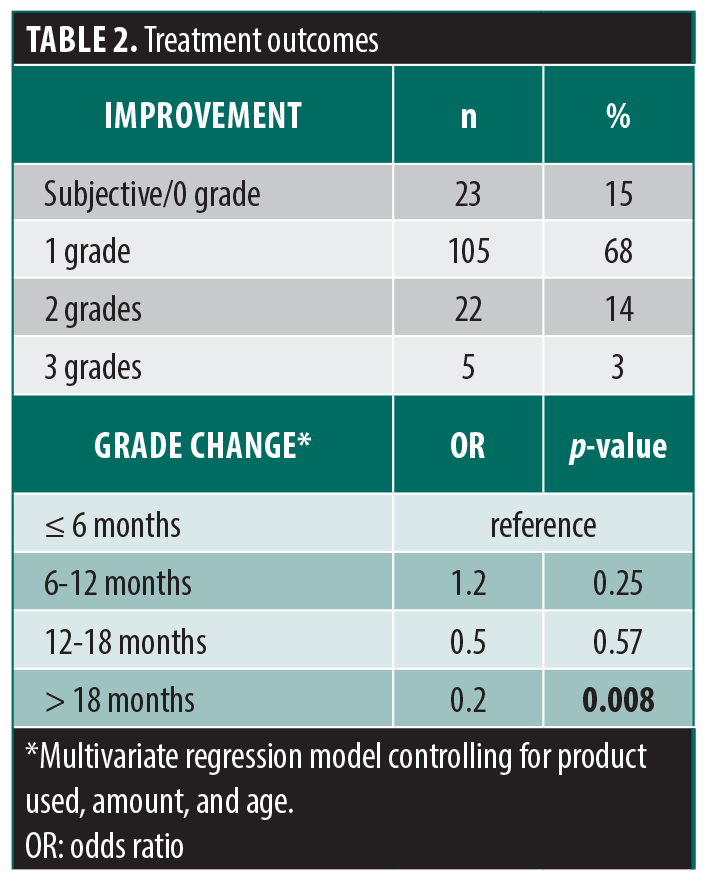
Studies show an average subjective effect duration of 10.8 months and an objective mean volume augmentation duration of 14.4 months, measured through 3D imaging.6 The results of our study demonstrate that TT-HA fillers offer a longer-lasting solution than previously thought, with significant results up to 18 months after treatment (Figure 1). Our clinical experience suggests that the results of TT-HA fillers may even be evident after 24 months (Figure 2). The extended longevity and product retention in the area is a common trait among most injectable HA gels. This phenomenon has been previously reported, with extended retention associated with the particular deformation characteristics of cross-linked HA.7
Most patients in our study were treated utilizing a 27G × 1″ cannula. Currently, there are no standard guidelines regarding TT- HA filler treatment with needle vs. cannula. A recent review study found no statistically significant differences in reported aesthetic results, patient satisfaction, or incidence of adverse events between needle and cannula.8 Conversely, in a meta-analysis of prospective clinical trials, the use of needles was associated with a significantly higher rate of ecchymosis, and cannula use was associated with a greater risk of edema.9 Nevertheless, a systematic review of patient satisfaction found similar patient satisfaction rates regardless of the technique used.10
The most common complications of infraorbital filler include ecchymosis, edema, blue-gray discoloration (Tyndall effect), and contour irregularities.11 TT-HA filler adverse effects typically manifest as mild and temporary, but there is the possibility of these effects to continue at long-term follow-ups or appear as delayed complications. A recent systematic review on delayed complications of TT fillers demonstrated that the most common delayed complication with any type of dermal filler was swelling.12 Other delayed complications reported included lumps or nodules, xanthelasma-like reaction, migration, and discoloration. On average, delayed complications occurred after 16.8 months, with xanthelasma-like reactions appearing earliest (10 months) and discoloration occurring later (52 months).12
The precise etiology of malar edema remains poorly understood, and various theories have been proposed.13 It is suspected that it may indicate a low-grade inflammatory reaction resulting from hypersensitivity to the HA gel, which in some cases is not properly degraded. Also, the drawing of fluid by the HA may be intensified in individuals with a tendency toward fluid retention. In terms of blue-gray dyschromia, light-skinned patients with thin skin are most susceptible to this adverse effect, and it may become more apparent after repeat injections.13 Deep injections, below the orbicularis oculi muscle or in the preperiosteal plane, with conservative filler volumes seem to decrease the likelihood of these occurrences.14 In some patients, delayed blue-gray discoloration may worsen as the filler product migrates anteriorly over time. This migration may also manifest as delayed contour irregularities, resembling an orbital ridge or nodules. Other potential explanations for delayed nodule formation are late capsular contraction of HA gels with higher resistance to deformation or a chronic granulomatous reaction to biofilm development.7,13,15 Oftentimes, these complications may be addressed through massage or hyaluronidase injection. Lastly, rare yet serious complications associated with TT-HA fillers include infections and ischemic adverse events, such as blindness.
TT-HA fillers are an effective and safe treatment to improve the appearance of TT deformities when performed by an experienced injector who comprehends the anatomy of the infraorbital region and employs the appropriate injection techniques for the patient in question. It is crucial to understand that the initial high patient satisfaction and low complication rates are most noticeable within the first 6 months posttreatment. However, the lasting permanence of the HA filler in this area may lead to long-term dissatisfaction. Over time, what was once a positive result may degrade into product visibility, edema, blue-gray discoloration (Tyndall effect), and contour irregularities. Based on the present understanding of the longevity of TT-HA fillers, it is important to exercise caution when treating the TT to avoid overfilling the area, mitigating delayed complications.
Limitations of this study include the evaluation of front-facing photographs only, restricting our ability to assess the changes to the three-dimensional architecture of the infraorbital hollows. Also, factors including patient satisfaction and treatments performed by other practitioners were not included. Additionally, as a retrospective study, inherent limitations include selection bias, variable follow-up intervals, and lack of standardized data collection. However, this practical approach represents real-world clinical outcomes and treatment longevity. Our results imply that TT-HA fillers may last longer in practice, even though pivotal US Food and Drug Administration trials usually report shorter durations. This is a significant consideration that calls for additional prospective research.
Conclusion
This retrospective study provides evidence supporting the extended results of TT-HA fillers. The comprehensive evaluation of 155 patients, challenges conventional wisdom, suggesting that TT-HA fillers offer a longer-lasting solution than previously thought with significant results up to 18 months after treatment. While longer duration offers advantages, it also increases the risk of delayed complications. Such insights contribute significantly to the field of cosmetics and can guide providers in offering more accurate and informed expectations to patients seeking infraorbital rejuvenation.
References:
- Trinh LN, Grond SE, Gupta A. Dermal fillers for tear trough rejuvenation: a systematic review. Facial Plast Surg. 2022;38(03):228-239.
- Biesman B, Verma A, Cheng N, Duncan A. Development and validation of a photonumeric scale for evaluation of infraorbital hollowing. J Drugs Dermatol. 2023;22(1):74-81.
- Anido J, Fernández JM, Genol I, Ribé N, Pérez Sevilla G. Recommendations for the treatment of tear trough deformity with cross-linked hyaluronic acid filler. J Cosmet Dermatol. 2021;20(1):6-17.
- Jiang J, Wang X, Chen R, et al. Tear trough deformity: different types of anatomy and treatment options. Postepy Dermatol Alergol. 2016;33(4):303-308.
- Liu X, Gao Y, Ma J, Li J. The efficacy and safety of hyaluronic acid injection in tear trough deformity: a systematic review and meta-analysis. Aesthetic Plast Surg. 2024;48(3):478-490.
- Donath AS, Glasgold RA, Meier J, Glasgold MJ. Quantitative evaluation of volume augmentation in the tear trough with a hyaluronic acid-based filler: a three-dimensional analysis. Plast Reconstr Surg. 2010;125(5):1515-1522.
- Dayan SH, Arkins JP, Somenek M. Restylane persisting in lower eyelids for 5 years. J Cosmet Dermatol. 2012;11(3):237-238.
- Rao BK, Berger LE, Reilly C, Alamgir M, Galadari H. Tear trough filler techniques utilizing hyaluronic acid: a systematic review. Plast Reconstr Surg. 2022;149(5):1079-1087.
- Nikolis A, Enright KM, Berros P, Sampalis JS. Safety of infraorbital hyaluronic acid injections: outcomes of a meta-analysis on prospective clinical trials. J Cosmet Dermatol. 2023;22(9):2382-2390.
- Gorbea E, Kidwai S, Rosenberg J. Nonsurgical tear trough volumization: a systematic review of patient satisfaction. Aesthet Surg J. 2021;41(8):NP1053-NP1060.
- Reddy S, Nguyen TA, Gharavi N. Complications associated with infraorbital filler injection. J Cosmet Laser Ther. 2020;22(6-8):226-229.
- Trinh LN, McGuigan KC, Gupta A. Delayed complications following dermal filler for tear trough augmentation: a systematic review. Facial Plast Surg. 2022;38(3):250-259.
- Mustak H, Fiaschetti D, Goldberg RA. Filling the periorbital hollows with hyaluronic acid gel: long-term review of outcomes and complications. J Cosmet Dermatol. 2018;17(4):611-616.
- Goldberg RA, Fiaschetti D. Filling the periorbital hollows with hyaluronic acid gel: initial experience with 244 injections. Ophthal Plast Reconstr Surg. 2006;22(5):335-341; discussion 341-343.
- Marusza W, Mlynarczyk G, Olszanski R, et al. Probable biofilm formation in the cheek as a complication of soft tissue filler resulting from improper endodontic treatment of tooth 16. Int J Nanomedicine. 2012;7:1441-1447.