J Clin Aesthet Dermatol. 2026;19(3):15–19.
by Chandler E. Johnson, MD; Kareem G. Elhage, MD; Riley K. Spencer, DO; Joy Q. Jin, MD; Samuel Yeroushalmi, MD; Mitchell S. Davis, MD; Marwa Hakimi, MD; Wilson Liao, MD; and Tina Bhutani, MD, MAS, FAAD
All authors are with the Department of Dermatology at the University of California San Francisco, San Francisco, California.
FUNDING: This study was sponsored by Regeneron Pharmaceuticals.
DISCLOSURES: Dr. Jin has received research grant funding from the National Psoriasis Foundation and institutional funding from the University of California, San Francisco. Dr. Bhutani has received research grant funding from Novartis and Regeneron and is a principal investigator for trials sponsored by AbbVie, Castle, CorEvitas, Dermavant, Galderma, Mindera, and Pfizer. Dr. Bhutani has also served as an advisor for AbbVie, Arcutis, Boehringer Ingelheim, Bristol Myers Squibb, Janssen, LEO Pharma, Lilly, Novartis, Pfizer, Sun, and UCB. Dr. Liao has received research grant funding from AbbVie, Amgen, Janssen, LEO Pharma, Novartis, Pfizer, Regeneron, and TRex Bio. The remaining authors have nothing to disclose.
ABSTRACT: Objective: This study aims to prospectively evaluate the emotional impact of dupliumab treatment on patients with atopic dermatitis (AD) using a validated psychometric instrument, the Psychological General Wellbeing (PGWB) index. This index is widely used in nondermatologic conditions and assesses self-representations of intrapersonal affective and emotional states reflecting a sense of subjective wellbeing or distress. Methods: This was a Phase IV, open-label clinical trial using dupilumab for the treatment of moderate-to-severe AD. The primary endpoint was the change in PGWB score at Week 16 of dupilumab treatment compared to baseline. Results: A total of 24 participants completed the study. The change in average PGWB score after 16 weeks of treatment with dupilumab compared to baseline was not statistically significant (5.8±19.7; P=0.15). There was significant improvement in PGWB score at Week 52 of treatment (8.5±17.4; P=0.03). Mean Eczema Area and Severity Index (EASI) improvements at Weeks 16 and 52 were 60.3% and 76.8%, respectively. Regarding Investigator Global Assessment (IGA), 29.2% and 41.7% of participants achieved a score of 0 or 1 at Weeks 16 and 52, respectively. Limitations: This study did not have a control arm and was limited by small sample size. Conclusion: Treatment with dupilumab for 52 weeks was associated with improvement of psychological wellbeing, suggesting that intervention with this interleukin (IL) 4/IL-13 inhibitor is capable of restoring quality of life in these patients. Trial Administration: NCT03667014 Keywords: Atopic dermatitis, dupilumab, clinical trials, quality of life, mental health
Introduction
Atopic dermatitis (AD) is a chronic, inflammatory skin condition characterized by severe pruritus and recurring eczematous lesions.1 The disease affects pediatric and adult patients alike, with an estimated prevalence of 1% to 20% worldwide.2,3 Furthermore, AD is associated with a plethora of physical and psychological comorbidities, including sleep disruption, pain, anxiety, and depression. Several treatments for AD may effectively manage symptoms of mild disease, including topical corticosteroids, emollients, Janus kinase (JAK) inhibitors, and calcineurin inhibitors.4,5 However, for moderate-to-severe disease, these agents may have limited efficacy, and phototherapy or systemic therapies are typically preferred.
Recent advances in our understanding of AD have elucidated a number of inflammatory pathways that contribute to its pathogenesis and disease severity. Interleukin (IL) 4 and IL-13 are two helper T-cell 2 cytokines implicated in many of the symptoms and signs associated with AD, including itch, inflammation, skin barrier disruption, and subsequent increased risk of infection.4 Dupilumab, a human monoclonal antibody that blocks both IL-4 and IL-13 signaling, has shown great efficacy in the treatment of moderate-to-severe AD in several clinical trials.1,6-11
In 2013, our group conducted a study on the psychological burden of moderate-to-severe plaque psoriasis using a nondermatologic quality of life questionnaire to collect data that could be compared to a broader range of conditions.12 Similarly, while previous clinical trials have employed quality of life questionnaires to measure symptoms and signs of AD, they have been largely limited to dermatology-specific and health-related quality of life instruments, which only measure satisfaction with life in patients affected by AD. This makes it difficult to objectively compare the psychological impact of living with AD to other conditions, such as heart disease, cancer, and diabetes.
The Psychological General Wellbeing (PGWB) index is a 22-item, self-administered quality of life survey that measures a person’s psychological wellbeing across 6 domains: anxiety, depressed mood, positive wellbeing, self-control, general health, and vitality.13 Importantly, it is designed to collect information regarding quality of life and mental health from the general population rather than the psychiatric population. Similar to many other psychometric instruments, the PGWB is graded on a Likert scale, with answers ranging from strongly disagree to strongly agree. Total scores range from 0 to 110, and higher scores indicate better mental health. Scores can be further broken down into severe distress (0–60), moderate distress (61–71), no distress (72–92), and psychological wellbeing (93–110).14 The PGWB is a validated instrument that has been adapted in many languages and is cross-culturally validated to evaluate psychological wellbeing in patients with various medical conditions. This questionnaire takes into account overall positive feelings and emotions, thus expanding on previous health-related quality of life instruments.
In this study, we sought to evaluate the improvement in psychological wellbeing after treatment with dupilumab using a validated nondermatologic psychometric instrument—the PGWB. Furthermore, as this was a real-world trial, participants were allowed to continue treatment with topical corticosteroids, phototherapy, or other systemic agents as deemed appropriate for the standard care of their AD by study investigators.
Methods
Study design. This was a Phase IV, single-center, open-label study conducted at the University of California, San Francisco (NCT03667014). Thirty-four participants with moderate-to-severe AD were recruited to receive treatment with dupilumab. Patients were enrolled from November 6, 2018, until August 25, 2021.
This study was conducted in accordance with the recommendations of the International Conference on Harmonization Good Clinical Practice and Declaration of Helsinki guidelines. The institutional review board reviewed and approved the protocol and informed consent form before any participants were enrolled, and participants consented using the approved informed consent form before participating in any study-related activities.
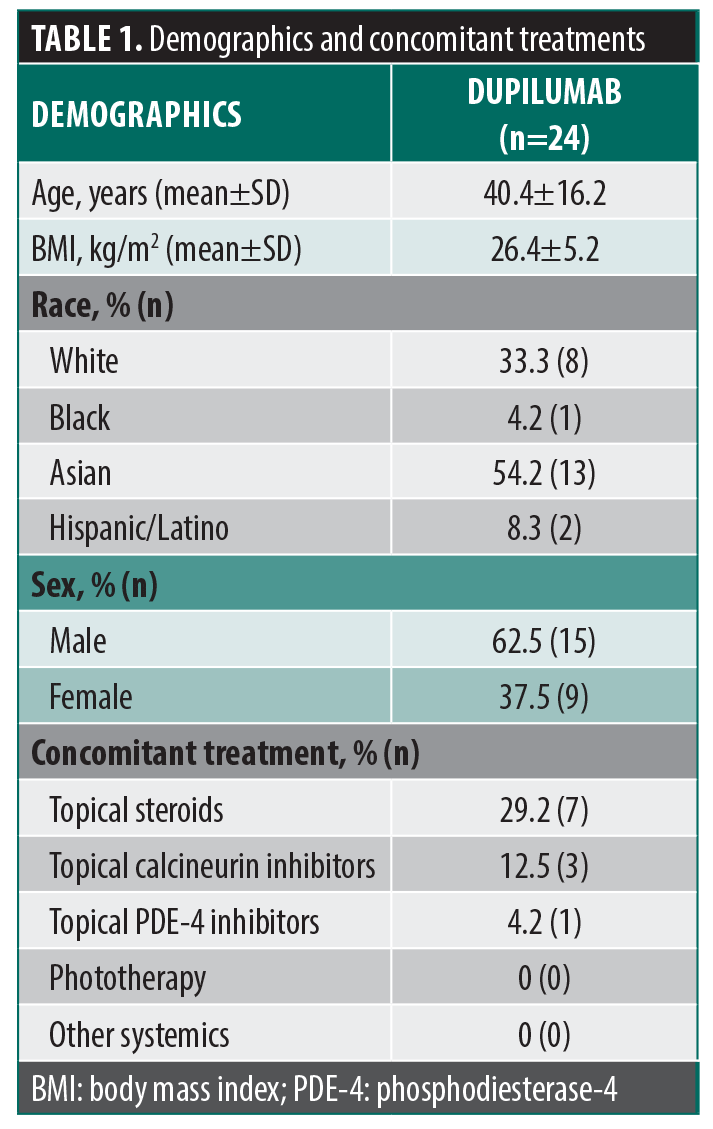
Each patient enrolled in the study underwent a screening visit. (Table 1) Demographics, medical history, and AD history were recorded. A physical exam was performed, during which AD severity was assessed using the Eczema Area and Severity Index (EASI) score and Investigator Global Assessment (IGA) score.
Once enrolled, scheduled visits occurred every 4 weeks for a total of 52 weeks. At each follow-up visit, AD severity was assessed. Patients completed the PGWB and Itch Numeric Rating Scale (INRS). Full body photographs were obtained at baseline and Weeks 16, 24, and 52.
Patient population. Inclusion criteria for study selection were as follows: ability to provide informed consent, aged 18 years or older, diagnosis of AD at least 6 months prior to study enrollment, stable disease for at least 2 months, no history of immunodeficiency, EASI score of 6 or greater, candidate for phototherapy or systemic therapy, and, for participants of childbearing potential, a negative urine pregnancy test 7 days prior to their first dose of dupilumab and use of a medically acceptable form of contraception. Participants were excluded if they did not meet the aforementioned inclusion criteria or if they exhibited evidence of other skin conditions that may interfere with the current study, well-controlled disease on their current regimen, previous or current dupilumab treatment, or have a history of intolerance to any component of dupilumab, immunocompromise, poorly controlled medical conditions, or drug or
alcohol abuse.
Treatment. All patients enrolled in the study self-administered dupilumab subcutaneously per the United States Food and Drug Administration-recommended dosing for adults with AD at an initial loading dose of 600 mg followed by 300 mg every other week through the duration of the study.
Participants were advised to continue concomitant treatments already in use before the start of the study, including topical corticosteroids, phototherapy, or other systemic agents as long as they were well established at stable doses. However, the use of other biologic therapies or addition of new treatments for AD was prohibited.
Outcomes. The primary endpoint was the change from baseline in participants’ psychological wellbeing as determined by the PGWB index at Week 16 of dupilumab treatment. Secondary endpoints included evaluating the change in the 6 PGWB subscores, mean EASI improvements, proportion of patients who achieved IGA scores of 0 or 1 (indicating clear or almost clear disease), and a ≥4 point reduction in INRS score at Weeks 16 and 52.
Statistical analysis. Patient characteristics were summarized using means and standard deviations for continuous variables, including age and body mass index (BMI). Tabulations and percentages were used to summarize categorical variables, including race and sex. The univariate associations between the total PGWB and subscores or change in total PGWB and subscores with the binary EASI (including 50%, 75%, and 90% improvement in EASI score [EASI 50, EASI 75, EASI 90]), IGA (0 or 1), or ≥4-point reduction in INRS scores at Weeks 16 and 52 were examined using nonparametric Wilcoxon tests. Multiple linear models controlling for age, sex, BMI, and ethnicity were used to examine the associations of changes in total PGWB and subscores with changes in EASI, IGA, and itch scores. P values less than 0.05 were considered statistically significant. All analyses were performed in SAS version 9.4.
Results
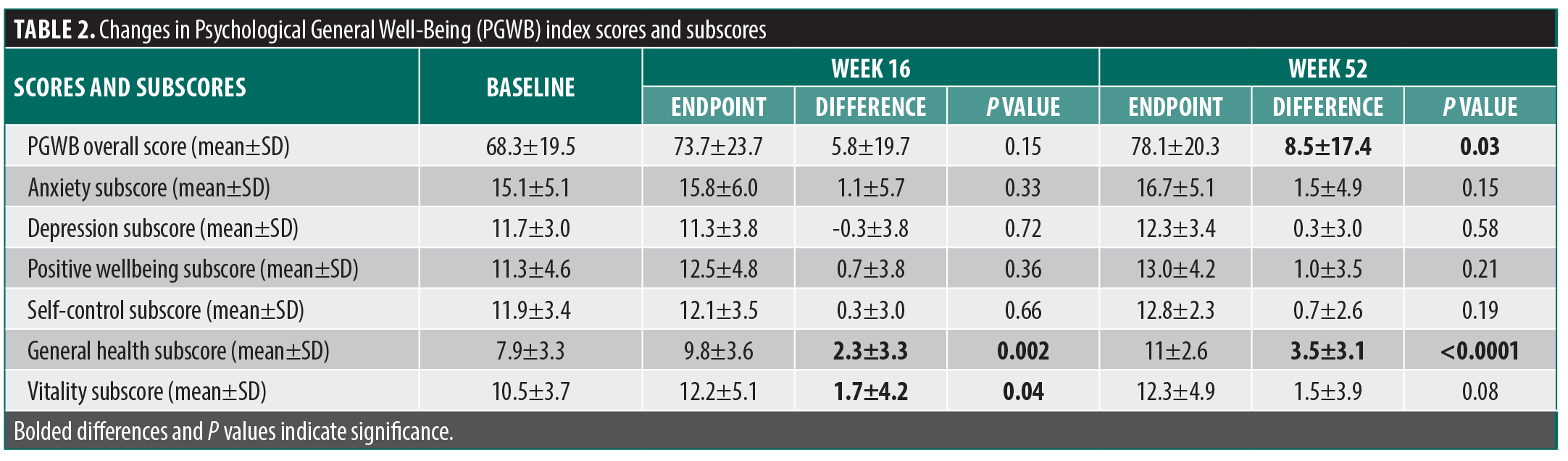
A total of 34 patients were recruited for participation in the study. Ten patients withdrew from the study prior to Week 52. Thus, 24 patients completed all 52 weeks and were included in the analysis. Patients withdrew for the following reasons: becoming pregnant (1), adverse effects (facial erythema, eye redness, eyelid swelling) (2), moving out of state (1), discomfort with in-person visits due to COVID-19 (1), and loss to follow-up (5). Seven participants were using topical medications—including clobetasol, triamcinolone, desonide, hydrocortisone, pimecrolimus, and tacrolimus—at enrollment and continued them for the duration of the study. None of the participants received concomitant phototherapy or other systemics at any point during the study period. The change in average PGWB score at Week 16 compared to baseline was not statistically significant (5.8±19.7; P=0.15). However, the change at Week 52 showed statistically significant improvement (8.5±17.4; P=0.03) (Table 2).
Over the course of the study, EASI score improved by 60.3% and 76.8% at Weeks 16 and 52, respectively. Mean IGA score also improved at Weeks 16 and 52, with 29.2% and 41.7% of participants achieving a score of 0 or 1, respectively. There was no significant association between change in total PGWB score and change in disease severity (EASI, INRS, and IGA) in both univariate analyses and multivariate regression models controlling for age, sex, ethnicity, and BMI.
For the PGWB subscores, only the changes in general health subscores at Weeks 16 and 52 and vitality subscores at Week 16 were significant compared to baseline. In multivariate analyses, when compared to their male counterparts, female participants were found to have a decrease in the positive wellbeing subscore at Week 52, indicating that female participants were more refractory to improvement in this domain (P=0.04). Again, there was no significant association between change in PGWB subscores and change in disease severity (EASI, INRS,
and IGA).
Discussion
Studies have confirmed improvement in quality of life after treatment with dupilumab. A randomized, placebo-controlled trial conducted by Simpson et al15 showed that AD patients taking 300 mg dupilumab every other week for 16 weeks showed significant improvements in anxiety, depression, and health-related quality of life. Another randomized, placebo-controlled trial conducted by Thaçi et al16 found that a dupilumab regimen of 300 mg every other week for 16 weeks led to significantly improved quality of life. Whereas health-related quality of life instruments measure a patient’s happiness and satisfaction with life in the setting of a disease such as AD, psychological wellbeing expands on this concept. It is a combination of both feeling good and functioning well; psychological wellbeing is the experience of positive emotions as well as the development of one’s potential, having some control over one’s life, having a sense of purpose, and experiencing positive relationships.17 It encompasses the perception that life is going well.
In this 52-week clinical trial studying the impact of dupilumab on psychological wellbeing, improvements were seen across several domains at both Weeks 16 and 52. Specifically, overall PGWB, anxiety, positive wellbeing, self-control, general health, and vitality scores improved at Week 16. By Week 52, improvements were seen across every domain. At Week 16, compared to baseline scores, statistically significant improvements were seen in general health and vitality subscores. This was also the case for the overall PGWB score and general health subscore at Week 52. Thus, our study showed that intervention with dupilumab is associated with improvement in psychological wellbeing in patients with AD, albeit at a later time period than other similar studies showing significant improvement by Week 16.15,16
Most previous clinical trials have focused on measures relating to symptoms of AD, such as the Dermatology Life Quality Index (DLQI) and Patient Oriented Eczema Measure (POEM) questionnaires. Thus, it was not possible to objectively compare the negative impact of AD to other nondermatologic conditions. Our study adds to the previous literature by focusing on the interplay between these symptoms and the PGWB index, which provides a more comprehensive evaluation and understanding of a patient’s self-perceived wellbeing and psychological health.18 Moreover, the PGWB is not specific to skin disease and has been used extensively to evaluate wellbeing in nondermatologic conditions, including hypertension, Turner syndrome, and gastroesophageal reflux disease.19–24 By using a validated questionnaire applicable to a broader range of medical conditions, we aimed to show that patients with moderate-to-severe AD experienced a similar—or more severe—psychological burden to those affected by the aforementioned conditions.
In our study, the mean PGWB before treatment with dupilumab was 68.3—considered indicative of moderate distress—and comparable to those of patients with nondermatologic serious conditions. For example, other studies of cohorts with type 2 diabetes, congestive heart failure, breast cancer, and inflammatory bowel disease reported mean PGWB scores ranging between 66.9 to 74.6, indicating comparable psychological burden.18,25–27 In our study, after 52 weeks of treatment with dupilumab, the mean PGWB score significantly improved to a level indicative of no distress. When comparing AD participants treated with dupilumab vs patients with psoriasis treated with adalimumab, the latter achieved statistically significant improvements in total PGWB by Week 4. These results indicate that AD patients may need longer periods to endorse improvement in psychological wellbeing.
This trial had several limitations. We did not have a placebo arm or control group. Ten patients dropped out of the study before reaching Week 36 for several reasons, including adverse effects, pregnancy, moving away, and discomfort with in-person visits due to COVID-19. This limited our sample size. Finally, PGWB data from our cohort were not directly compared to nondermatologic disease in a head-to-head fashion. However, previous studies in a psoriasis population faced similar obstacles while still yielding valuable findings.12,28
It is important to note that several studies have highlighted the mental health burden associated with the COVID-19 pandemic and quarantine.29,30 As this clinical trial took place during the height of the pandemic, it is possible that participant responses were negatively influenced, thereby decreasing overall PGWB scores and subscores due to an external factor.
Conclusion
In this clinical trial, participants taking dupilumab showed improvements across all PGWB subscores and overall PGWB score by the end of the study period. Statistically significant improvements from baseline scores were seen across overall PGWB score as well as the general health and vitality subscores.
References
- Beck LA, Thaçi D, Hamilton JD, et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N Engl J Med. 2014;371(2):130–139.
- Eckert L, Gupta S, Amand C, Gadkari A, Mahajan P, Gelfand JM. The burden of atopic dermatitis in US adults: health care resource utilization data from the 2013 National Health and Wellness Survey. J Am Acad Dermatol. 2018;78(1):54–61.e1.
- Silverberg JI, Gelfand JM, Margolis DJ, et al. Patient burden and quality of life in atopic dermatitis in US adults: a population-based cross-sectional study. Ann Allergy Asthma Immunol. 2018;121(3):340–347.
- Ring J, Alomar A, Bieber T, et al; European Dermatology Forum; European Academy of Dematology and Venereology; European Federation of Allergy; European Task Force on Atopic Dermatitis; European Society of Pediatric Dermatology; Global Allergy and Asthma European Network. Guidelines for treatment of atopic eczema (atopic dermatitis) part I. J Eur Acad Dermatol Venereol. 2012;26(8):1045–1060.
- Chovatiya R, Paller AS. JAK inhibitors in the treatment of atopic dermatitis. J Allergy Clin Immunol. 2021;148(4):927–940.
- Paller AS, Siegfried EC, Thaçi D, et al. Efficacy and safety of dupilumab with concomitant topical corticosteroids in children 6 to 11 years old with severe atopic dermatitis: a randomized, double-blinded, placebo-controlled phase 3 trial. J Am Acad Dermatol. 2020;83(5):1282–1293.
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2021;157(9):1047–1055.
- Blauvelt A, Simpson EL, Tyring SK, et al. Dupilumab does not affect correlates of vaccine-induced immunity: a randomized, placebo-controlled trial in adults with moderate-to-severe atopic dermatitis. J Am Acad Dermatol. 2019;80(1):158–167.e1.
- Guttman-Yassky E, Bissonnette R, Ungar B, et al. Dupilumab progressively improves systemic and cutaneous abnormalities in patients with atopic dermatitis. J Allergy Clin Immunol. 2019;143(1):155–172.
- de Bruin-Weller M, Thaçi D, Smith CH, et al. Dupilumab with concomitant topical corticosteroid treatment in adults with atopic dermatitis with an inadequate response or intolerance to ciclosporin A or when this treatment is medically inadvisable: a placebo-controlled, randomized phase III clinical trial (LIBERTY AD CAFÉ). Br J Dermatol. 2018;178(5):1083–1101.
- Simpson EL, Bieber T, Guttman-Yassky E, et al; SOLO 1 and SOLO 2 Investigators. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375(24):2335–2348.
- Bhutani T, Patel T, Koo B, Nguyen T, Hong J, Koo J. A prospective, interventional assessment of psoriasis quality of life using a nonskin-specific validated instrument that allows comparison with other major medical conditions. J Am Acad Dermatol. 2013;69(2):e79–88.
- Wenger NK, Mattson ME, Furberg CD, Elinson J. Assessment of quality of life in clinical trials of cardiovascular therapies. Am J Cardiol. 1984;54(7):908–913.
- The Psychological General Wellbeing Index (PGWBI) User Manual – PDF Free Download. Accessed February 28, 2023. https://docplayer.net/26261269-The-psychological-general-wellbeing-index-pgwbi-user-manual.html
- Simpson EL, Gadkari A, Worm M, et al. Dupilumab therapy provides clinically meaningful improvement in patient-reported outcomes (PROs): a phase IIb, randomized, placebo-controlled, clinical trial in adult patients with moderate to severe atopic dermatitis (AD). J Am Acad Dermatol. 2016;75(3):506–515.
- Thaçi D, Simpson EL, Beck LA, et al. Efficacy and safety of dupilumab in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical treatments: a randomised, placebo-controlled, dose-ranging phase 2b trial. Lancet. 2016;387(10013):40–52.
- Ruggeri K, Garcia-Garzon E, Maguire Á, Matz S, Huppert FA. Wellbeing is more than happiness and life satisfaction: a multidimensional analysis of 21 countries. Health Qual Life Outcomes. 2020;18(1):192.
- Grossi E, Groth N, Mosconi P, et al. Development and validation of the short version of the Psychological General Wellbeing Index (PGWB-S). Health Qual Life Outcomes. 2006;4(1):88.
- Omvik P, Thaulow E, Herland OB, Eide I, Midha R, Turner RR. Double-blind, parallel, comparative study on quality of life during treatment with amlodipine or enalapril in mild or moderate hypertensive patients: a multicentre study. J Hypertens. 1993;11(1):103–113.
- Walle PO, Westergren G, Dimenäs E, Olofsson B, Albrektsen T. Effects of 100 mg of controlled-release metoprolol and 100 mg of atenolol on blood pressure, central nervous system-related symptoms, and general well being. J Clin Pharmacol. 1994;34(7):742–747.
- Wiklund I, Berg G, Hammar M, Karlberg J, Lindgren R, Sandin K. Long-term effect of transdermal hormonal therapy on aspects of quality of life in postmenopausal women. Maturitas. 1992;14(3):225–236.
- Havelund T, Lind T, Wiklund I, et al. Quality of life in patients with heartburn but without esophagitis: effects of treatment with omeprazole. Am J Gastroenterol. 1999;94(7):1782–1789.
- Reddy P, White CM, Dunn AB, Moyna NM, Thompson PD. The effect of testosterone on health-related quality of life in elderly males – a pilot study. J Clin Pharm Ther. 2000;25(6):421–426.
- Boman UW, Bryman I, Halling K, Möller A. Women with Turner syndrome: psychological wellbeing, self-rated health and social life.
J Psychosom Obstet Gynaecol. 2001;22(2): 113–122. - Matza LS, Boye KS, Yurgin N. Validation of two generic patient-reported outcome measures in patients with type 2 diabetes. Health Qual Life Outcomes. 2007;5:47.
- Klocek M, Kubinyi A, Bacior B, Kawecka-Jaszcz K. Effect of physical training on quality of life and oxygen consumption in patients with congestive heart failure. Int J Cardiol. 2005;103(3):323–329.
- Bell RJ, Lijovic M, La China M, et al. Psychological wellbeing in a cohort of women with invasive breast cancer nearly 2 years after diagnosis. Support Care Cancer. 2010;18(8):921–929.
- Rapp SR, Feldman SR, Exum ML, Fleischer AB Jr, Reboussin DM. Psoriasis causes as much disability as other major medical diseases. J Am Acad Dermatol. 1999;41(3 Pt 1):401–407.
- The Lancet Psychiatry. COVID-19 and mental health. Lancet Psychiatry. 2021;8(2):87.
- Jones EAK, Mitra AK, Bhuiyan AR. Impact of COVID-19 on mental health in adolescents: a systematic review. Int J Environ Res Public Health. 2021;18(5):2470.