J Clin Aesthet Dermatol. 2025;18(11):41–43.
by Allison Lardieri, PharmD, BCPPS; Melissa Reyes, MD, MPH, FAAD; Karen E. Konkel, MD, MSHI; Ida-Lina Diak, PharmD, MS; and Lynda McCulley, PharmD, BCPS
All authors are with the Division of Pharmacovigilance, Office of Surveillance and Epidemiology at the FDA Center for Drug Evaluation and Research in Silver Spring, Maryland.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors report no conflicts of interest relevant to the content of this article.
ABSTRACT: Objective. We sought to characterize skin injuries following layperson application of chemical peel products (CPPs). Methods. Cases of skin injuries following layperson application of CPPs from August 31, 2018 through August 31, 2023 were retrieved from the United States Food and Drug Administration (FDA) Adverse Event Reporting System, the Human Foods Complaint System, the literature, and Amazon.com consumer product reviews with images. Results. We identified 18 skin injuries following layperson application of CPPs, including several cases using products labeled with concentrations typically used by healthcare professionals. Injuries involved burns with associated wounds, pain, swelling, and scars. Consequences included disfiguring scarring, reduced mobility, and need for emergency department, dermatologic, or surgical care. Analysis of Amazon.com demonstrated that CPPs are sold under many names with numerous therapeutic claims, list a variety of concentrated agents, and have thousands of reviews. Limitations: Limitations include underreporting of adverse events to the FDA and incomplete clinical information. With consumer product reviews, there was potential bias and poor replicability due to the dynamic nature of the Amazon website. Conclusion. Laypersons who apply CPPs of any strength without guidance from licensed and trained healthcare professionals may experience skin injury and long-term sequelae. Dermatologists are positioned to advise against application of CPPs without professional supervision. Keywords: Chemical burn, chemical peel, dermatology, self-medication, skin injury, topical product, unapproved product
Introduction
Epidermal and dermal regeneration and remodeling lead to improved skin appearance after controlled wounding, and chemical peel products (CPPs) aim to achieve skin improvement via topical application of one or more keratolytic agents.1,2 CPP penetration can be superficial, medium, or deep, depending on the concentration, pH, and active ingredient(s) in the product.1-4 Healthcare professionals are trained to: (1) select the appropriate chemical peel agent on a patient-level basis depending on Fitzpatrick skin type and treatment site; (2) provide counseling, pretreatment, and aftercare; (3) use correct application technique; (4) recognize and mitigate risks; and (5) anticipate and manage complications.1,5 In contrast, laypersons have an increased risk of complications when using these products due to a lack of training and understanding of the differences among products.
The American Academy of Dermatology considers CPPs generally safe when applied under dermatologist supervision for acne vulgaris, actinic keratoses, melasma, and scarring.2,6 However, no CPPs have been approved or evaluated for safety, efficacy, and product quality by the United States Food and Drug Administration (FDA). The FDA has warned consumers to avoid CPPs sold directly to laypersons to remove tattoos, warts, moles, and skin tags or to “reduce fine lines” or “stimulate collagen,” and it has issued warning letters to several companies for selling CPPs.3
Minor adverse reactions to CPP exposure include irritation, burning, erythema, pruritus, edema, blistering, infections, acneiform eruptions, and pigmentary changes.1 Rare, major complications include allergic reactions, laryngeal edema, toxic shock syndrome, cardiotoxicity, salicylism, kidney injury, corneal damage, scarring, and dyspigmentation.1 Serious outcomes have been reported after the application of superficial CPPs,7 but the landscape of CPP-associated injuries after layperson use has yet to be examined. Herein, we describe the largest analysis of skin injuries caused by layperson use of CPPs to date.
Methods
We searched the FDA Adverse Event Reporting System, the Human Foods Complaint System (which includes cosmetic products), the literature (PubMed and Embase for case reports), and Amazon.com consumer product reviews from August 31, 2018 through August 31, 2023 for skin injuries following layperson application of CPPs. We excluded cases with concurrent use of another skin procedure or where products were intended for application by a healthcare professional. Amazon reviews with over 1000 consumer ratings from “verified purchasers” for products that were not “sponsored” and that contained images of skin injury were included. The extent of each injury was classified as superficial, partial, full, or full with underlying injury.8
Results
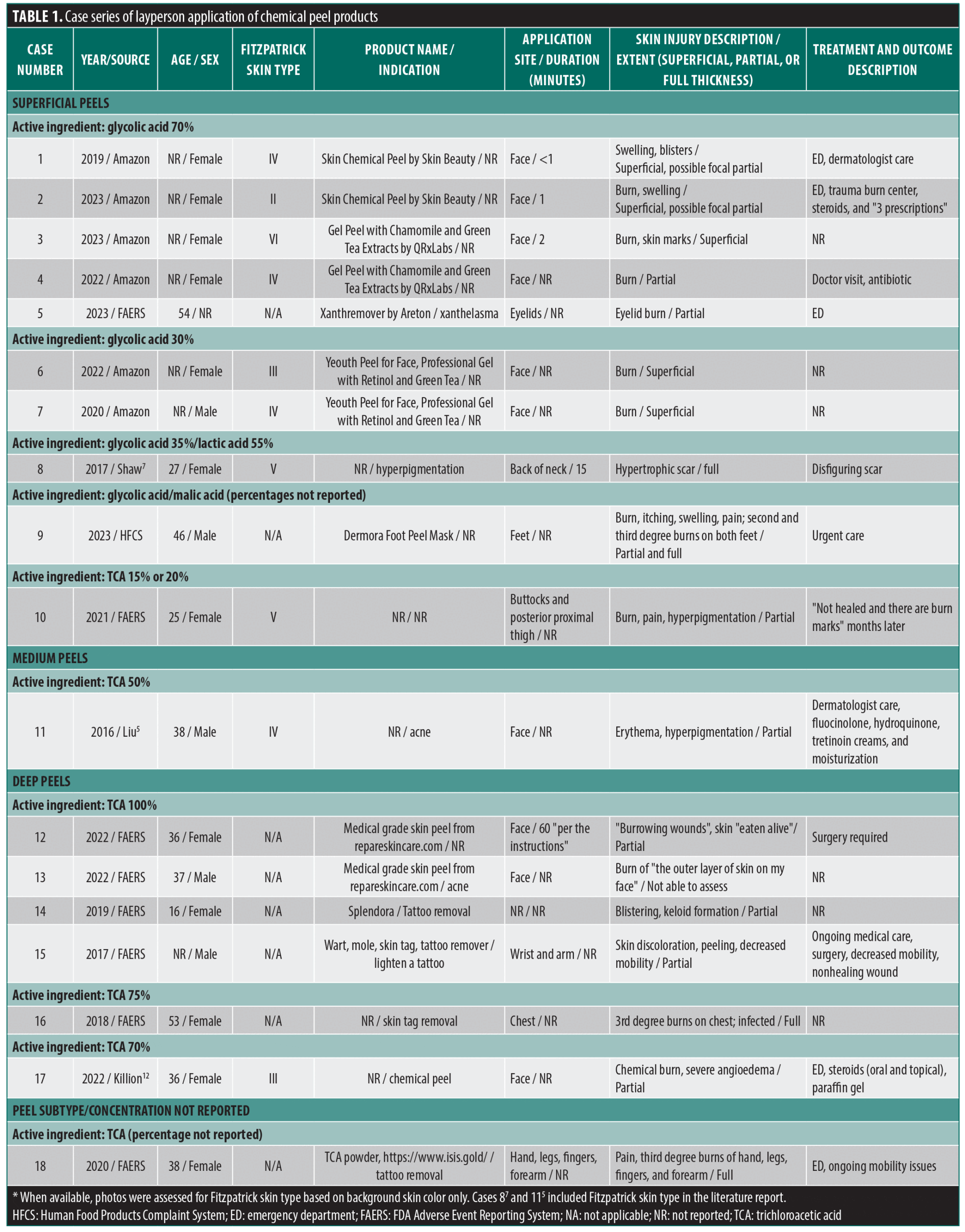
Eighteen skin injuries were identified, including 9 reported to the FDA, 6 from Amazon consumer product reviews, and 3 from the literature (Table 1). Most patients were female (12 of 18), and the age range, reported in 11 of 18 cases, was 16 to 54 years (median: 37 years). Fitzpatrick skin type was assessable in 10 cases and ranged from Type II to VI. Injuries involved burns with associated wounds, pain, swelling, and scars. Some cases resulted in disfigurement and reduced mobility, and half required specialty care. The extent of skin injury was superficial (3 cases), superficial with possible focal partial (2), partial (8), full and partial (1), full (3), or could not be classified (1).
Several cases reported medium or deep chemical peels labeled as containing active ingredient concentrations used by healthcare professionals (e.g., 70 to 100% trichloroacetic acid [TCA]). Seven cases reported partial or full thickness skin injury after superficial chemical peel use. Products were commonly applied to the face (10 of 18), but other target areas included eyelids, neck, feet, buttocks, wrist/arm, and chest. Of 9 cases reporting use of 30 to 70% glycolic acid, four cases reported applying the CPP for 1 to 15 minutes and none described neutralization steps.
Analysis of Amazon.com consumer product reviews demonstrated that CPPs are sold under many names and marketed with numerous therapeutic claims. CPPs list various potent active ingredients, lack instructions, and are widely accessible.
Discussion
To date, this is the largest case series describing CPP-related skin injuries after layperson use. Half of the injuries assessed in this series required emergency care or care from a dermatologist or surgeon. Injury severity varied across all chemical peel types. Many consumers bought CPPs online and applied them without any training or instructions on application technique, potential risks, risk mitigation, or management of complications associated with chemical peels. Some products instructed the consumer to apply the product to their eyelids for the purpose of eyelid tightening, which is concerning given the proximity to the eye. Amazon.com consumer product reviews provided pictorial evidence and descriptions of recent superficial to partial-thickness skin injury after consumer use of unapproved over-the-counter chemical peels. Multiple peeling agents are available for consumer purchase online, most commonly glycolic acid, a superficial-depth peel, and TCA, which can range from superficial to deep penetration depending on its concentration. Various chemical peel strengths were used on several Fitzpatrick skin types and body sites. Some agents may penetrate deeply if applied repeatedly to the same area or not neutralized (e.g., glycolic acid). No cases reporting glycolic acid application described neutralization steps. Medium and deep chemical peels are riskier if used on the lower third of the neck or nonfacial areas, or by individuals with Fitzpatrick skin types III to VI.9,10 Multiple reports describe consumers purchasing the highest strength of TCA (100%) and experiencing skin injury after at-home use. Two of 6 deep TCA peels were used on nonfacial areas.10 Use of CPPs with lower concentrations of active ingredient did not necessarily have less severe skin injury outcomes; 55.6% of cases in this series involved superficial CPPs (Table 1).
Limitations
The data sources in this case series have limitations, including significant underreporting. The FDA does not receive all adverse event reports because manufacturers and consumers are unlikely to report those related to unapproved products. Many cases did not contain photos, product name, user training, or details of peel application (e.g., number and duration). When available, photos were assessed for Fitzpatrick skin type and injury extent, which is subjective and depends on photo quality. Actual ingredients, concentrations, purity, and manufacturing practices are unknown for CPPs because they are marketed without FDA review. Consumer product reviews submitted to Amazon.com may provide incomplete information on the injury, be biased, or be edited.11 Additionally, the dynamic nature of the website limits replicability. For example, sellers may remove and re-post products with revised names or packaging, presumably to remove negative reviews.
Conclusion
This case series demonstrates that laypersons who apply CPPs of any strength may experience skin injury and long-term sequelae. In addition to recognizing and treating skin injuries caused by these products, dermatologists are positioned to advise laypersons against application of CPP products without professional supervision.
References
- Chemical peels: FAQs. American Academy of Dermatology Association. Accessed October 9, 2024. https://www.aad.org/public/cosmetic/younger-looking/chemical-peels-faqs
- Lee KC, Wambier CG, Soon SL, et al. Basic chemical peeling: Superficial and medium-depth peels. J Am Acad Dermatol. 2019;81(2):313-324.
- FDA warns against purchasing or using chemical peel skin products without professional supervision. US Food and Drug Association. Updated July 30, 2024. Accessed August 5, 2024. https://www.fda.gov/drugs/drug-safety-and-availability/fda-warns-against-purchasing-or-using-chemical-peel-skin-products-without-professional-supervision
- O’Connor AA, Lowe PM, Shumack S, Lim AC. Chemical peels: a review of current practice. Australas J Dermatol. 2018;59(3):171-181.
- Liu H, Khachemoune A, Rashid RM. Chemical burn following 50% trichloroacetic acid for acne: presentation of a case and a focused review. J Dermatol Surg. 2016;20(1):71-74.
- Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol. 1988;124(6):869-71.
- Shaw DJ, Higgins HW II, Lee KC. Development of a hypertrophic scar after application of an over-the-counter chemical peel. Dermatol Surg. 2017;43 Suppl 1:S91-s93.
- Rice P, Orgill D. Assessment and classification of burn injury. UpToDate. Updated February 20, 2023. Accessed November 13, 2023. https://www.uptodate.com/contents/assessment-and-classification-of-burn-injury
- Monheit G, Hrynewyca K. Chemical and mechanical skin resurfacing. In: Bolognia JV, Schaffer LC, editors. Dermatology. 5th ed. New York: Elsevier; 2024. p. 2633-2649.
- Vemula S, Maymone MBC, Secemsky EA, et al. Assessing the safety of superficial chemical peels in darker skin: A retrospective study. J Am Acad Dermatol. 2018;79(3):508-13.e2.
- Konkel K, Reyes M, Diak IL, et al. Serious skin injuries following exposure to unapproved mole and skin tag removers. J Clin Aesthet Dermatol. 2023;16(1):14-17.
- Killion L, Roche M. High strength chemical peels-not to be tried at home. Health Sci Rep. 2022 Jan;5(1):e491.