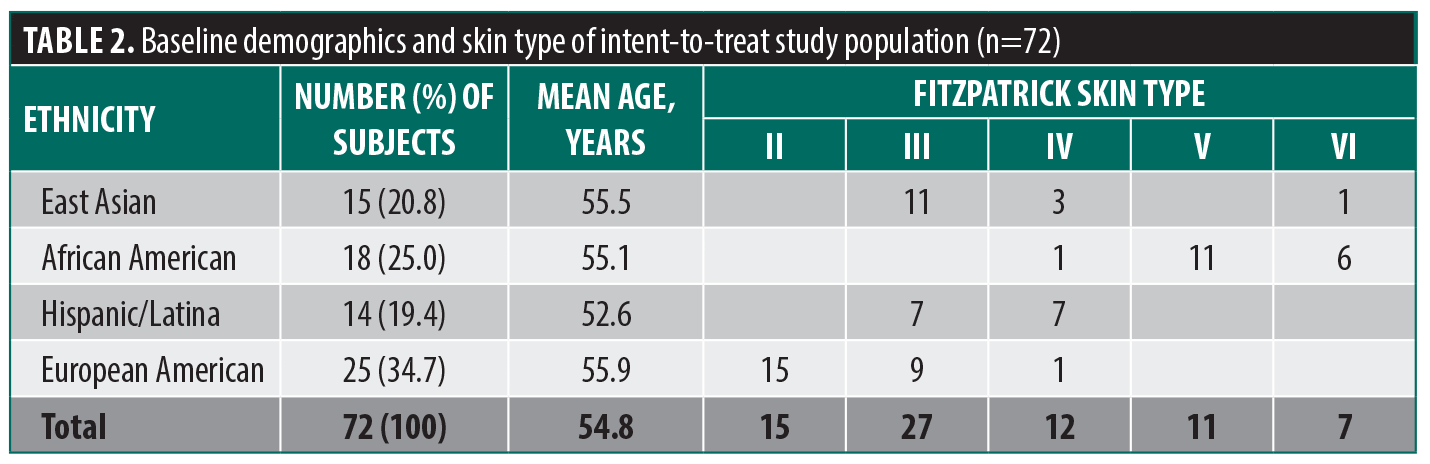J Clin Aesthet Dermatol. 2025;18(12):34–41.
by Elizabeth Bruning, BSc Hons, LLB; Edward (Ted) Lain, MD, MBA; Glynis Ablon, MD; Vivian Bucay, MD; Cheryl Burgess, MD; James Del Rosso, DO; Patricia K. Farris, MD; Jill Waibel, MD; Denise DiCanio, EdD, MBA; and Sarah Vickery, PhD
Ms. Bruning was with The Estée Lauder Companies in Melville, New York at the time of writing. Drs. DiCanio and Vickery are with The Estée Lauder Companies, Research and Development, in Melville, New York. Dr. Lain is with Sanova Dermatology in Austin, Texas. Dr. Ablon is with Ablon Skin Institute and Research Center in Manhattan Beach, California. Dr. Bucay is with Bucay Center for Dermatology and Aesthetics in San Antonio, Texas. Dr. Burgess is Assistant Clinical Professor of Dermatology at Georgetown University and George Washington School of Medicine and Health Sciences in Washington, District of Columbia. Dr. Del Rosso is with Del Rosso Dermatology Research Center in Las Vegas, Nevada. Dr. Farris is Clinical Associate Professor of Dermatology at Tulane University School of Medicine in New Orleans, Louisiana. Dr. Waibel is with Miami Dermatology and Laser Institute in Miami, Florida.
FUNDING: Funding for this study was provided by The Estée Lauder Companies, Melville, New York.
DISCLOSURES: Ms. Bruning, Dr. DiCanio, and Dr. Vickery are current or former employees of The Estée Lauder Companies. Drs. Lain, Ablon, Bucay, Burgess, Del Rosso, Farris, and Waibel have received honoraria from The Estée Lauder Companies.
ABSTRACT: Background: Efficacy evaluations of topical cosmetic products purported to produce younger-appearing skin require an objective assessment of perceived age. The aim of this study is to quantify the age-reversal efficacy of a facial skincare regimen containing sirtuin-targeting and other anti-aging ingredients in a multiethnic population in terms of change in perceived age, using a validated age prediction model. Methods: In this single-center, monadic study, eligible participants (n=72) applied a 3-product regimen (serum, soft crème, and eye crème) twice daily for 12 weeks. High-resolution digital photographs of subjects were graded for 7 facial parameters using an 11-point photonumeric scale (0=no signs of aging; 10=severe signs of aging) at baseline and 2, 4, 6, 8, and 12 weeks. Predicted Age scores derived from the model algorithm at each assessment point were compared with baseline scores to calculate change in Predicted Age in years. Results: Statistically significant decreases vs. baseline in calculated Predicted Age were observed as early as Week 2, with the difference increasing with continued product application (difference at Week 12 was –4.0 years). Each facial aging parameter improved at a different rate, with forehead lines and elongated cheek pores significantly improving as early as Week 2. Under eye lines and forehead lines showed the greatest calculated improvement over time. Conclusion: Based on the Predicted Age model, the 3-product face/eye skincare regimen applied twice daily resulted in a significant decrease in calculated Predicted Age starting as early as 2 weeks, with continued improvement over time, and a decrease of 4.0 years by Week 12. Keywords: Sirtuins, anti-aging, cellular longevity, age-reversal, skincare
Introduction
In the cosmeceuticals and aesthetics field, there has been a paradigm shift from addressing existing symptoms of skin aging to modulating the cellular processes that underlie the aging process, representing a focal shift from anti-aging to pro-longevity. This focal shift suggests that cosmeceuticals that prevent aging will form the foundation of the next generation of skincare products.1
One of the hallmarks of aging is cellular senescence, a state of irreversible cell cycle arrest in which cells no longer proliferate but remain metabolically active.2,3 Accumulation of senescent cells within the skin, a result of intrinsic (chronological) and extrinsic aging, is associated with epidermal thinning, flattening of the dermal-epidermal junction, hyperpigmentation, loss of melanocytes, and degradation of collagen and elastin.4,5 These physiological changes, in turn, are reflected in the visible signs of skin aging, such as laxity, wrinkles, and volume loss.5 Hence, inhibiting the senescence of skin cells and related cellular aging processes has the potential, in theory, to increase cellular longevity and prevent or delay the visible signs of skin aging.
Sirtuins are a family of 7 signaling proteins (SIRT1 through SIRT7) that function primarily as nicotinamide adenine dinucleotide-dependent histone deacetylases (Class III HDACs) and act as epigenetic regulators of cellular metabolism and homeostasis.6 Both intrinsically and extrinsically aged skin are characterized by reduced expression and downregulation of specific sirtuins.7-11 Intrinsically aged skin is characterized by increased histone acetylation and decreases in SIRT6 and other HDACs.7 In humans, SIRT1 expression in dermal fibroblasts decreases with chronological age9 as does SIRT6 expression.8 With respect to extrinsically aged skin, normal circadian expression of SIRT1, SIRT3, and SIRT6 in normal human epidermal keratinocytes (NHEK) is disrupted following UV-B exposure, coinciding with a decrease in adenosine triphosphate (ATP) production and an increase in reactive oxygen species,10 and expression of SIRT3 in NHEK decreases after exposure to ozone.11 Activation of specific sirtuin pathways in skin cells has been shown in in vitro and animal studies to protect cells against UV-induced senescence and oxidative stress, and to restore energy homeostasis in skin cells to extend cellular lifespan.12-19 However, very few studies have examined whether topical application of sirtuin-targeting compounds that are active at the cellular level can effect visible changes in skin.20,21
In our laboratories, several proprietary facial skincare formulations incorporating ingredients that promote SIRT1, SIRT3, and SIRT6 activity at the cellular level, along with other active ingredients, have been developed to reduce visible signs of skin aging and help users “look younger.” The efficacy of such anti-aging facial skincare formulations should be measured by an objective assessment of perceived age, that is, how old a person looks before and after formulation use over a given period of time. Clinical assessments of facial aging parameters by human graders to calculate an estimated age have been shown to be a valid method, with high inter-rater and intra-rater reliability.22-24 However, in these models, selection of the relevant facial aging parameters is based on observations from a population of a single ethnicity and/or a limited range of skin types and therefore may not be applicable to evaluations of products used by diverse populations. In addition, the broad scales used to grade the facial parameters in these models (ie, 4-point scales corresponding to none, low, moderate, and severe signs of aging) may not be sufficiently sensitive to detect the gradual changes that may occur with use of cosmetic formulations.
DiCanio et al25 developed a methodology to calculate perceived or apparent age based on clinical grading assessments of an optimized set of visible facial aging parameters, independent of ethnicity and skin type. Briefly, this model was developed based on an image data set of a large and diverse multiethnic population (n=2825 European American, African American, East Asian, and Hispanic/Latina women across Fitzpatrick skin types I-VI). Trained graders assessed facial aging markers for all subjects using an 11-point photonumeric scale. Multiple linear regression analysis was used to identify 7 universal face and eye parameters that contribute most to aging: nasolabial folds, under eye lines, elongated cheek pores, forehead lines, under eye puffiness, uneven skin tone, and marionettes. The algorithm derived from the multiple linear regression analysis could then be used to calculate a predicted age, with each facial parameter weighted according to its contribution to the overall age estimation.
The aim of the present study is to assess the effects of a facial skincare regimen containing sirtuin-targeting agents and other anti-aging ingredients on visible skin aging parameters in a multiethnic population, and to quantify these effects in terms of changes in perceived age using the age prediction model developed by DiCanio et al.25
Methods
Study design overview. This was a single-center, monadic, assessor-blinded cosmetic regimen study conducted by an independent clinical research organization (SGS North America, Inc.). Eligible subjects applied a facial skincare regimen consisting of 3 products (serum, soft crème, and eye crème) twice daily according to a standardized application procedure for 12 weeks. High-resolution digital photography of all subjects was obtained at baseline before application of the regimen and at 2, 4, 6, 8, and 12 weeks. A blinded, trained clinician-grader evaluated the digital photographs for 7 key facial parameters on an 11-point analog scale using methods previously described by DiCanio et al.25 Predicted Age was calculated at baseline and at all assessment points based on the model and algorithm derived by DiCanio et al25; age scores computed using the algorithm at each measurement were compared with baseline scores to calculate change in Predicted Age in terms of years.
Study subjects. The study enrolled European American, African American, East Asian, and Hispanic/Latina women aged 35 to 65 years (35 to 70 years for African American and East Asian subjects), with at least 12 subjects in each group, distributed across Fitzpatrick skin types I-VI, with evidence of mild to moderate photoaging (score of 2 to 6 on a modified Griffiths scale, where 0=none and 9=severe). Subjects could have normal, oily, or dry skin. Eligible subjects could not have had any facial treatments in the past 6 months (eg, facial peels, botulinum toxin, fillers, laser treatments) or any other treatment designed to improve the appearance or firmness of facial skin.
Key exclusion criteria included known allergies to skincare products; reactivity/sensitivity to sunflower seed, coffee, apple, black truffle, and cucumber (active ingredients in product regimen); history of skin cancer within the past 5 years; use of oral or topical prescription medications for acne within 3 months; use of oral isotretinoin within 6 months; use of prescription-strength skin-lightening products within 3 months; use of over-the-counter anti-wrinkle, skin-lightening, or other topical or systemic products known to affect skin aging or dyschromia within 2 weeks; and excess exposure to sun via outdoor activities (eg, sunbathing).
Written informed consent forms, including photograph release forms, were obtained from all subjects. The study was conducted in accordance with federal regulations and Good Clinical Practice guidelines. Institutional Review Board approval was not sought as the study involved cosmetic research with safety-tested and marketed products posing minimal risk to human subjects.
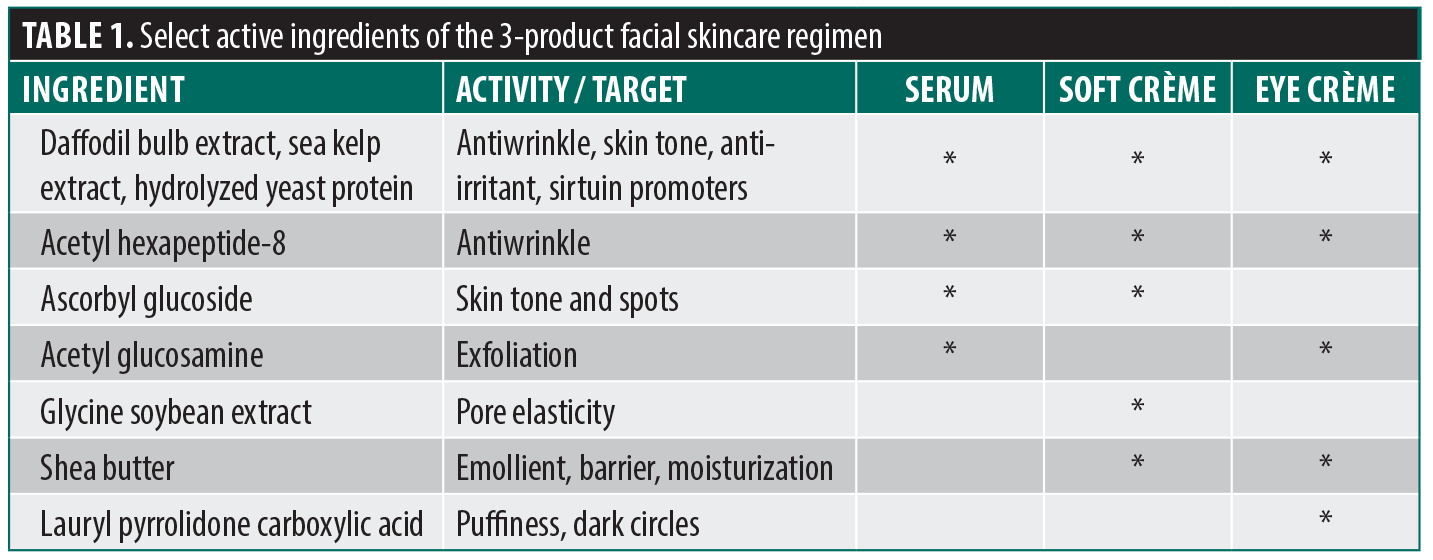
Product regimen and skin application. The facial skincare regimen tested included a serum, soft crème, and eye crème. The 3 products each contain patented ingredients that have been shown in internal studies to promote SIRT1, SIRT3, and SIRT6 activity, as well as other active ingredients with properties that enhance overall skin health and appearance (eg, moisturizing, anti-irritant, anti-wrinkle, skin tone improving, and antioxidant properties; Table 1). Our published and internal in vitro and ex vivo studies in normal human epidermal keratinocytes and human dermal fibroblasts have demonstrated effects of the SIRT1, SIRT3, and SIRT6 activators on aging-related processes at the cellular level, including increasing fibrillin production, mitophagy and autophagy rates, and mitochondrial ATP production; reducing NF-κB-mediated inflammation; improving cellular lifespan; protecting against UV-induced cellular senescence; and boosting collagen production.26 Although the 3 products contain common ingredients, including the 3 sirtuin activity promoters, each product also contains unique ingredients that are not formulated in the other 2 products in the regimen. The use of both face and eye products allowed for holistic treatment of the subjects’ entire face.
Subjects were instructed to first wash their face, pat the skin dry, and apply the serum, soft crème, and eye crème, in that order, twice daily (in the morning and evening) for 12 weeks. The serum and soft crème were to be applied to the entire face, avoiding the eye area, and allowed to absorb for at least 1 minute. The eye crème was applied to the eye area with the fingertips and then massaged around the eye area using an eye wand or roller. Written application instructions were provided to subjects to ensure proper use of the products. Subjects performed the first application of the test products in the clinic after completion of baseline evaluations.
Subjects were also provided with a standard facial cleanser and a broad-spectrum sunscreen (sun protection factor [SPF] 50) to use daily.
Digital photography. Digital images of the subjects (front facing and right and left ¾ side views) were obtained at baseline before application of the regimen and at 2, 4, 6, 8, and 12 weeks using a VISIA-CR™ photo station (Canfield Imaging Systems) with a Canon R5 Digital SLR camera (Canon Incorporated) and light-emitting diode (LED) flash illumination under the following lighting conditions: standard (visible [bright]), raked, cross-polarized, parallel polarized, and UV absorption.
Photographs were taken by reproducibly positioning the participant’s head (using stationary chin and forehead supports) and maintaining consistent camera and lighting settings at each study visit.
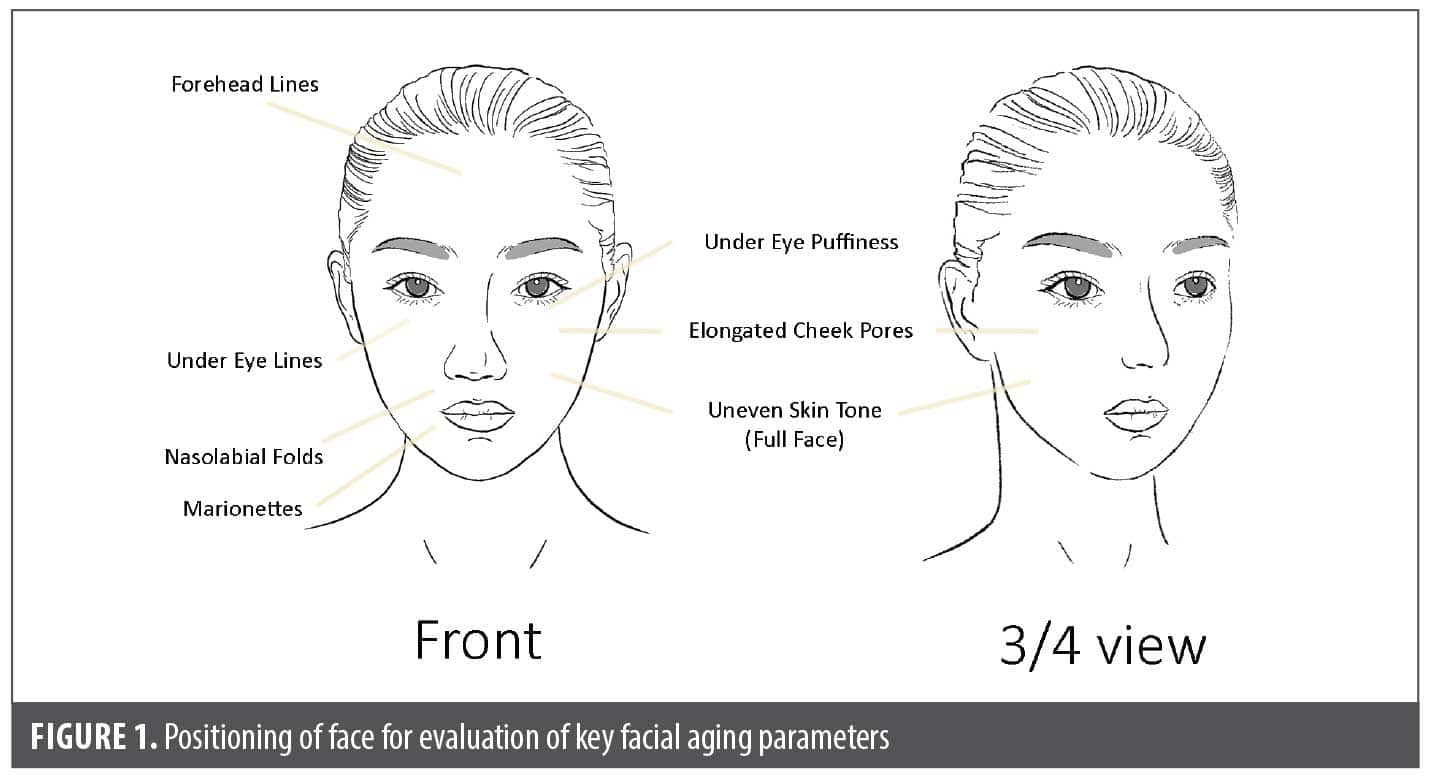
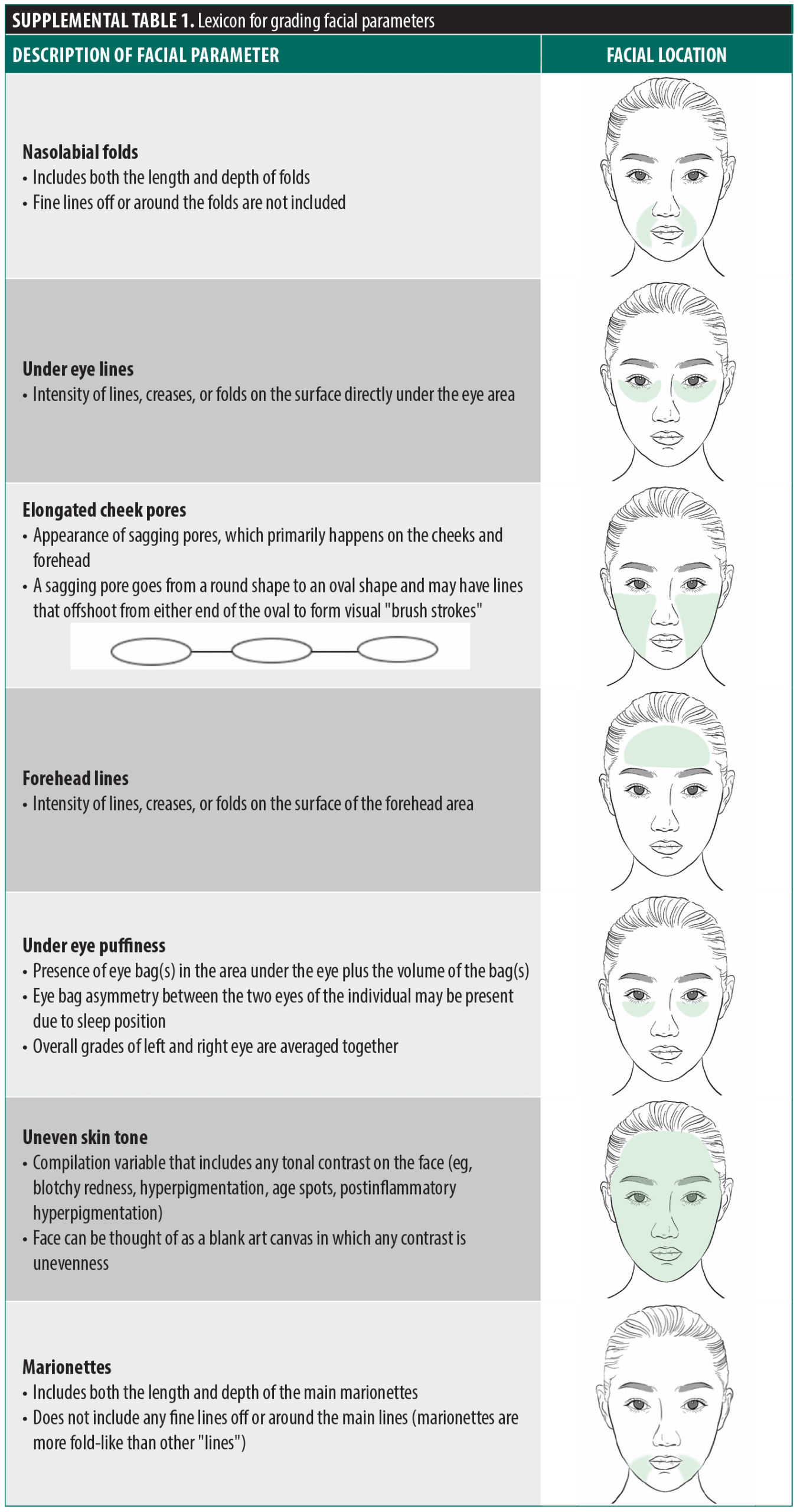
Facial parameter grading. Digital photographs of the subjects were graded by a blinded, trained clinician for 7 face and eye parameters on an 11-point photonumeric scale (0=no signs of aging; 10=severe signs of aging): nasolabial folds, under eye lines, elongated cheek pores, forehead lines, under eye puffiness, uneven skin tone, and marionettes. These 7 parameters are described in Supplemental Table 1. Grading increments of 0.5 were permitted. Visual grading scales (with a representation of photographs for each level of severity) were used as a reference to grade the 7 skin parameters as previously described by DiCanio et al.25 Front-view images were used to grade all markers, and right and left ¾ side view images were used to grade uneven skin tone and elongated cheek pores (Figure 1). The trained clinician grader was blinded to the week at which photographs were captured.
Statistical methods. Predicted Age score for each week was calculated using the graded values for the individual facial parameters and the algorithm by DiCanio et al.25 Predicted Age scores computed using the algorithm for each posttreatment measurement were compared with baseline scores to calculate change in Predicted Age in terms of years; statistical significance was determined using correlated t tests. Statistical analysis was conducted on the intent-to-treat (ITT) population, which included all subjects who received treatment, had at least 1 postbaseline visit and were not otherwise disqualified. All statistical tests were 2-sided at significance level α=0.05. Statistical analysis was performed using SPSS/PC+ V5.0.2 statistical software (IBM).
Results
Subject disposition and baseline demographics. A total of 77 subjects were enrolled in the study; 3 were lost to follow-up after the baseline visit and did not receive product. Five subjects were excluded from the analysis; 1 had rosacea-prone skin (exclusion criterion), 3 had multiple sun exposures (ie, noncompliance), and 2 subjects were discontinued from the study due to injuries to the face that were unrelated to the study product regimen (1 due to spider bite and 1 due to airbag deployment into face). The ITT population, therefore, included 72 patients. Three subjects withdrew from the study: 2 after 6 weeks and 1 after 8 weeks (1 due to illness and 2 for unknown reasons).
The study population was balanced with respect to ethnicity and Fitzpatrick skin type (Table 2) and similar in composition to the population on which the Predicted Age model was based.25 The mean (SD) age was 54.8 (8.4) years (range: 35 to 69 years), and the majority of subjects were Fitzpatrick skin type III. No subjects were Fitzpatrick skin type I.
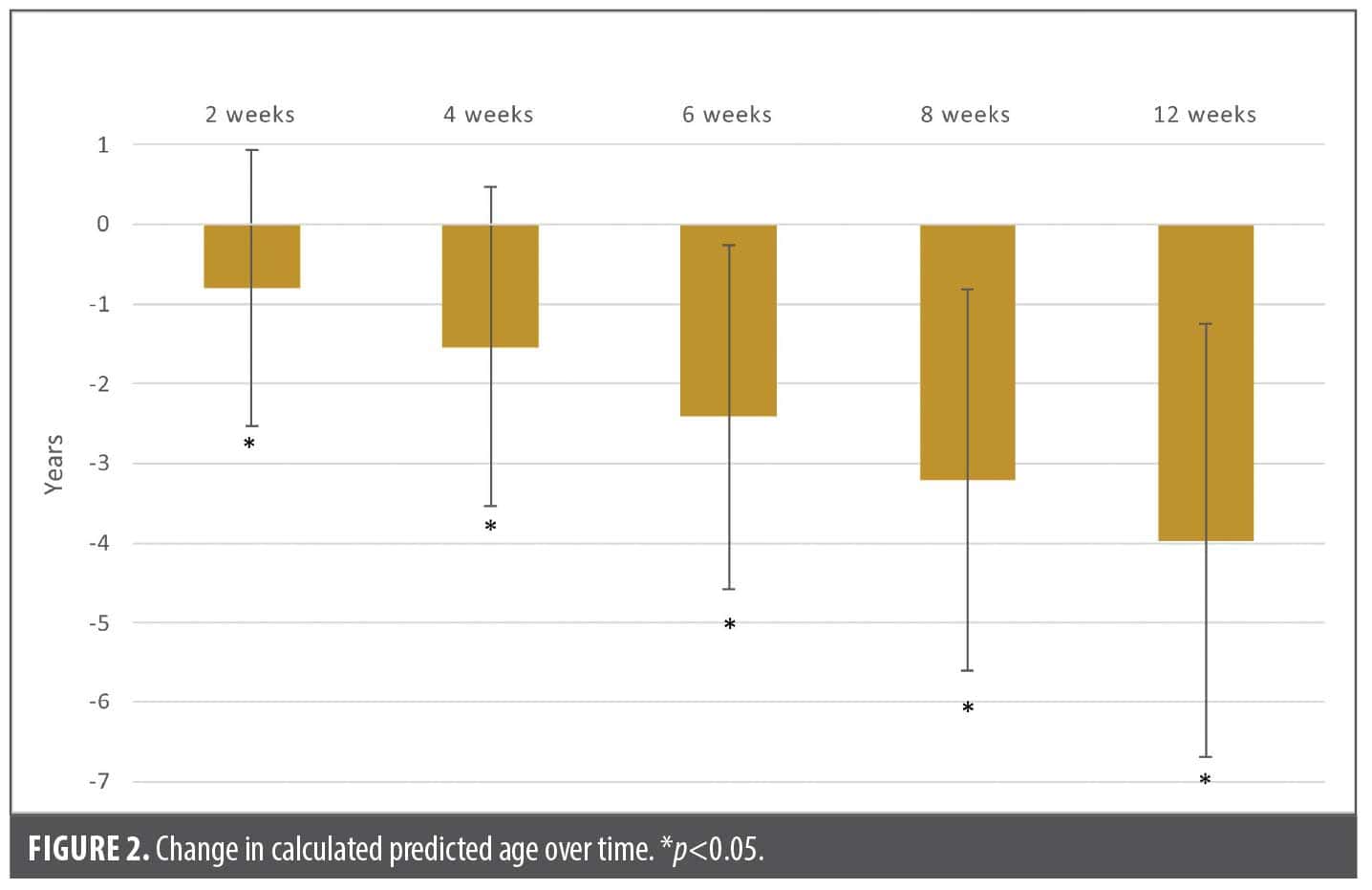
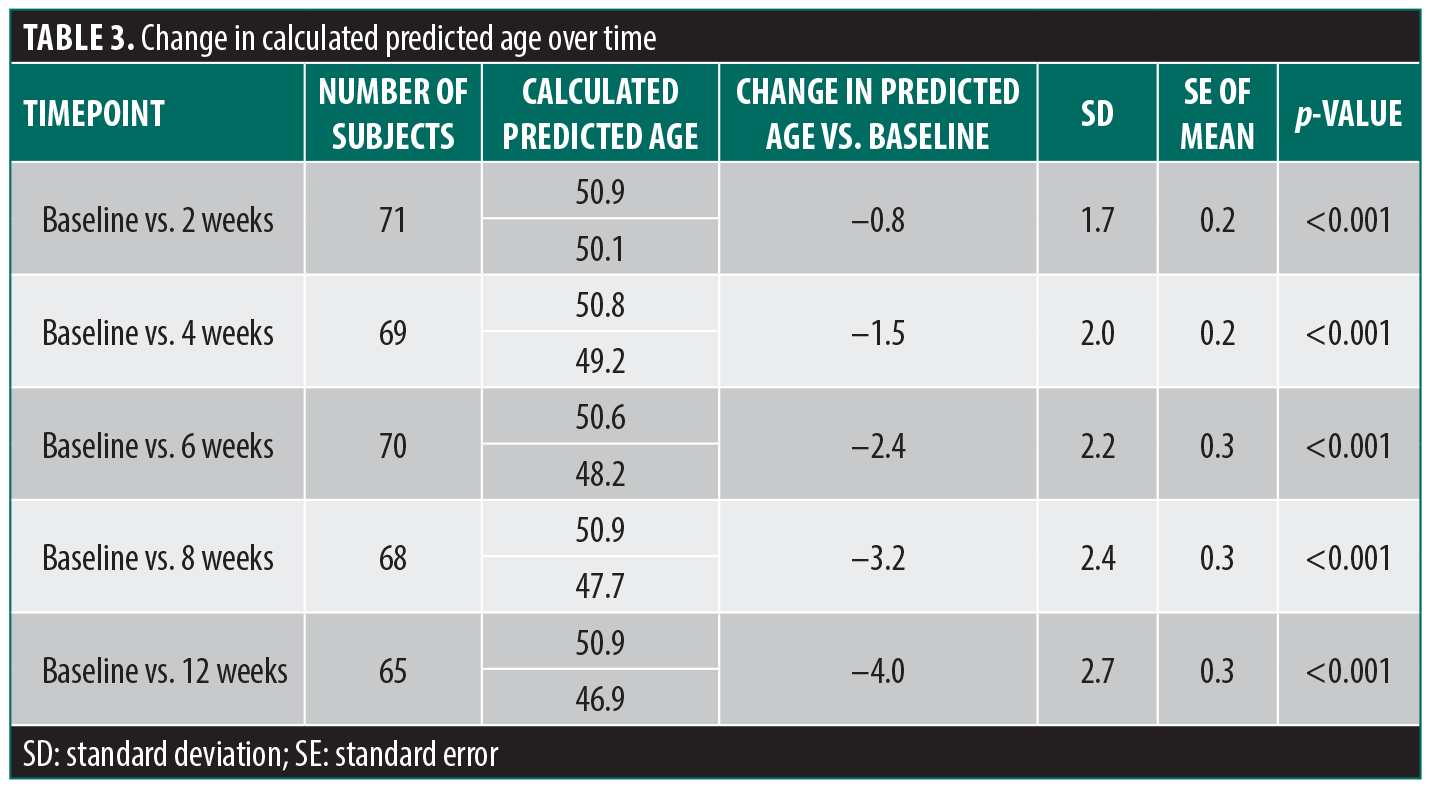
Primary endpoint: change in predicted age. Beginning as early as 2 weeks after twice-daily application of the 3-product facial skincare regimen, the decrease in predicted age versus baseline was statistically significant (Table 3, Figure 2). At each assessment point, the difference versus baseline continued to increase, indicating that calculated predicted age decreased progressively with continued application of the facial regimen. This significant predicted age-reversal effect continued until study end at 12 weeks, at which point the difference in predicted age versus baseline was –4.0 years (ie, at 12 weeks, subjects’ calculated predicted age was 4 years younger than at baseline).
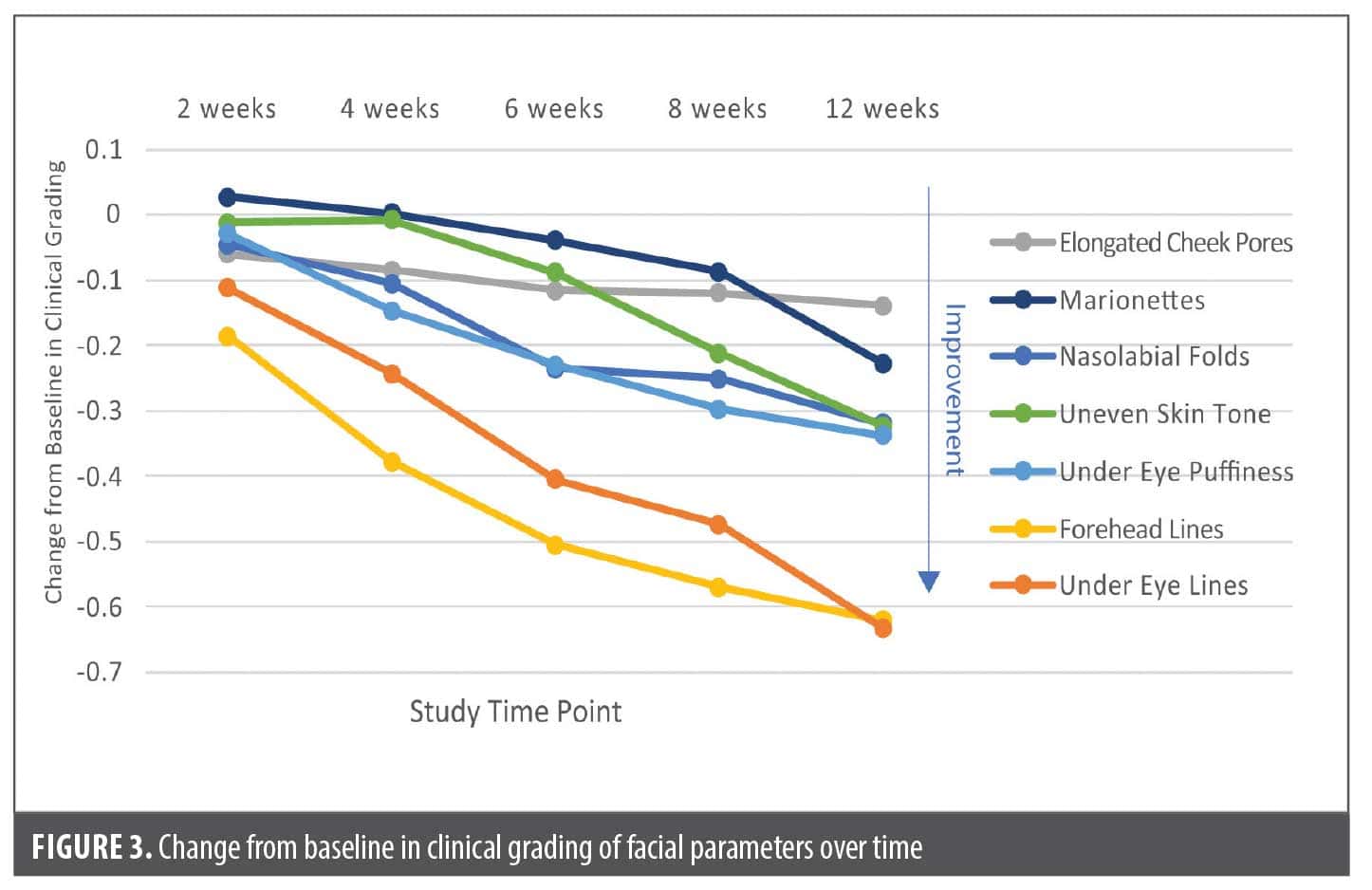
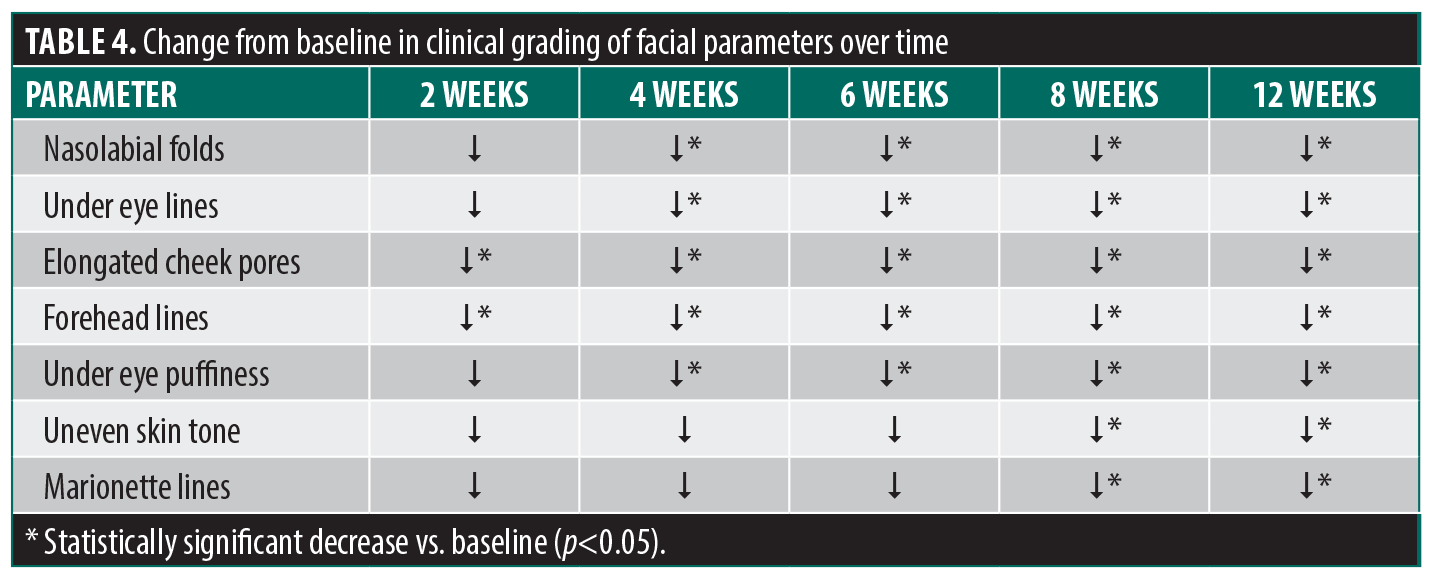
Secondary endpoint: change in individual facial aging parameters. Each of the 7 facial aging parameters improved with treatment but at different rates (Figure 3, Table 4). The fastest improving variables were forehead lines and elongated cheek pores, as statistically significant changes in clinical grading assessments were noted as early as 2 weeks into treatment. Under eye lines and forehead lines showed the greatest calculated improvement over time (Figure 3). Figure 4 shows a sample of images demonstrating changes in clinical grading of the 7 parameters at baseline vs. 12 weeks.
Adverse events. Two subjects experienced adverse events (AEs) deemed possibly related to the study product regimen. One subject experienced very mild pruritus, erythema, and hordeolum in the area under the right eye, and a second subject experienced moderate dryness and erythema on the nose. AEs in both subjects resolved within 5 to 6 days, and the subjects continued in the study without disruption to treatment.
One subject experienced a serious AE of influenza, pneumonia, and staphylococcal infection unrelated to the study regimen. The subject was discontinued from the study but included in the ITT population.
Discussion
Skin aging is caused by both intrinsic (eg, genetics and age-related cellular changes in the skin) and extrinsic (eg, UV exposure, pollution) factors. Both intrinsically and extrinsically aged skin are characterized by decreased expression of specific sirtuin proteins.8-12 Sirtuin proteins are expressed in all skin cells—fibroblasts, melanocytes, and keratinocytes. Activation of sirtuin signaling pathways in skin cells has been shown in in vitro and murine studies to protect cells against UV-induced DNA damage and photoaging as well as against intrinsic hallmarks of skin aging such as cellular senescence and chronic inflammation.12,14-17
The goal of the present study was to assess whether a facial skincare regimen that includes agents that enhance sirtuin activity as well as other active anti-aging ingredients can produce visibly younger-looking skin, as measured by a decrease in perceived age. Study results demonstrate that twice-daily use of the 3-product skincare regimen containing ingredients promoting SIRT1, SIRT3, and SIRT6 activity and other actives resulted in statistically significant visible improvements in 7 key facial parameters. Using the Predicted Age model developed by DiCanio et al,25 these changes could be quantified as a significant decrease in calculated predicted age beginning as early as 2 weeks after first product application, with an average decrease of 4 years after 12 weeks of use in this multiethnic population (ie, at 12 weeks, subjects’ average calculated predicted age was 4 years younger than at baseline). In addition, these age-reversal effects appeared to increase over time, suggesting that regular use of the 3-product regimen over a longer period could help to further improve the appearance of skin.
Due to the lack of a control arm, and all 3 products in the regimen having ingredients that promote sirtuin activity as well as moisturizing, anti-irritant, anti-wrinkle, skin tone improving, and antioxidant agents, we were unable to attribute the observed effects to sirtuin-targeted agents or other active anti-aging agents.
The study aimed to recruit individuals from 4 ethnicities (European American, African American, East Asian, and Hispanic) consistent with the demographics of the population of the United States to ensure representation across the range of skin types (Fitzpatrick I-VI). Demographic makeup of the study population was designed to mirror the multiethnic population on which the Predicted Age model and algorithm were based.25 However, no subjects with Fitzpatrick skin type I were enrolled in the present study, and the majority of subjects were of skin type III or darker. Individuals with lighter skin types are more prone to photoaging effects due to reduced melanin in the skin, with earlier development of wrinkles and sagging skin.27,28 The absence of subjects with Fitzpatrick skin type I, therefore, may have led to a greater than expected mean reduction in calculated predicted age following use of the 3-product facial skincare regimen.
In developing their age prediction model, DiCanio et al25 determined that nasolabial folds and under eye lines were the largest contributors to perceived facial age and that forehead lines and marionettes showed the largest changes with each progressive decade of life (elongated cheek pores showed the least change).25 In the present study, all 7 facial aging parameters studied significantly improved with use of the 3-product skincare regimen, with forehead lines and under eye lines showing the greatest improvement over time. The eye area is of particular relevance in evaluations of products claiming to have age-reversal properties. In a study in which untrained graders estimated the age of 173 Caucasian women based on digital photographs, the eye area was 1 of 2 facial attributes related to perceived age (the other was skin color uniformity).29 The 3-product regimen included an eye crème applied specifically to the eye area, which may have contributed to the enhanced effect on under eye lines.
Although elongated cheek pores demonstrated statistically significant improvements as early as 2 weeks after first use of the facial regimen, the overall magnitude of change was the lowest (–0.14) for this facial parameter during the 12-week study. This is consistent with the age prediction model of DiCanio et al,25 in which elongated cheek pores showed the lowest dynamic range of clinical grading scores across successive age cohorts (from 0 in the age 18 to 29 years cohort to 0.5 for the age ≥60 years cohort).25 Therefore, given the limited potential range of improvement in this variable, a statistically significant change of –0.14 in clinical grading score after 12 weeks is notable.
The 3-product skincare regimen was well tolerated, with only 2 subjects experiencing localized skin reactions that resolved after a few days and did not require disruption of the skincare regimen. These observations suggest that the AEs were not likely to be related to product use.
Limitations
The key limitations of this monadic study were the absence of a control group, use of a single human grader for the clinical grading assessments, and lack of biomarker or instrumental measurements to validate human grader assessments. Inclusion of a comparator or control could have been achieved through a split-face study design, a control group applying only facial cleanser and sunscreen, or a run-in period during which subjects applied facial cleanser and sunscreen only for several weeks followed by regimen products for 12 weeks.
A single trained clinician with approximately 10 years’ experience grading images using the proprietary visual scales used in this study performed the clinical grading assessments. DiCanio et al25 demonstrated excellent inter-rater consistency in grading assessments when developing the model on which the analysis of the present study is based25; hence, we believe similar results would have been obtained had a panel of trained graders been used. Future validation studies of the model by DiCanio et al25 are planned and will compare grading assessments performed by dermatologists versus those performed by trained nondermatologist graders. Artificial intelligence (AI)-based models may be a more objective way to grade facial parameters based on digital photographs. AI-based grading has been shown to correlate well with human expert grading on most, but not all, key facial parameters, with lower accuracy in grading of pigmentation signs, which are particularly relevant to skin of color.30-32 Measurement of biomarkers in biopsied skin tissue or instrumentation-based measurement of parameters such as forehead or under eye lines are possible methods of validating the trained grader’s assessments.33 However, these assessments would have been more invasive and burdensome for subjects.
Conclusion
Based on the Predicted Age model developed by DiCanio et al,25 twice-daily application of the 3-product facial and eye skincare regimen for 12 weeks resulted in younger-appearing skin, as measured by significant visible improvements in 7 key facial aging parameters and an average decrease of 4 years in calculated predicted age, in this multiethnic population. The predicted age-reversal effect was statistically significant starting at Week 2 of product application and was maintained until Week 12 of the study, with continued improvement over time. In addition, the 3-product regimen was well tolerated for the duration of the study.
References
- Wyles S, Mehta R, Mannick J, Day D. Skin longevity: a paradigm shift in aesthetics. J Cosmet Dermatol. 2024;23(9):2814-2815.
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: an expanding universe. Cell. 2023;186(2):243-278.
- Fitsiou E, Pulido T, Campisi J, Alimirah F, Demaria M. Cellular senescence and the senescence-associated secretory phenotype as drivers of skin photoaging. J Invest Dermatol. 2021;141(4S):1119-1126.
- Chin T, Lee XE, Ng PY, Lee Y, Dreesen O. The role of cellular senescence in skin aging and age-related skin pathologies. Front Physiol. 2023;14:1297637.
- Zhang J, Yu H, Man MQ, Hu L. Aging in the dermis: fibroblast senescence and its significance. Aging Cell. 2024;23(2):e14054.
- Garcia-Peterson LM, Wilking-Busch MJ, Ndiaye MA, et al. Sirtuins in skin and skin cancers. Skin Pharmacol Physiol. 2017;30(4):216-224.
- Lee Y, Shin MH, Kim MK, et al. Increased histone acetylation and decreased expression of specific histone deacetylases in ultraviolet-irradiated and intrinsically aged human skin in vivo. Int J Mol Sci. 2021;22(4):2032.
- Sharma A, Diecke S, Zhang WY, et al. The role of SIRT6 protein in aging and reprogramming of human induced pluripotent stem cells. J Biol Chem. 2013;288(25):18439-18447.
- Kalfalah F, Sobek S, Bornholz B, et al. Inadequate mito-biogenesis in primary dermal fibroblasts from old humans is associated with impairment of PGC1A-independent stimulation. Exp Gerontol. 2014;56:59-68.
- Pelle E, Dong K, Pernodet N. Temporal variations in sirtuin expression under normal and ultraviolet B-induced conditions and their correlation to energy levels in normal human epidermal keratinocytes. J Cosmet Sci. 2015;66(1):57-63.
- McCarthy JT, Pelle E, Dong K, et al. Effects of ozone in normal human epidermal keratinocytes. Exp Dermatol. 2013;22(5):360-361.
- Lee JS, Park KY, Min HG, et al. Negative regulation of stress-induced matrix metalloproteinase-9 by Sirt1 in skin tissue. Exp Dermatol. 2010;19(12):1060-1066.
- Fan L, Luan X, Jia Y, et al. Protective effect and mechanism of lycium barbarum polysaccharide against UVB-induced skin photoaging. Photochem Photobiol Sci. 2024;23(10):1931-1943.
- Kawahara TL, Michishita E, Adler AS, et al. SIRT6 links histone H3 lysine 9 deacetylation to NF-kappaB-dependent gene expression and organismal life span. Cell. 2009;136(1):62-74.
- Geng A, Tang H, Huang J, et al. The deacetylase SIRT6 promotes the repair of UV-induced DNA damage by targeting DDB2. Nucleic Acids Res. 2020;48(16):9181-9194.
- Wen SY, Ng SC, Chiu YT, et al. Enhanced SIRT1 activity by galangin mitigates UVB-induced senescence in dermal fibroblasts via p53 acetylation regulation and activation. J Agric Food Chem. 2024;72(42):23286-23294.
- Di Raimo R, Mizzoni D, Aloi A, et al. Antioxidant effect of a plant-derived extracellular vesicles’ mix on human skin fibroblasts: induction of a reparative process. Antioxidants (Basel). 2024;13(11):1373.
- Di Lorenzo R, Falanga D, Ricci L, et al. NAD-driven sirtuin activation by Cordyceps sinensis extract: exploring the adaptogenic potential to promote skin longevity. Int J Mol Sci. 2024;25(8):4282.
- Wang T, Qin Y, Qiao J, Liu Y, Wang L, Zhang X. Overexpression of SIRT6 regulates NRF2/HO-1 and NF-κB signaling pathways to alleviate UVA-induced photoaging in skin fibroblasts. J Photochem Photobiol B. 2023;249:112801.
- Moreau M, Neveu M, Stéphan S, et al. Enhancing cell longevity for cosmetic application: a complementary approach. J Drugs Dermatol. 2007;6(6 Suppl):s14-9.
- Choi HJ, Alam MB, Baek ME, Kwon YG, Lim JY, Lee SH. Protection against UVB-induced photoaging by Nypa fruticans via inhibition of MAPK/AP-1/MMP-1 signaling. Oxid Med Cell Longev. 2020;2020:2905362.
- La Padula S, Hersant B, SidAhmed M, Niddam J, Meningaud JP. Objective estimation of patient age through a new composite scale for facial aging assessment: the face-objective assessment scale. J Craniomaxillofac Surg. 2016;44(7):775-782.
- Sen S, Choudhury S, Gangopadhyay A, Halder C, Biswas P, Jain A. A clinical rating scale for the assessment of facial aging in Indian population. Indian J Dermatol Venereol Leprol. 2016;82(2):151-161.
- Rzany B, Carruthers A, Carruthers J, et al. Validated composite assessment scales for the global face. Dermatol Surg. 2012;38:294-308.
- Dicanio D, Lain T, Del Rosso J, et al. Development of a novel facial age assessment model in a multiethnic population for evaluation of topical anti-aging products. J Clin Aesthet Dermatol. 2025;18(11):24-29.
- Goyarts EC, Dong K, Pelle E, Pernodet N. Effect of SIRT6 knockdown on NF-κB induction and on residual DNA damage in cultured human skin fibroblasts. J Cosmet Sci. 2017;68(1):25-33.
- Vashi NA, de Castro Maymone MB, Kundu RV. Aging differences in ethnic skin. J Clin Aesthet Dermatol. 2016;9(1):31-38.
- Hudson C, Brissett A, Carniol P. Analysis and assessment of facial aging. Curr Otorhinolaryngol Rep. 2021;9(2):415-421.
- Nkengne A, Bertin C, Stamatas GN, et al. Influence of facial skin attributes on the perceived age of Caucasian women. J Eur Acad Dermatol Venereol. 2008;22(8):982-991.
- Flament F, Hofmann M, Roo E, et al. An automatic procedure that grades some facial skin structural signs: agreements and validation with clinical assessments made by dermatologists. Int J Cosmet Sci. 2019;41(5):472-478.
- Flament F, Lee YW, Lee DH, et al. The continuous development of a complete and objective automatic grading system of facial signs from selfie pictures: Asian validation study and application to women of three ethnic origins, differently aged. Skin Res Technol. 2021;27(2):183-190.
- Flament F, Jiang R, Houghton J, et al. Accuracy and clinical relevance of an automated, algorithm-based analysis of facial signs from selfie images of women in the United States of various ages, ancestries and phototypes: a cross-sectional observational study. J Eur Acad Dermatol Venereol. 2023;37(1):176-183.
- DiCanio D, Sparacio R, Declercq L, et al. Calculation of apparent age by linear combination of facial skin parameters: a predictive tool to evaluate the efficacy of cosmetic treatments and to assess the predisposition to accelerated aging. Biogerontology. 2009;10(6):757-772.