J Clin Aesthet Dermatol. 2025;18(12):45–49.
by Rachel M. Seifert, BA; Christiaan H. Noot, BS; Naiem T. Issa, MD, PhD; Christopher M. Hull, MD; Jamie L. W. Rhoads, MD, MS; Jennie T. Clarke, MD; Christopher B. Hansen, MD; John J. Zone, MD; Zachary H. Hopkins, MD
Ms. Seifert, Mr. Noot, and Drs. Hull, Rhoads, Clarke, Hansen, Zone, and Hopkins are with the University of Utah Spencer F. Eccles School of Medicine, Department of Dermatology, Autoimmune Skin Diseases Clinic in Salt Lake City, Utah. Dr. Issa is with Forefront Dermatology in Vienna, Virginia, the Dr. Phillip Frost Department of Dermatology and Cutaneous Surgery at the University of Miami School of Medicine in Miami, Florida, and the George Washington University School of Medicine and Health Science in Washington, District of Columbia.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Hopkins is supported by a career development award from the Dermatology Foundation and has received consultation fees from Priovant Therapeutics. The remaining authors have no conflicts of interest relevant to the contents of this article.
ABSTRACT: Background: Oral lichen planus (OLP) is an inflammatory disease involving the oral mucosa. It affects roughly 0.5% to 2% of the global population and has an associated risk of oral squamous cell carcinoma. Treatment of moderate to severe OLP often requires immunosuppression. The durability of immunosuppressive medication is currently unknown and is important for understanding therapeutic testing needs. Objective: We investigated traditional immunosuppressive drug survival in patients with OLP and evaluated potential discontinuation factors. Methods: We retrospectively analyzed patients with OLP treated with methotrexate, mycophenolate, azathioprine, or cyclosporine. Time to medication discontinuation was evaluated using the Kaplan-Meier estimator, and Cox proportional hazards regression was used to compare the risk of discontinuing a medication between medications and across patient demographic and disease factors. Results: We identified 125 treatment periods with mycophenolate (n=58), methotrexate (n=34), azathioprine (n=19), or cyclosporine (n=14). Most patients had erosive disease (92%), and median time (IQR) to discontinuation due to adverse events or inefficacy was 9.43 months (6.51-16.1). Overall, only cyclosporine was associated with higher risk of discontinuation compared to methotrexate (hazard ratio [HR]: 2.94; 95% confidence interval [CI]: 1.32-6.45). There was no evidence for risk differences across age or sex for the overall cohort. Within individual medication groups, age was associated with a small increased risk of discontinuing mycophenolate (HR: 1.05; 95% CI: 1.00-1.10) and a small decreased risk in cyclosporine (HR=0.94, 95% CI: 0.89-0.99). Otherwise, no demographic factors were associated with discontinuation. Treatment success was reported 8 times. Discussion: Immunosuppressive medications were frequently discontinued after short time periods, and few were discontinued due to success. These data highlight the need for better systemic therapy in OLP. Keywords: Autoimmune, lichen planus, oral lichen planus, immunosuppression, epidemiology, dermatology
Introduction
Oral lichen planus (OLP) is a mucocutaneous inflammatory disease thought to be driven by a T-cell immune response, potentially to unknown antigens.1 Approximately 0.5% to 2% of the global population is affected, and there has been an associated increased risk of oral squamous cell carcinoma, particularly among more severe inflammatory or ulcerative subgroups.2,3 OLP is highly morbid, with one qualitative study noting that 69% of patients experienced significant impact on their activities of daily living.4 Although first-line treatment involves the use of topical corticosteroids, the evidence for its efficacy is somewhat inconclusive, and our clinical experience suggests that many patients with moderate-to-severe OLP require additional systemic therapy. A Cochrane systematic review found no significant difference in clinical resolution between topical corticosteroids and placebo, though pain was more likely to be resolved using topical corticosteroids.5 If topical therapies prove insufficient, severe disease may require treatment with immunosuppressive medications such as mycophenolate, methotrexate, azathioprine, and cyclosporine.6 Currently, immunosuppressive medication durability in OLP is unknown. Medication durability can provide insight into tolerability and efficacy.7 We also assessed for factors associated with discontinuation of treatment in the overall group and medication subgroups.
Methods
We included patients seen at the University of Utah from January 1, 2010 to April 1, 2024 with a dermatologist-verified OLP diagnosis treated with methotrexate (MTX), mycophenolate mofetil (MMF), azathioprine (AZA), or cyclosporine (CsA). OLP diagnosis required OLP diagnosis over at least two visits and clinically congruent appearance. While we did not require histologic confirmation, most cases had confirmatory histology (68 of 91 [74.7%]). We excluded patients with concomitant systemic treatments (besides hydroxychloroquine/short-term oral corticosteroid), undocumented reason for treatment discontinuation, or multiple trials of the same immunosuppressant (only the first trial was included). In patients who used multiple unique immunosuppressants at nonoverlapping time points (eg, MTX that was transitioned to MMF due to lack of efficacy), each trial was considered individually. Descriptive statistics included medians/interquartile ranges (IQR) and counts or percentages. Group comparisons were made using Kruskal-Wallis (continuous variables) and chi-square test (categorical variables). Time to medication discontinuation was evaluated using Kaplan-Meier estimator, and Cox proportional hazards regression was used to compare the risk of discontinuing a medication between medications and across different demographic factors.8,9 Our primary outcome was hazard of medication discontinuation due to adverse event or medication failure. Secondary analyses included an age- and sex-adjusted model for the primary outcome, individual evaluations of failure due to adverse events, medication failure, and medication success (ie, patient remission). Analyses were performed across medication groups and the overall cohort, as well as by individual medications. All analyses were performed in Stata v17.1, StataCorp, and p<0.05 was considered significant.
Results
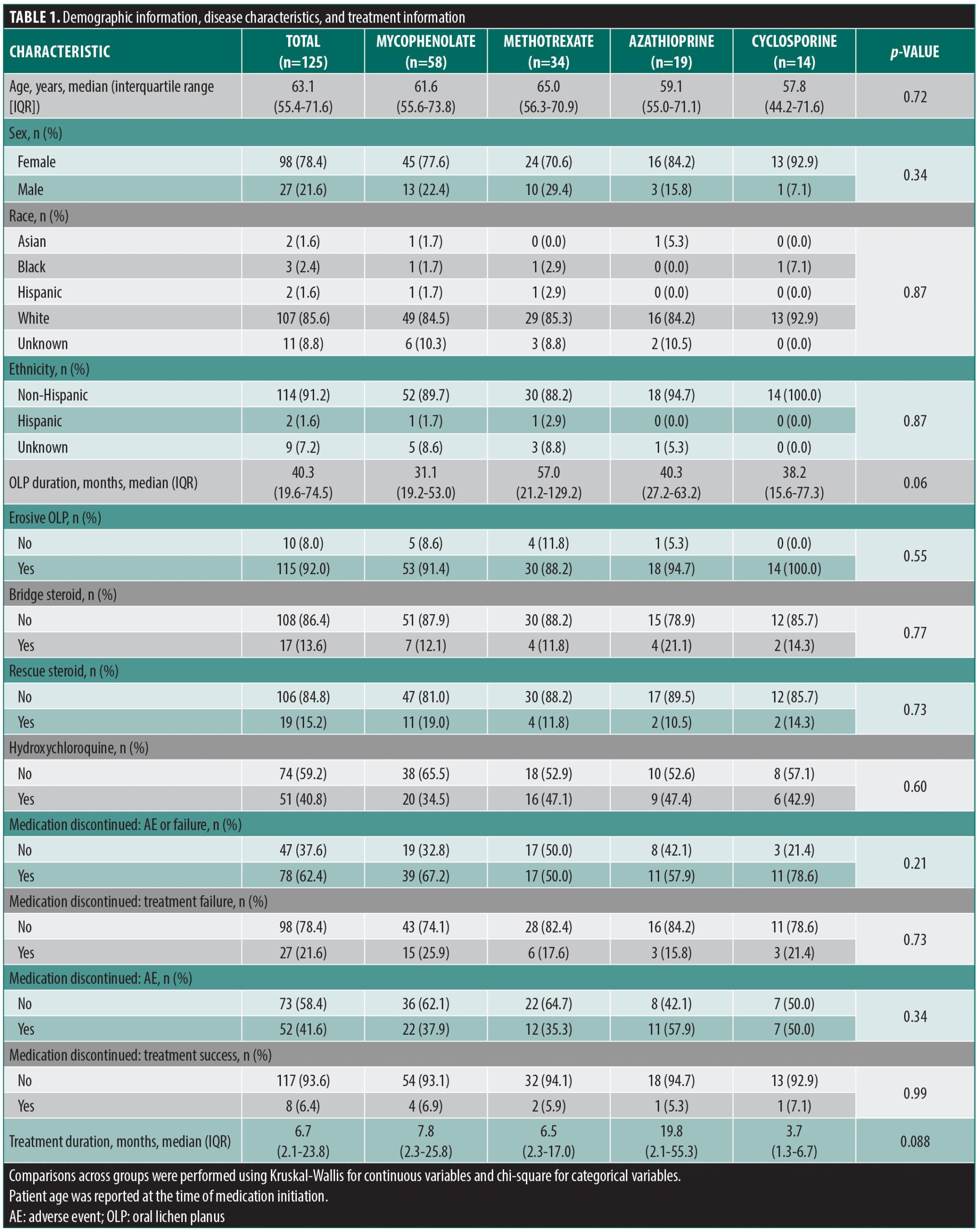
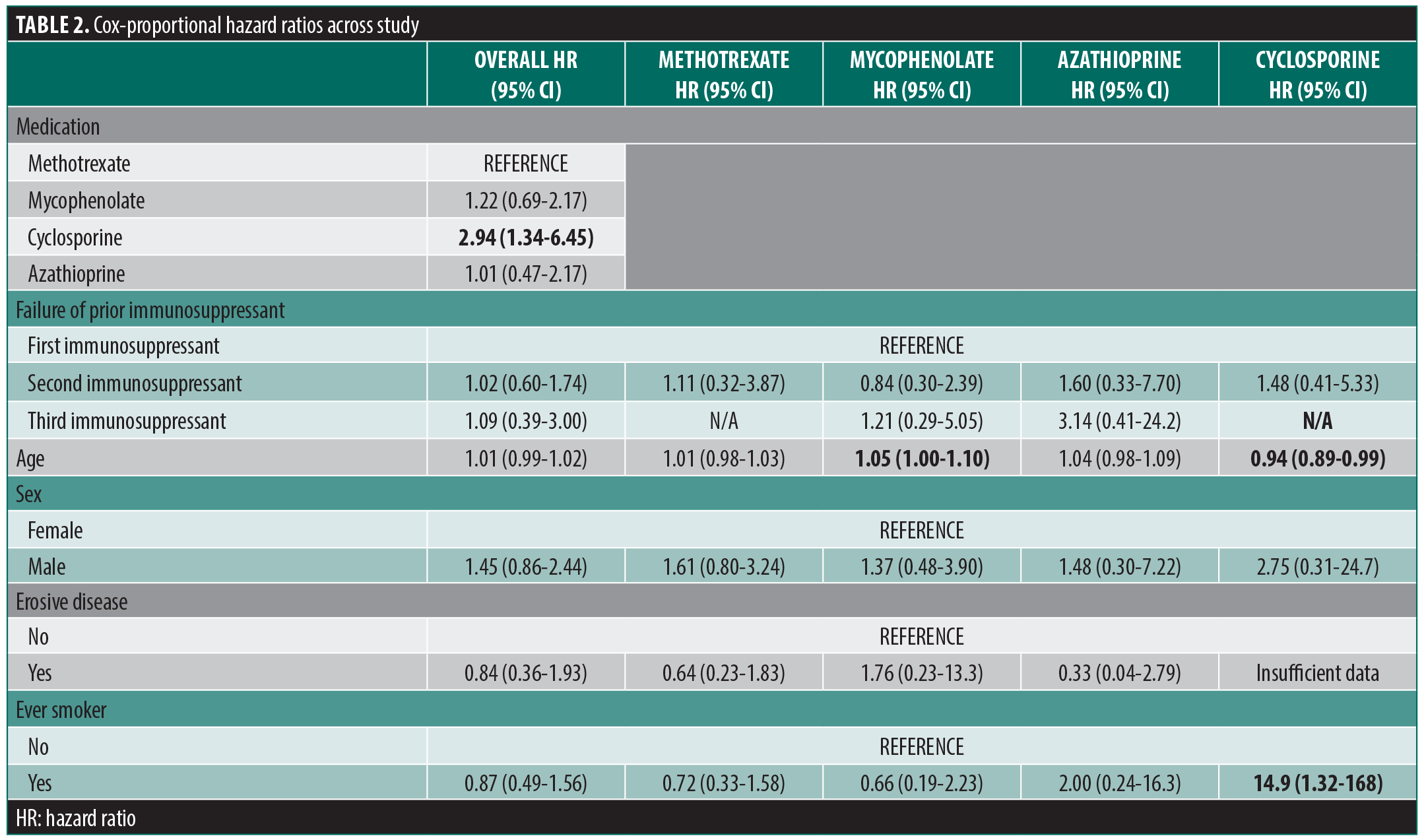
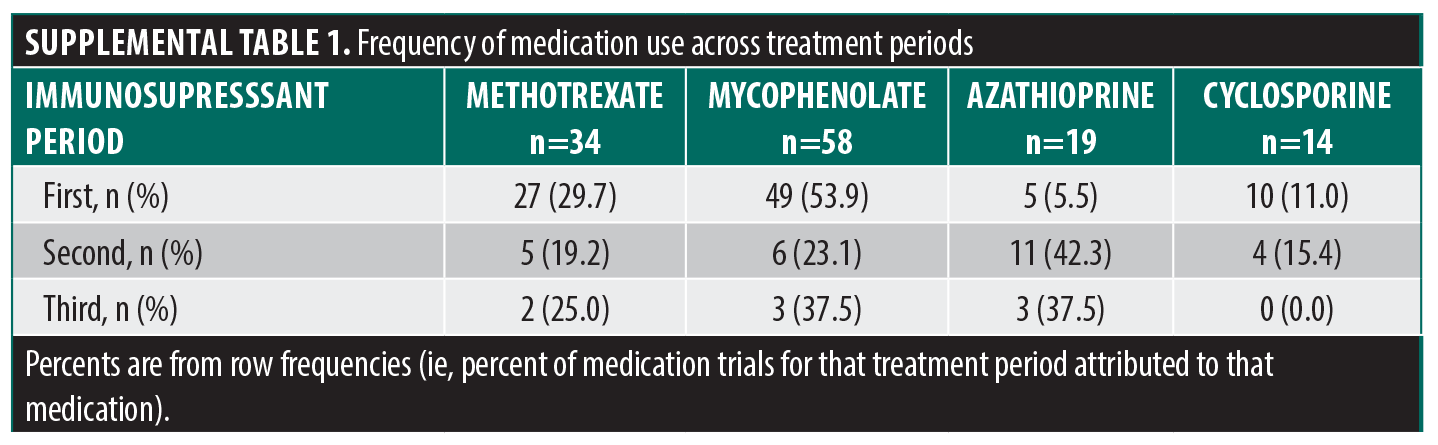
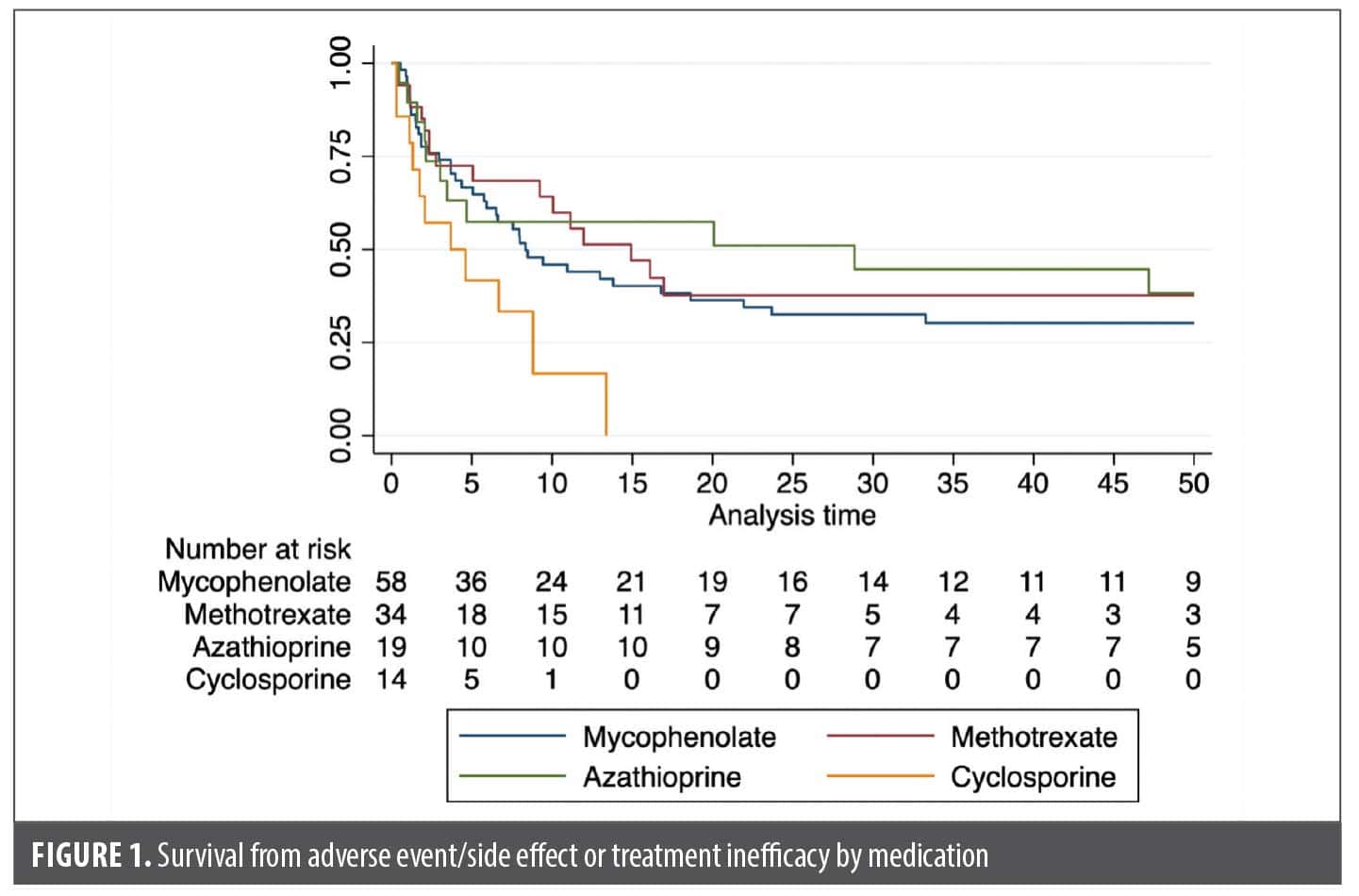
We identified 125 OLP treatment periods, which included treatment with MMF (n=58), MTX (n=34), AZA (n=19), and CsA (n=14), by 91 patients. Of these patients, 68 (74.7%) had confirmatory biopsies. The remaining patients had clinically consistent disease noted by a dermatologist with extensive OLP experience. Reasons for lack of confirmatory biopsy included: diagnoses that occurred prior to our current record availability, skin biopsies confirming lichen planus and OLP was assumed to be related, and nonconfirmatory biopsies but the clinician felt the oral disease was clinically consistent. Sixty-five patients had only one treatment period, 18 had two, and 8 had three treatment periods. MMF and MTX were used most during the first treatment period and overall, but AZA was most used during the second period (Supplemental Table 1). Overall, median (IQR) patient age was 63.1 years (55.4-71.6), 78.4% were female, and 85.6% were White (Table 1). Most had erosive disease (92.0%). Adjunctive therapy use (ie, hydroxychloroquine or oral corticosteroid) was similar between treatment cohorts. Treatment was discontinued in 113 treatment periods. Reasons for discontinuation included: adverse events or treatment failure (n=79), treatment success (n=8), lack of efficacy (n=27), and adverse events (n=52). Median (IQR) time to discontinuation overall was 9.43 months (6.51-16.1). Median time to discontinuation was highest for the first trialed immunosuppressant (10.1 months; 6.67-16.8), and decreased for the second (6.51 months; 3.45-47.2) and third (5.06 months; 0.89-N/A). Only CsA was associated with an increased risk for discontinuation compared to methotrexate (hazard ratio [HR]: 2.66; 95% CI: 1.25-5.65), including after adjusting for age and sex (HR: 2.94; 95% CI: 1.36-6.32) (Table 2, Figure 1). Across all medications and within each individual medication, we found no evidence for factors influencing hazard of discontinuation, save a small age effect in MMF and an imprecise, but large, impact on smoking for CsA (Table 2). We also evaluated if having failed one immunosuppressant increases the odds of failing the subsequent medication, but we did not find evidence for this (Table 2). Patients were concomitantly on hydroxychloroquine in 51 of 125 (40.8%) treatment periods. We found no evidence for decreased risk of discontinuation with hydroxychloroquine use (HR: 0.76; 95% CI: 0.48-1.19) nor an increased risk of treatment success (HR: 1.63; 95% CI: 0.36-7.41).
When analyses were stratified by individual medications, treatment discontinuation due to treatment failure occurred more often in men (HR: 1.91; 95% CI: 1.05-3.45) but otherwise estimates were similar. Corticosteroid rescue was identified 19 times. We identified no differences in steroid rescues across medications nor by patient and disease demographics. Discontinuation due to treatment success occurred in 8 treatment periods. We found no evidence for differences in chance of success across medication groups nor demographic factors.
Discussion
We found that immunosuppressive medications for this OLP cohort were frequently discontinued, due to adverse events and treatment failure but infrequently for treatment success. The median time to these events was 9.43 months, which is shorter than what has been observed when using these medications for other indications like bullous pemphigoid.10 For example, median drug survival in this study was 20.4 to 72.2 months compared to 9.43 months in our cohort.10
We generally found no evidence for differences in immunosuppressive durability between medications, except for CsA, suggesting that no one medication had better efficacy or safety profiles. We suspect that the difference seen in CsA may be driven in part by use of this medication as a short-term medication (with a goal to use for ≤12 months typically). However, this was difficult to fully assess in this database and survival times tended to be lower than 1 year. In a different OLP cohort, Myers et al11 similarly found no difference in adverse event prevalence between MMF, MTX, and AZA in OLP. However, their cohort did experience treatment-limiting adverse events less frequently (16.0-27.8%). The reason for this is unclear, but may stem from our cohort having a high prevalence of severe disease, as evidenced by the high prevalence of erosive disease. Although the male patients in our cohort appeared to have a higher hazard of treatment failure, we could not identify a clear reason for this.
This study has important limitations. First, all cases came from a single institution with limited racial or ethnic representation, limiting generalizability. There is also a potential for misclassification, given that 25% of cases did not have a biopsy available. Furthermore, most cases were severe with erosive subtype disease, which may alter drug survival. For example, with more severe erosive disease, smaller improvements may be considered grounds for discontinuation, whereas in more moderate disease, these improvements might be more acceptable and the medication may be more likely to be continued. We found no evidence for differences in drug survival by erosivity, but this confounding may remain, and generalizability is limited. The order of immunosuppressant usage may affect subsequent treatment periods. We attempted to account for this, but it is possible that other treatments that were not reported (ie, used by a referring physician or prior to our current electronic medical record time period). Lastly, direct comparisons between medications are limited by the retrospective nature and likely confounding by indication.
Overall, our results highlight that patients with OLP frequently encounter adverse events, treatment failure, and low immunosuppressive durability. These data underscore the difficulty of treating this condition and the need for more effective treatments with favorable safety profiles. Promisingly, cases of excellent treatment response to Janus kinase inhibitor (JAKi) therapy are emerging. For example, we recently reported a 10-patient case series reporting the effectiveness of upadacitinib, an oral JAKi, in the treatment of recalcitrant OLP,12 and additional cases of successful treatment using upadacitinib13 and deucravacitinib, a tyrosine kinase 2 inhibitor, have been recently reported.14 Conversely, a recent clinical trial of secukinumab failed to demonstrate efficacy in lichen planus subtypes, including OLP.15 However, further prospective clinical trials are needed.
References
- El-Howati A, Thornhill MH, Colley HE, Murdoch C. Immune mechanisms in oral lichen planus. Oral Dis. 2023;29(4):1400-1415.
- Nukaly HY, Halawani IR, Alghamdi SMS, et al. Oral lichen planus: a narrative review navigating etiologies, clinical manifestations, diagnostics, and therapeutic approaches. J Clin Med. 2024;13(17):5280.
- Roopashree MR, Gondhalekar RV, Shashikanth MC, et al. Pathogenesis of oral lichen planus—a review. J Oral Pathol Med. 2010;39(10):729-734.
- Mahon-Smith A, Clifford M, Batish A, et al. Patient experience of lichen planus: a qualitative exploration of signs, symptoms, and health-related quality of life impacts. Dermatol Ther (Heidelb). 2023;13(9):2001-2017.
- Lodi G, Manfredi M, Mercadante V, Murphy R, Carrozzo M. Interventions for treating oral lichen planus: corticosteroid therapies. Cochrane Database Syst Rev. 2020;2(2):CD001168.
- Louisy A, Humbert E, Samimi M. Oral lichen planus: an update on diagnosis and management. Am J Clin Dermatol. 2024;25(1):35-53.
- van den Reek JMPA, Kievit W, Gniadecki R, et al. Drug survival studies in dermatology: principles, purposes, and pitfalls. J Invest Dermatol. 2015;135(7):1-5.
- Bland JM, Altman DG. Survival probabilities (the Kaplan-Meier method). BMJ. 1998;317(7172):1572.
- Wakkee M, Hollestein LM, Nijsten T. Multivariable analysis. J Invest Dermatol. 2014;134(5):1-5.
- Awethe Z, Viveiros M, Kaffenberger J. Durability of long-term immunomodulating medications in the treatment of bullous pemphigoid. J Am Acad Dermatol. 2023;89(6):1287-1289.
- Myers EL, Hollis AN, Culton DA. Effectiveness and tolerability of systemic therapies in oral lichen planus: a retrospective cohort study. JAAD Int. 2024;15:136-138.
- Noot CH, Hansen AM, Frost Z, et al. Oral lichen planus treated with upadacitinib: a case series. JAAD Case Rep. 2025;63:125-127.
- Sheehan CA, Brownstone ND, Lee JB, Hsu S. Use of upadacitinib for recalcitrant oral erosive lichen planus. JAAD Case Reports. 2025;60:167-169.
- Vu M, Abdin R, Issa NT. Treatment of oral lichen planus using deucravacitinib. JAAD Case Rep. 2024;44:94-97.
- Passeron T, Reinhardt M, Ehst B, et al. Secukinumab in adult patients with lichen planus: efficacy and safety results from the randomized placebo-controlled proof-of-concept PRELUDE study. Br J Dermatol. 2024;191(5):680-690.