J Clin Aesthet Dermatol. 2025;18(11):48–57.
by Nicole Werpachowski, DO; Lyudmyla Susla, BS; and Karan Lal, DO, FAAD
Dr. Werpachowski is with Lenox Hill Hospital, Northwell Health in New York, New York. Ms. Susla is with the New York Institute of Technology College of Osteopathic Medicine in Old Westbury, New York. Dr. Lal is with Affiliated Dermatology in Scottsdale, Arizona.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors report no conflicts of interest relevant to the content of this article.
ABSTRACT: Objective: Infantile hemangioma (IH) is the most common benign childhood tumor. Timolol is a widely used treatment for IH due to its efficacy and safety. Although systemic absorption is rare, timolol has been detected in urine and blood, raising concerns about potential adverse effects. This study aims to systematically review the literature on reported adverse effects associated with topical timolol for IH treatment. Methods: A systematic review following 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines was conducted. PubMed and MEDLINE databases (2000-2024) were searched. Studies reporting treatment-related adverse effects of topical timolol for IH treatment were included. Results: Twenty articles met inclusion criteria, comprising 1780 patients. Local adverse effects occurred in 4.7% of patients, including irritation, scaling, ulceration, and pruritus. Systemic adverse effects occurred in 1.2% of cases, including bradycardia, bronchospasm, wheezing, hypothermia, and sleep disturbances. There was no evidence that the severity or frequency of local adverse effects predisposed patients to systemic ones. Similarly, there was no pattern to suggest that longer treatment durations were associated with increased systemic effects. Limitations: Limitations include heterogeneity of included studies and the exclusion of studies that did not report complications, which may overestimate the frequencies of local and systemic adverse effects. Conclusion: Topical timolol is generally well tolerated for IH treatment, with systemic adverse effects occurring infrequently. Preterm infants and those with ulcerated or deep IHs may be at increased risk for complications. Further research is warranted to better define risk factors for systemic absorption and establish optimal dosing guidelines for safe use in infants. Keywords: Timolol, topical timolol, infantile hemangioma, superficial, ulceration, adverse effects
Introduction
Infantile hemangiomas (IHs) are the most common benign vascular tumors, with a prevalence of 4 to 5% in mature neonates. Many of the identified risk factors for IH development include prematurity, low birth weight, placental anomalies or intrauterine complications, and family history.1,2 For instance, Goelz and Poets2 reported how the incidence of IHs increases up to 23% in neonates weighing less than 1000 grams at birth. The pathogenesis of IH involves a complex interplay of genetic predisposition, dysregulated vasculogenesis and angiogenesis, and environmental factors such as fetal hypoxia.3 Their growth is not linear, as IHs undergo a proliferative phase (until 4 to 18 months of age) followed by gradual involution (over 3 to 9 years on average).4 Most cases regress by 4 years of age, but deeper lesions may persist for several more years.5 Clinically, superficial IHs appear as red, lobulated plaques, and deep IHs extend into the reticular dermis and present as bluish tumors.6
While most IHs regress spontaneously, complications such as ulceration, bleeding, disfigurement, and functional impairment may necessitate treatment. In particular, ulceration, which can be painful, is one of the more common complications, with large, superficial, and segmental IHs at increased risk.7 These complications underscore the importance of timely and appropriate treatment to mitigate risks and prevent functional impairment. Systemic β-blockers like oral propranolol are the standard of care but carry potent systemic risks, including bradycardia, hypotension, or bronchospasm.8 Topical timolol is an effective alternative for smaller tumors or those borderline case necessitating systemic treatment, offering theoretically fewer systemic risks due to its localized mechanism of action. However, concerns about systemic absorption and associated adverse effects remain and have been reported, particularly with deep IHs. This systematic review aims to investigate the range of complications associated with topical timolol use in IH management and identify patient subgroups at higher risk of adverse effects, providing dermatologists and other clinicians with valuable insights to optimize treatment safety.
Methods
The 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines on the conduct of systematic reviews were adhered to. The PubMed (January 1, 2000 through December 31, 2024) database was queried with the following search terms: (( (( “Timolol/administration and dosage”[Mesh] OR “Timolol/adverse effects”[Mesh] OR “Timolol/therapeutic use”[Mesh] OR “Timolol/toxicity”[Mesh] OR timolol)) AND (“Hemangioma”[Mesh] OR hemangioma). The MEDLINE (2000-2024) database was queried with the following search terms: (timolol.af or timolol.hw) and (“infantile hemangioma”.af or hemangioma.af or hemangioma.hw). Both databases were adjusted with filters for language (English) and age: (infant: birth – 23 months). Publication duplicates were reconciled using Rayyan’s online platform. The results of the database search were independently screened by two researchers to ensure a comprehensive and unbiased selection process.
Studies met inclusion criteria if they used topical timolol specifically for the treatment of IHs and detailed adverse effects or complications related to treatment. Clinical trials, prospective/retrospective interventional studies, case-control studies, case series, and case reports documenting the treatment response of IH to any preparation of topical timolol were included. Abstracts, clinical studies, interventional studies, review articles, and cases that did not use topical timolol (i.e., other topical or systemic β-blockers) for the treatment of IHs or did not discuss treatment-related adverse effects were excluded. An additional five studies, not captured in the initial PubMed and MEDLINE database outputs, identified from review articles were also assessed for inclusion in this systematic review.
Results
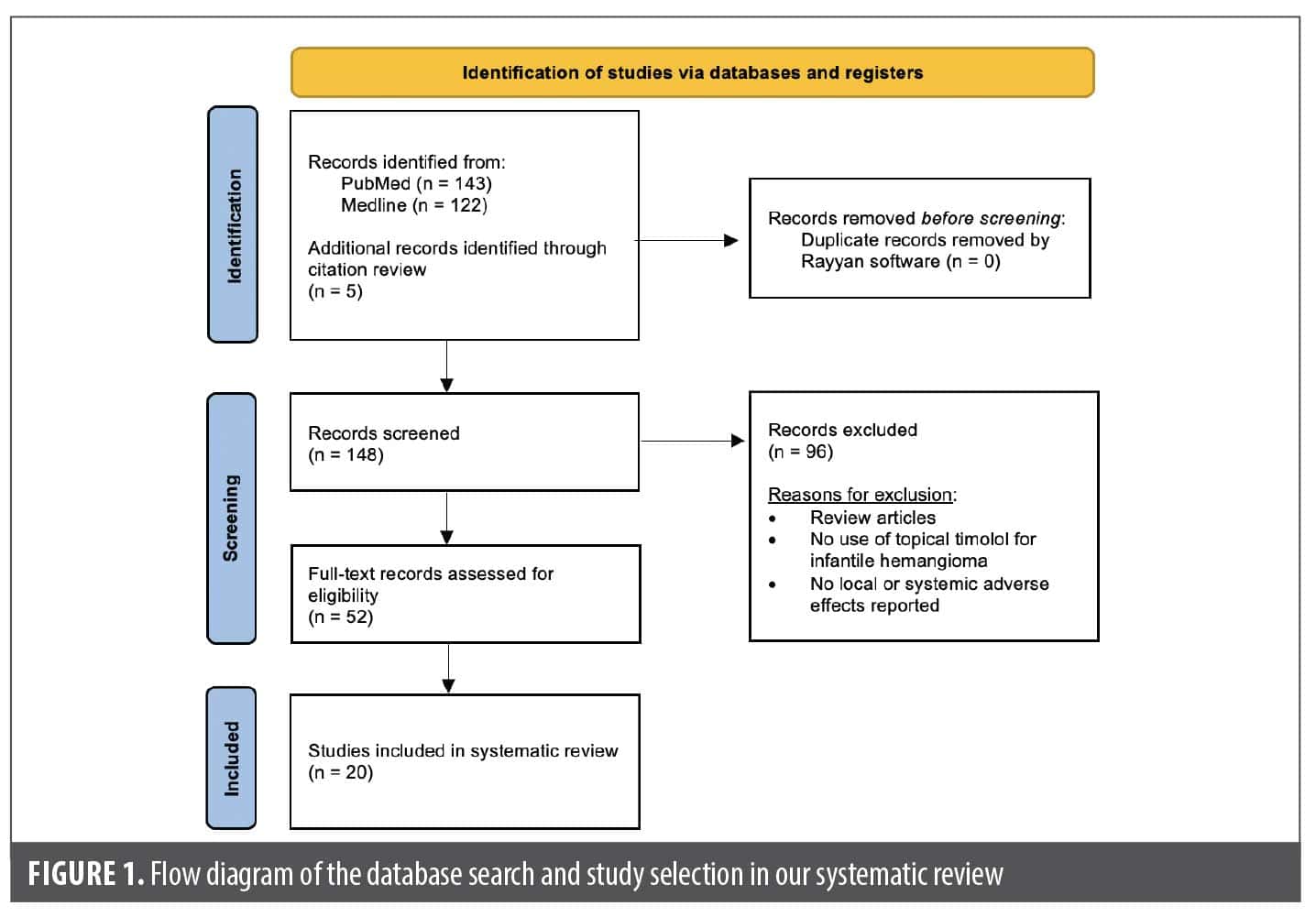
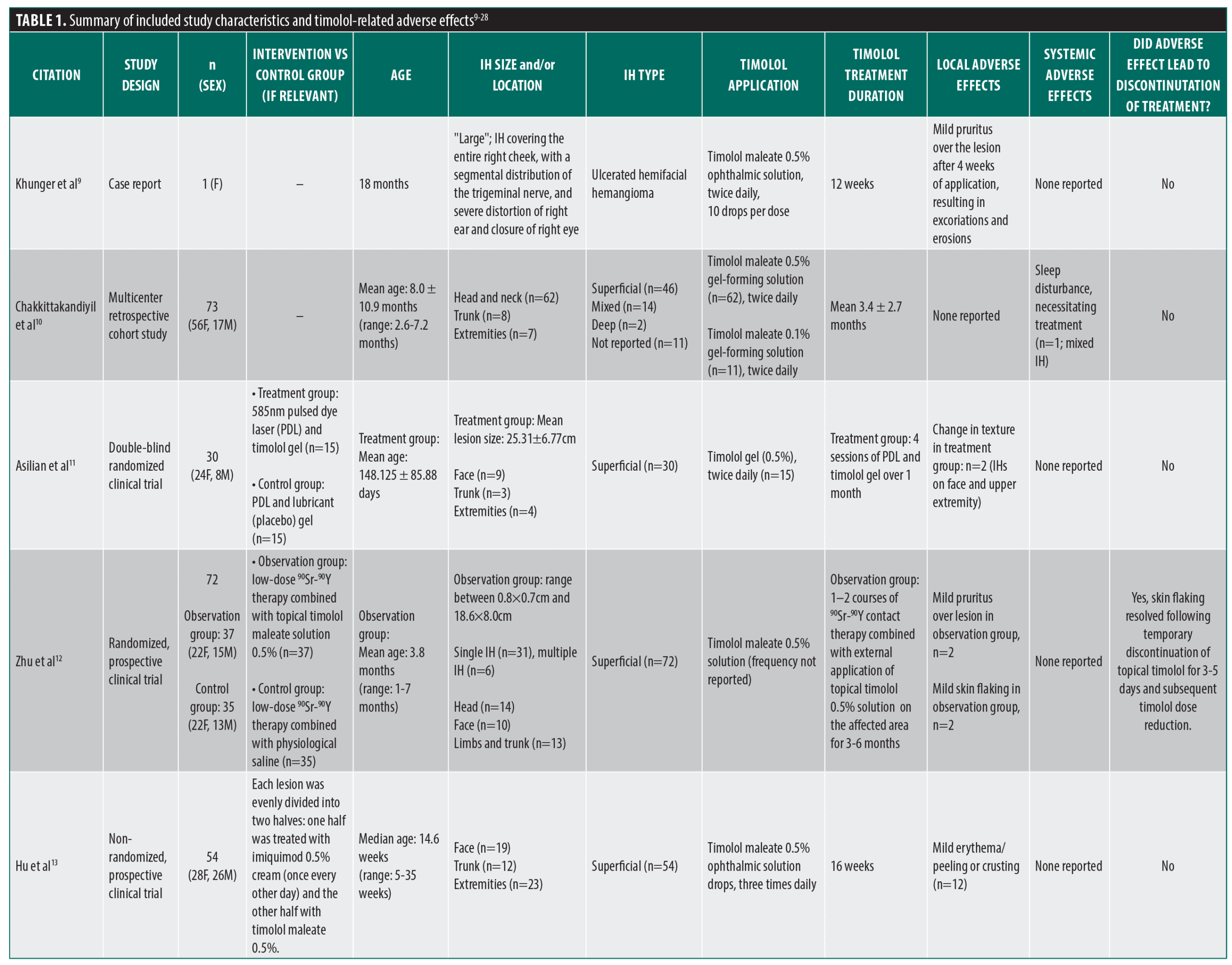
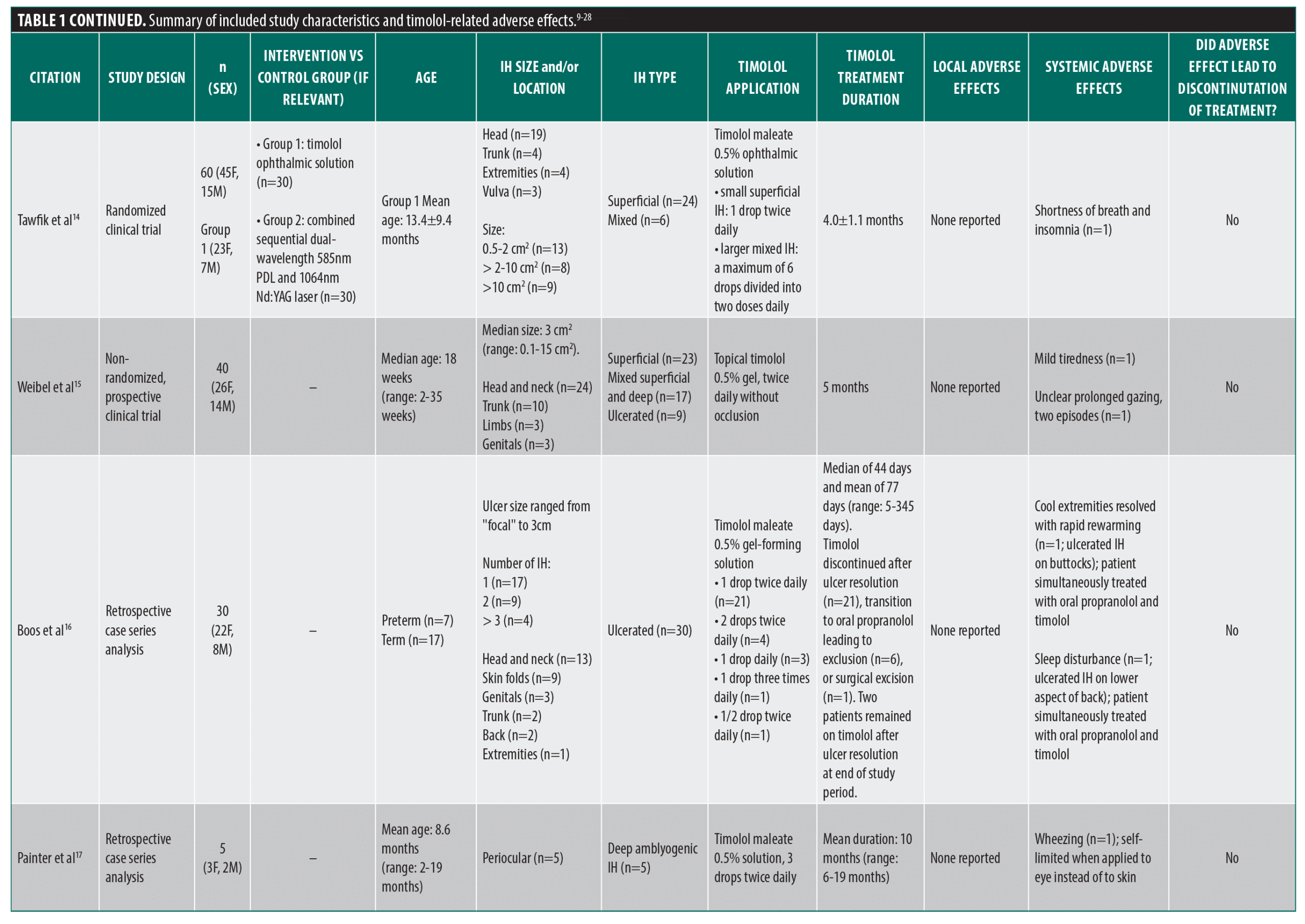
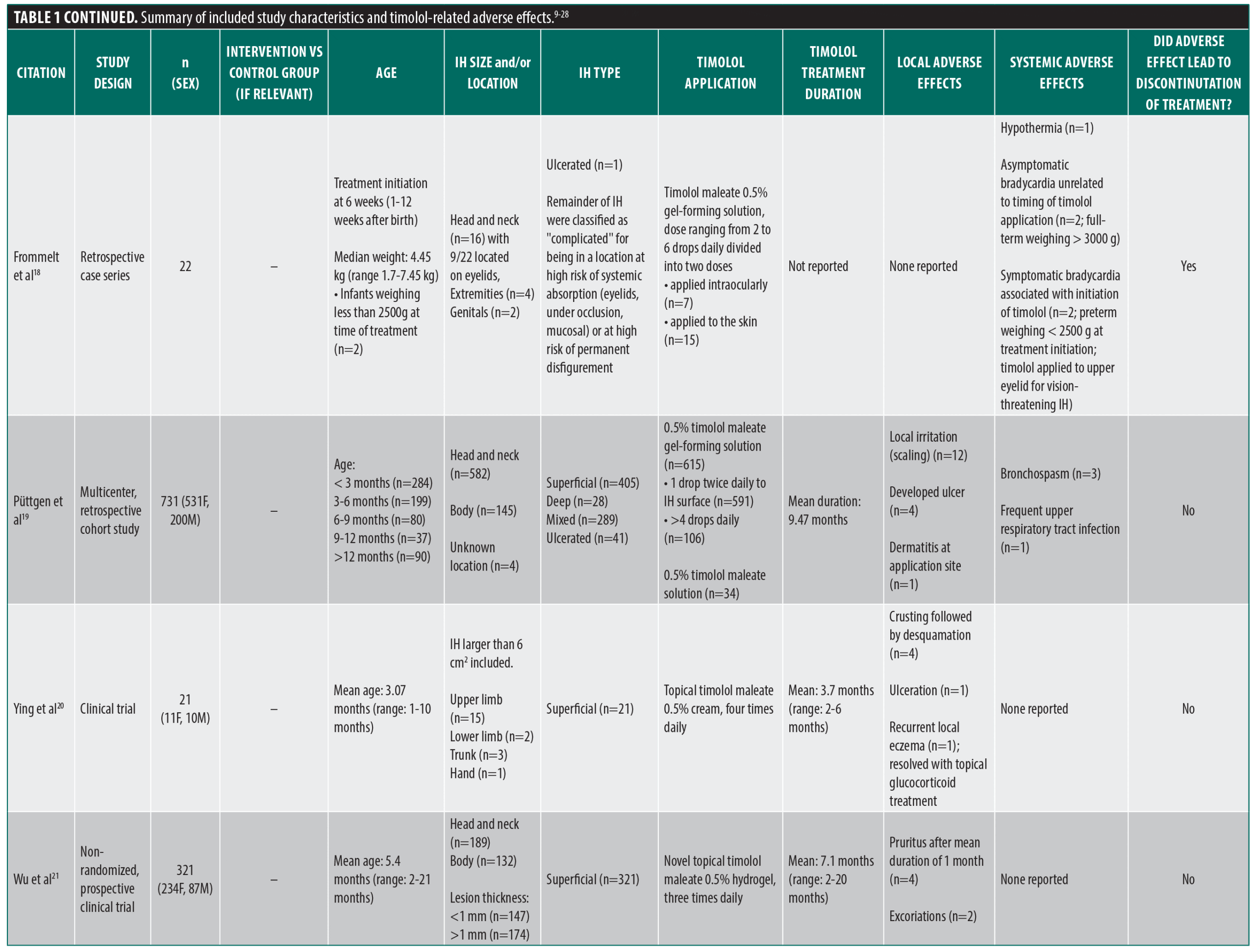
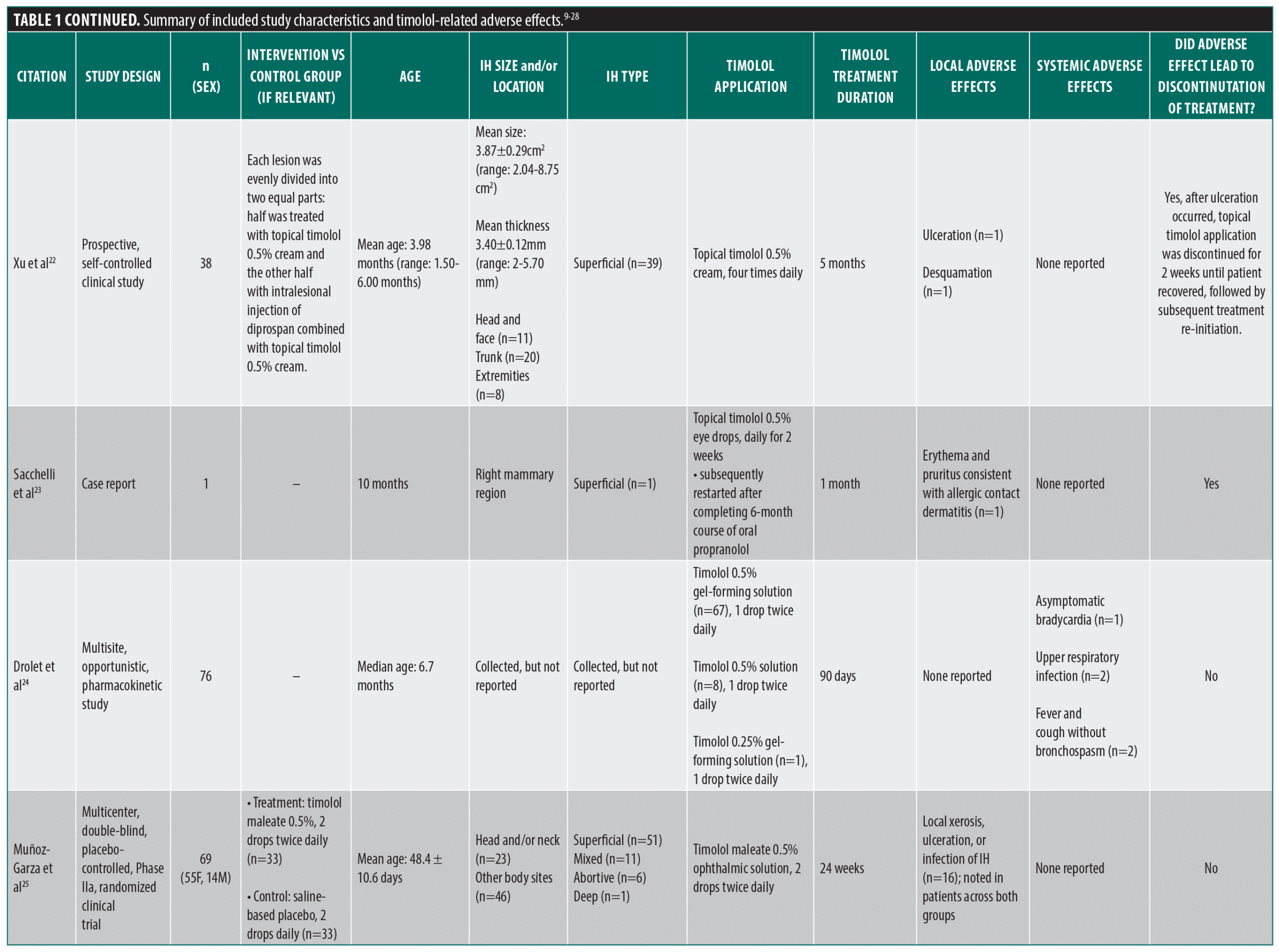
From the PubMed/MEDLINE database search, a total of 143 unique articles were reviewed for eligibility (Figure 1). Seventeen articles met the inclusion criteria, including 2 case reports, 4 retrospective cohort studies, 3 retrospective chart reviews, 4 clinical trials, 3 prospective interventional studies, and 1 pharmacokinetic study. An additional three unique articles that met inclusion criteria were identified from citations in review articles, including 1 clinical trial and 2 prospective studies. The 20 articles comprised a total of 2036 patients, of which 1780 patients were specifically on timolol (Table 1).9-28 All of the studies that met the inclusion criteria used topical timolol for IHs and reported treatment-related complications or adverse effects. For each included study, data extracted included patient demographics, IH characteristics (size, location, type), characteristics of timolol administration (dosage, frequency, application type), treatment duration, and local and/or systemic adverse effects related to treatment.
Of the reported 1909 total IHs, most were superficial (76.2%), followed by mixed (17.6%), ulcerated (4.3%), and deep (1.8%) IHs. Similarly, of the reported 1797 total IH locations, a majority were located in the head and neck region (67.2%). Most of the included studies used topical timolol ophthalmic solution unless specified otherwise. A variety of study designs were used, with some studies comparing timolol to other therapies and other assessing its use in combination treatment modalities. Only one randomized controlled trial compared topical timolol 0.5% twice daily with a saline-based placebo.25 One clinical trial used topical timolol in conjunction with pulsed dye laser (PDL),11 and another randomized prospective study used topical timolol in conjunction with low-dose strontium 90–yttrium 90 (90Sr-90Y) radiation therapy.12 Two clinical trials compared the efficacy of timolol ophthalmic solution to a different therapy: combined PDL and neodymium-doped yttrium aluminum garnet (Nd:YAG) laser therapy14 or oral propranolol.26 A retrospective cohort study investigated the efficacy of introducing timolol solution through various procedural methods, including a nanomicroneedle technique or dipped with a medical swab.27 In two studies, an IH lesion was divided evenly into two halves, whereby timolol was applied to one half.13,22 In one study, imiquimod cream 5% was applied to the other half.13 In the other study, intralesional injection of diprospan was administered in combination with topical timolol to the other half.22 Despite the potential for cross-contamination, some patients exhibited adverse effects on both treatment sides, whereas others had reactions only on the timolol-treated side, suggesting that the response was not influenced by imiquimod and/or diprospan. One study reported the use of a novel hydrogel formulation of topical timolol 0.5%.21 A majority of the included studies used topical timolol 0.5%. However, the 0.1% preparation of topical timolol was used in one study with a total of 11 patients10 while topical timolol 0.25% was used in one patient in a different study.24 One study compared the efficacy of varying formulations of topical timolol, including a gel-forming and a non-gel-forming solution.24 Some studies assessed the efficacy of timolol at varying doses, indicating varying frequencies of drops applied daily.16,18,19 The remainder of the studies included case reports or studies that employed the same concentration, dose, and formulation of topical timolol for IH for all included patients.9,15,17,20,23,28
Discussion
The primary goal of IH treatment is to prevent severe complications and minimize the risk of permanent functional or cosmetic disfigurement.29 β-blockers, specifically oral propranolol, have been widely recognized as the first-line therapy for IHs. Many have theorized its role in proliferation and growth arrest through proposed mechanisms, including apoptosis, vasoconstriction, downregulation of vascular endothelial growth factor signaling, and reduced activity of the renin-angiotensin system.30,31 While effective, it is associated with many severe adverse effects, the most notable include bronchospasm or wheezing, bradycardia, hypotension, and hypoglycemia.32,33 Although there are relatively few of these adverse effects reported for the large number of children treated, this prompted growing interest in topical β-blockers like timolol.
Increasingly reported as a therapeutic since 2010, topical timolol has been investigated as an effective and safe alternative to oral propranolol. Timolol is a nonselective β-antagonist whose mechanism of action is similar to that of systemic β-blockers.34 However, concerns persist regarding potential systemic absorption and its associated risks, especially in particular types of IH. Our systematic review explored the reported local and systemic adverse effects of topical timolol, and which patients can be at higher risk of these effects, in hopes of allowing physicians to decide which patients are appropriate candidates for this therapy.
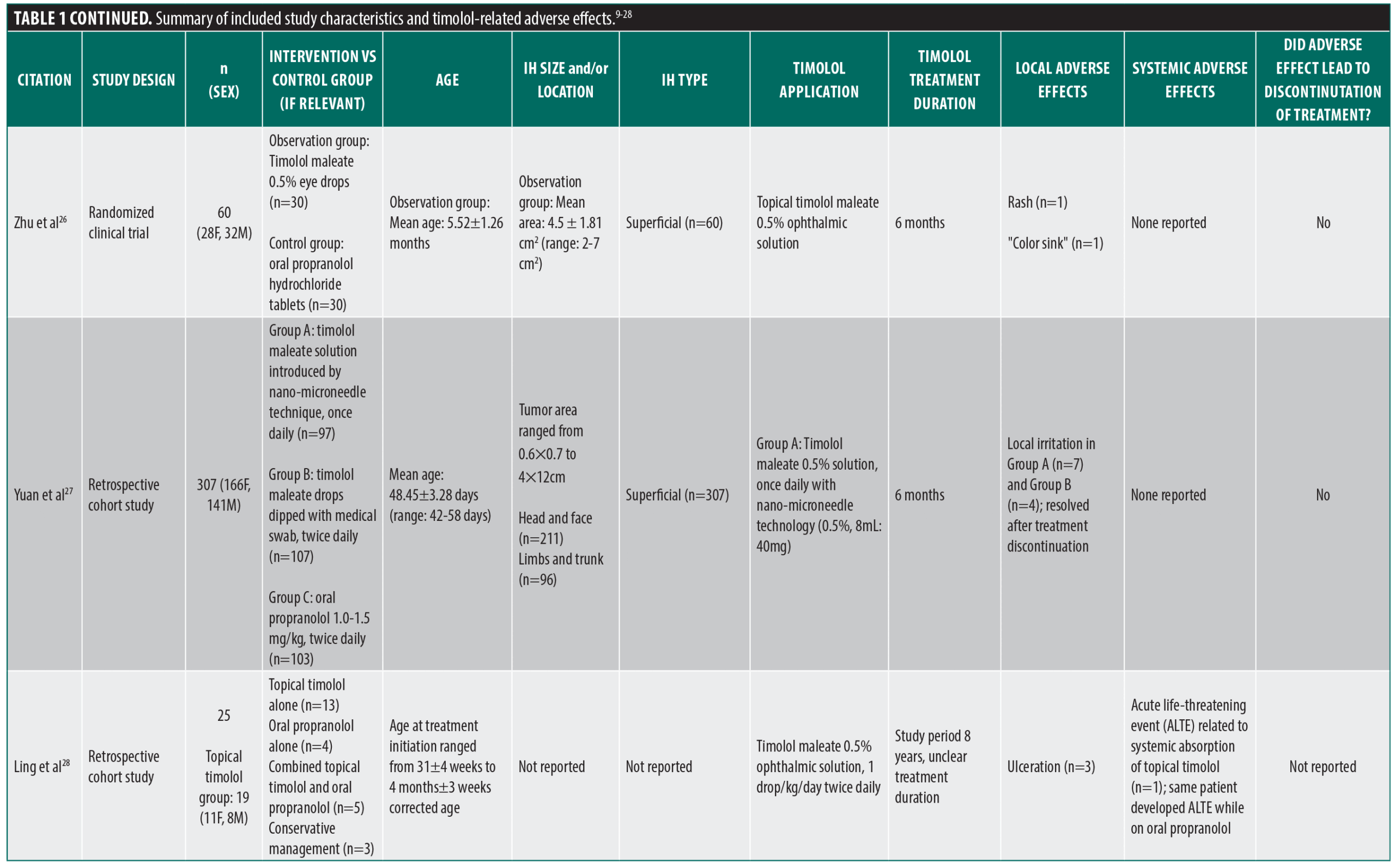
Local adverse effects. In our review, both local and systemic adverse effects were observed in patients treated with topical timolol. Among the 1780 patients on topical timolol included in the studies that met our inclusion criteria, local adverse effects were observed in 83 patients, accounting for 4.7% of cases. The most frequently reported local adverse effect was irritation at the application site, which commonly manifested as scaling, ulceration, and pruritus, occasionally progressing to excoriations or erosions. Additionally, patients experienced changes in skin texture, including flaking, peeling, crusting, desquamation, and rash. Three cases of allergic contact dermatitis were also reported. Notably, allergic contact dermatitis has been previously documented with timolol eye drops used for antiglaucoma therapy.35,36
Despite these local reactions, none were severe enough to necessitate permanent discontinuation of treatment. For example, in four patients (each presenting with skin flaking, ulceration, or allergic contact dermatitis) in two of the prospective studies, topical timolol was discontinued for 3 days to two weeks until recovery was achieved, after which treatment was either resumed at the original dose or adjusted accordingly.12,22 A possible explanation for local irritation may be attributed to individual sensitivity or excessive application, leading to skin hypersensitivity. This is supported by a study by Ying et al20, in which crusting and desquamation resolved within one week after the patients’ guardians were instructed to reduce the dose of timolol. Similarly, Weibel et al15 highlighted that unintentional excessive application of topical timolol, particularly due to a lack of awareness of systemic absorption risks, may contribute to adverse effects, as evidenced by detectable timolol levels in the urine of children with ulcerated IHs. Painter and Hildebrand17 further supported this notion, reporting a higher likelihood of skin irritation with increasing doses of topical timolol. Although local adverse effects were generally mild and manageable with dose modifications or temporary discontinuation, the potential for systemic absorption highlights the need to further examine potential systemic complications. β-blockers, in general, are known to dry out the skin; thus, use of emollients after application can help reduce risks of desiccation and potentially ulceration.
Systemic adverse effects. Systemic adverse effects were observed in 22 out of 1780 patients, accounting for 1.2% of cases. These effects were diverse, with cardiovascular, respiratory, and neurological complications reported. In addition, other less common systemic adverse effects were also noted, warranting further investigation into their potential links to topical timolol treatment.
Cardiovascular adverse effects were reported in seven patients, including bradycardia (n=5), hypothermia (n=1), and cool extremities (n=1). In the study by Frommelt et al,18 two infants with symptomatic bradycardia weighed less than 2500g and were preterm (postmenstrual age: 34 weeks and 37 weeks) at the initiation of therapy. Additionally, timolol was applied for a vision-threatening IH on the upper eyelid, where thinner skin may have facilitated increased systemic absorption, potentially leading to bradycardia. Thus, the authors concluded that initiating topical timolol treatment in infants younger than 44 weeks postmenstrual age and weighing less than 2500g may increase the risk of bradycardia, hypotension, apnea, and hypothermia.18 These findings suggest that infants with lower birth weights, particularly those receiving topical timolol on areas with thinner skin, may be at higher risk of cardiovascular effects.
Several studies have assessed cardiovascular parameters and evaluated the adverse effects of timolol and have found no significant effects. In 2013, Chan et al37 conducted a randomized clinical trial to examine the safety and efficacy of timolol maleate 0.5% gel for treating superficial IHs in 41 infants with a median age of 9 weeks. No adverse events were reported, and there were no significant differences in mean heart rate, systolic blood pressure, or diastolic blood pressure between the treatment and placebo groups. Similarly, Muñoz-Garza et al25 conducted a randomized clinical trial to evaluate the efficacy and safety of timolol maleate 0.5% solution for the early treatment of IHs in 69 infants younger than 60 days. They measured systolic and diastolic blood pressure, as well as heart rates at 1 hour posttreatment, and found no significant differences between the timolol and placebo groups. These findings suggest that topical timolol does not appear to have a significant impact on cardiovascular parameters in infants. The consistency of results across multiple studies reinforces its favorable safety profile in term infants with superficial IH. However, continued research with larger sample sizes and longer follow-up periods may further clarify any potential risks associated with its use in this population.
Respiratory adverse effects were noted in five patients, including shortness of breath (n=1), wheezing (n=1), and bronchospasm (n=3). Among the three cases of bronchospasm, no specific patient or IH characteristics were identified as risk factors for timolol-associated adverse effects. The single case of wheezing occurred following accidental ocular administration of timolol rather than cutaneous application. This reaction was self-limiting and did not recur when the medication was applied exclusively to the skin.17 However, caution should be exercised in children with a predisposition to asthma.
A neurological adverse effect of sleep disturbance occurred in three infants, one of whom was also receiving concomitant propranolol therapy. Given that sleep disturbance is a known adverse effect of oral propranolol, its occurrence with topical timolol suggests potential for systemic absorption despite its topical administration. In a separate study, mild fatigue (n=1) was reported in one patient on the first day of treatment, while another patient experienced two episodes of prolonged, unclear gazing.15 These symptoms resolved without intervention. Although in this study systemic absorption was confirmed through positive urinalysis in 24 patients and detectable serum levels in three infants, Weibel et al15 concluded that a direct link between mild fatigue, prolonged gazing, and timolol treatment remained uncertain. Interestingly, 18% of the enrolled patients in this study had ulcerated IHs and 35% of patients had mixed and deep IHs.15 These findings highlight the possibility of systemic absorption of topical timolol, particularly in patients with ulcerated IHs or those with deeper IHs necessitating a higher dosage of timolol, which may increase permeability. While the reported effects were mild and self-resolving, further research is needed to clarify the extent of systemic absorption and its clinical significance, especially in vulnerable populations such as infants.
Aside from the above systems, two patients experienced fever and cough, while three patients developed upper respiratory tract infections.19,24 Importantly, with the exception of the two patients who experienced symptomatic bradycardia,18 none of the other systemic adverse effects reported were severe enough to necessitate discontinuation of treatment. While local adverse effects were reported more frequently than systemic ones, our review did not find evidence that the severity or frequency of local adverse effects influences a predisposition to systemic adverse effects. Additionally, no consistent pattern emerged to suggest that longer treatment durations are associated with a higher occurrence of systemic adverse effects. These findings highlight the overall tolerability of topical timolol, but also underscore the need for continued monitoring.
Populations at increased risk and clinical implications. Taking into account the range of local and systemic adverse effects, despite their relatively low frequency, it is important to note whether certain groups may be more vulnerable to complications from topical timolol. Some studies have advised caution when treating ulcerated or large IHs due to the possibility of high permeability through the lesion, and suggest monitoring temperature, blood pressure, and heart rate 2 to 4 hours after application in preterm or young infants.15,21
One theory for the increased risk of timolol complications in mucosal or ulcerated sites can be related to metabolism. Timolol maleate is metabolized by cytochrome P450 and CYP2D6, whereby poor metabolizers have higher peak plasma levels and longer elimination half-lives, leading to heightened sensitivity to a drug’s therapeutic effects.19 Another theory is related to physiological stress, whereby children may be particularly vulnerable to adverse effects from β-blockers when their body’s ability to respond to stress is compromised, such as during fasting or illness. As a result, it could be important to consider discontinuing topical and oral β-blockers in cases of severe illness, reduced oral intake, bronchospasm, or other effects.38 To mitigate concerns regarding potential percutaneous absorption and toxicity, several authors recommend administering limited amounts of the medication, such as one drop administered two to three times daily.19,29 Our review aligns with this perspective, as systemic adverse effects were observed in studies that included mixed, deep, and ulcerated IHs. Overall, topical timolol appears to be well tolerated, with case retrospective reviews reporting relatively few adverse effects in infants receiving 1 or 2 drops per day.
Another vulnerable group that has potential for increased risk of adverse effects are premature infants, particularly when considering cardiovascular or respiratory effects. One of the included studies in our review documented two episodes of symptomatic bradycardia associated with timolol initiation in preterm infants weighing less than 2500g.18 There were also respiratory adverse effects (bronchospasm, wheezing, shortness of breath) noted in 5 patients from our included studies.14,17,19 Prematurity is a known risk factor for IH development, and premature infants often develop chronic lung disease (eg, apnea of prematurity) consequently. Thus, topical timolol should be used cautiously in preterm infants, especially those with a history of apnea or chronic lung disease.39 Careful monitoring and dosing can help mitigate these risks, especially if topical timolol would be of greater therapeutic benefit than risk.
Limitations. While our review provides valuable insights into the adverse effects related to the use of topical timolol for treating IHs, several limitations must be considered when interpreting the findings. One limitation involves the heterogeneity of the included studies, whereby not all of the studies specified IH characteristics (e.g., type, size, or location of IH) related to the adverse effects reported. More importantly, another limitation is the limited number of studies evaluating adverse effects, as not all adverse effects may have been adequately reported or captured in the excluded studies. A significant number of the reviewed studies did not meet our inclusion criteria and were excluded due to authors explicitly stating that topical timolol treatment was well tolerated without any major complications. Thus, it is worth noting that our reported frequencies for timolol-related local (4.7%) and systemic (1.2%) adverse effects may overestimate the true incidence, as studies that did not report adverse effects were excluded from our analysis.
Conclusion
Topical timolol is generally well tolerated for the treatment of IHs, offering a favorable safety profile compared to systemic β-blockers. Our systematic review highlights that while adverse effects are relatively uncommon, both local and systemic complications can occur. Local reactions, such as irritation, ulceration, and allergic contact dermatitis, were more frequently reported, whereas systemic effects, including cardiac, respiratory, and neurologic, were rare but still clinically relevant. The risk of systemic absorption appears to be higher in certain populations, particularly preterm infants and those with ulcerated, deep, or large hemangiomas. This increases the potential risk for associated adverse effects.
Given these findings, it is essential to carefully screen patients who may be at increased risk before prescribing topical timolol for IH management. In addition to these patient-specific considerations, conservative dosing and close monitoring for potential complications are crucial to optimize safety and efficacy. In conclusion, further research is needed to better define the safety profile of topical timolol, particularly in these high-risk populations, to establish standardized guidelines for its use in clinical practice.
References
- Munden A, Butschek R, Tom WL, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170(4):907-913.
- Goelz R, Poets CF. Incidence and treatment of infantile haemangioma in preterm infants. Arch Dis Child Fetal Neonatal Ed. 2015;100(1):F85-F91.
- Drolet BA, Frieden IJ. Characteristics of infantile hemangiomas as clues to pathogenesis: does hypoxia connect the dots? Arch Dermatol. 2010;146(11):1295-1299.
- Couto RA, Maclellan RA, Zurakowski D, Greene AK. Infantile hemangioma: clinical assessment of the involuting phase and implications for management. Plast Reconstr Surg. 2012;130(3):619-624.
- Bauland CG, Lüning TH, Smit JM, Zeebregts CJ, Spauwen PHM. Untreated hemangiomas: growth pattern and residual lesions. Plast Reconstr Surg. 2011;127(4):1643-1648.
- Chiller KG, Passaro D, Frieden IJ. Hemangiomas of infancy: clinical characteristics, morphologic subtypes, and their relationship to race, ethnicity, and sex. Arch Dermatol. 2002;138(12):1567-1576.
- Léauté-Labrèze C, Harper JI, Hoeger PH. Infantile haemangioma. Lancet. 2017;390(10089):85-94.
- Langley A, Pope E. Propranolol and central nervous system function: potential implications for paediatric patients with infantile haemangiomas. Br J Dermatol. 2015;172(1):13-23.
- Khunger N, Pahwa M. Dramatic response to topical timolol lotion of a large hemifacial infantile haemangioma associated with PHACE syndrome. Br J Dermatol. 2011;164(4):886-888.
- Chakkittakandiyil A, Phillips R, Frieden IJ, et al. Timolol maleate 0.5% or 0.1% gel-forming solution for infantile hemangiomas: a retrospective, multicenter, cohort study. Pediatr Dermatol. 2012;29(1):28-31.
- Asilian A, Mokhtari F, Kamali AS, Abtahi-Naeini B, Nilforoushzadeh MA, Mostafaie S. Pulsed dye laser and topical timolol gel versus pulse dye laser in treatment of infantile hemangioma: A double-blind randomized controlled trial. Adv Biomed Res. 2015;4:257.
- Zhu HJ, Liu Q, Deng XL, Guan YX. Efficacy of low-dose 90Sr-90Y therapy combined with topical application of 0.5% timolol maleate solution for the treatment of superficial infantile hemangiomas. Exp Ther Med. 2015;10(3):1013-1018.
- Hu L, Huang HZ, Li X, Lin XX, Li W. Open-label nonrandomized left-right comparison of imiquimod 5% ointment and timolol maleate 0.5% eye drops in the treatment of proliferating superficial infantile hemangioma. Dermatology. 2015;230(2):150-155.
- Tawfik AA, Alsharnoubi J. Topical timolol solution versus laser in treatment of infantile hemangioma: a comparative study. Pediatr Dermatol. 2015;32(3):369-376.
- Weibel L, Barysch MJ, Scheer HS, et al. Topical timolol for infantile hemangiomas: evidence for efficacy and degree of systemic absorption. Pediatr Dermatol. 2016;33(2):184-190.
- Boos MD, Castelo-Soccio L. Experience with topical timolol maleate for the treatment of ulcerated infantile hemangiomas (IH). J Am Acad Dermatol. 2016;74(3):567-570.
- Painter SL, Hildebrand GD. Topical timolol maleate 0.5% solution for the management of deep periocular infantile hemangiomas. J AAPOS. 2016;20(2):172-174.e1.
- Frommelt P, Juern A, Siegel D, et al. Adverse Events in Young and Preterm Infants Receiving Topical Timolol for Infantile Hemangioma. Pediatr Dermatol. 2016;33(4):405-414.
- Püttgen K, Lucky A, Adams D, et al. Topical Timolol Maleate Treatment of Infantile Hemangiomas. Pediatrics. 2016;138(3):e20160355.
- Ying H, Zou Y, Yu W, et al. Prospective, open-label, rater-blinded and self-controlled pilot study of the treatment of proliferating superficial infantile hemangiomas with 0.5% topical timolol cream versus 595-nm pulsed dye laser. J Dermatol. 2017;44(6):660-665.
- Wu HW, Liu C, Wang X, et al. Topical Application of 0.5% Timolol Maleate Hydrogel for the Treatment of Superficial Infantile Hemangioma. Front Oncol. 2017;7:137.
- Xu P, Yu Q, Huang H, Zhang W, Li W. A self-controlled study of intralesional injection of diprospan combined with topical timolol cream for treatment of thick superficial infantile hemangiomas. Dermatol Ther. 2018;31(3):e12595.
- Sacchelli L, Vincenzi C, La Placa M, Piraccini BM, Neri I. Allergic contact dermatitis caused by timolol eyedrop application for infantile haemangioma. Contact Dermatitis. 2019;80(4):255-256.
- Drolet BA, Boakye-Agyeman F, Harper B, et al. Systemic timolol exposure following topical application to infantile hemangiomas. J Am Acad Dermatol. 2020;82(3):733-736.
- Muñoz-Garza FZ, Ríos M, Roé-Crespo E, et al. Efficacy and safety of topical timolol for the treatment of infantile hemangioma in the early proliferative stage: a randomized clinical trial. JAMA Dermatol. 2021;157(5):583-587.
- Zhu H, Luo H, Lai W. Efficacy of timolol in the treatment of facial hemangioma and its effect on the proliferation and apoptosis of hemangioma stem cells. Cell Mol Biol (Noisy-le-grand). 2023;69(10):115-120.
- Yuan L, Wang J. Efficacy and safety of introduction timolol maleate by manometer microneedles among infantile hemangioma: a retrospective study in China. Eur J Pediatr Surg. 2024;34(3):261-266.
- Ling LCT, Aan MKJ. Beta-blockers for the treatment of infantile haemangiomas in premature infants. Australas J Dermatol. 2024;65(7):e194-e197.
- Krowchuk DP, Frieden IJ, Mancini AJ, et al. Clinical practice guideline for the management of infantile hemangiomas. Pediatrics. 2019;143(1):e20183475.
- Leaute-Labreze C, Taieb A. Efficacy of beta-blockers in infantile capillary haemangiomas: the physiopathological significance and therapeutic consequences. Ann Dermatol Venereol. 2008;135: 860–2.
- Itinteang T, Brasch HD, Tan ST, Day DJ. Expression of components of the renin-angiotensin system in proliferating infantile haemangioma may account for the propranolol-induced accelerated involution. J Plast Reconstr Aesthet Surg. 2011;64(6):759-765.
- Breur JM, de Graaf M, Breugem CC, Pasmans SG. Hypoglycemia as a result of propranolol during treatment of infantile hemangioma: a case report. Pediatr Dermatol. 2011;28(2):169-171.
- Bayliss SJ, Berk DR, Van Hare GF, et al. Propranolol treatment for hemangioma of infancy: risks and recommendations. Pediatr Dermatol. 2010;27(3):319-320.
- Painter SL, Hildebrand GD. Review of topical beta blockers as treatment for infantile hemangiomas. Surv Ophthalmol. 2016;61(1):51-58.
- Otero-Rivas MM, Ruiz-González I, Valladares-Narganes LM, Delgado-Vicente S, Rodríguez-Prieto MÁ. A case of contact dermatitis caused by timolol in anti-glaucoma eyedrops. Contact Dermatitis. 2015;73(4):256-257.
- Chernoff KA, Zippin JH. Allergic contact dermatitis to timolol: a report of 2 cases and review of the literature. Dermatitis. 2014;25(1):41-42.
- Chan H, McKay C, Adams S, Wargon O. RCT of timolol maleate gel for superficial infantile hemangiomas in 5- to 24-week-olds. Pediatrics. 2013;131(6):e1739-e1747.
- Léauté-Labrèze C, Hoeger P, Mazereeuw-Hautier J, et al. A randomized, controlled trial of oral propranolol in infantile hemangioma. N Engl J Med. 2015;372(8):735-746.
- McMahon P, Oza V, Frieden IJ. Topical timolol for infantile hemangiomas: putting a note of caution in “cautiously optimistic”. Pediatr Dermatol. 2012;29(1):127-130.