J Clin Aesthet Dermatol. 2026;19(1):7–10.
by Diego Ruiz Dasilva, MD; Graham H. Litchman, DO, MS; Alondra Soto-González, MS; Naiem Issa, MD, PhD; E. James Song, MD; Christopher G. Bunick, MD, PhD; and James Q. Del Rosso, DO
Dr. Dasilva is with Forefront Dermatology in Virginia Beach, Virginia and Eastern Virginia Medical School in Norfolk, Virginia. Dr. Litchman is with Vivida Dermatology in Las Vegas, Nevada and Touro University, Nevada College of Osteopathic Medicine in Henderson, Nevada. Ms. Soto-González is with Universidad Central del Caribe School of Medicine in Bayamón, Puerto Rico and the Department of Dermatology at the Perelman School of Medicine at the University of Pennsylvania in Philadelphia, Pennsylvania. Dr. Issa is with Forefront Dermatology in Virginia Beach, Virginia; the Dr. Phillip Frost Department of Dermatology and Cutaneous Surgery at the University of Miami School of Medicine in Miami, Florida; and the Department of Dermatology at George Washington University, School of Medicine and Health Sciences in Washington, District of Columbia. Dr. Song is with Frontier Dermatology in Mill Creek, Washington. Dr. Bunick is with the Department of Dermatology and Program in Translational Biomedicine at the Yale University School of Medicine in New Haven, Connecticut. Dr. Del Rosso is with JDR Dermatology Research in Las Vegas, Nevada and Touro University, Nevada College of Osteopathic Medicine in Henderson, Nevada.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Ruiz Dasilva: AbbVie, Arcutis, Dermavant, Galderma, Janssen, LEO Pharma, Lilly, Pfizer, Sanofi & Regeneron, UCB, Verrica. Dr. Issa: AbbVie, Bristol Myers Squibb, Castle Biosciences, Dermavant, DermTech, Galderma, Incyte, Jannsen, Journey, LEO Pharma, Lilly, Ortho Dermatologics, Pfizer, Regeneron, Sanofi, SUN Pharma, Verrica Pharmaceuticals. Dr. Bunick: AbbVie, Almirall, Alumis, Amgen, Apogee, Arcutis, Botanix, Connect BioPharma, Daiichi Sankyo, Dermavant, EPI Health/Novan, Galderma, Incyte, LEO Pharma, Lilly, Novartis, Ortho Dermatologics, Palvella, Pfizer, Regeneron, Sanofi, Sun Pharma, Takeda, Timber, Teladoc, Triveni, and UCB. Dr. Litchman: Arcutis, Castle Biosciences, Galderma, Pfizer, Sanofi & Regeneron, UCB. Dr. Song: AbbVie, Arcutis, Alphyn, Alumis, Amgen, Apogee, Bristol Myers Squibb, Boehringer Ingelheim, CorEvitas Registry, Dermavant, DermBiont, Galderma, Incyte, Janssen, LEO Pharma, Lilly, Moonlake, Novartis, Ortho Dermatologics, Pfizer, Sanofi & Regeneron, Sun Pharma, Target Registry, TIMBER, UCB, Takeda. Dr. Del Rosso: AbbVie, Almirall, Amgen, Anaptos Bio, Apogee, Arcutis, Bausch, Beiersdorf, Biofrontera, Botanix, Bristol Myers Squibb, Cage Bio, Cara, Cassiopea, Dermata, Dermavant (Organon), Ferndale, Galderma, Incyte, Johnson and Johnson, La Roche Posay, LEO Pharma, Lilly, L’Oréal, MC2, Moonlake, Nektar, Novan, Nutrafol, Pfizer, Ralexar, RBC Consultants, Regeneron, Sanofi, Sente, Sun Pharma, Takeda, UCB, Verrica, Vyne. Ms. Soto-González: no conflicts to disclose related to the contents of this article.
ABSTRACT: Objective: Oral Janus kinase inhibitors (JAKi) are effective in managing moderate-to-severe atopic dermatitis (AD), but concerns regarding venous thromboembolism (VTE) risk persist, particularly in female patients with overlapping risk factors such as oral contraceptive pill (OCP) use and nicotine exposure (primarily smoking). We evaluated VTE events in this population using data from the abrocitinib clinical trial program. Methods: We reviewed the Phase II and Phase III clinical trial data (JADE program) for abrocitinib in AD, focusing on VTE incidence in female patients with documented OCP use and nicotine exposure to contextualize thromboembolic risk. Results: VTE events were rare. Nonfatal VTE incidence was low, with dose-specific incidence rates less than 1.0 per 100 patient-years (PY). Among female patients taking OCPs and current or former smokers, no VTEs occurred (0 of 78). In the overall active study group, most VTEs arose in patients with multiple baseline risk factors (eg, obesity, immobilization, prior thrombosis). Discontinuation due to VTE-related adverse events was infrequent (n=7, 0.13/100 PY) in the overall study group. No VTE-related deaths were reported. Comparative rates were consistent with or lower than background risk in AD populations with similar demographics. Limitations: This review is limited by the lack of publicly available patient-level data, possible underreporting of lifestyle risk factors, inability to quantify total nicotine exposure, small sample size, and unmeasured confounding variables. Conclusion: VTE events in female patients with AD treated with abrocitinib, including those taking OCPs and with nicotine exposure, were rare and generally associated with multiple concurrent risk factors for VTE. No VTE events were noted in female patients treated with abrocitinib who were taking OCPs and with history of smoking/nicotine exposure. These findings may help contextualize VTE risk in real-world treatment decisions. Keywords: Atopic dermatitis, Janus kinase inhibitor, venous thromboembolism, oral contraceptives, nicotine, safety, abrocitinib
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin disease that most commonly presents with itch, eczematous lesions, and a relapsing and remitting course. Beyond the marked impact on quality of life, untreated moderate-to-severe AD is associated with increased rates of major adverse cardiovascular events and venous thromboembolism (VTE), likely due to the underlying state of chronic systemic inflammation in AD.1 Oral Janus kinase inhibitors (JAKi) have revolutionized the treatment of AD, given their rapid, targeted mechanism that modulates multiple key inflammatory cytokines (interleukin [IL] 4, IL-13, IL-31, thymic stromal lymphopoietin [TSLP], IL-22, interferon [IFN]-γ) in this heterogeneous disorder. The selective JAK1 inhibitors (abrocitinib, upadacitinib) are approved to treat moderate-to-severe AD in patients aged 12 years and older.2,3 However, in the United States, these medications are labeled with a boxed warning that reports “Thrombosis has occurred in patients treated with abrocitinib (or upadacitinib). Increased incidence of pulmonary embolism, venous and arterial thrombosis with another JAK inhibitor (JAKi) vs. tumor necrosis factor α (TNF-α) blockers.”4,5 This is extrapolated from the ORAL Surveillance study, which evaluated tofacitinib vs. TNF-α inhibitor risk profiles in high-risk patients with rheumatoid arthritis on concomitant methotrexate.6 Although VTE events in either drug in AD trials were rare and no signal was identified for VTE, this created significant hesitation for many prescribers. Herein, we focus on female patients taking oral contraceptive pills (OCPs) with a history of nicotine use (primarily smoking), given that both are independent and synergistic risk factors for VTE. This particular patient population was not excluded from the AD clinical trials; therefore, the outcomes can provide important and pragmatic information for clinicians.
Methods
Cohort data were analyzed from the clinical trial program for abrocitinib (Phase II and Phase III studies) in moderate-to-severe AD in women with nicotine exposure history (current or former smoking) and oral contraception use. The reported incidence rates of VTEs in this population of interest were compared with those without these risk factors.
Results
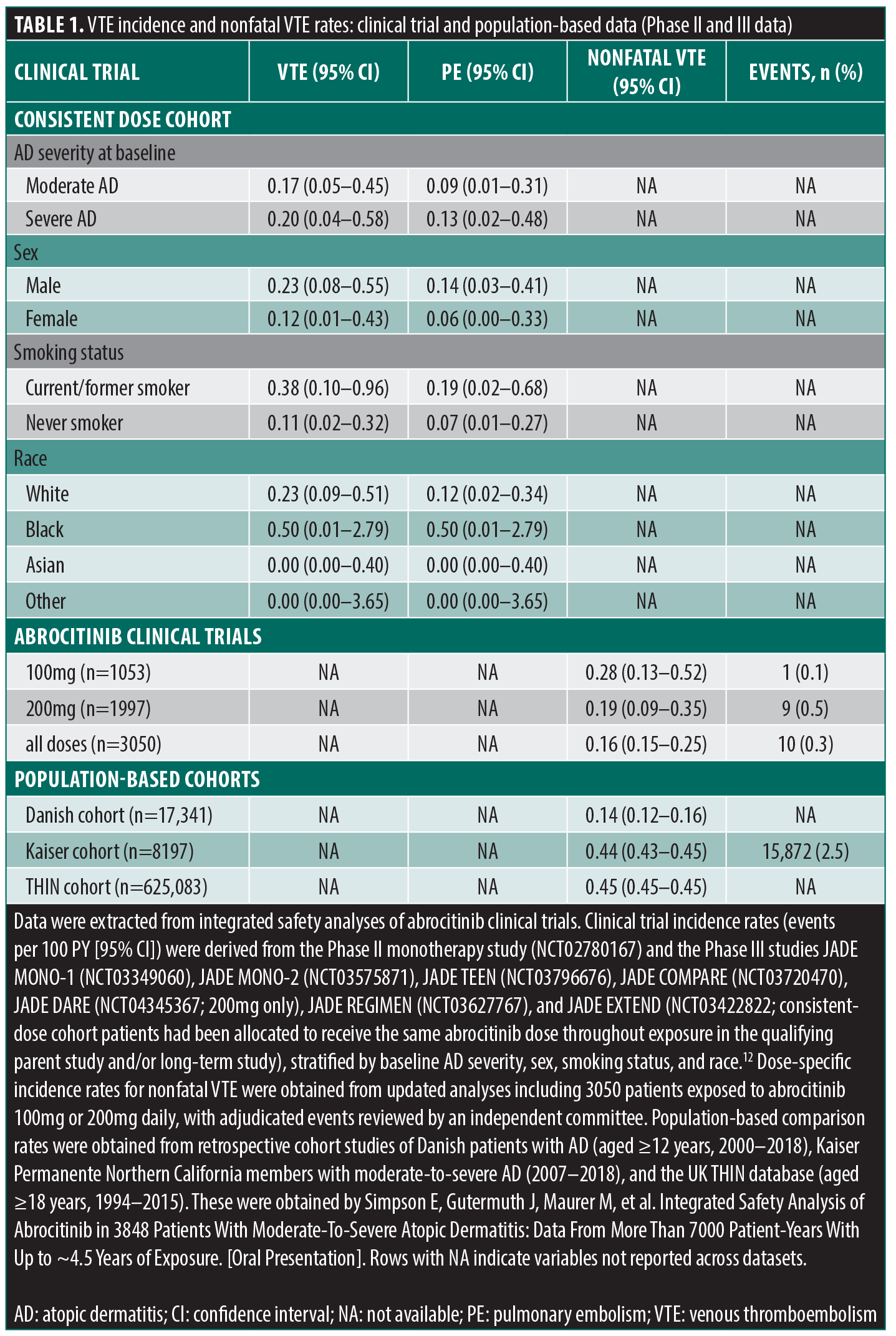
A total of 1384 female patients were in the abrocitinib JADE clinical program (including data from the Phase II monotherapy study and the Phase III studies: JADE MONO-1, JADE MONO-2, JADE TEEN, JADE COMPARE, JADE DARE, JADE REGIMEN, and JADE EXTEND). Of them, 405 (29.3%) were taking OCPs. Among OCP users, 78 (19.3%) were current or former smokers, and none developed VTE over 4.5 years (Table 1).7,8
Discussion
While clinical data support a low incidence rate of VTEs with long-term use of JAKi for the treatment of AD,1,3,6-9 there remains concern with the use of oral JAKi in female patients taking OCPs (particularly combined estrogen/progestin agents) who are also smokers due to perceived risk, given the class-wide boxed warning. However, the warning is extrapolated from a United States Food and Drug Administration (FDA)-mandated postmarketing safety study of oral tofacitinib, a pan-JAKi, in an enriched high-risk rheumatoid arthritis population treated in combination with other immunosuppressants vs TNF-α inhibitors, and it is not automatically generalizable to patients with AD (even more so female patients taking OCPs, considering patients were 50 years of age or older to be included in that study).6 The use of JAKi in this population is further complicated by a post hoc analysis of the same study, wherein it was demonstrated that only patients older than 65 years of age or who were current or former smokers had an appreciably higher risk with JAKi than TNF-α inhibitor use.10 Given the known increased risk of VTEs with OCPs and smoking, one may presume that the use of an oral JAKi in this population may further increase that risk. However, a prior meta-analysis including 6 abrocitinib Phase III dermatology clinical trials found no increased risk of adjudicated VTE with short-term oral or topical JAKi use.9
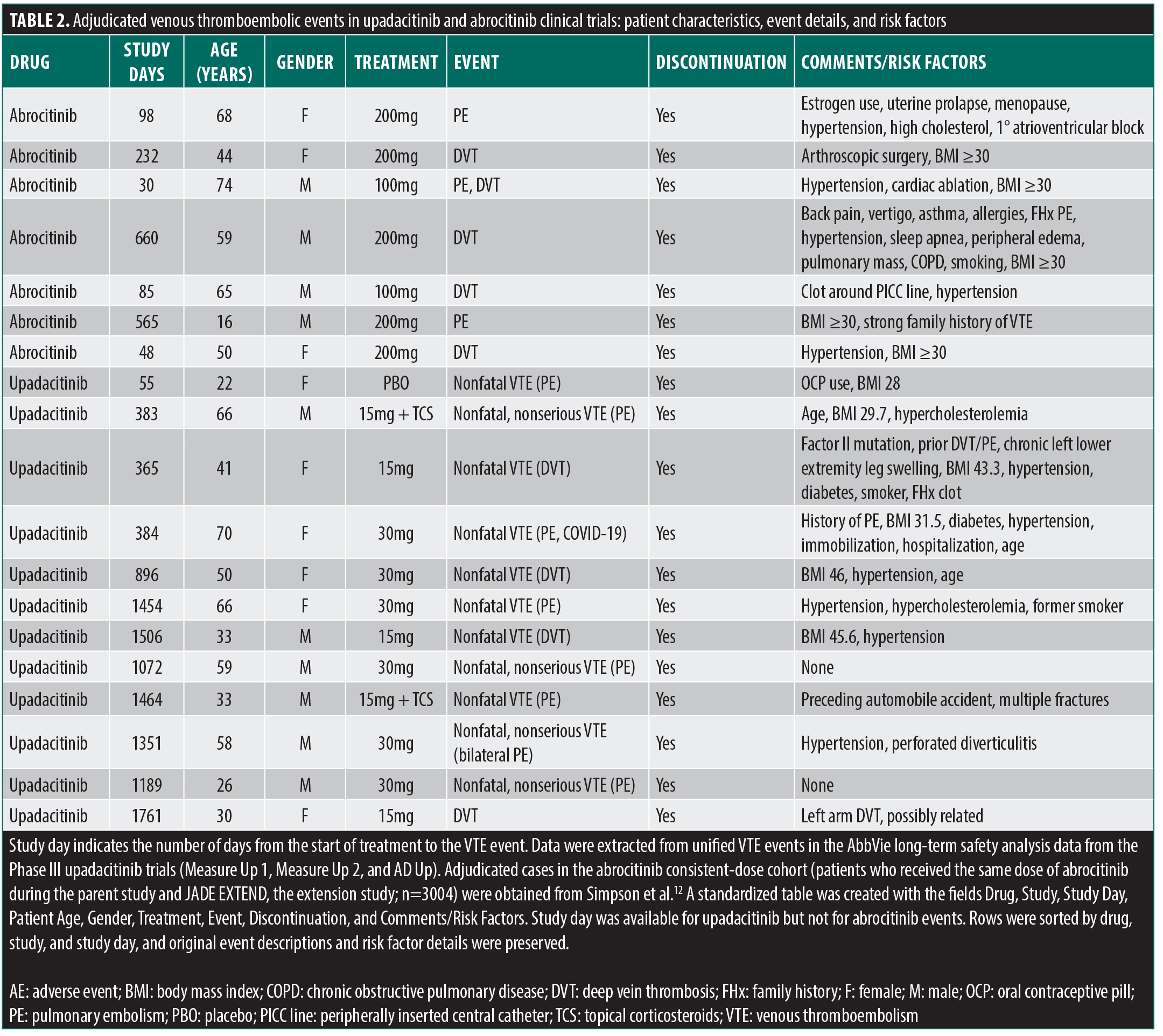
Nonetheless, there are limitations given the relatively small sample size that had both risk factors of OCPs and smoking history (n=78). Furthermore, the data were not granular enough to break down active smokers who are on a combined estrogen/progestin OCP, which would be the population at highest risk of VTE for this demographic. It is particularly noteworthy, however, that recent studies have shown JAKi-treated patients with AD have a lower rate of developing VTE compared to the background AD population and suggested that they may even be thromboprotective.1 JAKi mitigate proinflammatory cytokines, such as IL-6 and IFN-γ, as well as P-selectin and von Willebrand factor, resulting in decreased atherogenesis, coagulability, and leukocyte-endothelial adhesion.11 Among discontinuations in the AD studies for abrocitinib (including long-term extension), there were 7 cases of VTE in patients exposed up to 4 years (0.13 per 100PY).12 Though our focus is on abrocitinib, it is also worth mentioning that in the Phase III trials for upadacitinib, there were only 12 cases of VTE in patients exposed up to 6 years (≤0.2 per 100 PY), none of which were female patients taking OCPs with nicotine history.2 Risk factors in these patients included personal or family history of VTE, age over 65 years, hormone replacement therapy, recent surgery, history of smoking, and hypertension (Table 2). History of smoking and age over 65 years were the only risk factors that showed an increased hazard ratio, though both had overlapping confidence intervals.7
It is crucial that patients with AD are not undertreated and are provided with access to the most current and effective advanced systemic therapies, especially when certain reservations that limit prescribing are based on low-quality evidence and theoretical risk.13 Biologics and JAKi have markedly different mechanisms of action. AD biologics are human monoclonal antibodies that inhibit either IL-31, IL-4, and/or IL-13 function, while JAK1 inhibitors prevent the transduction of signals via the JAK/signal transducer and activator of transcription (STAT) pathway, which includes IL-31, IL-4, and IL-13, but also IL-22, IFN-γ, and TSLP, which are highly relevant cytokines for many subgroups with AD.14-16 There are many reasons a patient may prefer or require a JAKi over biologic for treatment of AD. This includes speed of onset of therapeutic response, depth of response, preference for oral medication, or failure or intolerance of a biologic. Moreover, strong rationale for oral JAKi use in AD is their performance in head-to-head clinical trials against dupilumab, where both upadacitinib and abrocitinib demonstrated superior skin and itch responses.17-19 Regarding traditional immunosuppressant agents, a comparison study noted equal or higher incidence of malignancy, VTE, and major adverse cardiac events per 100 PY among patients prescribed methotrexate, cyclosporine, or prednisone as compared to oral JAKi.20
We acknowledge significant limitations of these types of reviews, including paucity of publicly available patient-level data, potential underreporting of lifestyle risk factors, study design, and the inability to adjust for confounding variables. Nonetheless, our findings provide clinicians with additional data points to help in counseling on the usage of oral JAKi in women of childbearing age, especially because their contraceptive methods and/or nicotine habits may change over time.
While these data are encouraging, we emphasize the importance of recommending modification of all potentially adjustable risk factors for any patient considering an oral JAKi. Ultimately, the treatment of AD is becoming highly personalized, and shared decision-making is strongly recommended. This dialogue is particularly important, with recent data showing that JAKi are more likely to achieve optimal treat-to-target goals and minimal disease activity, and we hope to encourage future trials to include a larger number of patients with two or more cardiometabolic risk factors to enhance the generalizability of safety results.11
References
- Alani O, Wang D, Wahood S, et al. Is upadacitinib cardioprotective in chronic inflammatory diseases? A review of major adverse cardiovascular events and venous thromboembolism in atopic dermatitis. J Drugs Dermatol. 2025;24(5):530-533.
- Bunick CG, Silverberg JS, Chovatiya R, et al. Long-term upadacitinib safety in moderate‑to‑severe atopic dermatitis up to 6 years: an integrated analysis with over 9000 patient‑years of exposure. Poster presented at: 2024 Revolutionizing Atopic Dermatitis (RAD) Virtual Conference; December 8, 2024.
- Stein Gold L, Shahriari M, Guttman-Yassky E, et al. Patients with atopic dermatitis using oral contraceptive pills or hormone replacement therapy: long-term safety of upadacitinib for major adverse cardiovascular events, venous thromboembolism, or malignancy (excluding nonmelanoma skin cancer). Poster presented at: 2025 Revolutionizing Atopic Dermatitis Conference; June 6-7, 2025; Nashville, TN.
- Abrocitinib. Prescribing information. Pfizer; 2025.
- Upadacitinib. Prescribing information.AbbVie; 2025.
- Ytterberg SR, Bhatt DL, Mikuls TR, et al; ORAL Surveillance Investigators. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386(4):316-326.
- Thaçi D, de Bruin-Weller M, Cork M, et al. Baseline use of oral contraceptives or hormone replacement therapy in patients with moderate-to-severe atopic dermatitis treated with abrocitinib in the phase 2 and phase 3 JADE clinical trial program and reported venous thromboembolic outcomes. Poster presented at: European Academy of Dermatology and Venereology (EADV) Congress; October 11-14, 2023; Berlin, Germany. Abstract #2796.
- Simpson E, Gutermuth J, Maurer M, et al. Integrated safety analysis of abrocitinib in 3848 patients with moderate-to-severe atopic dermatitis: data from more than 7000 patient-years with up to ~4.5 years of exposure. Oral abstract presented at: European Academy of Allergy and Clinical Immunology Congress 2024; May 31- June 3, 2024; Valencia, Spain. Oral abstract #15.
- Ingrassia JP, Maqsood MH, Gelfand JM, et al. Cardiovascular and venous thromboembolic risk with JAK inhibitors in immune-mediated inflammatory skin diseases: a systematic review and meta-analysis. JAMA Dermatol. 2024;160(1):28-36.
- Kristensen LE, Danese S, Yndestad A, et al. Identification of two tofacitinib subpopulations with different relative risk versus TNF inhibitors: an analysis of the open label, randomised controlled study ORAL Surveillance. Ann Rheum Dis. 2023;82(7):901-910.
- Beckman JD, DaSilva A, Aronovich E, et al. JAK-STAT inhibition reduces endothelial prothrombotic activation and leukocyte-endothelial proadhesive interactions. J Thromb Haemost. 2023;21(5):1366-1380.
- Simpson EL, Silverberg JI, Nosbaum A, et al. Integrated safety update of abrocitinib in 3802 patients with moderate-to-severe atopic dermatitis: data from more than 5200 patient-years with up to 4 years of exposure. Am J Clin Dermatol. 2024;25(4):639-654.
- Silverberg JI, Gooderham M, Katoh N, et al. Combining treat-to-target principles and shared decision-making: international expert consensus-based recommendations with a novel concept for minimal disease activity criteria in atopic dermatitis. J Eur Acad Dermatol Venereol. 2024;38(11):2139-2148.
- Kamata M, Tada Y. Optimal use of Jak inhibitors and biologics for atopic dermatitis on the basis of the current evidence. JID Innov. 2023;3(3):100195.
- Del Duca E, He H, Liu Y, et al. Intrapatient comparison of atopic dermatitis skin transcriptome shows differences between tape-strips and biopsies. Allergy. 2024;79(1):80-92.
- Guttman-Yassky E, Irvine AD, Brunner PM, et al. The role of Janus kinase signaling in the pathology of atopic dermatitis. J Allergy Clin Immunol. 2023;152(6):1394-1404.
- Silverberg JI, Bunick CG, Hong HC, et al. Efficacy and safety of upadacitinib versus dupilumab in adults and adolescents with moderate-to-severe atopic dermatitis: week 16 results of an open-label randomized efficacy assessor-blinded head-to-head phase IIIb/IV study (Level Up). Br J Dermatol. 2024;192(1):36-45.
- Blauvelt A, Teixeira HD, Simpson EL, et al. Efficacy and safety of upadacitinib vs dupilumab in adults with moderate-to-severe atopic dermatitis: a randomized clinical trial. JAMA Dermatol. 2021;157(9):1047-1055.
- Silverberg JI, Simpson EL, Pink AE, et al. Switching from dupilumab to abrocitinib in patients with moderate-to-severe atopic dermatitis: a post hoc analysis of efficacy after treatment with dupilumab in JADE DARE. Dermatol Ther (Heidelb). 2025;15(2):367-380.
- Daniele S, Bunick C. JAK inhibitor safety compared to traditional systemic immunosuppressive therapies. J Drugs Dermatol. 2022;21(12):1298-1303.