J Clin Aesthet Dermatol. 2026;19(3):20–30.
by Joshua Burshtein, MD; Sara Nahon, MS; Diego Ruiz Dasilva, MD; and Graham Litchman, DO, MS
Dr. Burshtein is with the Department of Dermatology at the University of Illinois-Chicago, Chicago, Illinois. Ms. Nahon is with the University of Illinois, College of Medicine at Chicago, Chicago, Illinois. Dr. Dasilva is with Forefront Dermatology and Eastern Virginia Medical School, Virginia Beach, Virginia. Dr. Litchman is with the Department of Dermatology at Touro University Nevada, and Vivida Dermatology, Las Vegas, Nevada.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Burshtein and Ms. Nahon declare no conflicts of interest relevant to the content of this article. Dr. Dasilva is a consultant and/or speaker for AbbVie, Arcutis, Dermavant, Galderma, Janssen, LEO Pharma, Lilly, Pfizer, Sanofi & Regeneron, UCB, and Verrica. Dr. Litchman has served as a research investigator, clinical advisor, or speaker for AbbVie, Arcutis, Bristol Meyers Squibb, Castle Biosciences, Dermavant, Galderma, Novartis, Pfizer, Sanofi/Regeneron, Taketa, and UCB.
ABSTRACT: Background: Atopic dermatitis (AD) is a common chronic inflammatory disease. Despite its global prevalence, the current standard of care has remained unchanged for many years. Historically, first-line agents include topical corticosteroids (TCS) and topical calcineurin inhibitors (TCIs). However, these agents have significant limitations, including local and systemic adverse effects. In the last several years, novel topical therapeutic agents have been approved by the United States Food and Drug Administration (FDA) and more are being developed. Objective: The present review aims to summarize these topical therapeutic advances and report their efficacy and safety relative to the existing armamentarium. Methods: Three searches were conducted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses and the Agency for Healthcare Research and Quality Methods Guide for Effectiveness and Comparative Effectiveness Reviews guidelines. These included electronic searches of FDALabel (without date restriction) and PubMed and ClinicalTrials.gov (between April 8, 2020 and April 8, 2025). Results: A total of 52 nongeneric, prescription topical therapeutic agents are currently approved by the FDA for AD, the majority of which are TCS (n=11; 52.4%) and TCIs (n=4; 19.0%). There have been several agents with novel mechanisms of action (n=3; 7.6%) approved by the FDA in recent years, including Janus kinase (JAK) inhibitors, nonsteroidal aryl hydrocarbon receptor (AhR) modulators, and phosphodiesterase 4 (PDE4) inhibitors. Conclusion: In the last 5 years, important innovations in the therapeutic landscape of AD have emerged. Treatments include novel JAK inhibitors, AhR modulators, and PDE4 inhibitors. Adopting these therapies as part of clinical care can improve patients’ therapeutic outcomes and quality of life. Keywords: Atopic dermatitis, topical treatments, corticosteroids, nonsteroid, adverse effects, novel therapies
Introduction
Atopic dermatitis (AD) is one of the most prevalent chronic inflammatory skin conditions worldwide, with millions of adults and children affected globally.1 Historically, first-line therapy includes moisturizers and emollients to maintain skin hydration and barrier function, as well as topical corticosteroids (TCS).2 In AD, low- to mid-level potency TCS are recommended as initial therapy, which can be raised to a higher potency in moderate-to-severe cases.3 However, TCS are associated with many known cutaneous and systemic adverse effects (AE). Other historical topical therapies that do not contain corticosteroids include tacrolimus and pimecrolimus, topical calcineurin inhibitors (TCI), and crisaborole, a phosphodiesterase 4 (PDE4) inhibitor. These treatments have more favorable safety profiles and can be effective for anatomically sensitive areas (eg, face, eyelids).4 Recent advancements in the therapeutic landscape have markedly transformed the management of AD, offering more targeted, effective, and personalized options than were previously available.
Limitations of the Current Standard of Care
TCS are associated with numerous AEs, including skin atrophy, tachyphylaxis, permanent striae on the groin, inner thigh, and underarms, telangiectasias,5 delayed wound healing,6 perioral dermatitis, steroid acne, induction or exacerbation of rosacea, hypertrichosis, contact dermatitis, hypopigmentation, potential topical steroid withdrawal, and increased susceptibility to infections.7 Systemic adverse effects can also arise due to suppression of the hypothalamic-pituitary-adrenal (HPA) axis, leading to hormonal dysregulation (caused by adrenal insufficiency), which can escalate to iatrogenic Cushing’s disease.7 Additionally, TCS can lead to hyperglycemia, particularly in individuals with pre-existing diabetes. Avascular necrosis has also been reported. Other rare systemic adverse effects include ocular defects (glaucoma, cataracts), electrolyte imbalances (edema, hypertension, hypocalcemia), and metabolic defects (osteopathy, decreased growth rate).8 A recent expert consensus panel determined that the use of long-term TCS is associated with significant AEs as well as notable medicolegal risks for clinicians prescribing these medications.8 While the adverse effects of TCI are milder than TCS, they also present with both local and systemic adverse effects. Local AEs include irritation (burning, itching, stinging),9 erythema,10 photosensitivity, and increased risk for skin infections and folliculitis.11 The main theoretical systemic side effect of long-term TCI is lymphoma and skin cancers, for which there is a boxed warning.12
In the last decade, new insights into the mechanism of disease for AD have ignited research into finding new disease-specific therapeutic options for patients. As a result, several novel topical therapies have been approved by the United States (US) Food and Drug Administration (FDA) or are undergoing clinical trials. These advanced targeted topical therapies are reported to be safe and effective for treating AD.8 Currently, limited literature critically analyzes these novel topical therapeutics relative to the existing armamentarium of topical therapies. The present review seeks to provide an overview of novel and upcoming topical therapies in
the pipeline.
Methods
FDALabel. A query was launched using the FDALabel website with the following criteria:
-
- Labeling Types: “HumanRX” and “Human OTC”
- Labeling Section: “Simple search” for “atopic dermatitis” within “1 indications and usage”
- Routes of Administration: “Topical”
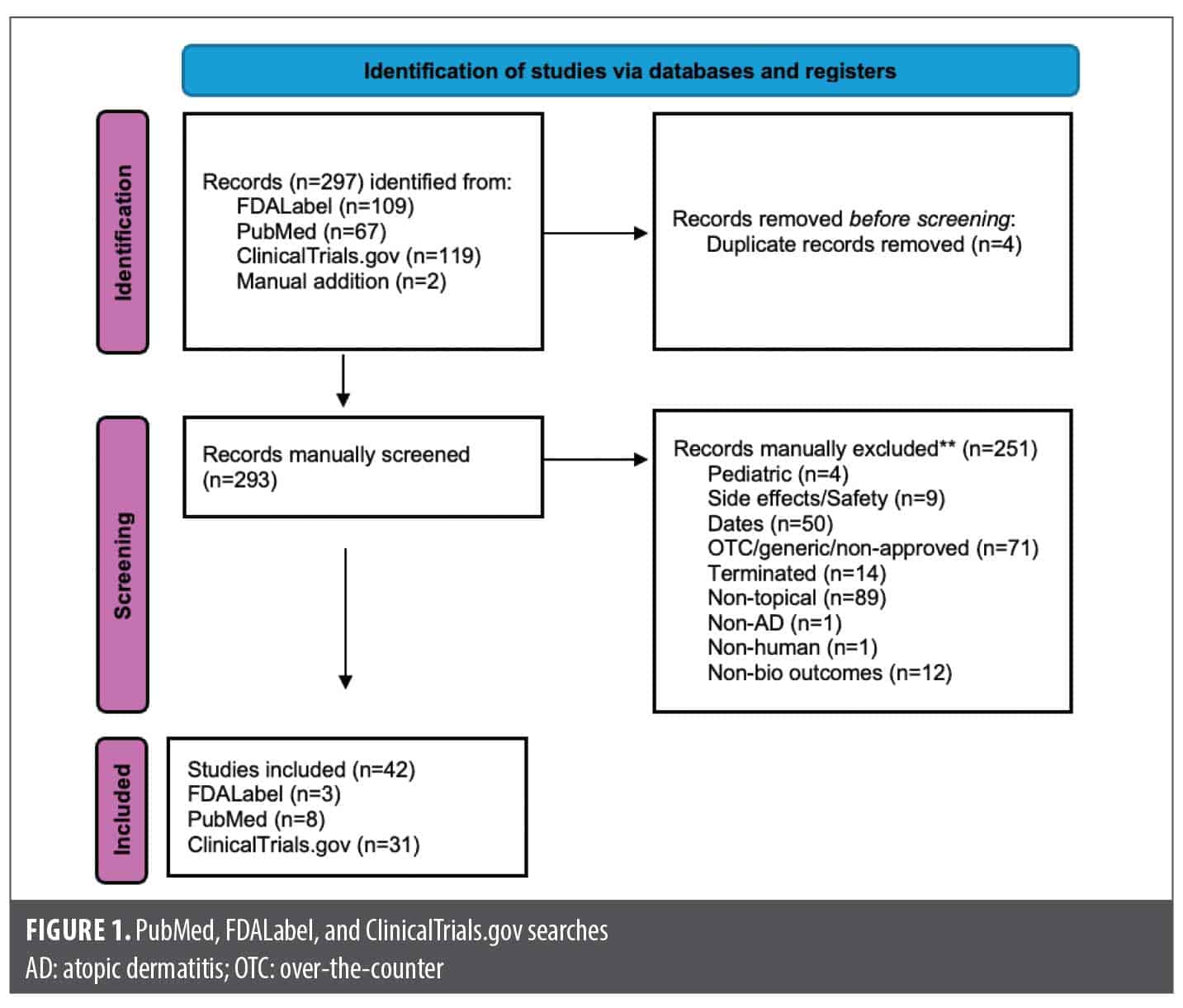
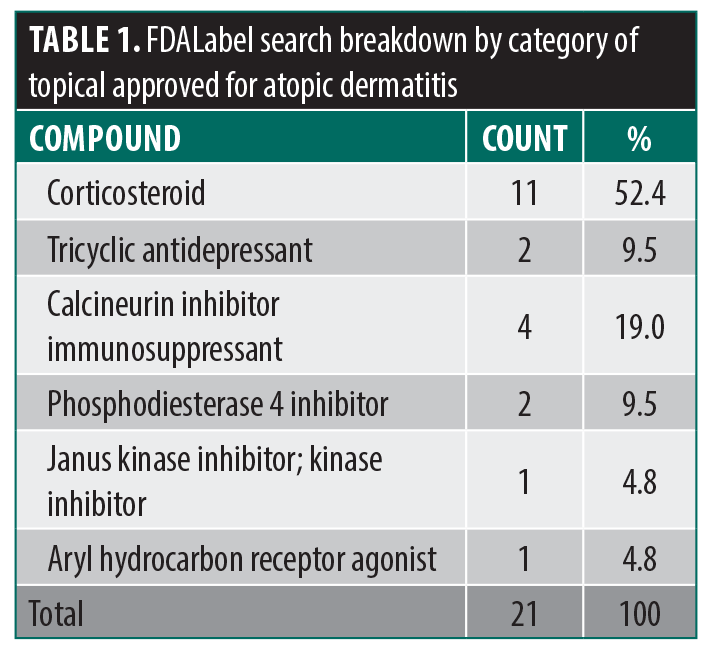
This search generated 109 results. Results were filtered to include marketing categories New Drug Application (NDA) only (ie, excluding Abbreviated NDA, NDA Authorized Generic, OTC Monograph Final, and Unapproved drug other). This left 21 results, for which AD approval dates were manually confirmed using a public search engine. Missing drug classes were manually added.
PubMed. A literature review was conducted on PubMed. The review was based on the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines13 and Agency for Healthcare Research and Quality (AHRQ) Methods Guide for Effectiveness and Comparative Effectiveness Reviews guidelines.14 An electronic search of PubMed between April 8, 2020 and April 8, 2025 using the terms (“Atopic Dermatitis” AND “Topical”) AND (“systematic review” OR “meta-analysis”) was conducted.
Inclusion criteria. Studies published in English, between April 8, 2020 and April 8, 2025, and available in full text were included.
Exclusion criteria. Preprints and studies examining nontopical delivery mechanisms, probiotics, non-FDA–approved or over-the-counter (OTC) treatments/home remedies, indications other than AD, nonapproved drugs in the last 5 years, pediatric patients, or nonhuman participants were excluded.
ClinicalTrials.gov. A search on ClinicalTrials.gov was conducted to understand the volume and classes of compounds in the pipeline for future novel therapeutic applications. A search was launched using the following criteria: Atopic Dermatitis | Completed studies | Adult (18 – 64) | Interventional studies | Studies with results | Study completion from April 8, 2020 to April 8, 2025.
Inclusion criteria. Studies published in English, between April 8, 2020 and April 8, 2025, and available in full text were included.
Exclusion criteria. Preprints and studies examining nontopical delivery mechanisms, terminated trials, non-FDA–approved or OTC treatments/home remedies, indications other than AD, or pediatric participants were excluded.
Results
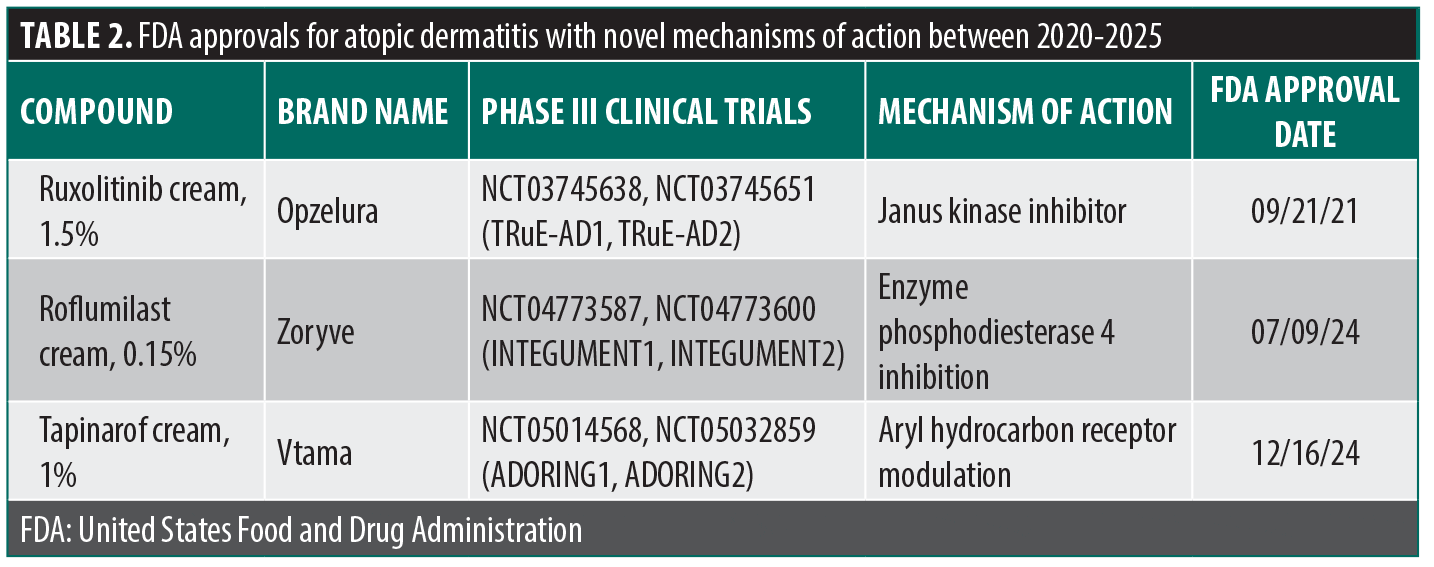
FDALabel. TCS represented the largest share of the available 21 NDA approvals (n=11; 52.4%), followed by TCIs (n=4; 19.0%). Several agents with novel mechanisms of action were identified, including Janus kinase (JAK) inhibitors (4.8%), nonsteroidal aryl hydrocarbon receptor (AhR) modulators (4.8%), and PDE4 inhibitors (9.5%) (Figure 1; Table 1; Table 2). Three agents (7.6%) were approved by the FDA in the last 5 years (Table 2).
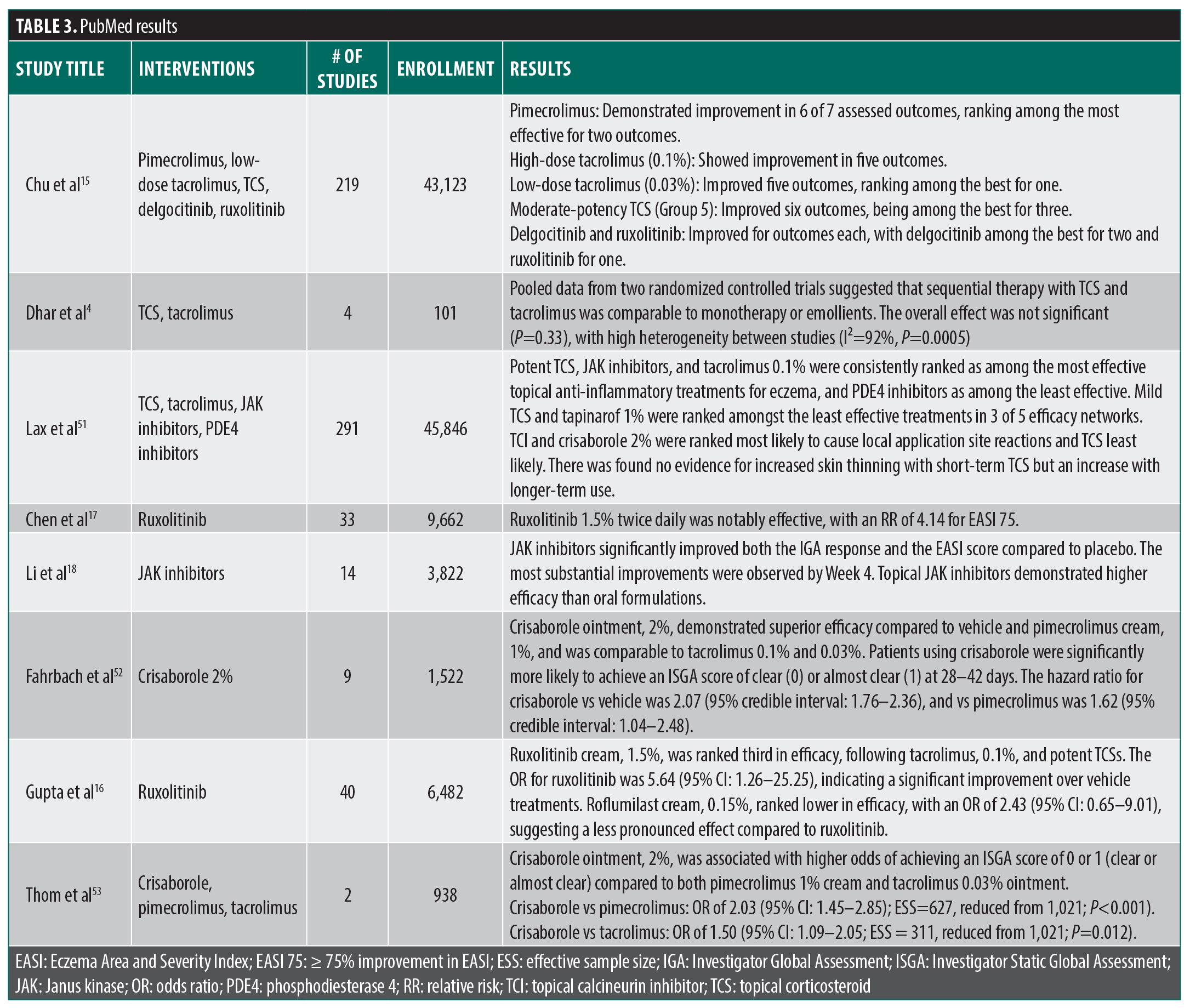
PubMed. Fifteen results matched the search criteria on PubMed and were filtered down to 8 (Figure 1; Table 3). Advanced targeted topical agents such as JAK inhibitors, AhR modulators, and PDE4 inhibitors were found to have efficacy for the treatment of AD with low rates of AEs.15–18
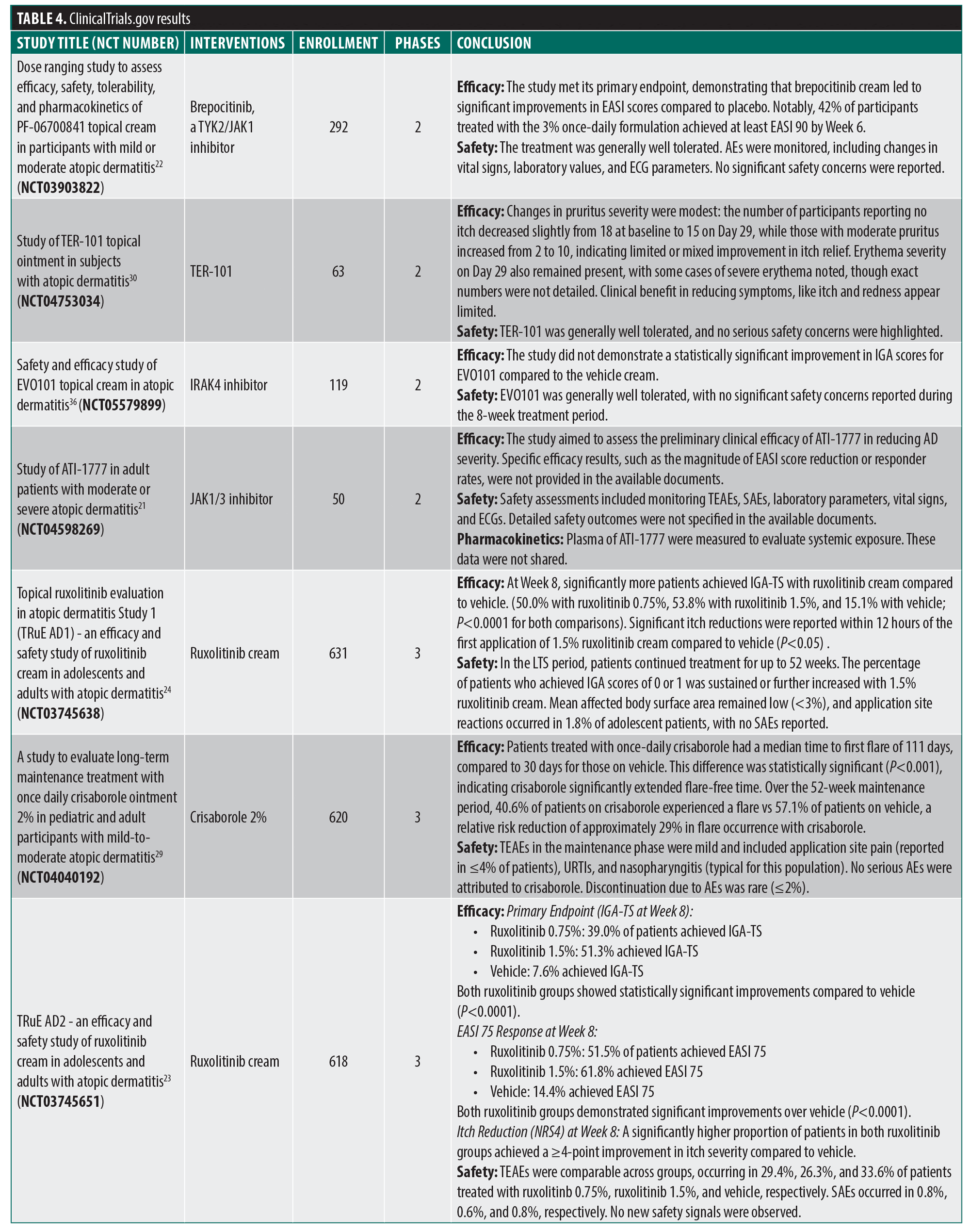
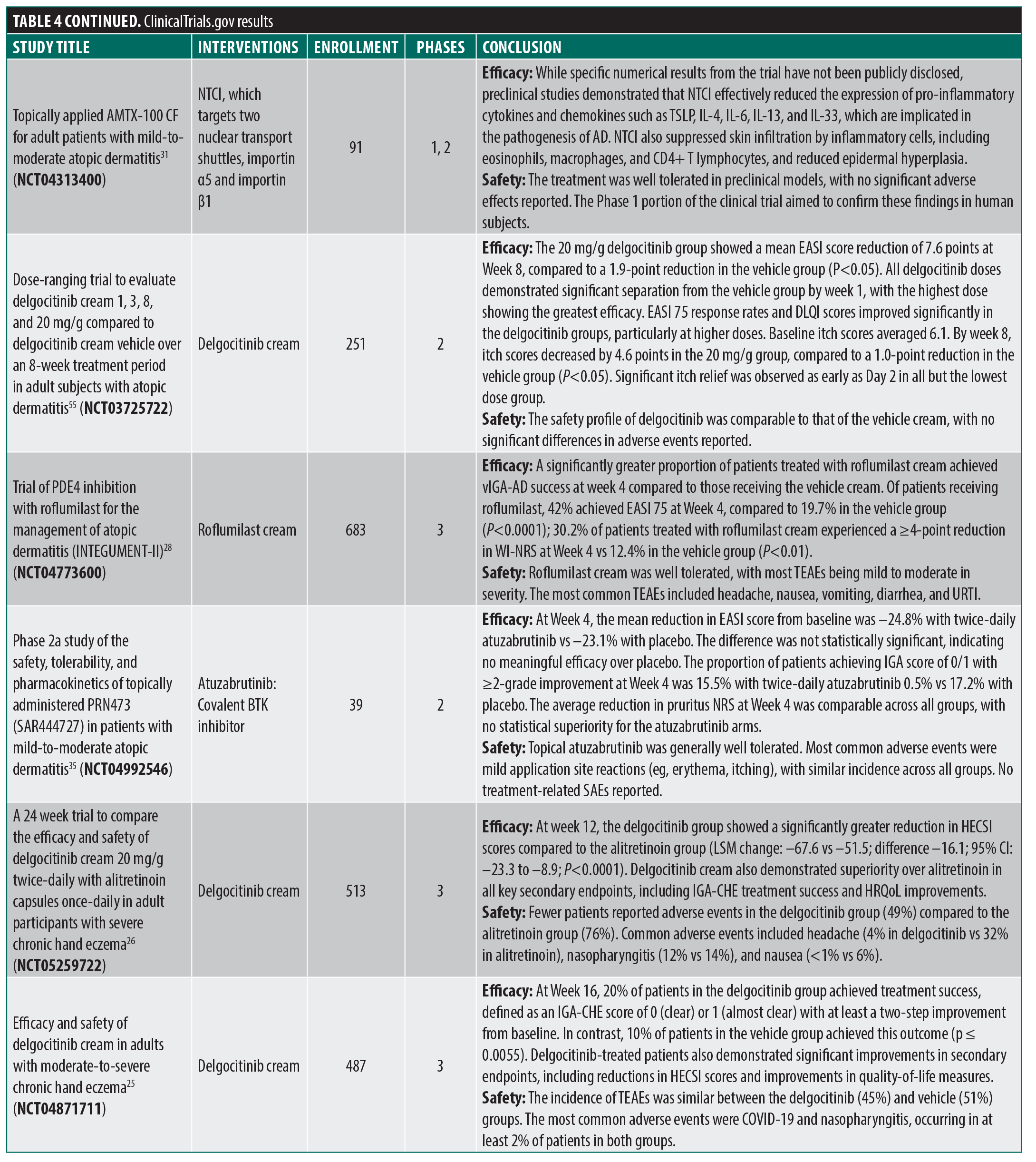
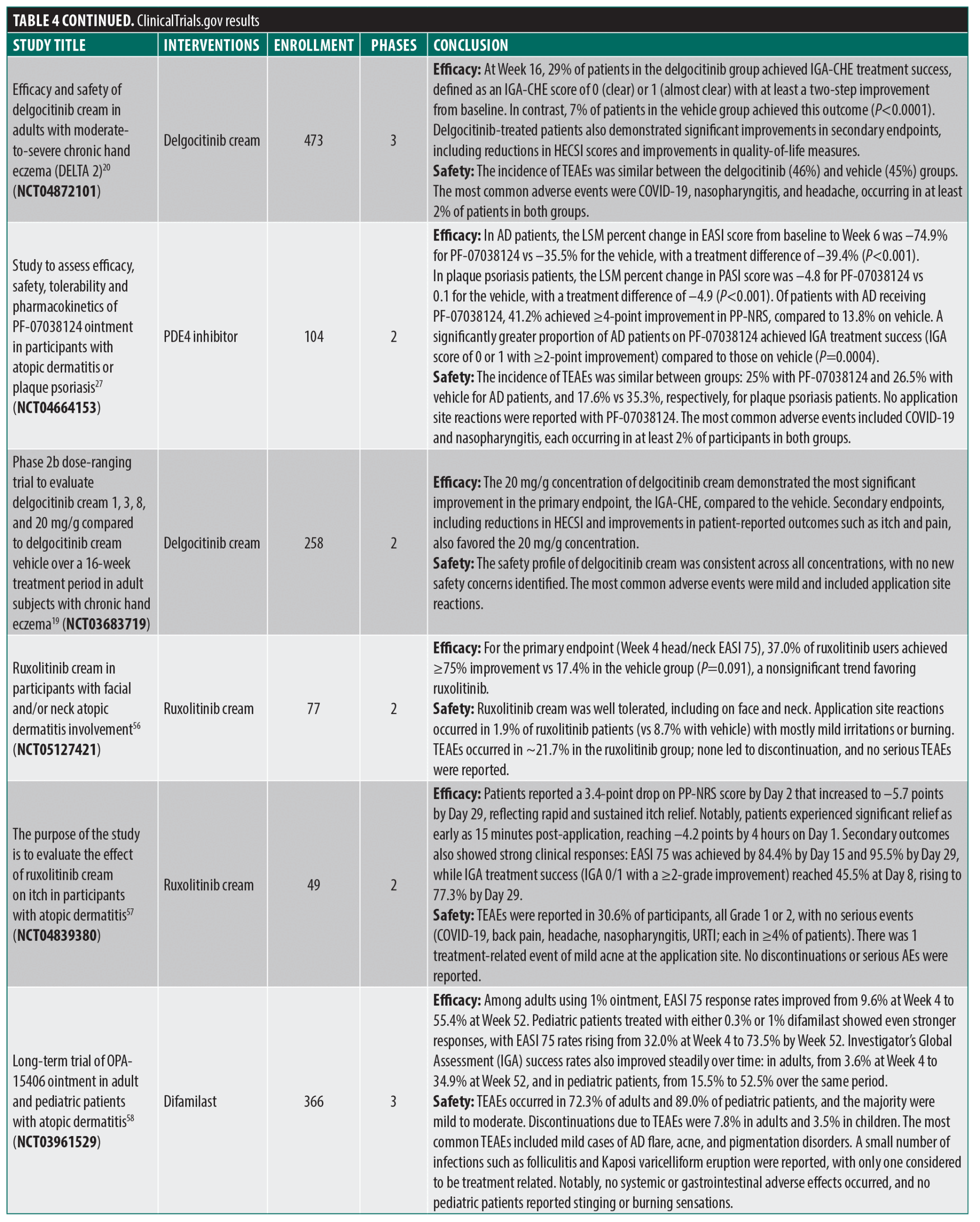
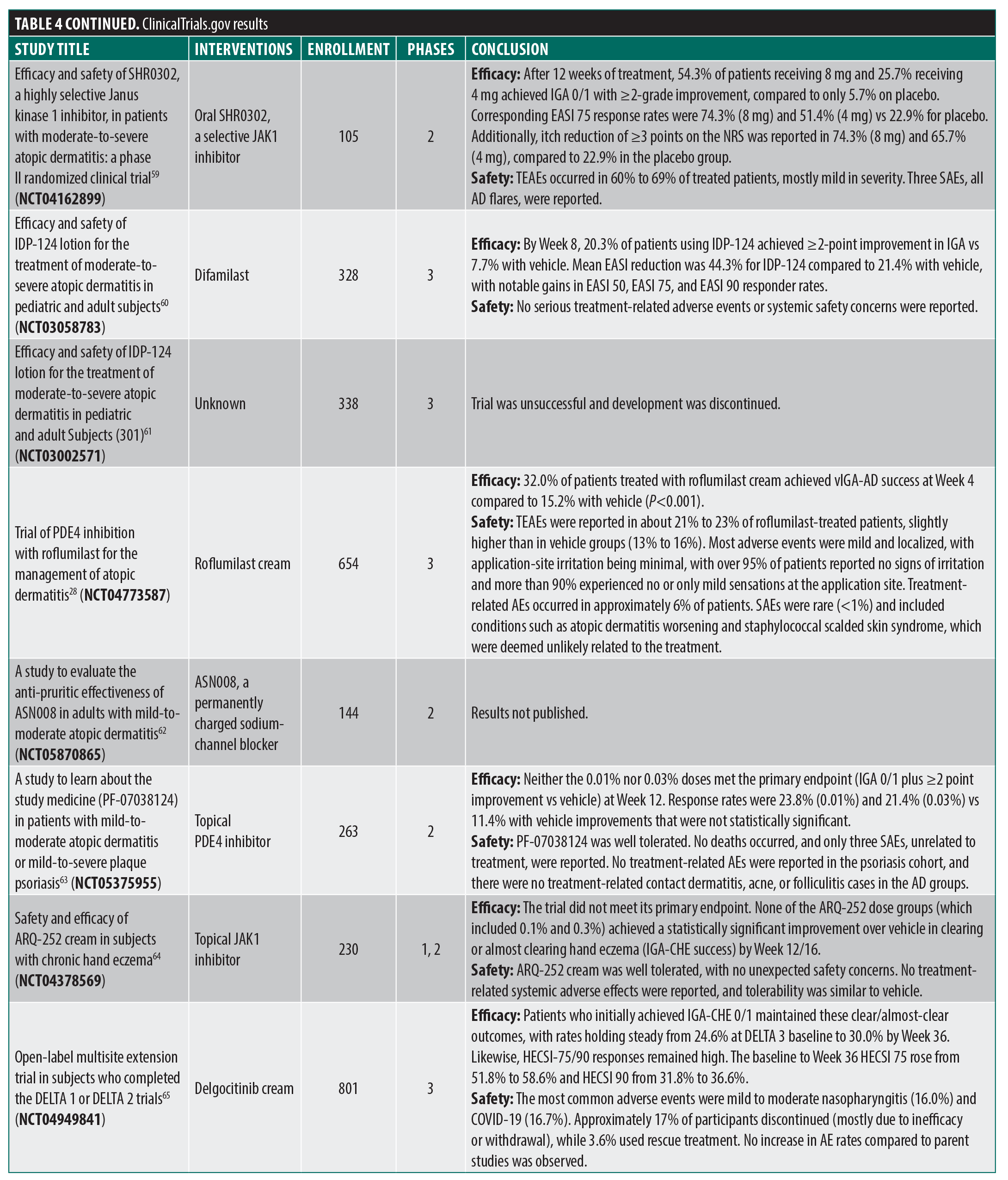
ClinicalTrials.gov. There were 119 results that were reduced to 31 trials upon secondary manual review (Figure 1; Table 4). Several novel JAK inhibitors were deemed safe and efficacious, including delgoticinib and ruxolitinib, while others failed to meet endpoints (ARQ-252).19–26 Two phase 3 (crisaborole and roflumilast) and one phase 2 (difamilast) trials for PDE4 inhibitors demonstrated statistically significant results.27–29 Other mechanisms of action were also examined with mixed findings and some AEs.30–35 For example, a phase 2 clinical trial for an interleukin 1 receptor-associated kinase 4 (IRAK4) inhibitor did not demonstrate statistically significant improvement relative to vehicle, ending development for that compound.36 Finally, 6 phase 3 trials were used to approve the agents tapinarof, roflumilast, and ruxolitinib (Table 2).23,24,37–41
Five studies (NCT04871711, NCT04872101, NCT05259722, NCT03683719, NCT04949841) examined delgoticinib, a JAK inhibitor, in phase 3 trials (DELTA1, DELTA2, and DELTA3) for the indication of chronic hand eczema (CHE).20, 25,26,42,43 Difamilast (PDE4 inhibitor) was another compound that was thoroughly investigated (NCT03961529).44
Discussion
Nonsteroidal AhR modulator. The AHR/AHR-nuclear translocator (ARNT) system is a sensitive sensor for exogenous and endogenous small–molecule chemicals.45 AhR expression has been shown to be increased in AD skin.46 Once activated, the AhR/ARNT axis strengthens skin barrier function and accelerates epidermal terminal differentiation by upregulating filaggrin expression.45 The FDA initially approved the first AhR modulator, tapinarof cream 1%, for plaque psoriasis in May 2022 and for AD in December 2024 in adults and children as young as 2 years. Two double-blind, randomized, vehicle-controlled phase 3 trials (NCT05014568, NCT05032859) were conducted.37 A total of 407 and 406 patients were randomized in ADORING 1 and 2, respectively. A significantly higher proportion of patients treated with tapinarof achieved a Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) score of 0 or 1 with at least a 2-grade improvement from baseline at Week 8 compared to those receiving the vehicle: 45.4% in the tapinarof arm compared with 13.9% with vehicle in ADORING 1 (P<0.0001) and 46.4% vs 18.0% in ADORING 2 (P<0.0001).37 A greater proportion of patients treated with tapinarof achieved at least 75% improvement in the Eczema Area and Severity Index (EASI 75) at Week 8 compared to the vehicle group: 55.8% of tapinarof-treated patients vs 22.9% for vehicle in ADORING 1 (P<0.0001) and 59.1% vs 21.2% in ADORING 2 (P<0.0001).37 Patients reported rapid and significant reductions in pruritus with tapinarof treatment compared to the vehicle.37 Common AEs (occurring in ≥5% of patients) included folliculitis, headache, and nasopharyngitis.37 Treatment-emergent adverse events (TEAEs) were reported in 70% of patients compared to 38% with vehicle, and these events were primarily mild or moderate in severity.37 Notably, fewer patients discontinued treatment due to AEs in the tapinarof group compared to the vehicle group.37
Two recent Japanese phase 3 studies evaluating tapinarof cream, 1%, for AD shared similar efficacy endpoints. In the ZBB4-1 randomized, double-blind, vehicle-controlled trial, the primary endpoint was the proportion of patients who achieved an Investigator’s Global Assessment (IGA) score of 0 (clear) or 1 (almost clear) with at least a 2-grade improvement at Week 8; this was reached by 20.24% of tapinarof-treated patients vs 2.24% on vehicle (P=0.0007).47 EASI 75 was achieved by 40.3% in the tapinarof group vs 4.3% on vehicle (P<0.0001).47 In the open-label, 52-week ZBB4-2 study, sustained efficacy was observed, with IGA success rates of 28.1% at Week 16, 32.3% at Week 24, and 41.3% at Week 52. EASI 75 response rates increased over time from 53.3% at Week 16 to 76.6% at Week 52.47 Across both trials, most AEs were mild or moderate. Common AEs included folliculitis, acne, and headache, and no new safety concerns were identified over the 52-week treatment period.47
JAK inhibitors. The JAK/signal transducer and activator of transcription (STAT) pathway is a significant cell signaling pathway involved in cytokine production, well characterized for mediating the inflammatory immune response, among other functions.48 It facilitates the production of inflammatory cytokines interleukin (IL) 4, IL-5, IL-13, and IL-31, which drive the activation of helper T-cell 2 cell-mediated immunity, a pathway crucial for the pathogenesis of AD.49 Blocking this inflammatory pathway has been shown to alleviate symptoms of AD.48,49 Regarding topical agents, research has indicated promise for both first-generation and second-generation JAK inhibitors for AD, including studies on delgocitinib (a pan-JAK inhibitor), ruxolitinib (a JAK1/JAK2 inhibitor), tofacitinib (a pan-JAK inhibitor), ifidancitinib (a JAK1/JAK3 inhibitor), upadacitinib (a JAK1 inhibitor), and brepocitinib (a TYK2/JAK1 inhibitor).48,49
Most recently, two phase 3 trials for ruxolitinib (TRuE-AD1 and TRuE-AD2) involving patients aged ≥12 years with mild-to-moderate AD found promising results.40 At Week 8, 53.8% to 54.5% of patients using ruxolitinib cream, 1.5%, twice daily achieved Investigator’s Global Assessment–Treatment Success (IGA–TS), defined as a score of 0 (clear) or 1 (almost clear) with ≥2-grade improvement from baseline, compared to 11.5% to 15.1% in the vehicle group.40 Additionally, 61.8% to 62.0% achieved EASI 75 vs 24.6% to 29.6% with vehicle.40 Treatment over 52 weeks resulted in 74.1% to 77.8% of patients achieving IGA 0 or 1.41 AEs were mostly mild; the most common were nasopharyngitis and application site reactions.40 After 1 year of treatment, only 7.4% experienced treatment-related AEs.41
Delgocitinib, a topical pan-JAK inhibitor, was approved in Japan in January 2020 for the treatment of AD in patients aged 16 years and older. In the US, delgocitinib received FDA approval for the treatment of moderate to severe CHE in adults in July 2025. In the European Union, delgocitinib cream received approval in September 2024 for the treatment of moderate to severe CHE in adults.
A phase 2B randomized, double-blind, vehicle-controlled study evaluated the efficacy and safety of brepocitinib, 1%, in adults with mild-to-moderate AD over 6 weeks.50 Patients treated with brepocitinib 1% twice daily achieved a 75% reduction in EASI scores at Week 6, compared to a 47.6% reduction in the vehicle group.50 Once-daily applications of brepocitinib 1% and 3% resulted in EASI score reductions of 70.1% and 67.9%, respectively, vs 44.4% in the once-daily vehicle group.50 A 90% reduction in EASI score (EASI 90) was observed in 27.0% to 41.7% of patients using brepocitinib, compared to 10.8% in the once-daily vehicle group and 8.3% with the twice-daily vehicle.50 Topical brepocitinib was well tolerated, with no serious treatment-related AEs reported. The most common adverse effects were mild application site reactions.50
PDE4 inhibitors. Roflumilast cream, 0.15%, was approved by the FDA for AD in patients aged 6 years and older in 2024. Clinical trials showed a significantly greater proportion of patients treated with roflumilast cream achieved vIGA-AD success at Week 4 compared to those receiving the vehicle cream.38,54 Of the patients treated with roflumilast, 42% achieved EASI 75 at Week 4, compared to 19.7% in the vehicle group (P<0.0001).38,54 Those treated with roflumilast also had a higher rate of achieving IGA success (32.0% vs 15.2%, P<0.001).38 Further, 30.2% of patients treated with roflumilast experienced a ≥4-point reduction in Worst Itch Numeric Rating Scale at Week 4 vs 12.4% in the vehicle group (P<0.01).38,54 Roflumilast cream was well tolerated, with most TEAEs being mild-to-moderate in severity.46,47 Serious AEs were experienced by 0.9% of patients, and 1.4% to 1.8% discontinued roflumilast due to any TEAE at 4 weeks.38,54 The most common TEAEs included headache, nausea, vomiting, diarrhea, and upper respiratory tract infection.38
Difamilast is another PDE4 inhibitor approved in Japan for AD. A phase 3 randomized, double-blind, vehicle-controlled study assessed the efficacy of difamilast ointment, 1%, applied twice daily for 4 weeks in adult patients with mild-to-moderate AD.44 At Week 4, 38.46% of patients achieved IGA 0 or 1 with a ≥2-grade improvement from baseline, compared to 12.64% in the vehicle group (P<0.0001).44 The difamilast group had statistically significant improvements across EASI reductions, including 58.24% vs 25.82% achieving at least 50% improvement in EASI (P<0.001), 42.86% vs 13.19% for EASI 75 (P<0.0001), and 24.73% vs 5.49% for EASI 90 (P<0.0001), respectively.44 Additionally, a significant mean percent change in overall EASI score from baseline was observed for the difamilast group compared with vehicle at Week 1 (–32.6% vs –10.4%, respectively; P<0.0001) and was sustained until Week 4 (–49.1% vs –10.5%, respectively; P<0.0001).44 TEAEs were mostly mild or moderate and occurred less frequently in the difamilast group than in the vehicle group.44
Conclusion
The therapeutic landscape for AD has been transformed through development of new, advanced targeted therapies. Short- and long-term use of TCS have substantial AEs, prompting a search for novel mechanisms of action to address this significant unmet need. Advanced agents have been introduced in the last five years using mechanisms such as JAK inhibition, PDE4 inhibition, and AhR modulation. Collectively, these advances mark a new era in the treatment of AD, offering more precise, effective, and safe options tailored to the disease’s immunologic complexity. Incorporating these therapies into treatment protocols may substantially improve disease management and patient quality of life.
References
- Tian J, Zhang D, Yang Y, et al. Global epidemiology of atopic dermatitis: a comprehensive systematic analysis and modelling study. Br J Dermatol. 2023;190(1):55–61.
- Frazier WL, Schneider L, Chu DK, Atopic dermatitis: updated guidelines from the AAAAI/ACAAI Joint Task Force on Practice Parameters. Am Fam Physician. 2024;110(4):437–439.
- Stacey SK, McEleney M. Topical corticosteroids: choice and application. Am Fam Physician. 2021;103(6):337–343.
- Dhar S, De A, Saha A, et al. Intermittent or sequential topical tacrolimus in atopic dermatitis: systematic review and meta-analysis. Cureus, 2023;15(12):e50640.
- Sreeraj K, Risana BU, Mathew SR, et al Optimized and safe use of topical corticosteroids in pediatric dermatology through interdisciplinary collaboration: a review. Discover Med. 2024;1:15.
- Mehta AB, Nadkarni NJ, Patil SP, Godse KV, Gautam M, Agarwal S. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82(4):371–378.
- Coondoo A, Phiske M, Verma S, Lahiri K. Side-effects of topical steroids: a long overdue revisit. Indian Dermatol Online J. 2014;5(4):416–425.
- Burshtein J, Chovatyia R, Golant A, et al. Risks of topical corticosteroid therapy and role for advanced targeted topical treatments for inflammatory skin diseases: an expert consensus panel. Dermatol Online J. 2025;31(1):1.
- Amiri D, Schwarz CW, Gether L, Skov L. Safety and efficacy of topical calcineurin inhibitors in the treatment of facial and genital psoriasis: a systematic review. Acta Derm Venereol. 2023;103:adv00890.
- Zhao Z, Gao XH, Li W, et al. Experts’ consensus on the use of pimecrolimus in atopic dermatitis in China: a TCS-sparing practical approach. Derm Ther (Heidelb). 2022;12(4):933–947.
- Siegfried ED, Jaworski JC, Kaiser JD, Hebert AA, Systematic review of published trials: long-term safety of topical corticosteroids and topical calcineurin inhibitors in pediatric patients with atopic dermatitis. BMC Pediatr. 2016;16:75.
- Ohtsuki M, Morimoto H, Nakagawa H. Tacrolimus ointment for the treatment of adult and pediatric atopic dermatitis: review on safety and benefits. J Dermatol. 2018;45(8):936–942.
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med. 2021;18(3):e1003583.
- AHRQ. Methods Guide for Effectiveness and Comparative Effectiveness Reviews. 2008: Rockville, MD.
- Chu DK, Chu AWL, Rayner DG, et al. Topical treatments for atopic dermatitis (eczema): systematic review and network meta-analysis of randomized trials. J Allergy Clin Immunol. 2023;152(6):1493–1519.
- Gupta S, Xiong G, Shenouda C, Keow S, Abu-Hilal M. Ruxolitinib vs roflumilast: ameta-analysis of the comparative efficacy of two topical therapies for atopic dermatitis. J Am Acad Dermatol. 2025;93(1):208–210.
- Chen Q, Cui L, Hu Y, Chen Z, Gao Y, Shi Y. Short-term efficacy and safety of biologics and Janus kinase inhibitors for patients with atopic dermatitis: a systematic review and meta-analysis. Heliyon. 2023;9(11):e22014.
- Li C, Sun X, Zhao K, et al. Efficacy and safety of Janus kinase inhibitors for the treatment of atopic dermatitis: a systematic review and meta-analysis. Dermatology. 2022;238(4):725–735.
- Phase 2b dose-ranging trial to evaluate delgocitinib cream 1, 3, 8, and 20 mg/g compared to delgocitinib cream vehicle over a 16-week treatment period in adult subjects with chronic hand eczema. Updated March 12, 2025. https://clinicaltrials.gov/study/NCT03683719
- Efficacy and safety of delgocitinib cream in adults with moderate-to-severe chronic hand eczema (DELTA 2). Updated April 8, 2025. https://clinicaltrials.gov/study/NCT04872101
- Study of ATI-1777 in adult patients with moderate or severe atopic dermatitis. Updated September 28, 2023. https://clinicaltrials.gov/study/NCT04598269
- Dose ranging study to assess efficacy, safety, tolerability, and pharmacokinetics of PF-06700841 topical cream in participants with mild or moderate atopic dermatitis. Updated March 29, 2021. https://clinicaltrials.gov/study/NCT03903822
- TRuE AD2 – an efficacy and safety study of ruxolitinib cream in adolescents and adults with atopic dermatitis. Updated September 9, 2023. https://clinicaltrials.gov/study/NCT03745651
- Topical ruxolitinib evaluation in atopic dermatitis Study 1 (TRuE AD1) – an efficacy and safety study of ruxolitinib cream in adolescents and adults with atopic dermatitis. Updated September 28, 2023. https://clinicaltrials.gov/study/NCT03745638
- Efficacy and safety of delgocitinib cream in adults with moderate-to-severe chronic hand eczema. Updated April 8, 2025. https://clinicaltrials.gov/study/NCT04871711
- A 24 week, randomised, assessor blinded, active-controlled, parallel group, Phase 3, 2 arm trial to compare the efficacy and safety of delgocitinib cream 20 mg/g twice-daily with alitretinoin capsules once-daily in adult participants with severe chronic hand eczema. 2022.
- Study to assess efficacy, safety, tolerability and pharmacokinetics of PF-07038124 ointment in participants with atopic dermatitis or plaque psoriasis. Updated August 26, 2022. https://clinicaltrials.gov/study/NCT04664153
- Trial of PDE4 inhibition with roflumilast for the management of atopic dermatitis (INTEGUMENT-II). Updated October 4, 2024. https://clinicaltrials.gov/study/NCT04773600
- A study to evaluate long-term maintenance treatment with once daily crisaborole ointment 2% in pediatric and adult participants with mild-to-moderate atopic dermatitis. Updated March 8, 2023. https://clinicaltrials.gov/study/NCT04040192.
- Study of TER-101 topical ointment in subjects with atopic dermatitis. Updated July 26, 2022. https://clinicaltrials.gov/study/NCT04753034.
- Two part, phase I/II, multi-center, double-blind, randomized, vehicle-controlled study of the safety and efficacy of topically applied AMTX-100 CF in adult patients with mild-to-moderate atopic dermatitis, R. Amarex Clinical, Editor. 2020.
- A Phase II, randomized, double-blind, vehicle controlled study of the efficacy, safety, and tolerability of B244 topical spray for the treatment of pruritus in adults with a history of atopic dermatitis, L.L.C. bioRasi, Editor. 2020.
- Multicenter, open label, uncontrolled clinical investigation on the performance and safety of DermoRelizema Ecofoam in the management of some dermatitis in the adult. 2021.
- A multicenter, open label, uncontrolled study for the evaluation of efficacy and safety by clinical parameters of Relizema Ecofoam in adult atopic and contact dermatitis. 2021.
- Phase 2a study of the safety, tolerability, and pharmacokinetics of topically administered PRN473 (SAR444727) in patients with mild-to-moderate atopic dermatitis. Updated September 10, 2025. https://clinicaltrials.gov/study/NCT04992546
- Safety and efficacy study of EVO101 topical cream in atopic dermatitis. Updated March 5, 2025. https://clinicaltrials.gov/study/NCT05579899
- Silverberg JI, Eichenfield LF, Hebert AA, et al. Tapinarof cream 1% once daily: significant efficacy in the treatment of moderate to severe atopic dermatitis in adults and children down to 2 years of age in the pivotal phase 3 ADORING trials. J Am Acad Dermatol. 2024;91(3):457–465.
- Simpson EL, Eichenfield LF, Alonso-Llamazares J, et al. Roflumilast cream, 0.15%, for atopic dermatitis in adults and children: INTEGUMENT-1 and INTEGUMENT-2 randomized clinical trials. JAMA Dermatol. 2024;160(11):1161–1170.
- Eichenfield LF, Serrao R, Prajapati VH, et al. Efficacy and safety of once-daily roflumilast cream 0.05% in pediatric patients aged 2-5 years with mild-to-moderate atopic dermatitis (INTEGUMENT-PED): a phase 3 randomized controlled trial. Pediatr Dermatol. 2025;42(2):296–304.
- Papp K, Szepietowski JC, Kircik L, et al. Efficacy and safety of ruxolitinib cream for the treatment of atopic dermatitis: results from 2 phase 3, randomized, double-blind studies. J Am Acad Dermatol. 2021;85(4):863–872.
- Papp K , Szepietowski JC, Kircik L, et al. Long-term safety and disease control with ruxolitinib cream in atopic dermatitis: results from two phase 3 studies. J Am Acad Dermatol. 2023;88(5):1008–1016.
- Bauer A, Thyssen JP, Buhl T, et al. Treatment with delgocitinib cream improves itch, pain and other signs and symptoms of chronic hand eczema: results from the Hand Eczema Symptom Diary in a phase IIb randomized clinical trial. Contact Dermatitis. 2023;89(1):46–53.
- Gooderham M, Molin S, Bissonnette R, et al. Long-term safety and efficacy of delgocitinib cream for up to 52 weeks in adults with chronic hand eczema: results of the phase 3 open-label extension DELTA 3 trial following the DELTA 1 and 2 trials. J Am Acad Dermatol. 2025;93(1):95–103.
- Saeki H, Ito K, Yokota D, Tsubouchi H. Difamilast ointment in adult patients with atopic dermatitis: a phase 3 randomized, double-blind, vehicle-controlled trial. J Am Acad Dermatol. 2022;86(3):607–614.
- Furue M, Hashimoto-Hachiya A, Tsuji G. Aryl hydrocarbon receptor in atopic dermatitis and psoriasis. Int J Mol Sci. 2019;20(21):5424.
- Kim HO, Kim JH, Chung BY, et al. Increased expression of the aryl hydrocarbon receptor in patients with chronic inflammatory skin diseases. Exp Dermatol. 2014;23(4):278–281.
- Igarashi A, Tsuji G, Fukasawa S, et al. Tapinarof cream for the treatment of atopic dermatitis: efficacy and safety results from two Japanese phase 3 trials. J Dermatol. 2024;51(11):1404–1413.
- Xue C, Yao Q, Gu X, et al. Evolving cognition of the JAK-STAT signaling pathway: autoimmune disorders and cancer. Signal Transduct Target Ther. 2023;8(1):204.
- Facheris P, Jeffery J, Del Duca E, Guttman-Yassky E. The translational revolution in atopic dermatitis: the paradigm shift from pathogenesis to treatment. Cell Mol Immunol. 2023;20(5):448–474.
- Landis MN, Arya, M, Smith S, et al. Efficacy and safety of topical brepocitinib for the treatment of mild-to-moderate atopic dermatitis: a phase IIb, randomized, double-blind, vehicle-controlled, dose-ranging and parallel-group study. Br J Dermatol. 2022;187(6):878–887.
- Lax SJ, Van Vogt E, Candy B, et al. Topical anti-inflammatory treatments for eczema: network meta-analysis. Cochrane Database Syst Rev. 2024;8(8):CD015064.
- Fahrbach K, Tarpey J, Washington EB, et al. Crisaborole ointment, 2%, for treatment of patients with mild-to-moderate atopic dermatitis: systematic literature review and network meta-analysis. Dermatol Ther (Heidelb). 2020;10(4):681–694.
- Thom H, Cheng V, Keeney E, et al. Matching-adjusted indirect comparison of crisaborole ointment 2% vs. topical calcineurin inhibitors in the treatment of patients with mild-to-moderate atopic dermatitis. Dermatol Ther (Heidelb). 2022;12(1):185–194.
- Simpson EL, Eichenfield LF, Papp KA, et al. Long-term safety and efficacy with roflumilast cream 0.15% in patients aged ≥6 years with atopic dermatitis: a phase 3 open-label extension trial. Dermatitis. 2025;37(1):75–83.
- Dose-ranging trial to evaluate delgocitinib cream 1, 3, 8, and 20 mg/g compared to delgocitinib cream vehicle over an 8-week treatment period in adult subjects with atopic dermatitis. Updated March 12, 2025. https://clinicaltrials.gov/study/NCT03725722
- Ruxolitinib cream in participants with facial and/or neck atopic dermatitis involvement. Updated August 15, 2024. https://clinicaltrials.gov/study/NCT05127421
- The purpose of the study is to evaluate the effect of ruxolitinib cream on itch in participants with atopic dermatitis. Updated October 31, 2023. https://clinicaltrials.gov/study/NCT04839380
- Long-term trial of OPA-15406 ointment in adult and pediatric patients with atopic dermatitis. Updated November 19, 2021. https://clinicaltrials.gov/study/NCT03961529
- Zhao Y, Zhang L, Ding Y, et al. Efficacy and safety of SHR0302, a highly selective Janus kinase 1 inhibitor, in patients with moderate to severe atopic dermatitis: a Phase II randomized clinical trial. Am J Clin Dermatol. 2021;22(6):877–889.
- A Efficacy and safety of IDP-124 lotion for the treatment of moderate-to-severe atopic dermatitis in pediatric and adult subjects. Updated May 8, 2023. https://clinicaltrials.gov/study/NCT03058783
- Efficacy and safety of IDP-124 lotion for the treatment of moderate-to-severe atopic dermatitis in pediatric and adult subjects (301). Updated May 8, 2023. https://clinicaltrials.gov/study/NCT03002571
- A study to evaluate the anti-pruritic effectiveness of ASN008 in adults with mild-to-moderate atopic dermatitis. Updated May 16, 2025. https://clinicaltrials.gov/study/NCT05870865
- A study to learn about the study medicine (PF-07038124) in patients with mild-to-moderate atopic dermatitis or mild-to-severe plaque psoriasis. Updated October 2, 2024. https://clinicaltrials.gov/study/NCT05375955
- Safety and efficacy of ARQ-252 cream in subjects with chronic hand eczema. Updated April 5, 2024. https://clinicaltrials.gov/study/NCT04378569
- Open-label multisite extension trial in subjects who completed the DELTA 1 or DELTA 2 trials. Updated April 8, 2025. https://clinicaltrials.gov/study/NCT04949841
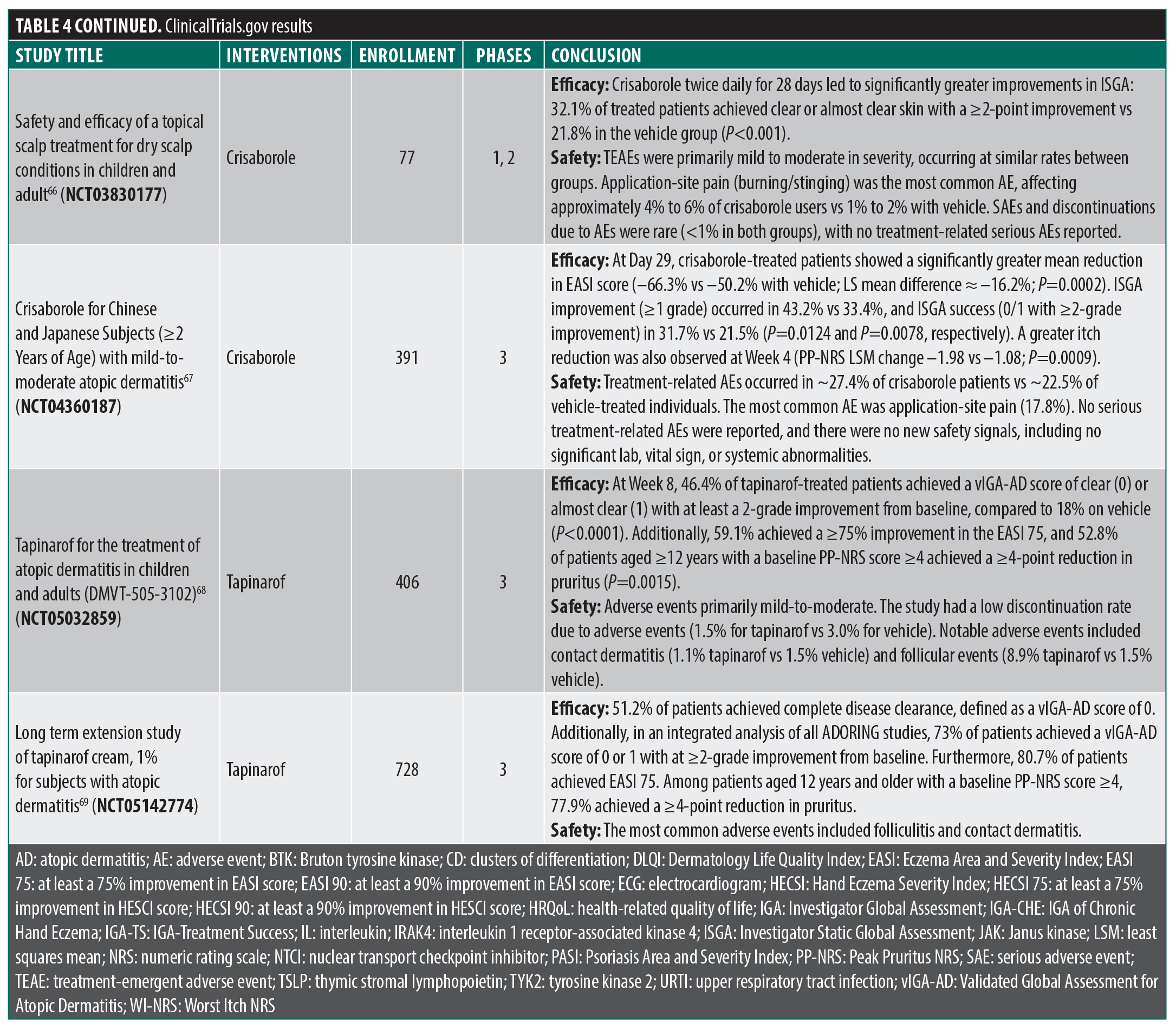
- Safety and efficacy of a topical scalp treatment for dry scalp conditions in children and adult. Updated February 24, 2025. https://clinicaltrials.gov/study/NCT03830177
- Crisaborole for Chinese and Japanese Subjects (≥2 Years of Age) With mild-to-moderate atopic dermatitis. Updated June 7, 2022. https://clinicaltrials.gov/study/NCT04360187
- Tapinarof for the treatment of atopic dermatitis in children and adults (DMVT-505-3102). Updated September 2, 2025. https://clinicaltrials.gov/study/NCT05032859
- Long term extension study of tapinarof cream, 1% for subjects with atopic dermatitis. Updated August 13, 2025. https://clinicaltrials.gov/study/NCT05142774