J Clin Aesthet Dermatol. 2026;19(2):26–32.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no conflict of interest to declare.
by Joshua K. Morales, BA; Jennifer Keelin, BS; Toan N. Vu, BS; Sabrina C. Camacho, BS; Selene M. Kizy, BS; Sarah Kazemeini, BS; Rebecca Metellus, BS; Naif Hebo, MS; and David G. Cotter, MD, PhD
Mr. Morales is with the Anne Marion Burnett School of Medicine at Texas Christian University in Fort Worth, Texas. Ms. Keelin is with the Florida International University Herbert Wertheim College of Medicine in Miami, Florida. Mr. Vu is with the University of Wisconsin School of Medicine and Public Health in Madison, Wisconsin. Ms. Camacho is with Texas Tech University Health Sciences Center School of Medicine in Lubbock, Texas. Ms. Kizy is with Oakland University William Beaumont School of Medicine in Rochester, Michigan. Ms. Kazemeini is with the Kirk Kerkorian School of Medicine at UNLV in Las Vegas, Nevada. Ms. Metellus is with Geisinger Commonwealth School of Medicine in Scranton, Pennsylvania. Mr. Hebo is with the University of Arizona College of Medicine Phoenix in Phoenix, Arizona. Dr. Cotter is with Las Vegas Dermatology and the University of Nevada, Las Vegas School of Medicine in Las Vegas, Nevada.
ABSTRACT: Glucagon-like peptide-1 receptor agonists (GLP-1 RAs), commonly prescribed for type 2 diabetes and obesity, have demonstrated potential anti-inflammatory and immunomodulatory effects that may be beneficial in chronic inflammatory skin conditions such as psoriasis and hidradenitis suppurativa (HS). A systematic review of the literature was conducted, focusing on prospective studies, case reports, and systematic reviews that evaluated the impact of GLP-1 RAs on these diseases. In psoriasis, GLP-1 RAs, particularly liraglutide, have been associated with improvements in the Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index (DLQI), especially among patients with T2D. Reported benefits include enhanced glycemic control, weight reduction, and decreased levels of inflammatory markers, suggesting that GLP-1 RAs may modulate immune pathways and proinflammatory cytokine activity involved in the pathogenesis of psoriasis. Similarly, in HS, GLP-1 RAs such as liraglutide and semaglutide have shown promising results, including decreased lesion severity, improved quality of life, and reduced systemic inflammation. Weight loss induced by these agents may also contribute to symptom improvement by reducing mechanical stress in intertriginous areas and mitigating inflammatory responses associated with HS. Although preliminary evidence suggests that GLP-1 RAs may play a role in managing psoriasis and HS through both metabolic and immunologic mechanisms, current data are limited to early-phase studies and case reports. Further large-scale randomized controlled trials, some of which are ongoing, with diverse study populations are necessary to better understand their efficacy, safety, and long-term impact in the treatment of these chronic inflammatory skin conditions. Keywords: GLP-1, psoriasis, hidradenitis suppurativa, liraglutide, semiglutide
Introduction
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are highly effective pharmacologic agents for managing type 2 diabetes (T2D) and obesity. Their efficacy also extends beyond glycemic control. GLP-1 RAs mimic the endogenous incretin hormone secreted by intestinal L-cells. This helps promote insulin release, delays gastric emptying, and enhances feelings of satiety.1 Studies have shown that GLP-1 RAs have favorable pharmacokinetics and demonstrate anti-inflammatory properties, making them potentially useful beyond
glycemic control.1,2
Psoriasis and hidradenitis suppurativa (HS) are immune-mediated dermatologic disorders that are frequently comorbid with obesity and insulin resistance. Both are associated with systemic inflammation and share overlapping cytokine profiles. Psoriatic disease is characterized by inflammation of the skin and joints and is associated with an increased risk of cardiovascular comorbidities such as stroke, myocardial infarction, and hypertension.2 HS is characterized by recurrent painful nodules and abscesses in intertriginous areas and is frequently associated with comorbidities such as obesity, insulin resistance, and cardio-vascular disease.3,4
Due to the overlap in chronic inflammatory pathophysiology, GLP-1 RAs may serve as a potential therapeutic candidate for the management of these skin disorders, particularly those co-occurring with metabolic dysfunction. These skin conditions not only affect physical health but can also have a serious toll on patients’ quality of life. Notably, clinical improvements in skin lesions have been observed in some studies, independent of weight loss or glycemic control.5,6 This raises the possibility that these medications may have a more direct role in calming inflammation in the skin.
Although recent studies have begun to explore the dermatologic applications of GLP-1 RAs, the literature remains limited. Most evidence comes from small trials or case reports. Nonetheless, the overlapping metabolic and immune pathways in diseases like psoriasis and HS indicate a need to explore the role of GLP-1 RAs in dermatology. This review aims to synthesize the available literature on the use of GLP-1 RAs in HS and psoriasis, evaluate their emerging clinical relevance, and identify areas where more research is needed.
Methods
A systematic literature search was conducted using PubMed, MEDLINE Ultimate, Scopus, and Web of Science, following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to identify studies examining the use of GLP-1 RAs in the treatment of psoriasis and HS. The following key terms were searched across all four databases: (“Psoriasis” OR “Hidradenitis Suppurativa” OR “Acne Inversa”) AND (“Glucagon-Like Peptide 1” OR “GLP-1 Agonists” OR “GLP-1 Receptor Agonists” OR “Liraglutide” OR “Semaglutide”). Search results were imported into Covidence for screening and review. Two reviewers independently screened titles and abstracts to determine eligibility. Additionally, a full-text assessment was conducted when required. Any disagreements were resolved by consensus between the two reviewers. Articles were included if they had full-text availability through either Florida International University Herbert Wertheim College of Medicine or the Anne Burnett Marion School of Medicine at Texas Christian University. Articles were included if they were published in the last 10 years, were written in the English language, and had a specific focus on the use of GLP-1 RAs in patients with psoriasis and/or HS. Articles were excluded if they were not written in the English language, did not have a registered digital object identifier (DOI), were a letter to the editor or an abstract-only publication, did not directly assess the use of GLP-1 RAs in psoriasis or HS, were duplicate studies, or were inaccessible through institutional resources.
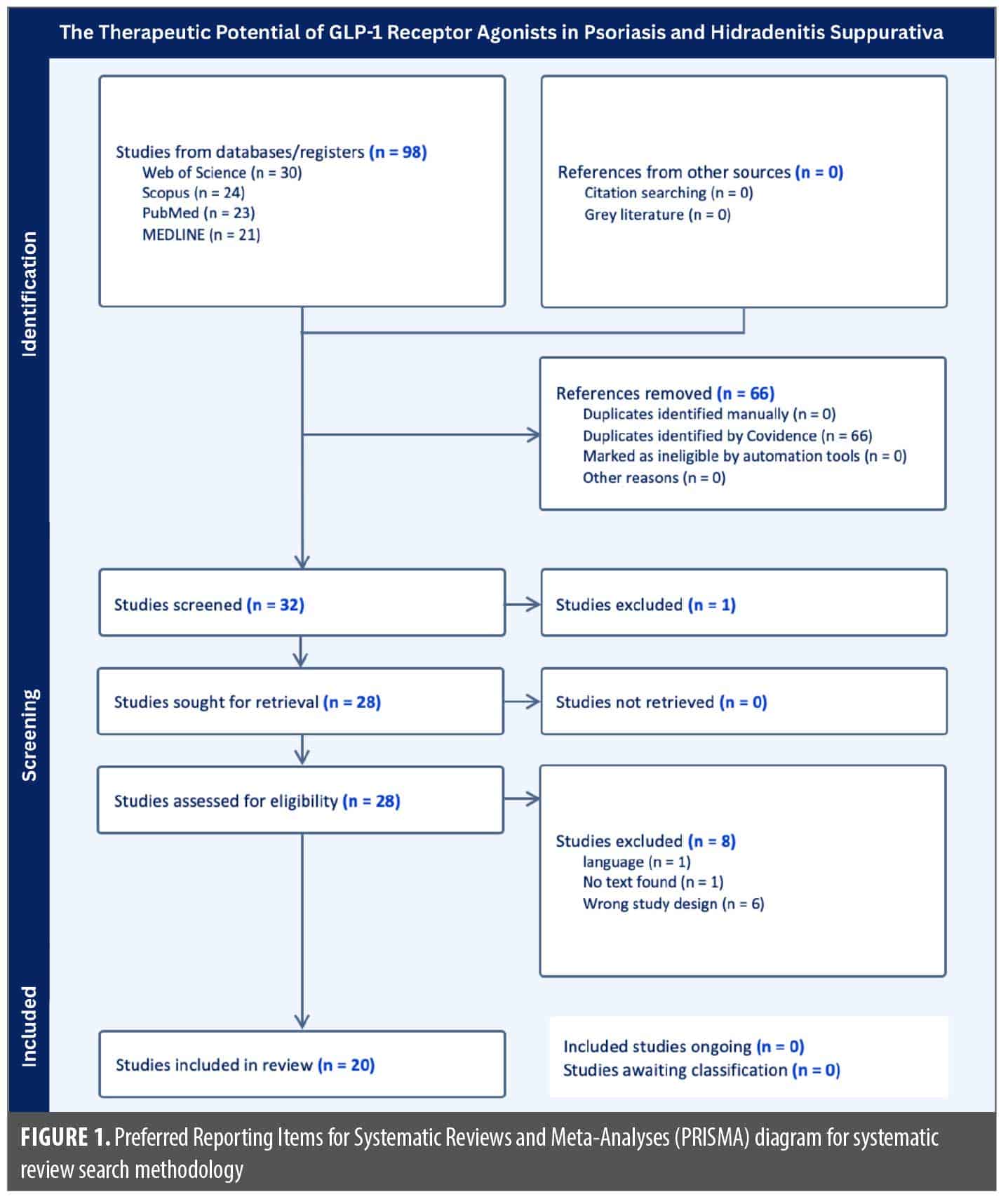
A total of 98 articles were identified across all four databases. After removal of 66 duplicates, 32 unique articles were screened. Following a full-text review, 20 studies met the inclusion criteria and were included in the final references. The PRISMA flow diagram (Figure 1) outlines the selection process.
GLP-1 RAs in Psoriasis
Mechanism of action. GLP-1 RAs improve psoriasis through immunologic effects and metabolic regulation. These agents reduce the amount of proinflammatory cytokines, such as interleukin (IL) 17A and tumor necrosis factor-α (TNF-α) that play a role in keratinocyte hyperproliferation and the inflammation that characterizes psoriatic plaques.2 Reductions have been seen in lesional skin and in bloodstream circulation as measured by gene expression analysis and enzyme-linked immunosorbent assay (ELISA).2 This supports a role in suppressing helper T-cell 17 (TH17) activity and disrupting the IL-23/IL-17 signaling pathway.
In addition to immunologic pathways, GLP-1 RAs also address the metabolic disturbances in patients with moderate-to-severe psoriasis. Many individuals with this disease meet the criteria for metabolic syndrome, especially insulin resistance and excess abdominal fat. Treatment with GLP-1 RAs has been associated with improved insulin sensitivity, as reflected by reductions in Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) scores, and these changes often align with improvements in psoriasis severity.1 For example, liraglutide treatment led to substantial PASI score reduction alongside improvements in both body mass index (BMI) and hemoglobin A1C (HbA1c).7 Semaglutide therapy reduced PASI from 12.0 at baseline to 4.0 at 4 months (−66.7%) and to 0.2 at 10 months (−98.3%), reflecting rapid improvement followed by near-complete clearance of lesions.7 Although metabolic changes were present, the rapid dermatologic response raised the possibility of additional mechanisms, such as direct modulation of cutaneous immune pathways.7 This presents the idea that GLP-1 RAs exert effects beyond weight loss or glycemic control, perhaps acting locally on immune cells or keratinocytes. Although the exact pathways remain unclear, there is growing interest in the role of cutaneous GLP-1 receptor expression.
These findings support the idea that GLP-1 RAs have a therapeutic effect in psoriasis. They can address metabolic dysfunction and may directly affect skin-specific immune pathways. Thus, they may lead to clinical improvement even in patients without significant shifts in metabolism. GLP-1 RAs are a promising treatment option for patients whose disease is characterized by a combination of metabolic and immune dysregulation.
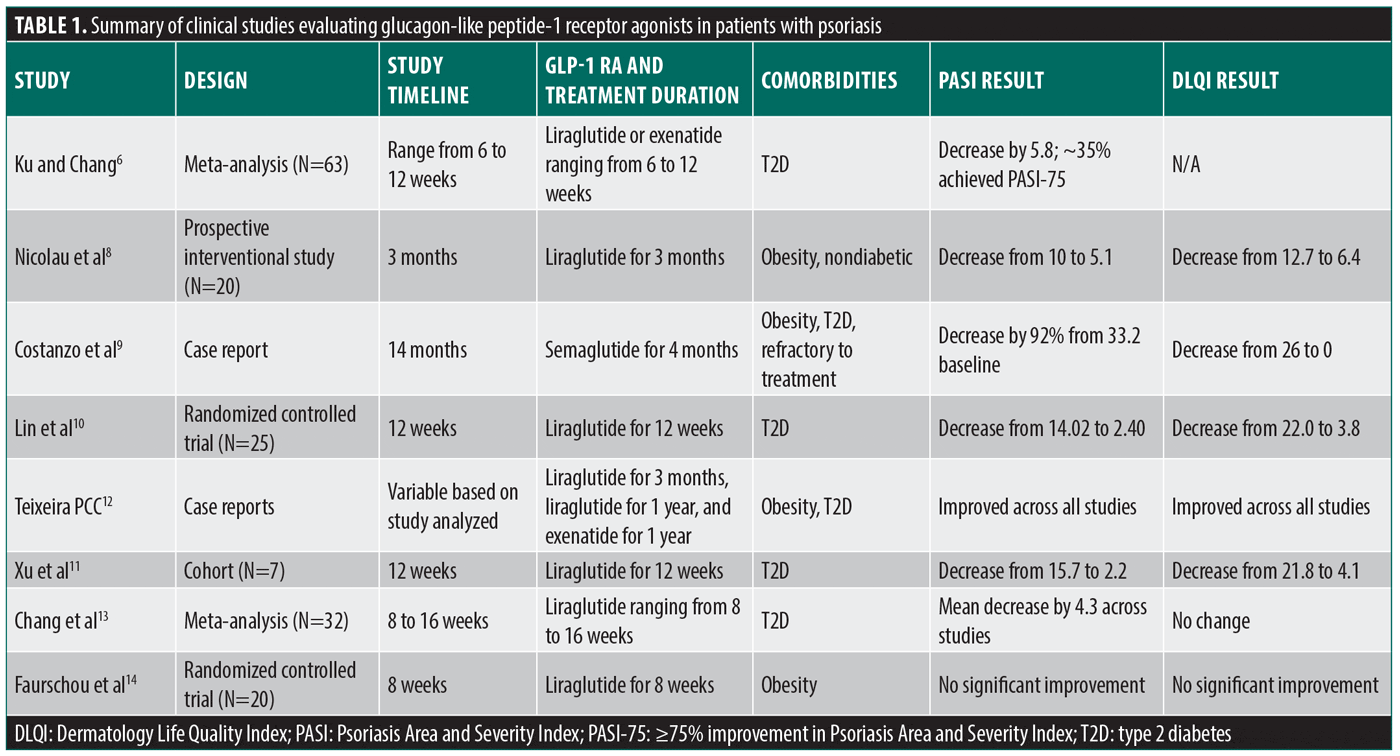
Evidence summary. GLP-1 RAs have been associated with significant clinical improvements in psoriasis severity, particularly in patients with T2D and/or obesity. Table 1 shows the results of GLP-1 RA use on psoriasis patients across randomized controlled trials, case reports, cohort studies, and meta-analyses. The results highlight that GLP-1 RA treatment was consistently linked to reductions in PASI scores and, in most cases, improvements in DLQI. Notably, Nicolau et al8 reported a significant PASI reduction from 10 to 5.1 and DLQI improvement from 12.7 to 6.4 in patients with obesity without diabetes treated with liraglutide. These findings were further supported by Nicolau et al5, who demonstrated similar clinical improvements in a broader cohort with extended follow-up, thereby reinforcing the potential use of GLP-1 RAs even in patients without metabolic comorbidities. Interestingly, in a case report by Costanzo et al9, a patient with severe psoriasis that did not respond to treatment experienced a 92% reduction in PASI and complete resolution of DLQI (back to normal values) following semaglutide therapy. Randomized data from Lin et al10 further confirmed significant improvement in both PASI and DLQI in patients with psoriasis and T2D receiving liraglutide compared to the control.
Beyond these clinical outcomes, other test results support a direct anti-inflammatory effect of GLP-1 RAs in psoriatic skin. Reductions in proinflammatory cytokines such as IL-17, IL-23, and TNF-α as detected by protein expression levels in immunohistochemistry, alongside histological improvements in epidermal thickness, support this anti-inflammatory mechanism.6,10,11 They further suggest that GLP-1 RAs may exert effects on psoriasis that are independent of metabolic changes. In fact, several analyses observed skin improvements preceding or occurring independently of weight loss or glycemic control.8,12,13 A meta-analysis by Ku and Chang6 reported PASI improvement in both diabetic and nondiabetic groups (over the 6- to 12-week periods of the 4 prospective cohort studies analyzed), with nearly 35% achieving a 75% or greater improvement in PASI (PASI-75) from their respective baselines, and a proposed immunomodulatory mechanism. Altogether, the literature suggests that GLP-1 RAs offer dermatologic benefits in tandem with their metabolic improvements. Thus, these medications deserve consideration as adjunctive therapies for psoriasis, especially in patients with metabolic comorbidities.
Limitations. Although GLP-1 RAs have garnered interest for the treatment of immune-mediated skin conditions such as psoriasis, there are notable limitations in the current literature. Many of the studies available are case reports or clinical trials with small sample sizes or short durations. For instance, a randomized placebo-controlled trial conducted by Faurschou et al14 examined the effect of liraglutide on psoriasis in glucose-tolerant patients. This study followed 20 patients over an 8-week treatment period. Nonetheless, this study demonstrated that liraglutide did not significantly change PASI and DLQI in these patients when compared with placebo. However, such results must be interpreted cautiously, as the small sample size and short study durations preclude comment on the long-term effects and safety of GLP-1 RAs in the treatment of psoriasis, ultimately limiting the ability to draw definitive, robust conclusions.
Another prospective cohort study by Nicolau et al8 explored the effects of liraglutide among patients with psoriasis and obesity. The study also included a sample size of 20, but it featured a treatment duration of 3 months. However, a major limitation of this study was the lack of a control group. Without a comparator, it is difficult to make definitive conclusions about causality and the effect of liraglutide on psoriasis outcomes, as confounding factors can influence observed results.
GLP-1 RA studies in psoriasis demonstrate consistent bias in patient selection, with the majority of studies targeting patients with comorbid T2D and/or obesity. For example, a study conducted by Teixeira PCC12 explored the role of GLP-1 RAs in psoriasis patients with obesity and T2D. As participants in this study had T2D, it is difficult to apply the findings to nondiabetic patient populations. Additionally, the bias in patient selection across the literature raises the probability that improvements in systemic metabolic function, rather than direct immunomodulation, may underlie the observed improvements in psoriatic skin lesions. These limitations underscore the need for large-scale randomized controlled trials with diverse patient populations and longer treatment durations to accurately assess the effects of GLP-1 RAs in the management of psoriasis.
GLP-1 RAs in HS
Mechanism of action. In recent years, GLP-1 RAs have emerged as potential therapeutic agents in HS. Their appeal lies in their dual mechanism of action: improving systemic metabolic parameters while simultaneously modulating immune responses. This integrated approach may offer benefits beyond traditional HS therapies, which often target either metabolic comorbidities or immune pathways in isolation.
One of the central mechanisms by which GLP-1 RAs may benefit patients with HS is through systemic metabolic modulation. Obesity is a well-recognized risk factor for HS, and excess adiposity contributes to mechanical friction, skin occlusion, and a proinflammatory state driven by adipokines and cytokines. GLP-1 RAs promote weight loss, enhancing satiety, reducing caloric intake, and delaying gastric emptying, which together reduce both the mechanical burden in intertriginous areas and systemic inflammation.3 These agents have also been shown to lower levels of circulating proinflammatory cytokines such as TNF-α and IL-6, both implicated in HS pathogenesis, as well as C-reactive protein (CRP), a marker of systemic inflammation that correlates with disease severity.4 By reducing the inflammatory load and metabolic stress that perpetuates HS lesions, GLP-1 RAs may interrupt the cycle of flare and progression that characterizes the disease.
Beyond systemic metabolic effects, GLP-1 RAs may also modulate immune responses relevant to HS, though the precise mechanisms remain to be elucidated. HS is increasingly recognized as a disease driven by systemic inflammation and metabolic dysfunction, with evidence supporting the use of treatments that address both pathways.3,4 These metabolic comorbidities, such as obesity, insulin resistance, and cardiovascular disease, exacerbate disease severity and complicate management. This dual burden emphasizes the need for therapeutic strategies that extend beyond localized skin treatments. In this context, GLP-1 RAs, which have demonstrated systemic metabolic benefits and potential immunological effects, represent promising adjuncts in the treatment of HS. While encouraging, these findings remain anecdotal and insufficient to establish a causal relationship between GLP-1 RA therapy and disease remission. Concerns about off-label prescribing and access, particularly with the rising demand for these medications in obesity and diabetes care, have also been raised.3
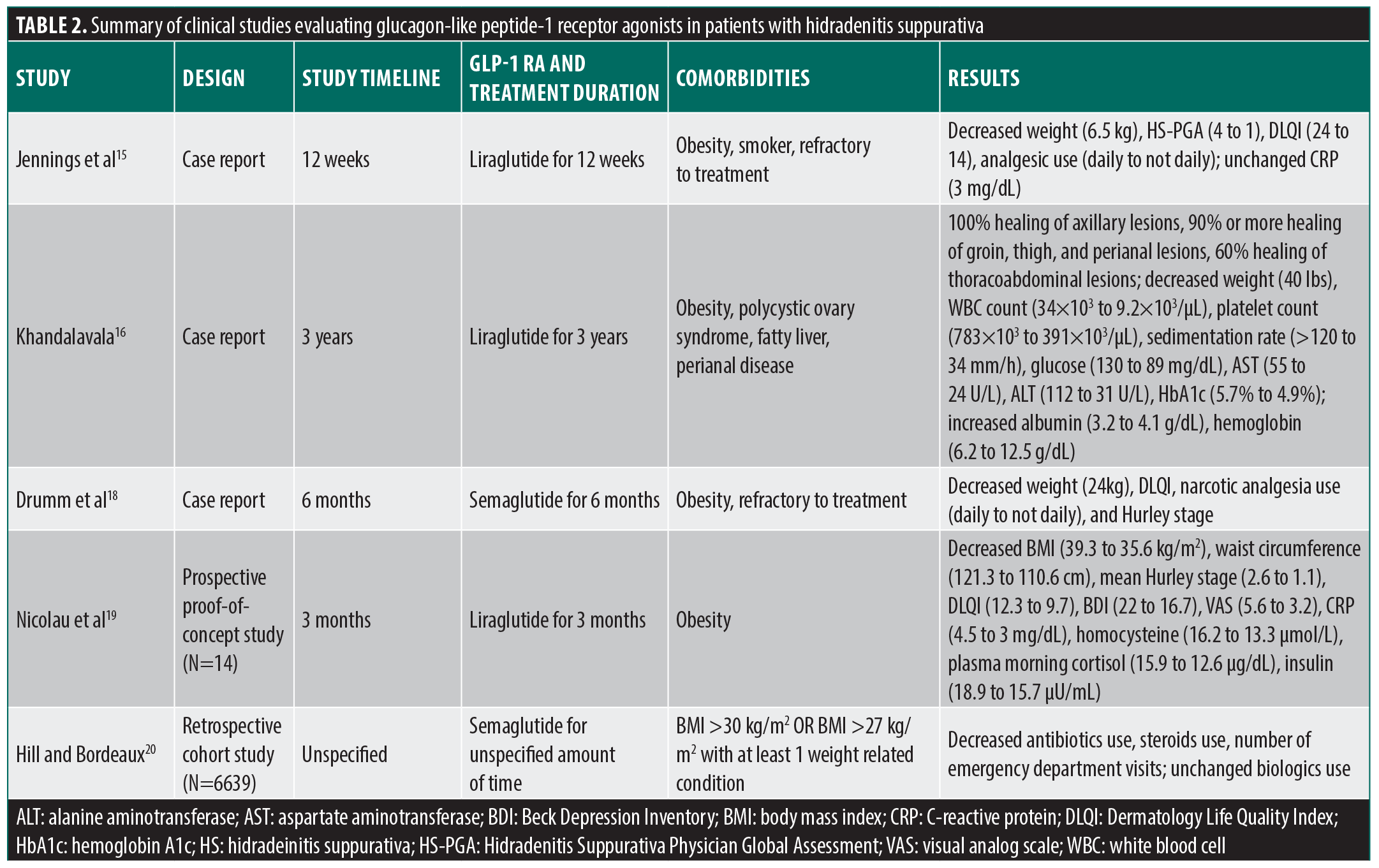
Evidence summary. GLP-1 RAs, particularly liraglutide and semaglutide, have been shown to have a generally positive effect in the treatment of HS. These medications are approved by the Food and Drug Administration (FDA) and are typically prescribed to patients with either a BMI greater than 30 kg/m2 (obese) or a BMI greater than 27 kg/m2 with a weight-related comorbidity as adjuncts to lifestyle modifications.3 Table 2 highlights the results of the use of GLP-1 RAs in treating HS from multiple case reports, a prospective proof-of-concept study, and a retrospective cohort study. The overall results show that GLP-1 RAs were successful in decreasing BMI, lesion severity and presence, and inflammatory markers while also increasing quality of life. Both case reports regarding the first uses of liraglutide in treating HS revealed a significant decrease in lesion severity and presence.15,16 Jennings et al15 demonstrated a significant decrease in weight (6.5 kg), HS Physician’s Global Assessment (HS-PGA) (from 4 to 1), DLQI (from 24 to 14), and analgesic use (from daily to not daily) but no decrease in CRP (from 3 mg/dL) after eight weeks of treatment. Khandalavala16 showed 100% healing of axillary lesions, 90% or more healing of groin, thigh, and perianal lesions, and 60% healing of thoracoabdominal lesions. There was also a decrease in weight (40 lbs), white blood cell (WBC) count (from 34×103 to 9.2×103/µL), platelet count (from 783×103 to 391×103/µL), sedimentation rate (from >120 to 34 mm/h), glucose (from 130 to 89 mg/dL), aspartate aminotransferase (AST) (from 55 to 24 U/L), alanine aminotransferase (ALT) (from 112 to 31 U/L), and HbA1c (from 5.7% to 4.9%), as well as an increase in albumin (from 3.2 to 4.1 g/dL) and hemoglobin (from 6.2 to 12.5 g/dL) after three years of treatment.15-17 The first case report documenting the successful use of semaglutide in treating HS by Drumm et al18 also showed a reduction in weight (24 kg), DLQI, narcotic analgesia use (from daily to not daily), and Hurley stage after six months of treatment. A prospective proof-of-concept study by Nicolau et al19 found that liraglutide treatment for three months resulted in a decrease in BMI (from 39.3 to 35.6 kg/m2), waist circumference (from 121.3 to 110.6 cm), mean Hurley stage (from 2.6 to 1.1), DLQI (from 12.3 to 9.7), Beck Depression Inventory (BDI) (from 22 to 16.7), and visual analog scale (VAS) for pain (from 5.6 to 3.2), as well as a decrease in inflammatory markers such as CRP (from 4.5 to 3 mg/dL), homocysteine (from 16.2 to 13.3 µmol/L), plasma morning cortisol (from 15.9 to 12.6 µg/dL), and insulin (from 18.9 to 15.7 µU/mL). Lastly, a retrospective cohort study by Hill and Bordeaux20 showed reduced HS resource utilization (HSRU), including decreased use of antibiotics and corticosteroids and the number of emergency department visits, but no change in the use of biologics after semaglutide treatment for an unspecified amount of time. Therefore, the literature suggests that GLP-1 RAs are effective in managing associated obesity, lesions, and inflammation in patients with HS, resulting in an improved quality of life.
Limitations. GLP-1 RAs have shown promising effects in the treatment of HS, but limitations exist in the current literature. Data were primarily collected through a small number of case reports, conference abstracts, and retrospective analyses. Furthermore, there is a lack of randomized controlled trials, standardized outcome measures, and long-term follow-up reports. For example, a literature review performed in 2024 found only two case reports of GLP-1 RAs for treating HS that met the inclusion criteria.17 The first case report by Jennings et al15 found that in a 31-year-old female patient with HS, treatment with liraglutide for eight weeks resulted in reductions in BMI, HS-PGA, DLQI, and analgesic use. However, there was no mention of follow-up results for this patient after the initial treatment period. A different case report of a 19-year-old female patient with HS by Khandalavala16 found that liraglutide in combination with metformin, levonorgestrel-ethinyl estradiol, dapsone, and finasteride for three years resulted in 60% to 100% improvement in lesions after a three-month delay in symptom improvement as well as a normalization of hematologic, metabolic, and liver values after a three year-long regimen. Follow-ups on a monthly basis were conducted for two years after treatment to confirm the regimen’s effectiveness, and no new lesions were reported for at least six months after the initial three-year treatment regimen.
Both patients in these two case reports were female, prompting the question of whether the same results would have been found in male patients. Moreover, using liraglutide alone vs. with other medications, different treatment durations, and different patient outcome measures were used, making it difficult to assess whether the GLP-1 RA had similar effects in both patients. Without randomized, long-term studies on the use of GLP-1 RAs in a large sample size of HS patients with a wide range of ages, comorbidities, risk factors, and sexes and without any confounding factors, it is difficult to conclusively determine whether GLP-1 RAs are a definitive treatment for HS.
There is also a lack of definitive literature regarding the adverse effects and contraindications of GLP-1 RAs for the treatment of HS. Adverse effects such as tachycardia, infections, headaches, dizziness, nausea, vomiting, diarrhea, and dyspepsia, as well as contraindications such as a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2, hypersensitivity, severe gastrointestinal diseases, renal impairment, and pregnancy have been noted. However, these recommendations lack sufficient evaluation of the risk-benefit profile in HS-specific populations, thereby justifying further safety studies.3 Additional studies are needed to assess the benefits and potential risks of using GLP-1 RAs in HS treatment in patients with and without comorbidities to determine whether GLP-1 RAs should be included in a standardized regimen.
Discussion
GLP-1 RAs have demonstrated immuno-modulatory effects, presenting a promising therapeutic potential in psoriasis and HS.2 However, the majority of publications presented these results as a secondary outcome in studies targeting the GLP-1 RA effect on patients with diabetes and patients with obesity. Although most current studies are confounded by metabolic comorbidities, recent findings by Nicolau et al5 begin to address this gap by evaluating the effects of GLP-1 RAs in metabolically healthy patients, providing preliminary support for a direct immunologic mechanism.5 Such trials will be essential to clarify the primary immunologic effects of GLP-1 RAs, offering clearer insight into their therapeutic potential beyond glycemic control.
This limitation reflects a broader gap in the literature. Haran et al2 underscore that the observed improvements in inflammatory skin disease, particularly psoriasis, often coincide with systemic metabolic changes, which confound direct attribution to immune effects.2 Similarly, Krajewski et al4 report reductions in HS lesion counts and inflammatory markers such as CRP but emphasize that these are closely linked to metabolic improvements, including weight loss and enhanced insulin sensitivity. This interplay complicates the interpretation of GLP-1 RA efficacy and underscores the need for dedicated dermatology-focused trials.
Mechanistic studies provide some evidence supporting the immunomodulatory capacity of GLP-1 RAs, showing reductions in key proinflammatory cytokines such as IL-17 and dermal T-cell infiltration in psoriatic lesions.2 However, the lack of randomized controlled trials specifically designed to evaluate dermatologic outcomes warrants caution in extrapolating these findings to clinical benefit. The heterogeneity of study designs, from single case reports to retrospective database analyses, further challenges causal inference. Moreover, concurrent use of other treatments and inconsistent use of validated clinical severity measures compromise cross-study comparisons and generalizability.
Safety considerations also warrant attention. Strong and Driscoll3 highlight that while GLP-1 RAs are generally tolerated, gastrointestinal adverse effects such as nausea and diarrhea are frequent and may necessitate dosage adjustments. Though concerns about pancreatitis and thyroid neoplasms have not been substantiated in large human studies, dermatology-specific safety guidelines remain absent. This gap is particularly relevant given the common use of systemic immunomodulators in dermatology patients, creating challenges for clinicians considering GLP-1 RAs in polypharmacy contexts.
From a broader immunologic perspective, Karacabeyli and Lacaille1 note that GLP-1 RAs may influence shared inflammatory pathways involved in immune-mediated inflammatory diseases (IMIDs), including psoriasis and inflammatory arthritis. Their review, however, identified no studies stratifying the potential role of GLP-1 RAs across overlapping dermatologic and rheumatologic conditions, suggesting that future multidisciplinary trials should incorporate these factors.
Overall, GLP-1 RAs appear promising as adjunctive therapies for HS and psoriasis, especially in patients with coexisting metabolic disease. Yet, to delineate their immunomodulatory potential distinct from metabolic effects, rigorous, randomized controlled trials in metabolically healthy dermatologic populations are essential. Only through such focused research can GLP-1 RAs be accurately positioned within evidence-based treatment frameworks for inflammatory skin diseases.
Conclusion
Although GLP-1 RAs are indicated for T2D and obesity, they have demonstrated promising effects in patients with psoriasis and HS. The reviewed literature consistently shows reductions in lesion severity, improvements in quality of life, and decreases in systemic inflammatory markers following treatment with agents such as liraglutide and semaglutide. In some instances, skin symptom improvements occurred even in the absence of significant weight loss or improved glycemic control. This suggests that GLP-1 RAs may also act through direct immunomodulatory mechanisms.
Despite these findings, evidence is limited. Most studies involve small sample sizes or include patients receiving concurrent treatments. Additionally, much of the research has focused on individuals with metabolic comorbidities, which leaves unanswered questions about the effectiveness of GLP-1 RAs in different populations. Although GLP-1 RAs are not currently approved for dermatologic indications, their combined metabolic and immunologic effects support their potential use in inflammatory skin diseases. Emerging evidence further supports the potential role of GLP-1 RAs in improving dermatologic outcomes independently of metabolic improvements, suggesting a dual therapeutic mechanism.5 These agents may offer therapeutic value for select patients with psoriasis or HS, especially among those whose disease burden is influenced by metabolic factors.
References
- Karacabeyli D, Lacaille D. Glucagon-like peptide 1 receptor agonists in patients with inflammatory arthritis or psoriasis: a scoping review. J Clin Rheumatol. 2024;30(1):26-31.
- Haran K, Johnson CE, Smith P, et al. Impact of GLP-1 receptor agonists on psoriasis and cardiovascular comorbidities: a narrative review. Psoriasis (Auckl). 2024;14:143-152.
- Strong J, Driscoll MS. Obesity in hidradenitis suppurativa: are GLP-1 receptor agonists the new rontier? Am J Clin Dermatol. 2025;26(2):175-182.
- Krajewski PK, Złotowska A, Szepietowski JC. The therapeutic potential of GLP-1 receptor agonists in the management of hidradenitis suppurativa: a systematic review of anti inflammatory and metabolic effects. J Clin Med. 2024;13(21):6292.
- Nicolau J, Nadal A, Sanchís P, et al. Effects of six months treatment with liraglutide among patients with psoriasis and obesity, beyond metabolic control? Med Clin (Barc). 2025;164(11):106941.
- Ku SC, Chang HC. Efficacy of glucagon-like peptide-1 receptor agonists for psoriasis: an updated systematic review and meta-analysis. J Dtsch Dermatol Ges. 2024;22(8):1148-1152.
- Malavazos AE, Meregalli C, Sorrentino F, et al. Semaglutide therapy decreases epicardial fat inflammation and improves psoriasis severity in patients affected by abdominal obesity and type-2 diabetes. Endocrinol Diabetes Metab Case Rep. 2023;2023(3):23-0017.
- Nicolau J, Nadal A, Sanchís P, Pujol A, Nadal C, Masmiquel L. Effects of liraglutide among patients living with psoriasis and obesity. Med Clin (Barc). 2023;161(7):293-296.
- Costanzo G, Curatolo S, Busà B, Belfiore A, Gullo D. Two birds one stone: semaglutide is highly effective against severe psoriasis in a type 2 diabetic patient. Endocrinol Diabetes Metab Case Rep. 2021;2021:21-0007.
- Lin L, Xu X, Yu Y, et al. Glucagon-like peptide-1 receptor agonist liraglutide therapy for psoriasis patients with type 2 diabetes: a randomized-controlled trial. J Dermatolog Treat. 2022;33(3):1428-1434.
- Xu X, Lin L, Chen P, et al. Treatment with liraglutide, a glucagon-like peptide-1 analogue, improves effectively the skin lesions of psoriasis patients with type 2 diabetes: a prospective cohort study. Diabetes Res Clin Pract. 2019;150:167-173.
- Teixeira PCC. Psoriasis patients with obesity and type 2 diabetes: the effect of glucagon-like peptide 1 (GLP-1) receptor agonists. Order No. 29150355 ed. Universidade do Porto (Portugal); 2015.
- Chang G, Chen B, Zhang L. Efficacy of GLP-1rA, liraglutide, in plaque psoriasis treatment with type 2 diabetes: a systematic review and meta-analysis of prospective cohort and before-after studies. J Dermatolog Treat. 2022;33(3):1299-1305.
- Faurschou A, Gyldenløve M, Rohde U, et al. Lack of effect of the glucagon-like peptide-1 receptor agonist liraglutide on psoriasis in glucose-tolerant patients–a randomized placebo-controlled trial. J Eur Acad Dermatol Venereol. 2015;29(3):555-559.
- Jennings L, Nestor L, Molloy O, Hughes R, Moriarty B, Kirby B. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177(3):858-859.
- Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9(2):70-78.
- Parvathaneni A, McQuitty EN, Slawin B, et al. Exploring the therapeutic potential of GLP-1 agonists in hidradenitis suppurativa. J Drugs Dermatol. 2025;24(4):422-423.
- Drumm C, Clowry J, Healy ML, Wynne B. Successful treatment of hidradenitis suppurativa with semaglutide. J Am Acad Dermatol. 2023;89(3):AB89.
- Nicolau J, Nadal A, Sanchís P, Pujol A, Masmiquel L, Nadal C. Liraglutide for the treatment of obesity among patients with hidradenitis suppurativa. Med Clin (Barc). 2024;162(3):118 122.
- Hill MA, Bordeaux JS. Semaglutide use for decreasing hidradenitis suppurativa resource utilization: a retrospective cohort study utilizing TriNetX. J Am Acad Dermatol. 2025;92(4):896-897.