J Clin Aesthet Dermatol. 2026;19(3):32–37.
by Zoe Diana Draelos, MD, FAAD; Jacqueline Watchmaker, MD, FAAD; and Diane B. Nelson, RN, MPH
Dr. Draelos is with Dermatology Consulting Service, PLLC in High Point, North Carolina. Dr. Watchmaker is with Southwest Skin Specialists in Scottsdale, Arizona. Ms. Nelson is a former employee of Skinbetter Science, a Dermatological Beauty Brand of L’Oreal USA, Inc.
FUNDING: No funding was provided for this article.
DISCLOSURES: Drs. Draelos and Watchmaker were study investigators, and Ms. Nelson was an employee of skinbetter science, a Dermatological Beauty Brand of L’Oréal USA, Inc. This study was sponsored by skinbetter science, a Dermatological Beauty Brand of L’Oréal USA, Inc.
ABSTRACT: Background: Persistent facial erythema is one of the most common subtypes of rosacea, often prompting patients to seek treatment. Characterized by multiple factors including adaptive immune and vascular dysregulation, genetics, and environmental influences, skincare measures aimed at reducing erythema and strengthening the skin barrier are fundamental to treatment. Plant adaptogens are rich phytochemicals that promote homeostasis, helping to increase resistance to stress, mitigate inflammation, support the skin barrier, and prevent premature aging. A serum containing plant adaptogens demonstrated significant improvements from baseline in global skin quality in subjects with mild-to-severe facial photodamage over 12 weeks. Objective: We sought to evaluate the safety and efficacy of a serum containing plant adaptogens (MYS) in subjects with mild-to-moderate persistent centrofacial erythema. Methods: A multicenter, open-label trial enrolled male or female subjects 30 to 65 years of age with mild (2) to moderate (3) persistent centrofacial erythema based on a Modified Clinical Erythema grading scale (M-CEA; 0=clear to 4=severe). A subset of subjects enrolled was taking oral or topical medications for rosacea (≥6 months) with breakthrough erythema. Subjects with >5 papules or pustules were excluded from participation. The investigators assessed erythema based on the M-CEA scale and global inflammatory lesions utilizing a 5-point grading scale (0=none to 4=severe) at baseline, 2, 4, 8, and 12 weeks. Subjects assessed erythema, dryness/flaking, itching, stinging, and burning using a 5-point grading scale (0=none to 4=severe) at baseline, 2, 4, 8, and 12 weeks. Subjects also completed self-assessment questionnaires. Digital photography and adverse events (AEs) were captured throughout the study. In addition to the study product (MYS [AM/PM]), subjects used a standardized skincare regimen that included a gentle cleanser, moisturizer, and sunscreen. Results: Fifty-five subjects were enrolled and 51 subjects completed the study: 38 in the Non-Medication Treatment Group (N-MTG) and 13 in the Medication Treatment Group (MTG). Significant mean reductions in the appearance of erythema of –17%, –25%, –32%, and –33% were demonstrated from baseline at Weeks 2, 4, 8, and 12 (all P<0.001), respectively, with each group demonstrating significant reductions over 12 weeks. Subject-assessed erythema demonstrated comparable results for both groups combined, with –22%, –30%, –34%, and –35% significant mean reductions occurring from baseline at Weeks 2, 4, 8, and 12 (all P<0.001). Significant mean reductions also occurred in papules/pustules at Weeks 4 (–29%), 8 (–43%), and 12 (–58%) (n=34; P<0.05). Subjects reported improvements in dryness/flaking, itching, stinging, and burning over the study period. After 12 weeks, subjects (94%) reported that flare-ups were not as intense and their skin looked less blotchy and looked healthier. Subjects (88%) reported feeling less self-conscious about their skin’s appearance. One mild, possibly related hypersensitivity reaction on the neck was reported and resolved without sequela. No subject discontinued the study due to an AE. Conclusion: Twice-daily use of a serum comprised of plant adaptogens in subjects with mild-to-moderate persistent centrofacial erythema demonstrated significant reductions in the appearance of erythema from baseline as assessed by investigators and subjects over 12 weeks. Significant reductions from baseline also occurred in papules/pustules. Subjects reported decreases in skin sensitivity and healthier-looking skin. Keywords: Plant adaptogens, rosacea, erythema, homeostasis, papules/pustules
Introduction
Rosacea is a chronic, inflammatory facial dermatosis that affects approximately 5% of individuals worldwide.1 While the etiology remains unknown, it is believed that the clinical manifestations relate to an underlying multivariate disease process that may involve adaptive immune and vascular dysregulation, genetics, and environmental influences.1–5 Persistent facial erythema, with or without papules and pustules, is one of the most common subtypes of rosacea and often prompts patients to seek treatment owing to its substantial burden to quality of life.6–9 However, survey data indicate that few patients use the available treatments for rosacea or are suboptimally satisfied with available pharmacotherapies.10,11
The 2019 update by the National Rosacea Society (NRS) Expert Committee of the American Academy of Dermatology (AAD) identified both topical and oral pharmacotherapies for the management of specific rosacea symptoms, along with intense pulsed light therapy or laser therapy for more persistent and severe cases of erythema.5 In addition to pharmacotherapies, the AAD emphasized that patient education and effective skincare, including a gentle cleanser, moisturizer, and broad-spectrum sunscreen (SPF ≥30) that affords broad protection against UV-A, UV-B, infrared radiation (IR), and blue light, are essential components of the overall treatment strategy.5 Similarly, the most recent Global ROSacea Consensus (ROSCO) Committee guidelines emphasized the importance of using sunscreen, gentle cleansers, and skincare products that can repair and maintain the stratum corneum barrier function, enhance skin hydration, and minimize the likelihood of skin irritation.12 As such, skincare measures aimed at reducing erythema and strengthening and preserving the skin barrier, while avoiding fragrances and harsh exfoliation, are foundational to the treatment of rosacea.13,14
Plant-derived adaptogens are rich phytochemicals that promote homeostasis, helping to increase resistance to stress, mitigate inflammation, support the skin barrier, and prevent premature aging.13 They have multifunctional properties and work on multiple pathways and molecular targets in the skin, possessing an innate ability to defend against stressors.15–18 Plant adaptogens have developed the ability, via adaptation and evolution, to defend and protect themselves against environmental insults and other stressors.15,16,18–26 They are increasingly used in aesthetic skincare products to target aging skin, along with products used in the management of dermatological conditions such as psoriasis, atopic dermatitis, and acne.16 Combinations of certain adaptogens may provide synergistic benefits that further increase resistance to stress, minimize inflammation, and address manifestations of premature aging.17 Toward this end, a novel serum (MYS) was purposely developed using Plant Adaptogens Targeting Homeostasis (P.A.T.H.) technology to support the natural ability of skin to adapt and achieve homeostasis and visibly improve skin quality (Table 1).
This research evaluates the safety and efficacy of MYS serum in subjects with mild-to-moderate persistent centrofacial erythema associated with rosacea.
Methods
This multicenter trial was conducted under institutional review board approval in conjunction with current Good Clinical Practice (cGCP) guidelines. Two board-certified dermatologists recruited and enrolled study subjects across 2 clinical research sites. All study subjects provided written informed consent prior to study participation.
The study enrolled male or female subjects, aged 30 to 65 years, with mild (2) to moderate (3) persistent centrofacial erythema associated with rosacea based on a Modified Clinical Erythema Assessment scale (M-CEA; 0=clear, no erythema to 4=severe, marked erythema) and a history of flares, facial flushing, or blushing. A subset of enrolled subjects was taking oral or topical medications for rosacea (≥6 months) with breakthrough erythema (Medication Treatment Group [MTG]). At least 50% of enrolled subjects reported having sensitive skin.
Subjects were deemed eligible for inclusion in the Non-Medication Treatment Group (N-MTG) following a 2-week washout period of any topical, nonprescription acne or rosacea treatments and following a 4-week washout period of prescription or nonprescription retinoids or retinol products or of prescription topical or systemic corticosteroids. Subjects who were pregnant, breastfeeding or planning a pregnancy; had recently started hormonal replacement therapy (HRT) or hormonal contraceptives (HT) within the previous 3 months; or planned to start, stop, or change doses of HT during the course of the study were excluded. Subjects who had used oral isotretinoin or energy-based devices on their face within the past 6 months were excluded from study participation. Subjects with severe persistent facial erythema, severe quantity of telangiectasias, >5 inflammatory facial papules/pustules, or phymatous or ocular rosacea were also excluded from study participation. Subjects were instructed to apply the study product (MYS) to their face every morning and evening for 12 weeks after gently cleansing their face with the supplied cleanser. Additional products included a moisturizer (morning and evening application) and sunscreen (morning application).
Investigator and subject assessments occurred at baseline and at 2, 4, 8, and 12 weeks. Investigators assessed facial erythema utilizing the 5-point M-CEA grading scale (0=clear, no erythema to 4=severe, marked erythema); global inflammatory lesions utilizing a 5-point grading scale (0=no papules/pustules to 4=severe, numerous small and/or large papules/pustules); and tolerability (dryness/flaking) utilizing a 5-point grading scale (0=none to 4=severe). Subjects assessed facial redness utilizing a 5-point grading scale (0=no redness to 4=marked redness), and tolerability (dryness/flaking, itching, stinging, and burning) utilizing a 5-point grading scale (0=none to 4=severe, marked amount). In addition, subjects completed self-assessment questionnaires at 2, 4, 8, and 12 weeks. Adverse events (AEs) were captured throughout the study.
Results
Demographics. Fifty-five subjects were enrolled, and 51 subjects completed the study (N-MTG, n=38; MTG, n=13). Three subjects discontinued the study, and 1 subject was lost to follow-up. The majority of subjects were female (91%), White (92%) and non-Hispanic (92%), with an overall mean age of 52 years. The majority of enrolled subjects had Fitzpatrick skin type 2 (62%), and 77% of subjects reported having sensitive skin.
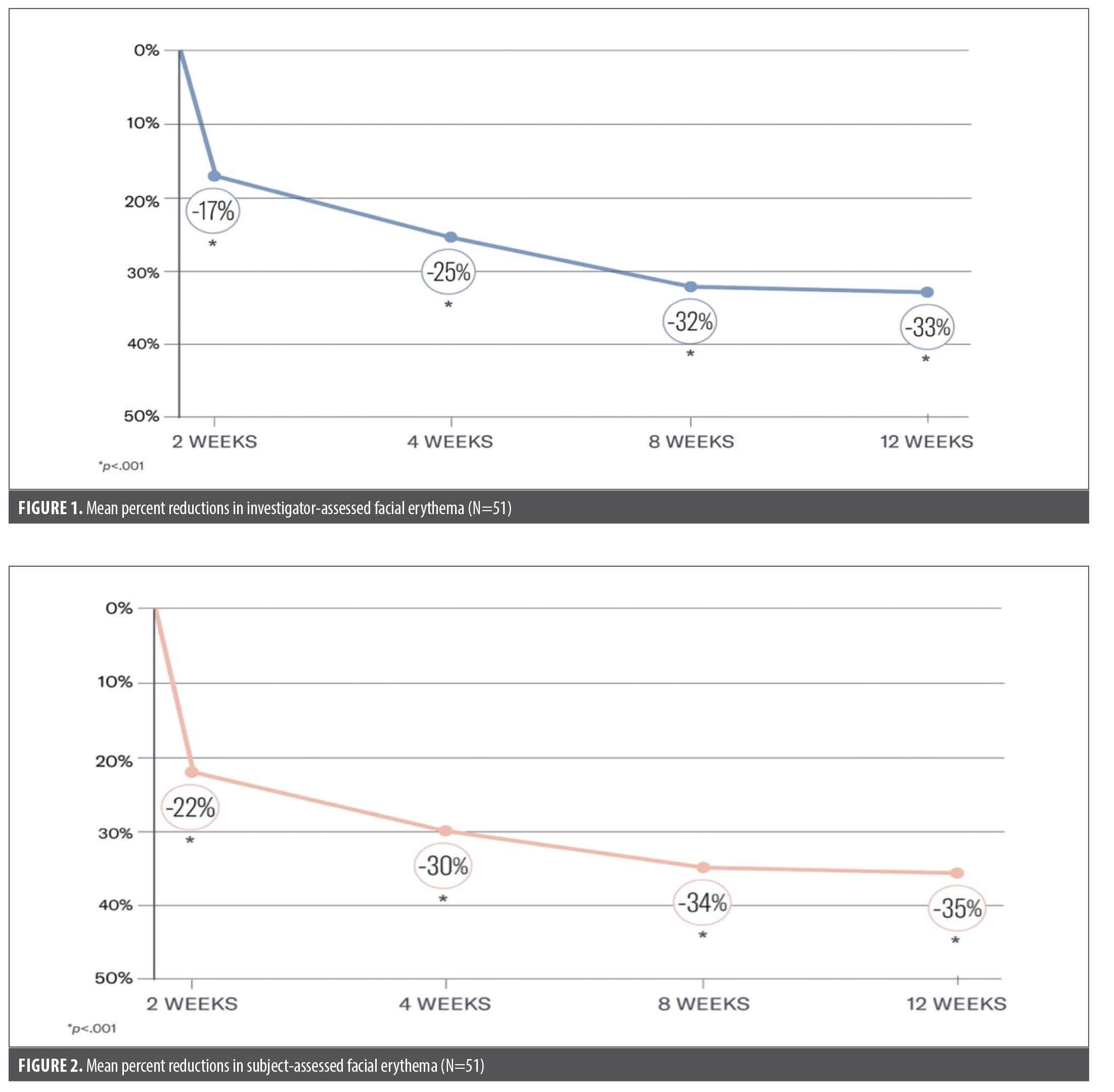
Investigator assessments. Significant mean progressive reductions were demonstrated from baseline in the appearance of facial erythema of –17%, –25%, –32%, and –33% at 2, 4, 8, and 12 weeks, respectively (all P<0.001) in both groups combined (Figure 1). Each group demonstrated significant reductions at 12 weeks, with –34% reduction in the N-MTG (P<0.001) and –29% reduction in the MTG (P<0.016).
In the 34 patients who presented with papules/pustules at baseline, significant mean reductions occurred at 4 (–29%; P<0.01), 8 (–43%; P<0.001), and 12 weeks (–58%; P≤0.001). Subjects in the MTG demonstrated a –87% reduction (P=0.004) in papules/pustules from baseline, with a –46% reduction (P=0.004) in the N-MTG.
No dryness/flaking was observed by the investigators. One mild, possibly related hypersensitivity reaction on the neck was reported and resolved without sequelae, and no subject discontinued the study due to an AE.
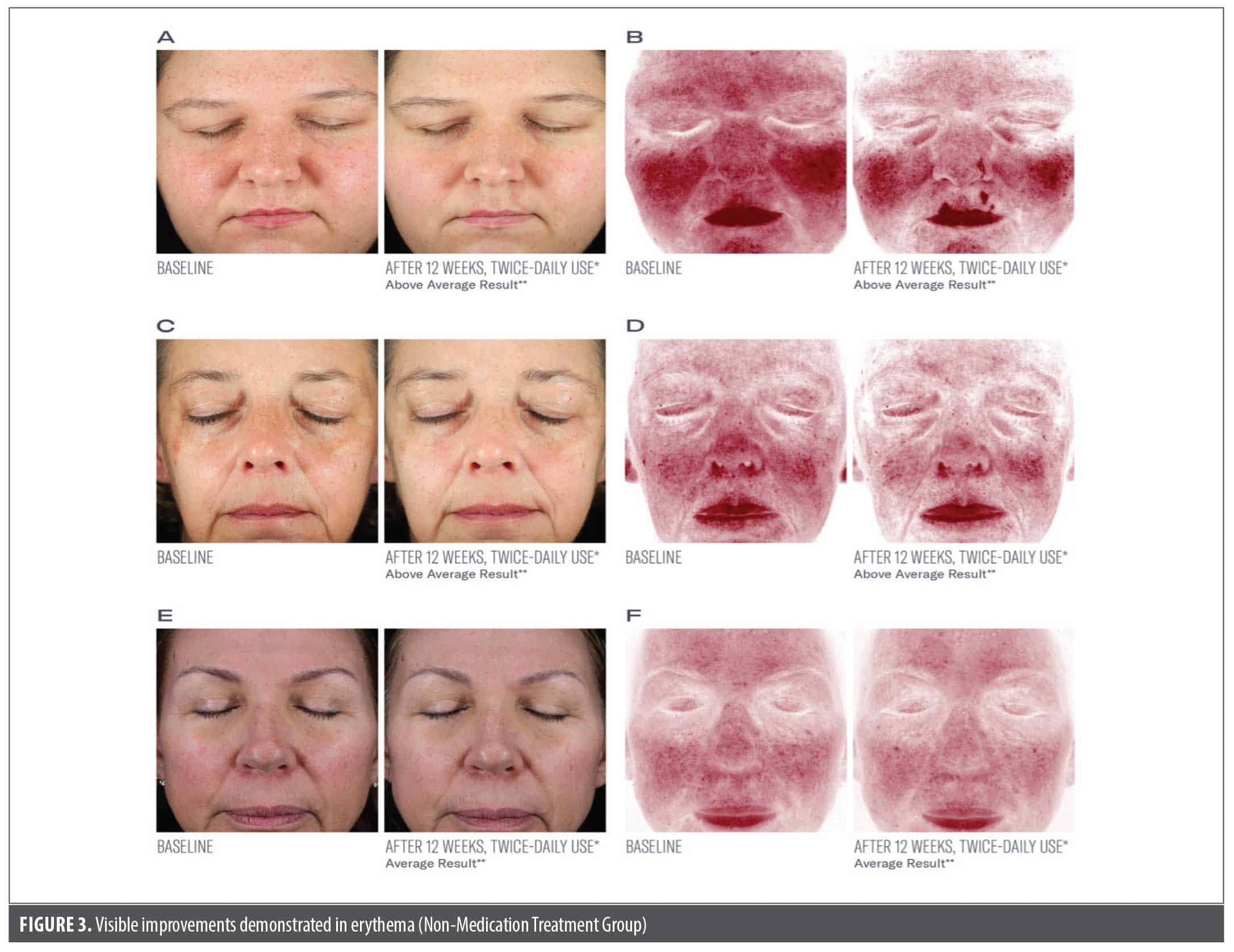
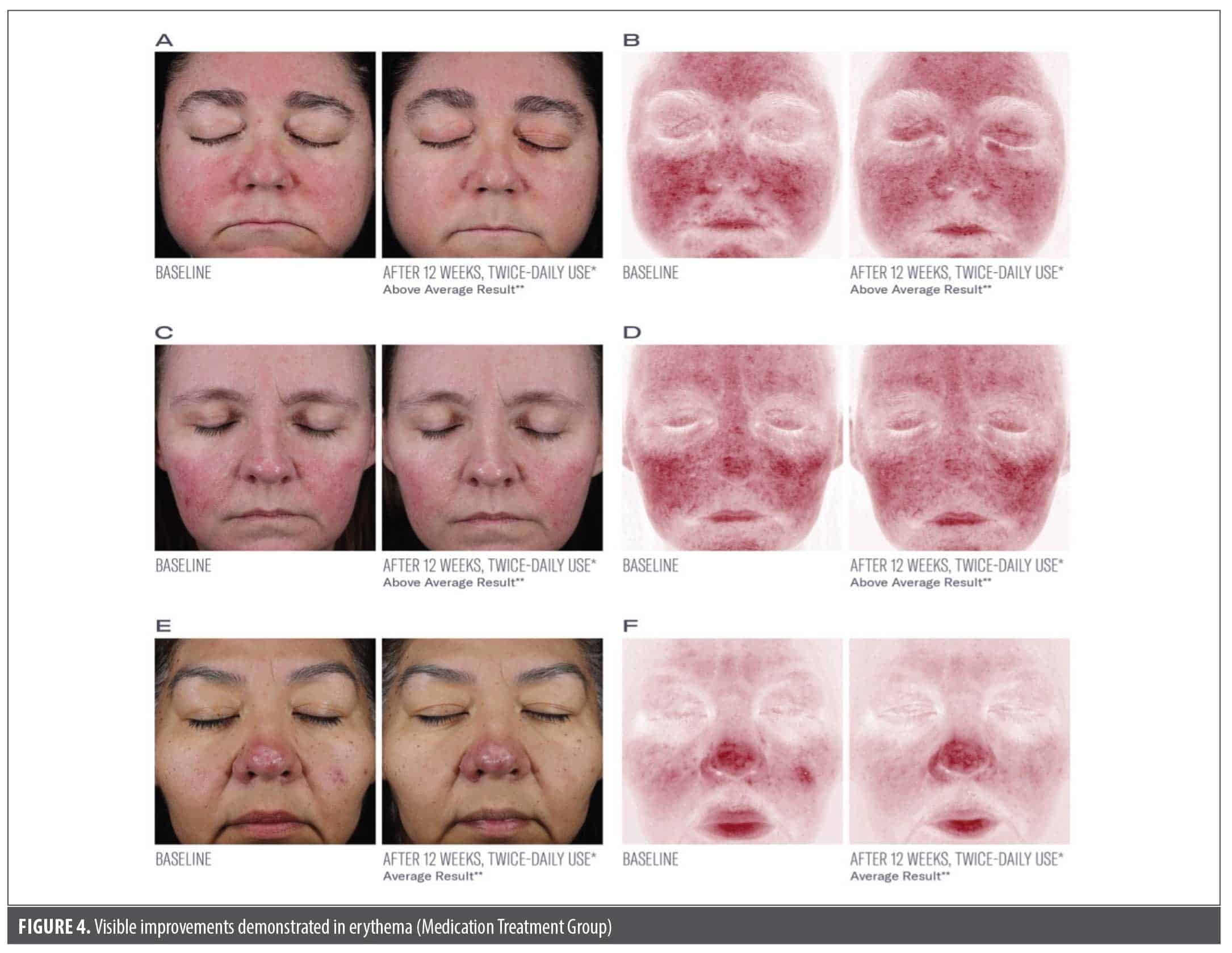
Subject assessments. Significant mean reductions were demonstrated in subject-assessed erythema of –22%, –30%, –34%, and –35% from baseline at 2, 4, 8, and 12 weeks, respectively (all P<0.001; Figure 2). Results were similar between the groups (Figure 3, Figure 4).
Subjects reported high tolerability, with significant reductions reported in dryness/flaking (–38%; P=0.001), itching (–52%; P=0.033), and stinging (–63%; P=0.038) at 12 weeks. A –28% reduction in burning was also reported at 12 weeks (non-significant).
After 12 weeks, 100% of participants reported that their skin burned and stung less. Patients reported that their skin felt softer, calmer, and more comfortable (98%) and that it looked healthier and less blotchy, with less intense flare-ups (94%). Patients (93%) also noted that the small, red blood vessels around their nose and/or cheeks were less visible. With regard to quality of life, 88% of subjects reported feeling less self-conscious about their skin’s appearance at the conclusion of the study.
Discussion
The treatment of persistent centrofacial erythema associated with rosacea is a complex, challenging condition to manage and may require a multimodal approach, including prescription and nonprescription modalities. Foundational to treatment is the incorporation of safe, tolerable, and effective skincare products. The innate ability of skin to adapt to internal and external stresses and to achieve and maintain homeostasis is compromised in patients with rosacea and also decreases with age and continued exposures to the stressors. The multifunctional properties of plant adaptogens and their ability to support multiple pathways and molecular targets in the skin enhance homeostasis, increase resistance to stress, mitigate inflammation, and reinforce the skin barrier.23 Plant adaptogens are able to activate the extracellular matrix, complement system, and intracellular adaptive stress response signaling pathways and to inhibit oxidative stress pathways.16,27
A previous in vitro study conducted analyzing gene expression in a human epidermal skin model reported increased expression of aryl hydrocarbon receptor, thioredoxin reductase 1, and metallothionein (MT)-1A, MT1F, and MT1G and reduced expression of chloride channel accessory 2 after 48-hours of topical exposure to MYS serum.28 These genes play a role in supporting skin homeostasis, and the skin’s adaptive response to stress. These effects also translated into clinical improvements in global skin quality, notably including improvements in erythema as well as skin dullness and texture, as demonstrated in an in vivo study.15
In light of these prior in vivo and in vitro findings,15,28 we hypothesized that MYS serum would be beneficial for adults with persistent centrofacial erythema associated with rosacea. In our current findings, subjects demonstrated significant reductions from baseline in both erythema and papules/pustules after 12 weeks of twice-daily use of MYS serum, with significant reductions in erythema occurring as early as Week 2. Both the MTG and N-MTG experienced significant reductions in the appearance of erythema. In addition to reductions in dryness/flaking, itching, stinging, and burning over the course of the study, subjects also observed similar reductions in erythema to those observed by the investigators. Further, subjects reported high levels of satisfaction with MYS serum, noting it improved the appearance of their skin, made it look healthier with less blotching, and reduced the intensity of their flares. Notably, a high percentage of subjects (88%) reported feeling less self-conscious about the appearance of their skin. This was a meaningful finding based on the negative burden rosacea has on emotional wellbeing and the substantial impact tolerable and efficacious skincare can play in improving patient quality of life. It is particularly beneficial for adults with rosacea to have a streamlined skincare regimen comprised of products that are also pleasant to use, feel good on sensitive skin, support the skin barrier, and contribute to global improvements in skin quality.
Limitations of this study include the absence of a control group or placebo comparison, the small sample size, and lack of demographic diversity. Future studies are warranted that include a control group and a more diverse population to better understand safety and efficacy outcomes in a broader population.
Conclusion
Twice-daily use of a serum containing plant adaptogens and P.A.T.H. technology in subjects with mild-to-moderate persistent centrofacial erythema associated with rosacea demonstrated significant reductions in the appearance of erythema and papules and pustules as assessed by investigators after 12 weeks. Significant improvements were observed in both groups of subjects, those who used the skincare regimen only and those who used the skincare regimen in combination with established prescription medications for rosacea treatment.
Acknowledgements
We would like to thank Lynne Kolton Schneider, PhD for her editorial assistance on this manuscript.
References
- Hua NJ, Chen J, Geng RSQ, Sibbald RG, Sibbald C. Efficacy of treatments in reducing facial erythema in rosacea: a systematic review. J Cutan Med Surg. 2025;29(1):43–50.
- Chen C, Wang P, Zhang L, et al. Exploring the pathogenesis and mechanism-targeted treatments of rosacea: previous understanding and updates. Biomedicines. 2023;11(8):2153.
- Galluccio G, D’Onghia M, Malvaso D, et al. Advances in the pathogenesis and treatment of rosacea: a phenotype-based therapeutic approach. Cosmetics. 2024; 11(1):11.
- Paiva-Santos AC, Gonçalves T, Peixoto D, et al. Rosacea topical treatment and care: from traditional to new drug delivery systems. Mol Pharm. 2023;20(8):3804–3828.
- Thiboutot D, Anderson R, Cook-Bolden F, et al. Standard management options for rosacea: the 2019 update by the National Rosacea Society Expert Committee. J Am Acad Dermatol. 2020;82(6):1501–1510.
- Steinhoff M, Schauber J, Leyden JJ. New insights into rosacea pathophysiology: a review of recent findings. J Am Acad Dermatol. 2013;69(6 suppl 1):S15–S26.
- Loyal J, Carr E, Almukhtar R, Goldman MP. Updates and best practices in the management of facial erythema. Clin Cosmet Invest Dermatol. 2021;14:601–614.
- Tan J, Almeida LM, Bewley A, et al. Updating the diagnosis, classification and assessment of rosacea: recommendations from the global ROSacea COnsensus (ROSCO) panel. Br J Dermatol. 2017;176(2):431–438.
- Tan J, Steinhoff M, Berg M, et al. Shortcomings in rosacea diagnosis and classification. Br J Dermatol. 2017;176(1):197–199.
- Zeichner JA, Eichenfield LF, Feldman SR, Kasteler JS, Ferrusi I. Quality of life in individuals with erythematotelangiectatic and papulopustular rosacea: findings from a web-based survey. J Clin Aesthet Dermatol. 2018;11(2):47–52.
- Chiu C-W, Tsai J, Huang YC. Health-related quality of life of patients with rosacea: a systematic review and meta-analysis of real-world data. Acta Derm Venereol. 2024:104:adv40053.
- Schaller M, Almeida LM, Bewley A, et al. Rosacea treatment update: recommendations from the global ROSacea COnsensus (ROSCO) panel. Br J Dermatol. 2017;176(2):465–471.
- Sharma A, Kroumpouzos G, Kassir M, et al. Rosacea management: a comprehensive review. J Cosmet Dermatol. 2022;21(5):1895–1904.
- Johnson SM, Berg A, Barr C. Managing rosacea in the clinic: from pathophysiology to treatment—a review of the literature. J Clin Aesthet Dermatol. 2020;13(4 Suppl 1):S17–S22.
- Draelos ZD, Grimes PE, Watchmaker J, Nelson DB. A multi-center trial evaluating a serum comprised of plant-based adaptogens targeting skin quality. J Clin Aesthet Dermatol. 2024;17(2):15–19.
- Panossian AG, Efferth T, Shikov AN, et al. Evolution of the adaptogenic concept from traditional use to medical systems: pharmacology of stress- and aging-related diseases. Med Res Rev. 2021;41(1):630–703.
- Pomatto LCD, Davies KJA. The role of declining adaptive homeostasis in ageing. J Physiol. 2017;595(24):7275–7309.
- Ray A, Gulati K, Anand R. Stress, adaptogens and their evaluation: an overview. J Pharma Reports. 2016;1(2):110.
- Barbalho SM, de Sousa Gonzaga HF, de Souza GA, de Alvares Goulart R, de Sousa Gonzaga ML, de Alvarez Rezende B. Dermatological effects of Curcuma species: a systematic review. Clin Exp Dermatol. 2021;46(5):825–833.
- Hwang E, Park S-Y, Yin CS, Kim HT, Kim TM, Yi TH. Antiaging effects of the mixture of Panax ginseng and Crataegus pinnatifida in human dermal fibroblasts and healthy human skin. J Ginseng Res. 2017;41(1):69–77.
- Lima EP, Gonçalves OH, Ames FQ, et al. Anti-inflammatory and antioxidant activity of nanoencapsulated curcuminoids extracted from Curcuma longa L. in a model of cutaneous inflammation. Inflammation. 2021;44(2):604–616.
- Park MY, Han SJ, Moon D, Kwon S, Lee JW, Kim KSl. Effects of red ginseng on the elastic properties of human skin. J Ginseng Res. 2019;44(5):738–746.
- Pham QL, Jang H-J, Kim K-B. Anti-wrinkle effect of fermented black ginseng on human fibroblasts. Int J Mol Med. 2017;39(3):681–686.
- Saboori S, Falahi E, Rad EY, Asbaghi O, Khosroshahi MZ. Effects of ginseng on C-reactive protein level: a systematic review and meta-analysis of clinical trials. Complement Ther Med. 2019;45:98–103.
- Vaughn AR, Branum A, Sivamani RK. Effects of turmeric (Curcuma longa) on skin health: a systematic review of the clinical evidence. Phytother Res. 2016;30(8):1243–1264.
- Yang Y, Ren C, Zhang Y, Wu XD. Ginseng: an nonnegligible natural remedy for healthy aging. Aging Dis. 2017;8(6):708–720.
- Zorina A, Zorin V, Kudlay D, Kopnin P. Molecular mechanisms of changes in homeostasis of the dermal extracellular matrix: both involutional and mediated by ultraviolet radiation. Int J Mol Sci. 2022;23(12):6655.
- Draelos ZD, Grimes PE, Watchmaker J, Nelson DB. Gene expression analysis of a topical serum comprised of plant-based adaptogens developed to support homeostasis and skin quality. J Clin Aesthet Dermatol. 2024;17(2):
43–46.