J Clin Aesthet Dermatol. 2026;19(1):41–46.
by Dawn Merritt, DO, FAAD; Jon Lyons, PhD, MBA; Mouna Mikati, PhD; Christian Geils, BS; and Todd Schlesinger, MD, FAAD
Dr. Merritt is with OhioHealth in Columbus, Ohio. Dr. Lyons and Dr. Mikati are with Biofrontera Inc Medical Affairs in Woburn, Massachusetts. Mr. Geils is with the University of South Carolina in Columbia, South Carolina. Dr. Schlesinger is with the Clinical Research Center of the Carolinas in Charleston, South Carolina.
FUNDING: Medical writing support was provided by 7 West Communications, supported by Biofrontera Inc. The authors received no honoraria related to the development of
this publication.
DISCLOSURES: Dr. Merritt is an investigator for Aclaris, Incyte, Lilly, Novartis, Regeneron, and Sanofi; is a speaker for AbbVie, Almirall, Amgen, Biofrontera, BMS, Lilly, OrthoDermatologics, Pfizer, Sun, and UCB; is on the advisory board for BMS, Biofrontera, Pfizer, Sun, and UCB; and is a consultant for Biofrontera and Novartis. Drs. Lyons and Mikati are employees of Biofrontera. Dr. Schlesinger is a speaker, consultant, and/or investigator for AbbVie, Almirall, Apogee, Arcutis Biotherapeutics, Benev, Biofrontera, Bristol Myers Squibb, Crown Aesthetics, Flint Clinical, Genentech, Janssen, LEO Pharma, Lilly, Pfizer, Regeneron Pharmaceuticals, Sun Pharma, Verrica, and UCB; has served as an investigator for AbbVie, Allergan, Arcutis Biotherapeutics, Aslan Pharmaceuticals, Biofrontera, Boehringer Ingelheim, Cara Therapeutics, Castle Biosciences. ChemoCentryx, Coherus Biosciences, Concentrics, Concert Pharmaceuticals (acquired by Sun Pharma), Cutanea, Dermavant Sciences, Galderma, Highlightll, Incyte, Janssen, Lilly, Nimbus Therapeutics, Novartis, Processa, Prolacta, Regeneron Pharmaceuticals, Sanofi, Takeda, Trevi Pharmaceuticals, and Verrica; is a shareholder of Bristol Myers Squibb, Lilly, and Remedly; and is a consultant for Beiersdorf, Dermsquared, HTL Biotechnology, L’Oreal, MJH Life Sciences, and RBC Consultants. Mr. Geils has no conflicts of interest relevant to the contents of this article.
ABSTRACT: Background: Photodynamic therapy (PDT) is an effective field treatment for actinic keratoses (AKs). It is an ideal option for patients who prefer a minimally invasive procedure conveniently administered in their physician’s office. Anecdotal clinician insights suggest that it may not be used at the same rate year-round. Objective: This study investigates whether PDT and cryosurgery usage is affected by season and geographic location in the United States. Methods: The data source is an aggregate of closed and open United States medical procedural claims (Common Procedural Code [CPT] and Healthcare Common Procedure Coding System [HCPCS]) from a broad selection of commercial payers and Centers for Medicare & Medicaid Services (CMS) from 2015 to 2022. Patient records associated with cryosurgery and PDT CPT codes were extracted. A total of 79.1 million patients from 50 states and 4 territories between 2015 to 2022 were included in the analysis. The data were further stratified using meteorologically defined seasons and geographic locations. Results: PDT utilization is affected by season and geographic location. Procedural claims peak during cooler months and decrease during warmer months, particularly where seasonal changes are more drastic. This trend repeats throughout the study period, including the COVID-19 pandemic. In contrast, use of cryosurgery remains unchanged. Limitations: The dataset does not capture claims from all commercial payers and CMS. Conclusion: To our knowledge, this study provides the first evidence of PDT seasonal usage patterns across the nation. This pattern could be attributed to several factors and the findings could serve as a learning opportunity for clinicians to reevaluate their approach in treating AKs. Keywords: Actinic keratosis, photodynamic therapy, PDT, cryosurgery, procedural claims, CMS, seasonality, real-world evidence
Introduction
Actinic keratosis (AK) is one of the most common skin conditions treated by dermatologists in the United States (US), with AKs diagnosed in more than 10% of outpatient dermatology visits and reported in up to 26% of Americans aged 30 years or older.1,2 Early detection and treatment of AK is important, as the lesions can progress into cutaneous squamous cell carcinomas (cSCCs), the second most common type of nonmelanoma skin cancers (NMSC). Because of its high prevalence and potential for malignant transformation, AK is an important health concern and presents a financial impact on the healthcare system. In 2013, the estimated cost of treating AK in the US was $1.68 billion,3 while the annual cost of treating nonmelanoma skin cancers (NMSCs) was estimated at $4.8 billion from 2007 to 2011.4
There are multiple treatment options available for the management of AK in the US, including cryosurgery, topicals, and photodynamic therapy (PDT), and while there isn’t a universally established standard of care, cryosurgery has long been a first-line option due to its ease of in-office administration and reported efficacy.5,6 Cryosurgery, however, can only be performed in a lesion-directed manner and is therefore not suitable for addressing field cancerization. It is also associated with potential adverse effects, such as blistering, long-term scarring, and hypopigmentation.5 PDT is an in-office, light/energy-based treatment that offers a noninvasive approach to AK treatment. PDT has some of the highest reported clearance rates, including sustainable long-term efficacy at 12 months, and is associated with enhanced cosmetic outcomes and high patient satisfaction.7-12 The biggest advantage of PDT is that it can be performed in a field-directed manner in which individual visible lesions as well as subclinical damage are treated simultaneously. This addresses field cancerization, which helps minimize the recurrence of AK lesions and could arguably help prevent their potential progression to cSCC12-15
—though long-term studies are yet to be conducted to conclusively show a prophylactic effect of PDT. In addition, PDT has shown very good short- and long-term efficacy for the treatment of basal cell carcinoma (BCC) and Bowen’s disease (SCC in situ [SCCis]).7,12,16-19 While approved in Europe, PDT is still off-label for the treatment of NMSC in the US.20,21 The major adverse effect of PDT is pain during treatment, and there are many practical pain management strategies to help improve patient comfort.21 In addition, recent studies and protocol amendments have sought to decrease patient discomfort by either reducing the incubation time of the aminolevulinic acid (ALA) or using daylight or artificial daylight as a light source, essentially using a light source with decreased energy fluence while increasing the illumination duration to activate the photosensitizer.22,23
PDT received Medicare coverage for the treatment of AK in 2001, which provided millions of Americans with an additional in-office and cost-effective therapy to treat lesions and help mitigate their risk of progression to cSCC.24,25 With 29.3% of Medicare beneficiaries receiving at least one AK diagnosis between 2009 and 2018, and an estimated 72% of cSCCs originating from preexisting AKs, the need for efficacious and cost-effective treatments is dire.26,27 PDT has been shown to be one of the most cost-effective in-office treatments, thus increasing its implementation footprint and frequency could help alleviate the growing healthcare burden associated with AK and NMSCs in the US.3,28,29
Although the efficacy of PDT is well documented in numerous published studies, anecdotal clinician insights suggest that it may not be used at the same rate year-round and that this “PDT seasonality” may vary by geographic location. In contrast, this seasonal trend has not been observed or reported with cryosurgery. This study aims to analyze PDT usage patterns as compared to cryosurgery to investigate whether PDT use is affected by season and geographic location in the US. The findings will provide insights on physician behavior and a learning opportunity for better use of PDT across the nation that would capitalize on the advantages offered to patients and the healthcare system year-round.
Methods
The data source is an aggregate of closed and open US medical procedural claims (Common Procedural Code [CPT] and Healthcare Common Procedure Coding System [HCPCS]) from a broad selection of commercial payers and Centers for Medicare and Medicaid Services (CMS) from 2015 to 2022. Patient records associated with CPT codes 17000, 17003, and 17004 for cryosurgery and 96567, 96573, and 96574 for PDT were extracted. A total of 79.1 million patients from 50 states and 4 territories between 2015-2022 were included in the analysis. The descriptions for each CPT code can be found in Table 1.
The results were further stratified based on meteorologically defined seasons: December 1 to February 28 (winter), March 1 to May 31 (spring), June 1 to August 31 (summer), and September 1 to November 30 (fall). Sinusoidal regression in the datasets was used to determine whether seasonality exists in the administration of cryosurgery and PDT in the US. Due to the impact of the COVID-19 pandemic, particularly on the administration of in-office procedures, data from the year 2020 were considered outliers and were excluded from the analysis.
Results
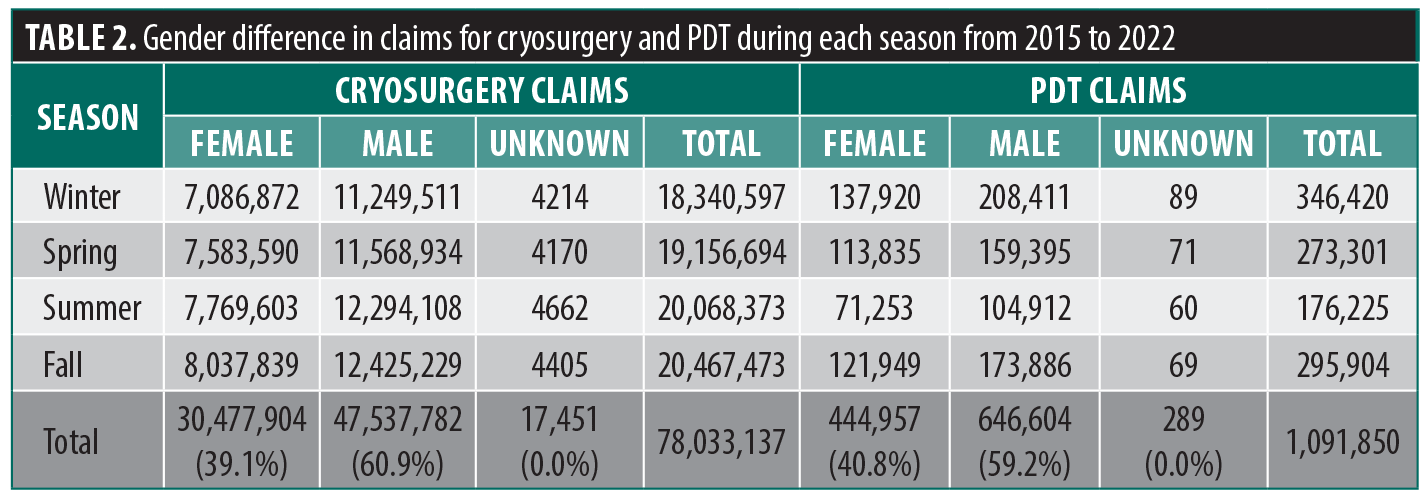
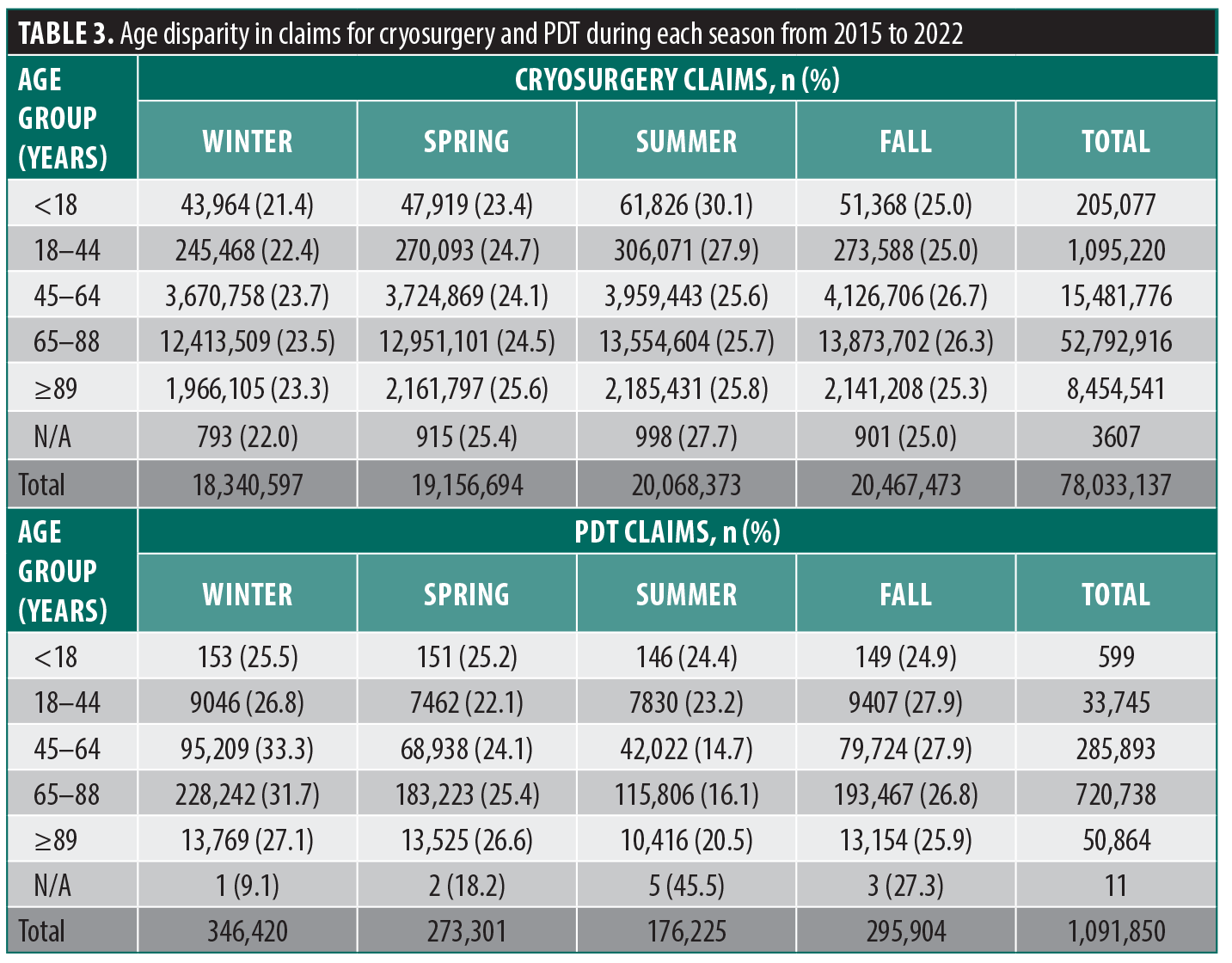
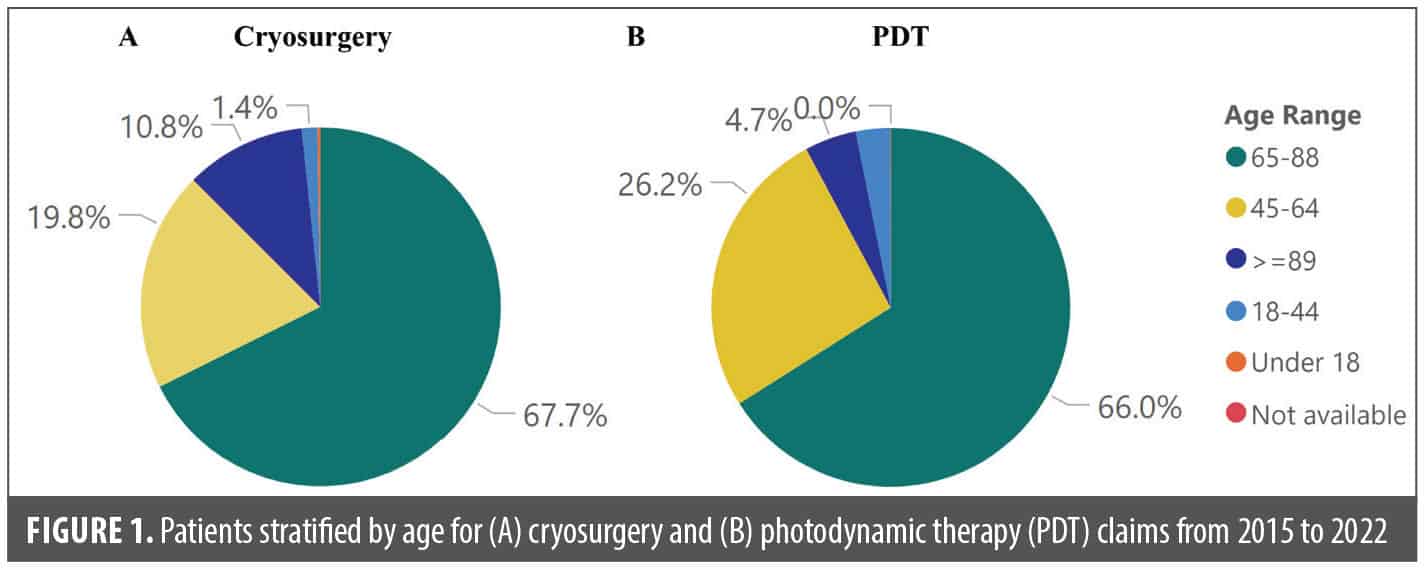
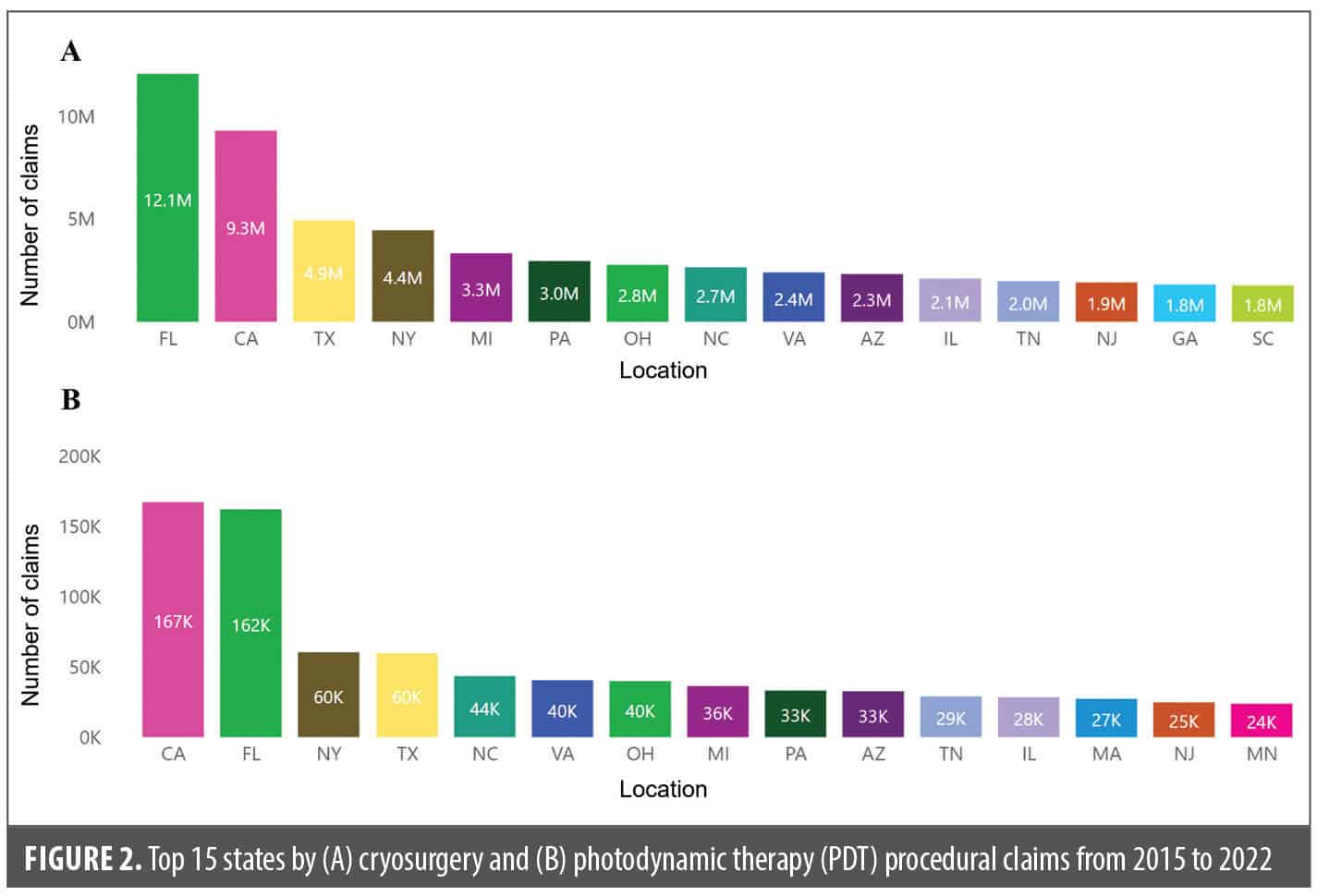
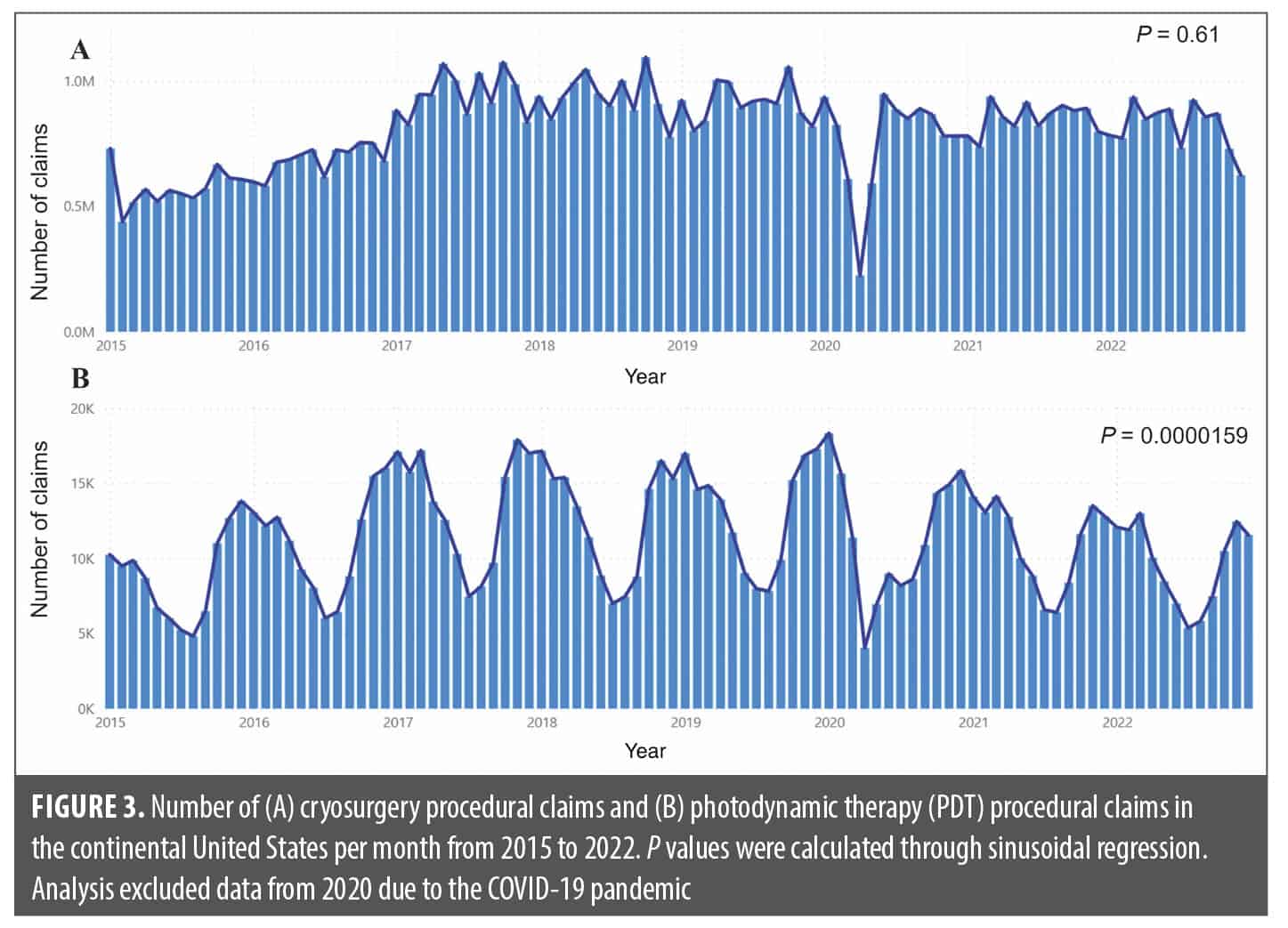
Of the 79.1 million patient records analyzed from 2015 to 2022, the majority were men, accounting for 60.9% of cryosurgery claims and 59.2% of PDT claims (Table 2). Patients aged 65 to 88 years accounted for the highest number of claims for both cryosurgery and PDT, closely followed by those aged 45 to 64 years (Table 3; Figure 1). Cryosurgery claim numbers peaked in the fall while PDT claims peaked in the winter (Table 3). Florida, California, Texas, and New York were the top four states with the highest number of procedural claims for both cryosurgery and PDT between 2015 and 2022 (Figure 2).
There was an increase in cryosurgery claims from 2015 to 2017, after which they stabilized. A significant decline in claims was observed in April 2020, aligning with the impact of COVID-19 pandemic lockdowns that restricted office visits that year. As such, all data from 2020 were excluded from the seasonality regression analysis. Despite slight monthly variations in claims, data analysis revealed no statistically significant seasonal trend in cryosurgery procedural claims (p=0.61) (Figure 3A). In contrast, PDT procedural claims showed cyclical seasonality, significantly increasing during the cooler months, with a peak around November and December, and decreasing during the warmer months, particularly around July (p=0.0000159) (Figure 3B). This seasonal pattern recurred annually from 2015 to 2022.
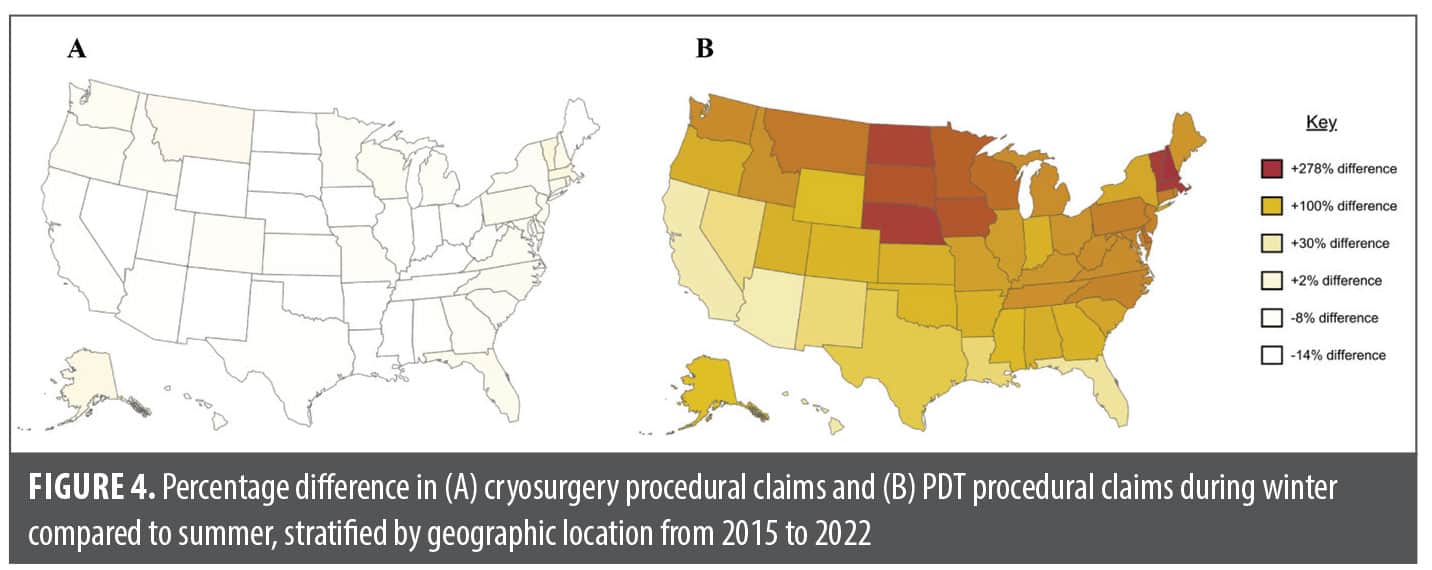
When comparing procedural claims from winter (December to February) and summer months (June to August) across the US, cryosurgery claims exhibited minimal differences across geographic locations (Figure 4A). In contrast, PDT claims display differences of up to 278% in winter months compared to summer months (Figure 4B). These variations are particularly pronounced in northern states such as North Dakota, South Dakota, Montana, Nebraska, Minnesota, Iowa, Wisconsin, and New Hampshire. Conversely, southern states including California, Arizona, Nevada, Louisiana, and Florida exhibited the least amount of change in claims between winter and summer months.
Throughout all seasons from 2015 to 2022, men consistently accounted for the majority of claims for both PDT and cryosurgery concerning premalignant skin lesions, and most claims were made by patients aged 45 and 88 years (Table 2, Figure 1). Both results are expected due to
the higher prevalence of AK in men and elderly patients.2,30,31
Discussion
This is the first time that seasonal trends in PDT and cryosurgery in the US were studied, with evidence provided on the disparity in PDT utilization across the US. Our data showed that there was no significant treatment seasonality for cryosurgery treatments, with minimal differences in claims between cooler and warmer months across the different states (Figure 3A, Figure 4A). In contrast, there was a cyclical seasonality with winter peaks and summer lows that persisted for PDT from 2015 to 2022 (Figure 3B, Figure 4B). When PDT claims are compared between winter and summer across the US, warmer states with fewer fluctuations in weather throughout the year, such as California, Arizona, New Mexico, Texas, Louisiana, and Florida, exhibited the least change in PDT claim numbers between the two seasons (Figure 4B).32 However, states with significant temperature variations throughout the year that also experience colder winters showed the most considerable differences in
the number of PDT procedures between winter and summer.32
Multiple factors could be attributed to the observed seasonality in PDT administration. Many physicians recommend undergoing dermatologic treatments, including PDT, in the winter months to avoid complications due to sun exposure in the typically sunnier summer months and due to patients wearing lighter, less sun-protective clothing.33 People tend to engage more in outdoor activity during the summer months, and PDT safety protocol involves patients avoiding sunlight and artificial UV exposure for 40 to 48 hours post-procedure due to the increased photosensitivity experienced post-treatment.34 Individuals residing in northern states may opt to maximize outdoor activities or vacations in the summer, hence preferring to undergo PDT in the winter. Also, there typically is an increase in social activities, such as weddings, outdoors gatherings, concerts, and travel, during the summer months, and the adverse effects of PDT (eg, erythema, swelling, scaling) on the treated area might deter people from getting this therapy during the busiest social season of the year.
PDT holds an advantage over cryosurgery because it offers a field-directed approach that targets field cancerization and multiple AKs on sun-damaged skin without the long-term adverse effects attributed to cryosurgery (eg, scarring and hypopigmentation). This makes it a desirable treatment option for cosmetically sensitive areas such as the face and the hands. Furthermore, a recent review focusing on the efficacy, cost, and adherence of the various field-directed therapies for AK determined PDT to be the most cost-effective treatment along with 4% 5-fluorouracil (5-FU) and stipulated that the high patient adherence of PDT would provide an added advantage in its improved real-world efficacy and long-term clearance.28 In fact, the tolerability profile of PDT makes it a strong central player in the network of AK treatments since it can be combined with other available AK therapies and benefit patients who have received treatments limited to target lesions, such as cryosurgery, or who have failed previous monotherapies.35 Considering these advantages, a restricted use of PDT could lead to reduced benefits for both patients and the healthcare system, while administering PDT year-round may optimize patient outcomes and mitigate the economic impact on a financially strained healthcare system.
Physicians can overcome barriers to treatment in the summertime by implementing simple tactics such as picking a favorable day of the week and working around their patients’ planned activities when scheduling PDT sessions. They can provide detailed post-treatment care instructions, emphasizing the use of protective measures, such as a tinted sunscreen and protective gear, to shield from UV and daylight exposure. Additionally, it is important to properly counsel patients about the risks of not following these UV safety measures to effectively minimize adverse events. While especially helpful in the summer months, these tactics are applicable and beneficial any time that PDT is administered.
Conclusion
Despite its well-documented efficacy and safety as a year-round treatment for AK, our study shows that PDT is underutilized during the summer season, particularly in northern states. Anecdotal insights gathered from healthcare professionals implied the existence of such seasonality in PDT use. Our findings could serve as a learning opportunity for clinicians to reevaluate their approach in treating AKs and to implement PDT as a year-round treatment in practice, either alone or in combination with other AK treatments (including cryosurgery) because of the advantages it offers to the affected patient population and the healthcare system as an efficacious, cost-effective, field-directed treatment. There is an overarching need for education on the practical implementation of PDT for the benefit of HCPs and patients alike. Future studies should seek to bridge gaps in the existing clinical practices and published guidelines36 and to provide real-world recommendations for improving use of this therapy.
References
- Li Y, Wang J, Xiao W, et al. Risk factors for actinic keratoses: a systematic review and meta-analysis. Indian J Dermatol. 2022;67(1):92.
- Reinehr CPH, Bakos RM. Actinic keratoses: review of clinical, dermoscopic, and therapeutic aspects. An Bras Dermatol. 2019;94(6):637-657.
- Lim HW, Collins SAB, Resneck JS Jr, et al. The burden of skin disease in the United States.J Am Acad Dermatol. 2017;76(5):958-972.e2.
- Aggarwal P, Knabel P, Fleischer AB Jr. United States burden of melanoma and nonmelanoma skin cancer from 1990 to 2019. J Am Acad Dermatol. 2021;85(2):388-395.
- Yoo SA, Kim YH, Han JH, et al. Treatment of actinic keratosis: the best choice through an observational study. J Clin Med. 2022;11(14):3953.
- Arisi M, Guasco Pisani E, Calzavara-Pinton P, Zane C. Cryotherapy for actinic Kkratosis: basic principles and literature review. Clin Cosmet Investig Dermatol. 2022;15:357-365.
- Reinhold U. A review of BF-200 ALA for the photodynamic treatment of mild-to-moderate actinic keratosis. Future Oncol. 2017;13(27):2413-2428.
- Reinhold U, Dirschka T, Ostendorf R, et al. A randomized, double-blind, phase III, multicentre study to evaluate the safety and efficacy of BF-200 ALA (Ameluz®) vs. placebo in the field-directed treatment of mild-to-moderate actinic keratosis with photodynamic therapy (PDT) when using the BF-RhodoLED® lamp. Br J Dermatol. 2016;175(4):696-705.
- Martínez-Carpio PA, Alcolea-López JM, Vélez M. Efficacy of photodynamic therapy in the short and medium term in the treatment of actinic keratosis, basal cell carcinoma, acne vulgaris and photoaging: results from four clinical trials. Laser Ther. 2012;21(3):199-208.
- Patel G, Armstrong AW, Eisen DB. Efficacy of photodynamic therapy vs other interventions in randomized clinical trials for the treatment of actinic keratoses: a systematic review and meta-analysis. JAMA Dermatol. 2014;150(12):1281-1288.
- Radakovic-Fijan S, Blecha-Thalhammer U, Kittler H, Hönigsmann H, Tanew A. Efficacy of 3 different light doses in the treatment of actinic keratosis with 5-aminolevulinic acid photodynamic therapy: a randomized, observer-blinded, intrapatient, comparison study. J Am Acad Dermatol. 2005;53(5):
823-827. - Farberg AS, Marson JW, Soleymani T. Advances in photodynamic therapy for the treatment of actinic keratosis and nonmelanoma skin cancer: a narrative review. Dermatol Ther (Heidelb). 2023;13(3):689-716.
- Griffin LL, Lear JT. Photodynamic therapy and nonmelanoma skin cancer. Cancers (Basel). 2016;8(10):98.
- Jetter N, Chandan N, Wang S, Tsoukas M. Field cancerization therapies for management of actinic keratosis: a narrative review. Am J Clin Dermatol. 2018;19(4):543-557.
- Liew YCC, De Souza NNA, Sultana RG, Oh CC. Photodynamic therapy for the prevention and treatment of actinic keratosis/squamous cell carcinoma in solid organ transplant recipients: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34(2):251-259.
- Morton CA, Dominicus R, Radny P, et al. A randomized, multinational, noninferiority, phase III trial to evaluate the safety and efficacy of BF-200 aminolaevulinic acid gel vs. methyl aminolaevulinate cream in the treatment of nonaggressive basal cell carcinoma with photodynamic therapy. Br J Dermatol. 2018;179(2):309-319.
- Alique-García S, Company-Quiroga J, Sánchez Campos A, Hernández Núñez A, Borbujo J. Treatment of superficial basal cell carcinoma with photodynamic therapy. Observational study in 22 patients with 5-aminolaevulinic acid and methyl aminolaevulinate. Photodiagnosis Photodyn Ther. 2019;26:
190-192. - Cervantes JA, Zeitouni NC. Photodynamic therapy utilizing 10% ALA nano-emulsion gel and red-light for the treatment of squamous cell carcinoma in-situ on the trunk and extremities: pilot study and literature update. Photodiagnosis Photodyn Ther. 2021;35:102358.
- Shim PJ, Zeitouni NC. Long-term follow up of ALA 10% gel and red-light photodynamic therapy for the treatment of squamous cell carcinoma in situ. Photodiagnosis Photodyn Ther. 2023;41:103211.
- Ameluz. Summary of product characteristics. Biofrontera Bioscience; 2024.
- Metvix. Summary of product characteristics. Galderma; November 3, 2022. https://www.medicines.ie/medicines/metvix-cream-32846/spc
- Zeitouni NC, Bhatia N, Ceilley RI, et al. Photodynamic therapy with 5-aminolevulinic acid 10% gel and red light for the treatment of actinic keratosis, nonmelanoma skin cancers, and acne: current evidence and best practices. J Clin Aesthet Dermatol. 2021;14(10):E53-E65.
- Neittaanmäki-Perttu N, Karppinen TT, Grönroos M, Tani TT, Snellman E. Daylight photodynamic therapy for actinic keratoses: a randomized double-blinded nonsponsored prospective study comparing 5-aminolaevulinic acid nanoemulsion (BF-200) with methyl-5-aminolaevulinate. Br J Dermatol. 2014;171(5):1172-1180.
- US Department of Health & Human Services. Medicare Coverage Issues Manual. Centers for Medicare & Medicaid Services; 2001. Transmittal 145.
- Siegel JA, Korgavkar K, Weinstock MA. Current perspective on actinic keratosis: a review. Br J Dermatol. 2017;177(2):350-358.
- Navsaria LJ, Li Y, Nowakowska MK, et al. Incidence and treatment of actinic keratosis in older adults with Medicare coverage. JAMA Dermatol. 2022;158(9):1076-1078.
- Czarnecki D, Meehan CJ, Bruce F, Culjak G. The majority of cutaneous squamous cell carcinomas arise in actinic keratoses. J Cutan Med Surg. 2002;6(3):207-209.
- Lampley N 3rd, Rigo R, Schlesinger T, Rossi AM. Field therapy for actinic keratosis: a structured review of the literature on efficacy, cost, and adherence. Dermatol Surg. 2023;49(2):124-129.
- Yoon J, Phibbs CS, Chow A, Weinstock MA; Veterans Affairs Keratinocyte Crcinoma Chemoprevention Trial Group. Impact of topical fluorouracil cream on costs of treating keratinocyte carcinoma (nonmelanoma skin cancer) and actinic keratosis. J Am Acad Dermatol. 2018;79(3):501-507.
- Flohil SC, van der Leest RJT, Dowlatshahi EA, Hofman A, de Vries E, Nijsten T. Prevalence of actinic keratosis and its risk factors in the general population: the Rotterdam Study.J Invest Dermatol. 2013;133(8):1971-1978.
- Marques E, Chen TM. Actinic Keratosis. Updated 2023 Aug 17. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. https://www.ncbi.nlm.nih.gov/books/NBK557401/
- National Centers for Environmental Information. Climate at a glance: statewide mapping. Accessed November 2, 2023. https://www.ncei.noaa.gov/access/monitoring/climate-at-a-glance/statewide/mapping/110/tavg/202308/1/value
- Von Neuschatz D. Are you ready for the summer? New York Social Diary. January 31, 2019. Accessed November 2, 2023. https://www.newyorksocialdiary.com/are-you-ready-for-the-summer/
- National Cancer Institute. Photodynamic therapy to treat cancer. Accessed November 5, 2023. https://www.cancer.gov/about-cancer/treatment/types/photodynamic-therapy
- Steeb T, Wessely A, Leiter U, French LE, Berking C, Heppt MV. The more the better? An appraisal of combination therapies for actinic keratosis.
J Eur Acad Dermatol Venereol. 2020;34(4):727-732. - Eisen DB, Asgari MM, Bennett DD, et al. Guidelines of care for the management of actinic keratosis: executive summary. J Am Acad Dermatol. 2021;85(4):945-955.