 by Jason Mathis, MD; and Sherrif F. Ibrahim, MD, PhD
by Jason Mathis, MD; and Sherrif F. Ibrahim, MD, PhD
Drs. Mathis and Ibrahim are with the Department of Dermatology at the University of Rochester Medical Center in Rochester, New York.
FUNDING: No funding was provided for this study.
DISCLOSURES: Dr. Ibrahim is a paid consultant and/or speaker for Sciton, Genentech and Regeneron. Dr. Mathis reports no conflicts of interest relevant to the content of this article.
ABSTRACT: Background.Rhinophyma is a dermatologic condition that can lead to severe disfiguration and psychological distress. Many therapies, both medical and surgical, have been reported, but few with acceptable and reproducible cosmesis.
Objective.We assessed the efficacy of erbium-doped yttrium aluminium garnet (Er:YAG) laser resurfacing as a treatment modality for rhinophyma of all degrees.
Design, Setting, and Participants.Eleven Caucasian male patients were classified into categories of mild, moderate, or severe rhinophyma and were treated at the University of Rochester Medical Center with a dual mode Er:YAG fully ablative laser.
Measurements.Severity was graded as mild, moderate, or severe, based on the degree of distortion of normal anatomy.
Results. The Er:YAG laser allowed for increased ablative precision, a decreased risk of complications, shorter downtime, and improved outcomes compared to currently available treatments.
Conclusion. Our results suggest Er:YAG laser resurfacing is an effective treatment modality, with low risk and excellent, reproducible cosmetic outcomes, for patients with rhinophyma of any severity.
KEYWORDS: Er:YAG, resurfacing, rhinophyma, rosacea
J Clin Aesthet Dermatol. 2019;12(7):28–33
First described by Von Hebra in 1856,1 rhinophyma presents as erythematous, nodular, bulbous, irregularly enlarged nasal tissue. Telangiectasias, atrophic skin, and dilated pilosebaceous pores are prominent.2 Rhinophyma is considered the fourth and end-stage of rosacea.3 Its precise etiology is unknown, though numerous theories have been explored, including vitamin deficiencies, stress, androgenic hormones and Demodex folliculorum mite invasion.3 Given its historic and erroneous association with alcohol use, rhinophyma has been referred to as “whiskey nose,” “rum nose,” and “gin blossom.”4 Rhinophyma most often affects Caucasian men aged 50 to 70 years.4 The male-to-female ratio for rhinophyma is 5:1 to 30:1, despite the fact that rosacea has a female predominance.5
Rhinophyma is a cosmetically disfiguring and psychologically damaging condition that might compromise physiological function. The majority of patients with erythematous rosacea report moderate-to-severe impairment in quality of life, with those suffering from phymatous rosacea reporting the greatest impairment.6 The psychosocial impact of severely disfiguring rhinophyma cannot be underestimated. Sequelae can include depression, agoraphobia, functional impairment via nasal airway obstruction,3 social stigma, and employment discrimination.7 In fact, rosacea might adversely affect quality of life to the same degree that psoriasis, diabetes, arthritis, epilepsy, and coronary artery disease do, according to indirect comparison between rosacea and these diseases via psoriasis using the Dermatology Life Quality Index (DLQI) and the European Quality of Life-5 Dimension (EQ-5D) surveys.6,8–10 The level of impairment in quality of life has been shown to be similar between rosacea and psoriasis, which has been demonstrated to impair quality of life on a scale similar to that in patients with cancer and other major chronic diseases, according to the EQ-5D.11,12 In addition, coinhabitants living with patients with psoriasis have elevated and similar rates of anxiety and depression when compared to controls.13 It is reasonable to infer, therefore, that this same relationship exists among coinhabitants of patients with rhinophyma as well, as the association was elucidated using the same quality of life index (DLQI) that showed a similar impairment in quality of life between psoriasis and rosacea.6,13 Due to the significant burden of disease rhinophyma places on patients and their coinhabitants, successful treatment can have far-reaching, dramatic, and unanticipated benefits.
Numerous treatment options for rhinophyma have been attempted and reported in the literature to date.3 Unfortunately, current treatment options are limited by many factors, including subtherapeutic results, high rates of side effects, scarring, bleeding, lack of available modalities or trained operators, excessively invasive procedure requirements, and prolonged recovery time.14 Medical management is limited and largely ineffective, necessitating more destructive and/or mechanical approaches. In our clinical experience, destructive therapeutic modalities, such as excision and dermabrasion, can result in subjectively suboptimal cosmetic outcomes. Argon and carbon dioxide laser treatment of rhinophyma were first reported in the 1980s as promising new options. Two small case series, one from Fincher et al4 in 2004 and another from Orenstein et al15 in 2001, evaluated patients with moderate-to-severe rhinophyma who were treated with the Er:YAG laser. Both case series reported efficacy, with good to excellent results and minimal complications. In this article, we present the largest series to date of patients with rhinophyma of varying severity treated with a dual-mode Er:YAG laser.
Methods
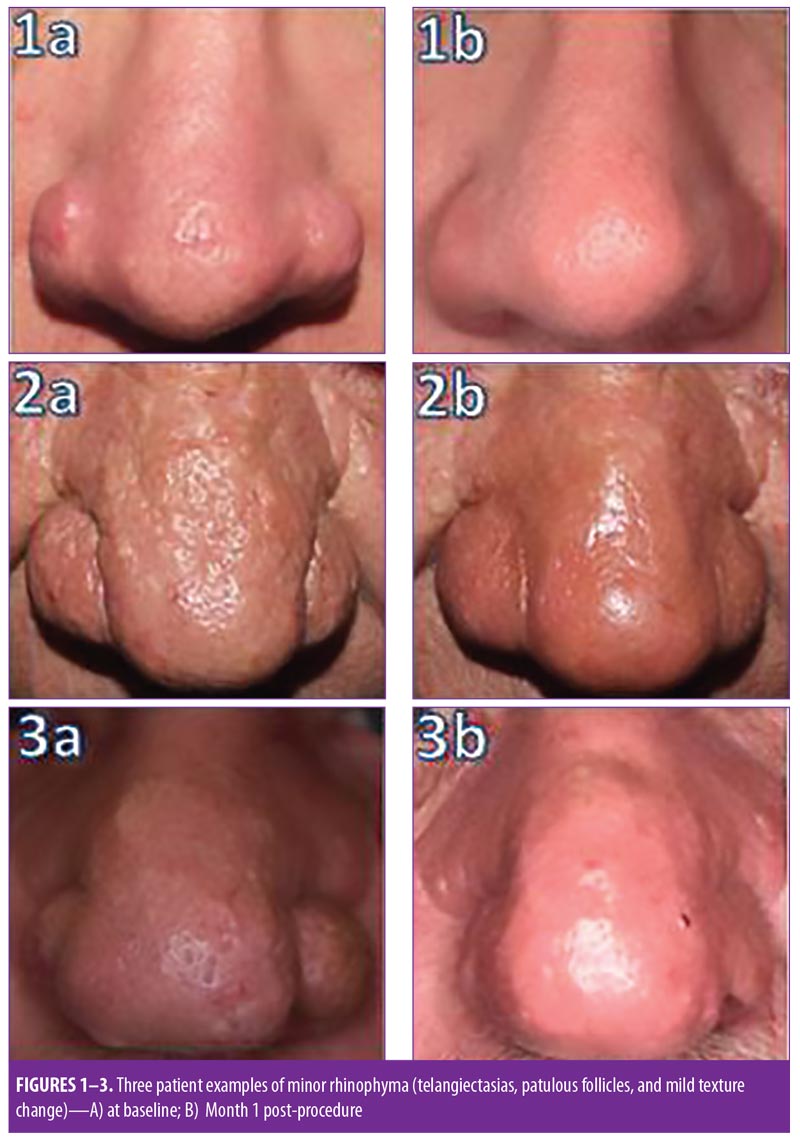
Eleven men with Fitzpatrick Skin Types I to II were treated on an outpatient basis at the Department of Dermatology at the University of Rochester Medical Center. Patients were categorized as having minor, moderate, or major rhinophyma in the same manner as Orenstein et al employed.15 The minor category included patients with telangiectasias, patulous follicles, and mild texture change. The moderate category added thickening of the nose and contour changes, and the major category added nodules to the previous findings. The mean duration of disease was six years. None of the patients had undergone prior treatment with a laser of any type, and many reported failed trials of topical rosacea medications, though this was unreliably reported or recalled by patients. Preprocedure photographs and signed consent were obtained from all patients. This study was approved by the University of Rochester Medical Center Research Study Review Board (approval no. RSRB00066655).
The skin was prepped with 4% chlorhexidine gluconate. All patients were treated under local anesthesia with direct infiltration of 1% lidocaine containing a 1:100,000 dilution of epinephrine buffered with sodium bicarbonate. Protective eye pads were applied. All patients were then treated with a dual-mode, 2940-nm Er:YAG laser (Joule; Sciton, Inc., Palo Alto, California). A 4-mm spot size was used with an ablative depth of 100 microns and 50 micrometers of coagulation per pulse at a frequency of 7Hz. Continuous passes were performed to precisely sculpt and restore the desired nasal contour. Saline-soaked gauze was used to remove debris as needed throughout the treatment. The most severely affected patient was pretreated with debulking by cold steel knife prior to ablation to shorten the procedure time to a tolerable length. One patient required 5mg of oral diazepam prior to the procedure and hydrocodone/acetaminophen for pain control afterward. Postoperatively, a semiocclusive, polyurethane foam, adhesive dressing was applied for 24 hours, followed by the daily application of petrolatum until the treated area re-epithelialized 8 to 13 days following treatment. The patients were evaluated at Postoperative Days 2, 5, 7, 14, and 30 to assess their responses to treatment.
Results
Of the 11 patients, three were categorized as having minor rhinophyma, two as moderate, and six as severe, according to the rating scale.In regard to treatment outcomes, a “good” result was defined as acceptable nasal contour with minimal scarring or color mismatch, while an “excellent” rating was described as displaying normal nasal contour, satisfactory color match, and no scarring. All but the most severe patient (Patient 11) had results rated “excellent,” while Patient 11’s outcome was rated as “very good” due to mild scarring that occurred as a result of the surgical blade debulking performed prior to laser resurfacing. It is possible, in Patient 11, that the deep level of ablation required to achieve normal contour resulted in the textural aberration. None of the other patients experienced this scarring though the other patients with severe rhinophyma all required significant ablation.
Regardless, all patients reported being completely satisfied with their cosmetic outcome. Baseline and 30-day post-treatment figures are presented in Figures 1 to 11, with the exception of one patient who refused follow-up. Complete re-epithelialization occurred by Postoperative Day 13 in all patients. There were no complications of infection, bleeding, significant scarring, alar rim retraction, prolonged erythema, or pigmentary alterations attributable to the Er:YAG system. None of our patients required additional treatments.
Regarding the need for surgical debulking prior to laser ablation in Patient 11, we believe that, with pure ablation only, the procedure time would be lengthened, but the resultant textural change would not have occurred. Multiple, shorter encounters could be used, rather than surgical debulking, to contain procedure time to tolerable limits in patients with massive rhinophyma. Patient 9 presented with the most severe functional impairment due to rhinophyma, stating that, prior to the procedure, he was not able to breathe through his nose while lying on his back or side. Figure 9B demonstrates an immediate change in patency of the nares post-treatment in this patient, with restoration of a functioning airway achieved following the laser procedure.



Discussion
Rhinophyma can have severe psychosocial and functional implications for patients, as illustrated by several patients in our case study. At least one of our patients reported symptoms of depression with agoraphobia as a result of their appearance. Additionally, at least one of our patients reported being denied treatment of their rhinophyma by other clinics prior to presenting at our facility. Restoration of normal appearance can produce almost immediate improvement in patient quality of life.
Medical treatments for rhinophyma and lifestyle changes have been unsuccessful.3,16–18 A variety of invasive techniques have been reported. Surgical excision with primary closure or healing by secondary intention has been employed since 1845, with skin grafting first used in 1912.3 This type of full-thickness excision can lead to scar formation, poor texture and color match, and suboptimal cosmesis. It has since been replaced by partial-thickness modalities, with incremental improvements in outcomes.4 Injection of fibrolysin, X-ray therapy, and electrocautery were utilized early in the 20th century, but these modalities did not allow for precise control of the depth of tissue destroyed and are suboptimal.3,19
Other surgical techniques have been used in the treatment of rhinophyma, including partial-thickness shaving with a cold steel knife and dermabrasion, dermaplaning with a dermatome, and heated knives and loops, such as the heated Shaw knife.3,17,20–22 Cryosurgery has also been performed on patients with rhinophyma. Cold steel excision enables precise sculpting, but can lead to extensive bleeding due to vascularization of the rhinophymatous skin and scar formation. Heat-based devices are thought to have higher rates of delayed healing, scarring, and unpredictable cosmetic outcomes due to uncontrolled thermal damage.4 More revently, hydrosurgery systems and ultrasonic scalpels have emerged as treatment options for rhinophyma.1,5,23
Ablative laser therapy has been used to treat rhinophyma since the 1980s, with the carbon dioxide (CO2) and argon lasers among the first to be used.3 Ablative lasers are especially suited for treating rhinophyma, due to the ability to vaporize excess tissue in a controlled manner. Moreover, ablative lasers cause collagen remodeling, which is beneficial due to the dermal dysregulation present in rhinophyma.4,24
Initially, the CO2 laser was considered one of the better therapies treating rhinophyma, and is still considered an effective treatment option for rhinophyma. As water is the primary target chromophore, there is a nonselective ablation of tissue as pulses produce thermal energy which nonselectively destroys anything near the chromophore (water) as collateral damage; hair, sebaceous glands, cartilage, bone, etc. This high thermal energy can be advantageous in that it provides a coagulative effect and, when slightly defocused, a potentially bloodless field. 4,25 The relatively deep penetrance also limits the ability to obtain accurate and precise tissue contouring and can lead to delayed healing. Newer pulsed, ultrapulsed, and scanning CO2 devices, however, have reduced the complication rate.4
The Er:YAG laser, with a wavelength of 2,940nm, has a better absorption coefficient of water and a smaller depth of tissue ablation per pass compared to the CO2 laser. This allows for more precise, controlled, and accurate tissue contouring than CO2 lasers.26 With a shorter pulse duration, the Er:YAG laser yields little collateral thermal damage, thus reducing nonspecific thermal injury, which reduces risk of pain and scarring, shortens recovery, and lowers risk of dyspigmentation. These advantages have been demonstrated in head-to-head comparisons with CO2 lasers in facial resurfacing studies.26–28
An early series of patients with rhinophyma treated with a first-generation, single-mode Er:YAG laser reported excellent results and no scarring, but topical epinephrine soaks and electrocautery were required for complete hemostasis.15 Newer dual-mode Er:YAG lasers combine the standard short-pulse ablative energy with a train of lower-energy, longer pulse-width pulses that deliver thermal energy for improved coagulation.4 These units mitigate the hemostasis disadvantage that Er:YAG lasers previously had compared to CO2 devices. This disadvantage can be further reduced by using stacked pulses rather than the scanning head and by increasing the coagulation setting to 25 to 50 micrometers. In our experience, these adjustments provide the hemostasis required for precise sculpting and a faster treatment time.
Conclusion
Our results indicate that the Er:YAG laser is an effective treatment option for the patients with mild-to-severe rhinophyma, due to its increased ablation precision, decreased risk of complications, reduced pain, and shorter recovery time compared to a CO2 laser. The patients in our series demonstrated satisfactory correction of their rhinophyma without adverse outcomes. The Er:YAG therapy was well tolerated without scarring or prolonged erythema and with improvements in texture. All patients reported being satisfied with the treatment outcomes, with responses ranging from increased self-esteem and social comfort to life-changing improvements. All patients tolerated the postoperative recovery well. The functional recovery noted in Patient 9 is particularly noteworthy in demonstrating that treatment with the Er:YAG laser not only can provide psychosocial benefits, but might also provide medical benefits.
References
- Taghizadeh R, Mackay SP, Gilbert PM. Treatment of rhinophyma with the versajet hydrosurgery system. J Plast Reconstr Aesthet Surg. 2008;61(3):330–333.
- Aloi F, Tomasini C, Soro E, Pippione M. The clinicopathologic spectrum of rhinophyma. J Am Acad Dermatol. 2000;42(3):468–472.
- Apikian M, Goodman GJ, Roberts S. Management of mild to moderate rhinophyma with a 1,450-nm diode laser: report of five patients. Dermatol Surg. 2007;33(7):847–850.
- Fincher EF, Gladstone HB. Use of a dual-mode erbium:YAG laser for the surgical correction of rhinophyma. Arch Facial Plast Surg. 2004;6(4):267–271.
- Tenna S, Gigliofiorito P, Langella M, et al. Treatment of rhinophyma with ultrasonic scalpel: case report. J Plast Reconstr Aesthet Surg. 2009;62(6):e164–e165.
- Moustafa F, Lewallen RS, Feldman SR. The psychological impact of rosacea and the influence of current management options. J Am Acad Dermatol. 2014;71(5):973–980.
- National Rosacea Society. Rosacea and employment. Available at: https://www.rosacea.org/weblog/rosacea_and_employment. Accessed February 1, 2018.
- Bewley A, Fowler J, Schofer H, et al. Erythema of rosacea impairs health-related quality of life: results of a meta-analysis. Dermatol Ther (Heidelb). 2016;6(2):237–247.
- Cresce ND, Davis SA, Huang WW, Feldman SR. The quality of life impact of acne and rosacea compared to other major medical conditions. J Drugs Dermatol. 2014;13(6):692–697.
- Lewis V, Finlay AY. 10 years experience of the dermatology life quality index (DLQI). J Investig Dermatol Symp Proc. 2004;9(2):169–180.
- Moller AH, Erntoft S, Vinding GR, Jemec GB. A systematic literature review to compare quality of life in psoriasis with other chronic diseases using EQ-5D-derived utility values. Patient Relat Outcome Meas. 2015;6:167–177.
- Rapp SR, Feldman SR, Exum ML, et al. Psoriasis causes as much disability as other major medical diseases. J Am Acad Dermatol. 1999;41(3 Pt 1):401–407.
- Martinez-Garcia E, Arias-Santiago S, Valenzuela-Salas I, et al. Quality of life in persons living with psoriasis patients. J Am Acad Dermatol. 2014;71(2):302–307.
- Sadick H, Goepel B, Bersch C, et al. Rhinophyma: diagnosis and treatment options for a disfiguring tumor of the nose. Ann Plast Surg. 2008;61(1):114–120.
- Orenstein A, Haik J, Tamir J, et al. Treatment of rhinophyma with er:YAG laser. Lasers Surg Med. 2001;29(3):230–235.
- Singh S, Peterson JD, Friedman PM. Management of mild to moderate rhinophyma using ablative fractional photothermolysis. Dermatol Surg. 2013;39(7):1110–1113.
- Rohrich RJ, Griffin JR, Adams WP Jr. Rhinophyma: review and update. Plast Reconstr Surg. 2002;110(3):860–69; quiz 870.
- Wiemer DR. Rhinophyma. Clin Plast Surg. 1987;14(2):357–365.
- Fisher WJ. Rhinophyma: its surgical treatment. Plast Reconstr Surg. 1970;45(5):466–470.
- Gupta S, Handa S, Saraswat A, Kumar B. Conventional cold excision combined with dermabrasion for rhinophyma. J Dermatol. 2000;27(2):116–120.
- Zide MF. Surgical removal of rhinophyma. J Oral Maxillofac Surg. 2008;66(10):2168–2177.
- Eisen RF, Katz AE, Bohigian RK, Grande DJ. Surgical treatment of rhinophyma with the shaw scalpel. Arch Dermatol. 1986;122(3):307–309.
- Dufresne RG, Zienowicz RJ, Rozelle A, Whalen JD. An introduction of the ultrasonic scalpel: utility in treatment of rhinophyma. Plast Reconstr Surg. 1996;98(1):160–162.
- Ross EV, McKinlay JR, Anderson RR. Why does carbon dioxide resurfacing work? A review. Arch Dermatol. 1999;135(4):444–454.
- el-Azhary RA, Roenigk RK, Wang TD. Spectrum of results after treatment of rhinophyma with the carbon dioxide laser. Mayo Clin Proc. 1991;66(9):899–905.
- Rostan EF, Fitzpatrick RE, Goldman MP. Laser resurfacing with a long pulse erbium:YAG laser compared to the 950 ms pulsed CO2 laser. Lasers Surg Med. 2001;29(2):136–141.
- Manuskiatti W, Iamphonrat T, Wanitphakdeedecha R, Eimpunth S. Comparison of fractional erbium-doped yttrium aluminum garnet and carbon dioxide lasers in resurfacing of atrophic acne scars in Asians. Dermatol Surg. 2013;39(1 Pt 1):111–120.
- Khatri KA, Ross V, Grevelink JM, et al. Comparison of erbium:YAG and carbon dioxide lasers in resurfacing of facial rhytides. Arch Dermatol. 1999;135(4):391–397.
