J Clin Aesthet Dermatol. 2025;18(11):16–20.
by Miranda An, BA, and Peter Lio, MD
Ms. An is with the University of California, Irvine School of Medicine in Irvine, California. Dr. Lio is with the Department of Dermatology at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Lio reports research grants/funding from AbbVie, AOBiome; is on the speaker’s bureau for AbbVie, Arcutis, Galderma, Hyphens Pharma, Incyte, La Roche-Posay/L’Oreal, Lilly, MyOR Diagnostics, ParentMD, Pfizer, Pierre-Fabre Dermatologie, Regeneron/Sanofi Genzyme, Verrica; reports consulting/advisory boards for Alphyn, AbbVie, Almirall, Amyris, Arcutis, ASLAN, Boston Skin Science, Bristol-Myers Squibb, Burt’s Bees, Castle Biosciences, Codex Labs, Concerto Biosci, Dermavant, Galderma, Janssen, Johnson & Johnson, Kimberly-Clark, LEO Pharma, Lilly, Lipidor, L’Oreal, Merck, Micreos, MyOR Diagnostics, Regeneron/Sanofi Genzyme, Skinfix, Theraplex, UCB, Unilever, Verrica, Yobee Care; stock options with Codex, Concerto Biosciences, and Yobee Care. In addition, Dr. Lio has a patent pending for a Theraplex product with royalties paid and is a Board Member and Scientific Advisory Committee Member of the National Eczema Association. Ms. An has no conflicts of interest to declare.
ABSTRACT: Atopic dermatitis (AD), a chronic inflammatory skin condition affecting up to 20% of children and 7% of adults in the United States, significantly impacts quality of life through recurrent flares and persistent symptoms. This commentary introduces the ESTAR framework—Efficacy, Safety, Tolerability, Accessibility, and Remission/Remittive Effects—as a modernized tool for shared decision-making in AD management. By addressing critical factors such as efficacy (onset speed, depth, durability, and reliability), safety, tolerability, and remission potential, this framework can aid in evaluating emerging therapies with a patient-centered approach. The ESTAR framework emphasizes balancing immediate symptom relief with sustainable long-term disease control, offering tools such as a radar chart to help clinicians and patients navigate complex treatment options, improve adherence, and optimize outcomes in AD care. Keywords: Atopic dermatitis, eczema, topical phosphodiesterase-4 (PDE-4) inhibitors, Janus kinase (JAK), dupilumab, efficacy, safety, tolerability, accessibility, remission
Introduction
Atopic dermatitis (AD), or eczema, is a common chronic inflammatory skin condition affecting up to 20% of children and 7% of adults in the United States (US).1,2 Its complex nature arises from an interplay of genetic, environmental, and immunologic factors—including a compromised skin barrier, immune dysfunction, and microbial imbalance—resulting in a continual and significant impact on quality of life. As AD is a chronic, relapsing condition without a cure, treatment focuses on two key objectives: managing flares (exacerbations or active disease) and minimizing the frequency and severity of flares to maintain remission and prevent future episodes (maintenance).3
The treatment of AD has been largely shaped by the introduction of corticosteroids in the 1950s. This revolutionary breakthrough not only provided patients with reliable relief for the first time but also sparked a deeper understanding of the condition. While topical corticosteroids remain the cornerstone of anti-inflammatory treatment to this day, associated safety and tolerability concerns have inspired the search for nonsteroidal treatments. This has paved the way for the development of other topical anti-inflammatory therapies.4 After a long period of drought, a rapidly expanding array of innovative therapies for AD is now emerging. Recently approved therapies, including topical phosphodiesterase-4 (PDE-4) inhibitors, Janus kinase (JAK) inhibitors (topical and oral), and systemic biologics targeting cytokines such as interleukins IL-4, IL-13, and IL-31, have significantly expanded the treatment landscape for AD (Table 1).5,6
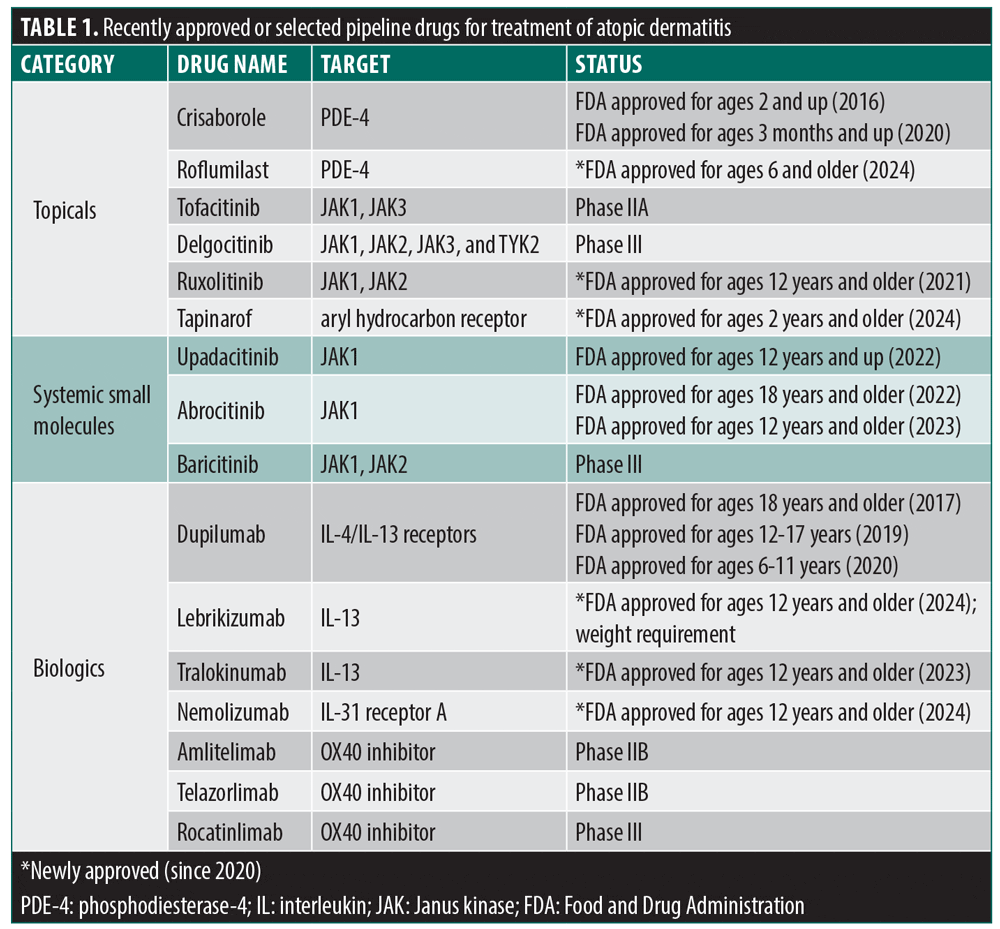
While these innovations offer new opportunities for tailored treatment, they also add complexity to the previously more straightforward decisions between topical corticosteroids and, for refractory AD, off-label systemic therapies such as methotrexate or cyclosporine. An update to the EAST model,7 the ESTAR framework—Efficacy, Safety, Tolerability, Accessibility, and Remission/Remittive Effects—aims to modernize shared decision-making in AD management. As the therapeutic landscape continues to evolve with novel biologics, topicals, and small molecules, the ESTAR assessment provides a comprehensive approach that incorporates detailed efficacy factors and remission potential, empowering patients and physicians to make more informed, individualized decisions. It begins by helping navigate the increasing pressure to select treatments, moving towards precision medicine given the multiple different aspects of the treatments and individual patient preferences and needs.
Selecting More Precisely
With the existing conventional therapies and the advent of numerous new agents, both the American Academy of Dermatology (AAD) and the Allergy, Asthma, and Immunology Joint Task Force (JTF) recently updated their AD guidelines to reflect the evolving landscape of approved nonsteroidal topicals, systemic biologics, and JAK inhibitors.3,8,9 Both guidelines were shaped by evidence-based practices assessing systemic reviews with a standardized GRADE (Grading of Recommendations Assessment, Development, and Evaluation) approach, emphasizing shared decision-making between patients and physicians and incorporating patient values and preferences.
For mild-to-moderate AD, both the AAD and JTF recommend regular moisturizer use to maintain skin hydration, identification and avoidance of triggers, and proper bathing practices to support overall skin health. In addition, topical corticosteroids are strongly recommended as the first-line anti-inflammatory agents, followed by topical calcineurin inhibitors and topical PDE-4 inhibitors.
For more moderate-to-severe cases, representing approximately 40% of adults and one-third of children with AD in the US,10,11 advanced treatment with phototherapy or a systemic therapy may be required. Both the AAD and JTF recommend dupilumab (an IL-4 and IL-13 inhibitor) and tralokinumab (an IL-13 inhibitor) as biologics for moderate-to-severe AD, followed by oral JAK inhibitors upadacitinib and abrocitinib for when biologics are not sufficient or appropriate. JAK inhibitor therapies require careful patient selection and monitoring due to potential adverse effects such as infections and thromboembolic events.
Lebrikizumab (an IL-13 inhibitor similar to tralokinumab) and nemolizumab (an IL-31 receptor inhibitor) were approved after the publication of the guidelines but are important additions with unique attributes. Additionally, phototherapy with narrowband ultraviolet B (NB-UVB) is recommended as a noninvasive option for moderate-to-severe disease, particularly for contraindications for systemic therapy or patients who prefer to avoid biologics. While cyclosporine is recommended for short-term use during severe flares, other systemic immunosuppressants, such as methotrexate, azathioprine, and mycophenolate mofetil, were conditionally recommended against in the JTF guideline documents given their numerous potential safety issues and relative lack of rigorous efficacy data. Most importantly, however, both the AAD and JTF guidelines emphasize tailoring therapies based on disease severity, patient preferences, comorbidities, and the risk-benefit profile of the treatments.
Choosing the right therapy for an individual with AD involves more than determining which agent is objectively “better.” Rather, a more nuanced approach is required, ideally guided by a shared decision-making process unique to each patient. To differentiate AD therapies effectively, it may be beneficial to assess them based on five key criteria: Efficacy (including onset, depth, durability, and reliability), Safety, Tolerability, Accessibility, and Remission/Remittive Effects—collectively referred to as the ESTAR framework.
Efficacy
A medication may excel in various metrics, such as affordability, a favorable safety profile, and ease of use, but the central question remains: How effective is it? While efficacy and potency graphs provide valuable insights into pharmacodynamics, the practical consideration prevails: Will it effectively address the patient’s condition? When evaluating medications, it is crucial to consider their effectiveness across diverse patient populations, recognizing the significant variability in individual responses.
Most therapies exhibit a wide range of effects, influenced by factors such as dose dependency and potential synergy with other treatments. These variables further shape their practical utility and overall efficacy, emphasizing the need for a comprehensive and adaptable approach to assessment. Broadly speaking, efficacy may be assessed through speed of onset, depth of effect, durability of effect, and reliability. Key questions to include are: What percentage of patients will experience some effect? What is the distribution of the effect, considering variability among individuals? These factors collectively form a nuanced framework for understanding therapeutic effectiveness. Unfortunately, clinicians rarely have sufficient data to answer such questions, let alone comparative evidence for more subtle distinctions across therapies. As such, these remain more subjective, experiential, and descriptive concepts rather than clearly defined objective metrics.
As an example, among the most rapid-acting therapies for AD are the oral JAK inhibitors. These agents demonstrate rapid onset of action, with significant improvements in symptoms, such as itch, seen as early as 1 to 2 days after initiation of treatment. This quick effect makes them particularly attractive for severe or refractory cases, where immediate symptom relief is of highest concern to the patient. The oral JAK inhibitors also have some of the best efficacy data in terms of achieving clinical endpoints, such as improvement in the Eczema Area and Severity Index (EASI) score, an aspect we might refer to as “depth” of effect, as measured across therapies using a Bayesian network meta-analysis approach.12
Safety
Safety, rooted in the principle of nonmaleficence, one of medicine’s foundational pillars, remains a key consideration, and often a decisive factor, in evaluating therapies. When assessing the safety profiles of AD therapies, both short- and long-term considerations are crucial. Frustratingly, many unanswered questions about safety remain, including vehement disagreements about how it is framed, sometimes going against a substantial amount of evidence, as in the case of the topical calcineurin inhibitors. These agents continue to have boxed warnings that specifically mention malignancy (skin and lymphoma), which can be tremendously off-putting for patients and families despite compelling evidence that this may no longer be warranted.13,14
Ruxolitinib (a JAK1/JAK2 inhibitor) cream provides a localized option with reduced systemic risks, though concerns about rare but potential systemic absorption and associated risks remain, represented by the boxed warning on this treatment as well. Discussing the actual safety risk with a patient can be extremely challenging, as these remain somewhat theoretical and class-based concerns, ignoring the specific drug, route of administration, and personalized attributes of the individual patient. This was illustrated recently by a disagreement between the AAD and JTF guidelines around topical ruxolitinib; the AAD guideline strongly recommends it, while the JTF conditionally suggests against topical ruxolitinib. Importantly, despite reviewing the same evidence, the JTF cites the safety profile and noted: “The panel inferred that most patients with mild-to-moderate AD would prefer to avoid the uncertain increase in death, cancer, thrombosis, and serious infections, particularly when there are multiple safer treatment options with larger certain benefits and higher certainty for safety.”
Notably, both guidelines conditionally recommend against systemic corticosteroids given their numerous safety concerns, especially in the longer term. Conventional immunosuppressants, such as cyclosporine and methotrexate, are associated with significant risks, including nephrotoxicity and hepatotoxicity, limiting their use primarily to severe, refractory cases and short-term flares. Biologics generally demonstrate more favorable safety profiles, with far fewer safety signals than conventional immunosuppressants.15,16
Systemic JAK inhibitors also carry a boxed warning for serious risks, including thromboembolism, malignancy, and serious infections, necessitating careful patient selection and laboratory monitoring. Phototherapy with NB-UVB is generally safe but can cause burns and, with prolonged use, an increased risk of photodamage.3,8 The expanding treatment landscape underscores the importance of individualized risk assessment and patient preferences, weighing safety as part of this larger framework to optimize outcomes in AD management.
Tolerability
Tolerability, distinct from safety, plays a critical role in treatment adherence and overall patient satisfaction. While tolerability issues, such as injection site pain or stinging/burning with application, may not constitute true safety concerns, they can significantly influence a patient’s willingness to initiate or continue treatment.
For example, crisaborole, despite its relatively favorable safety profile compared to topical corticosteroids, is reported to cause application site pain in 4 to 31% of patients, affecting its overall tolerability.17,18 Similarly, data from biologics like dupilumab indicate a favorable safety profile but highlight specific tolerability issues such as conjunctivitis, which has been reported in 9 to 22% of patients.8,19 Topical ruxolitinib has minimal tolerability issues but can cause mild application site reactions, and topical tapinarof, while possessing a very reassuring safety profile, has associated follicular events in approximately 10% of patients.20 Systemic therapies like JAK inhibitors, while highly effective, can carry tolerability concerns such as gastrointestinal upset and headache.3,8,9 Such experiences underscore that even mild adverse effects can influence adherence and treatment selection.
Given these considerations, a nuanced understanding of tolerability is essential for shared decision-making. Patients should be informed about potential adverse effects, their likelihood, and their impact on treatment experience to align therapy choices with individual preferences and lifestyle factors, ensuring better adherence and outcomes.
Accessibility
The cost of a drug is an important factor in understanding its accessibility but remains only part of a web that continues to complicate getting a medication in the American healthcare system. There is often a significant gap between the listed price of a medication and what patients ultimately pay, influenced by factors such as insurance coverage, pharmacy benefit managers, and healthcare provider policies. For many patients, affordability depends on navigating the intricate processes of insurance coverage and prior authorizations, which have become increasingly arduous with the introduction of biologics and other high-cost therapies.
Accessibility, therefore, goes beyond cost alone and includes the practical challenges of obtaining a medication. For example, a 60g tube of the generic calcineurin inhibitor cream pimecrolimus is listed for as much as $399 on GoodRx.com, a commonly used tool for drug pricing information.21 In contrast, while dupilumab is listed as up to $4,000 for two doses on the same site, patients with insurance coverage may pay nothing for the latter and possibly the full $399 for the former, making dupilumab surprisingly accessible for a newer, powerful systemic agent.21 While this example illustrates one potential scenario, the actual out-of-pocket cost for patients can vary substantially due to factors such as insurance coverage, deductibles, availability of manufacturer assistance programs, and regional pricing differences. Indeed, newer branded medications like roflumilast, though significantly pricier than older generics, are paradoxically often more readily accessible due to specialty pharmacies, manufacturer discount programs, and coupon offers.22
The time and effort required for prior authorization, combined with uncertainties about approval outcomes, often add additional barriers for patients and clinicians. These challenges are particularly evident in the age of biologics, where high upfront costs and coverage variability can hinder access even for insured patients. Given the rapid changes in drug pricing, regional differences, and the impact of individual insurance plans, the accessibility and affordability of AD treatments are highly individualized. Patients and healthcare providers must navigate this complex landscape to identify the most cost-effective and appropriate treatment options on a case-by-case basis.
Remission/remittive Effects
Achieving sustained remission is a key goal in the management of AD, particularly for patients with moderate-to-severe disease. Remission is admittedly somewhat difficult to define as it has a number of potential meanings. Perhaps the most basic definition is a period of minimal or no disease activity. However, while a patient is on the same dose of a medication, we could classify this simply as a durable response.
Ideally, remission would refer to maintained improvement without pharmacotherapy, or complete cessation of the use of a medication. Decreasing the dose, the frequency of use, or the use of adjunctive therapies such as topical medications is also meaningful and, in our definition, constitutes types of remission. In addition to alleviating symptoms of active disease, therapies with remittive effects may extend the time between flares, reduce the overall burden of treatment, and enhance patient quality of life. Patients in remission from AD may exhibit unique patterns of inflammation, offering valuable insights into the biological pathways underpinning sustained disease control.23
Some systemic therapies have demonstrated a remittive effect, potentially altering the course of AD by addressing subclinical inflammation that may be missed with topical agents.24 Emerging evidence suggests that while dupilumab does not have an approved extended dosing interval, many patients can maintain disease control at longer intervals, with some even achieving sustained remission even after complete discontinuation of the medication.25
Tralokinumab, lebrikizumab, and now nemolizumab all feature extended dosing intervals as part of their approved dosing regimens for patients who achieve adequate clinical response. Tralokinumab and lebrikizumab increase the spacing from every two weeks to monthly dosing, while nemolizumab increases from monthly dosing to every 8 weeks dosing, all while maintaining improvement in the vast majority of treated patients.26-28
Furthermore, the gradual discontinuation of adjunctive treatments, such as topical corticosteroids or calcineurin inhibitors, in patients on systemic therapies can indicate something beyond durable disease control and shed light on achieving remission through reduced reliance on combination therapies.29 While the definition of remission may still be debated, it is evident that these outcomes hold significant value for both patients and clinicians, demonstrating that improvements initially achieved with more frequent dosing can often be sustained at far less than that in many patients.
Complicating this landscape, however, is the inherently relapsing-remitting nature of AD. Some patients may achieve transient, spontaneous remission,30 reflecting the waxing and waning disease trajectory characterized by cycles of escalation and quiescence. Consequently, reduced treatment schedules or discontinuation of therapy might be feasible during periods of low disease activity, although this remains a subject of ongoing investigation.
Beyond this, the concept of disease modification is increasingly discussed, perhaps prompted by advancements in the OX40 pathway, where research suggests that longer-term remission and disease modification may be possible with regulatory T cell-targeted immunomodulatory therapy.31 Understanding these dynamics is crucial as new therapies emerge; these strategies not only alleviate the treatment burden on patients but also mitigate the long-term risks associated with prolonged medication use, contributing to an improved quality of life and more sustainable disease management.
Practical Implications
Combining the ESTAR framework with shared decision-making, prioritizing patient preferences and values, is essential for balancing immediate symptom relief with sustainable long-term disease control. While there are no fixed metrics for these parameters, clinicians can form individualized assessments of treatments to guide their considerations.
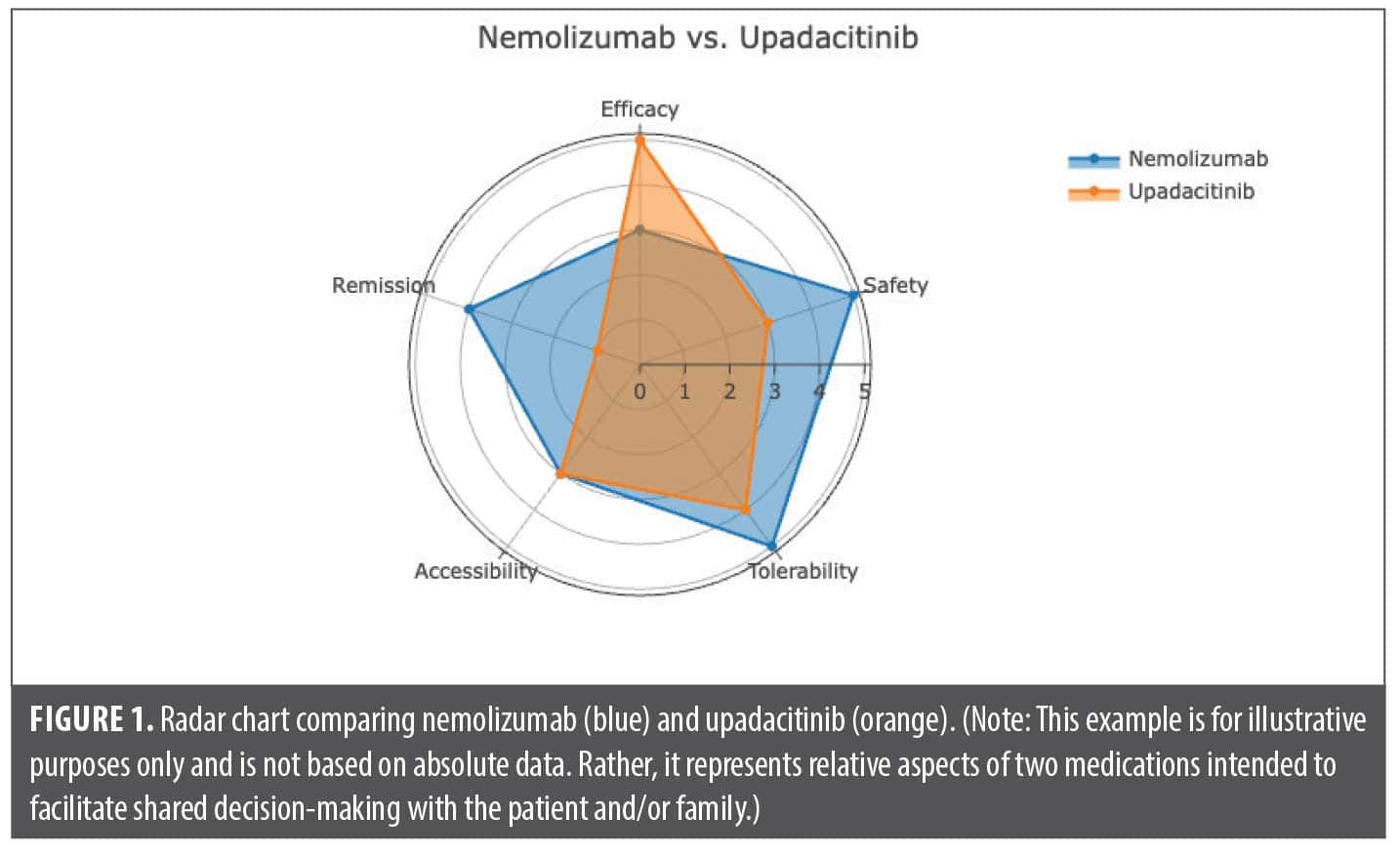
We present a radar chart as an example of a useful visual aid for comparing medications, mapping various attributes on an arbitrary scale of 0 to 5 (Figure 1). This figure illustrates our conceptualization of a comparison between nemolizumab and upadacitinib for the treatment of AD. It is important to recognize that interpretations of these parameters may vary among clinicians. Engaging in such evaluations and sharing them with patients can facilitate discussions and support treatment decisions, particularly as new therapeutic options continue to emerge and become available.
References
- Langan SM, Irvine AD, Weidinger S. Atopic dermatitis. Lancet. 2020;396(10247):345-360.
- Abuabara K, Magyari A, McCulloch CE, et al. Prevalence of atopic eczema among patients seen in primary care: data from the Health Improvement Network. Ann Intern Med. 2019;170(5):354-356.
- Chu DK, Schneider L, Asiniwasis RN, et al; AAAI/ACAAI JTF Atopic Dermatitis Guideline Panel. Atopic dermatitis (eczema) guidelines: 2023 American Academy of Allergy, Asthma and Immunology/American College of Allergy, Asthma and Immunology Joint Task Force on Practice Parameters GRADE- and Institute of Medicine-based recommendations. Ann Allergy Asthma Immunol. 2024;132(3):274-312.
- Gallagher M. The origin of eczema and the centuries-old history behind it. National Eczema Association. March 6, 2023. Accessed December 26, 2024. https://nationaleczema. org/blog/the-origin-of-eczema-and-the-centuries-old-history-behind-it/
- Lovell K, Patel N, Rao S, Strowd LC. The future of atopic dermatitis treatment. Adv Exp Med Biol. 2024;1447:227-244.
- Nakashima C, Yanagihara S, Otsuka A. Innovation in the treatment of atopic dermatitis: emerging topical and oral Janus kinase inhibitors. Allergol Int. 2022;71(1):40-46.
- Lio P, Rangwani S. Look to the EAST: shared decision-making in the era of new therapies. Practical Dermatol. 2020:10;24-30.
- Davis DMR, Drucker AM, Alikhan A, et al. Guidelines of care for the management of atopic dermatitis in adults with phototherapy and systemic therapies. J Am Acad Dermatol. 2024;90(2):e43-e56.
- Sidbury R, Alikhan A, Bercovitch L, et al. Guidelines of care for the management of atopic dermatitis in adults with topical therapies. J Am Acad Dermatol. 2023;89(1):e1-e20.
- Chiesa Fuxench ZC, Block JK, Boguniewicz M, et al. Atopic Dermatitis in America Study: a cross-sectional study examining the prevalence and disease burden of atopic dermatitis in the US adult population. J Invest Dermatol. 2019;139(3):583-590.
- Silverberg JI, Simpson EL. Associations of childhood eczema severity: a US population-based study. Dermatitis. 2014;25(3):107-114.
- Drucker AM, Lam M, Prieto-Merino D, et al. Systemic immunomodulatory treatments for atopic dermatitis: living systematic review and network meta-analysis update. JAMA Dermatol. 2024;160(9):936-944.
- Paller AS, Fölster-Holst R, Chen SC, et al. No evidence of increased cancer incidence in children using topical tacrolimus for atopic dermatitis. J Am Acad Dermatol. 2020;83(2):375-381.
- Devasenapathy N, Chu A, Wong M, et al; AAAAI/ACAAI Joint Task Force on Practice Parameters for Atopic Dermatitis Guideline Development Group. Cancer risk with topical calcineurin inhibitors, pimecrolimus and tacrolimus, for atopic dermatitis: a systematic review and meta-analysis. Lancet Child Adolesc Health. 2023;7(1):13-25.
- Mazaud C, Fardet L. Relative risk of and determinants for adverse events of methotrexate prescribed at a low dose: a systematic review and meta-analysis of randomized placebo-controlled trials. Br J Dermatol. 2017;177(4):978-986.
- de Bruin-Weller M, Thaçi D, Smith CH, et al. Dupilumab with concomitant topical corticosteroid treatment in adults with atopic dermatitis with an inadequate response or intolerance to ciclosporin A or when this treatment is medically inadvisable: a placebo-controlled, randomized phase III clinical trial (LIBERTY AD CAFÉ). Br J Dermatol. 2018;178(5):1083-1101.
- Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75(3):494-503.e6.
- Lin CPL, Gordon S, Her MJ, Rosmarin D. A retrospective study: application site pain with the use of crisaborole, a topical phosphodiesterase 4 inhibitor. J Am Acad Dermatol. 2019;80(5):1451-1453.
- Simpson EL, Bieber T, Guttman-Yassky E, et al. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016;375(24):2335-2348.
- Silverberg JI, Eichenfield LF, Hebert AA, et al. Tapinarof cream 1% once daily: significant efficacy in the treatment of moderate to severe atopic dermatitis in adults and children down to 2 years of age in the pivotal phase 3 ADORING trials. J Am Acad Dermatol. 2024;91(3):457-465.
- Prescription Prices. GoodRx: Coupons & Pharmacy Information; 2024; Available from: GoodRx.com.
- Dover JS, Mariwalla K. The Business of Dermatology. 1st ed: Thieme Medical Publishers; 2020.
- Sánchez A, Caraballo A, Alvarez L, et al. Molecular characteristics of atopic dermatitis patients with clinical remission. World Allergy Organ J. 2024;17(11):100983.
- Lio PA. Considerations in weaning or withdrawing dupilumab therapy—nothing is forever. JAMA Dermatol. 2020;156(2):119-120.
- Treister AD, Lio PA. Remittive effect of dupilumab in atopic dermatitis. Dermatol Ther. 2018;31(6):e12711.
- Weidinger S, Bewley A, Hong HC, et al. Predicting success with reduced dosing frequency of tralokinumab in patients with moderate-to-severe atopic dermatitis. Br J Dermatol. 2025;192(3):410-419.
- Silverberg J, Wollenberg A, Legat FJ, et al. Maintenance of efficacy and safety with nemolizumab at week 48: results from two global phase 3 pivotal studies (ARCADIA 1 and ARCADIA 2) in patients with moderate-to-severe atopic dermatitis. Abstract presented at: American Academy of Dermatology Annual Meeting; March 8-12, 2024; San Diego, CA. Abstract 56887.
- Silverberg JI, Guttman-Yassky E, Thaci D, et al; Advocate1 and Advocate2 Investigators. Two phase 3 trials of lebrikizumab for moderate-to-severe atopic dermatitis. N Engl J Med. 2023;388(12):1080-1091.
- Watanabe A, Kamata M, Okada Y, et al. Possibility of maintaining remission with topical therapy alone after withdrawal of dupilumab in Japanese patients with atopic dermatitis and their characteristics in the real world. Exp Dermatol. 2024;33(9):e15175.
- Kang Y, Hwang J, Lai Y-C, Choi H, Do Y. A nonlinear transient-dynamics approach to atopic dermatitis: role of spontaneous remission. Chaos Solitons Fractals. 2024;179:114464.
- Nahm DH. Regulatory T cell-targeted immunomodulatory therapy for long-term clinical improvement of atopic dermatitis: hypotheses and perspectives. Life (Basel). 2023;13(8):1674.
Reaching for the Stars in Atopic Dermatitis: Efficacy, Safety, Tolerability, Accessibility, and Remission/Remittive Effects (ESTAR)
Categories:
J Clin Aesthet Dermatol. 2025;18(11):16–20.
by Miranda An, BA, and Peter Lio, MD
Ms. An is with the University of California, Irvine School of Medicine in Irvine, California. Dr. Lio is with the Department of Dermatology at Northwestern University Feinberg School of Medicine in Chicago, Illinois.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Lio reports research grants/funding from AbbVie, AOBiome; is on the speaker’s bureau for AbbVie, Arcutis, Galderma, Hyphens Pharma, Incyte, La Roche-Posay/L’Oreal, Lilly, MyOR Diagnostics, ParentMD, Pfizer, Pierre-Fabre Dermatologie, Regeneron/Sanofi Genzyme, Verrica; reports consulting/advisory boards for Alphyn, AbbVie, Almirall, Amyris, Arcutis, ASLAN, Boston Skin Science, Bristol-Myers Squibb, Burt’s Bees, Castle Biosciences, Codex Labs, Concerto Biosci, Dermavant, Galderma, Janssen, Johnson & Johnson, Kimberly-Clark, LEO Pharma, Lilly, Lipidor, L’Oreal, Merck, Micreos, MyOR Diagnostics, Regeneron/Sanofi Genzyme, Skinfix, Theraplex, UCB, Unilever, Verrica, Yobee Care; stock options with Codex, Concerto Biosciences, and Yobee Care. In addition, Dr. Lio has a patent pending for a Theraplex product with royalties paid and is a Board Member and Scientific Advisory Committee Member of the National Eczema Association. Ms. An has no conflicts of interest to declare.
ABSTRACT: Atopic dermatitis (AD), a chronic inflammatory skin condition affecting up to 20% of children and 7% of adults in the United States, significantly impacts quality of life through recurrent flares and persistent symptoms. This commentary introduces the ESTAR framework—Efficacy, Safety, Tolerability, Accessibility, and Remission/Remittive Effects—as a modernized tool for shared decision-making in AD management. By addressing critical factors such as efficacy (onset speed, depth, durability, and reliability), safety, tolerability, and remission potential, this framework can aid in evaluating emerging therapies with a patient-centered approach. The ESTAR framework emphasizes balancing immediate symptom relief with sustainable long-term disease control, offering tools such as a radar chart to help clinicians and patients navigate complex treatment options, improve adherence, and optimize outcomes in AD care. Keywords: Atopic dermatitis, eczema, topical phosphodiesterase-4 (PDE-4) inhibitors, Janus kinase (JAK), dupilumab, efficacy, safety, tolerability, accessibility, remission
Introduction
Atopic dermatitis (AD), or eczema, is a common chronic inflammatory skin condition affecting up to 20% of children and 7% of adults in the United States (US).1,2 Its complex nature arises from an interplay of genetic, environmental, and immunologic factors—including a compromised skin barrier, immune dysfunction, and microbial imbalance—resulting in a continual and significant impact on quality of life. As AD is a chronic, relapsing condition without a cure, treatment focuses on two key objectives: managing flares (exacerbations or active disease) and minimizing the frequency and severity of flares to maintain remission and prevent future episodes (maintenance).3
The treatment of AD has been largely shaped by the introduction of corticosteroids in the 1950s. This revolutionary breakthrough not only provided patients with reliable relief for the first time but also sparked a deeper understanding of the condition. While topical corticosteroids remain the cornerstone of anti-inflammatory treatment to this day, associated safety and tolerability concerns have inspired the search for nonsteroidal treatments. This has paved the way for the development of other topical anti-inflammatory therapies.4 After a long period of drought, a rapidly expanding array of innovative therapies for AD is now emerging. Recently approved therapies, including topical phosphodiesterase-4 (PDE-4) inhibitors, Janus kinase (JAK) inhibitors (topical and oral), and systemic biologics targeting cytokines such as interleukins IL-4, IL-13, and IL-31, have significantly expanded the treatment landscape for AD (Table 1).5,6
While these innovations offer new opportunities for tailored treatment, they also add complexity to the previously more straightforward decisions between topical corticosteroids and, for refractory AD, off-label systemic therapies such as methotrexate or cyclosporine. An update to the EAST model,7 the ESTAR framework—Efficacy, Safety, Tolerability, Accessibility, and Remission/Remittive Effects—aims to modernize shared decision-making in AD management. As the therapeutic landscape continues to evolve with novel biologics, topicals, and small molecules, the ESTAR assessment provides a comprehensive approach that incorporates detailed efficacy factors and remission potential, empowering patients and physicians to make more informed, individualized decisions. It begins by helping navigate the increasing pressure to select treatments, moving towards precision medicine given the multiple different aspects of the treatments and individual patient preferences and needs.
Selecting More Precisely
With the existing conventional therapies and the advent of numerous new agents, both the American Academy of Dermatology (AAD) and the Allergy, Asthma, and Immunology Joint Task Force (JTF) recently updated their AD guidelines to reflect the evolving landscape of approved nonsteroidal topicals, systemic biologics, and JAK inhibitors.3,8,9 Both guidelines were shaped by evidence-based practices assessing systemic reviews with a standardized GRADE (Grading of Recommendations Assessment, Development, and Evaluation) approach, emphasizing shared decision-making between patients and physicians and incorporating patient values and preferences.
For mild-to-moderate AD, both the AAD and JTF recommend regular moisturizer use to maintain skin hydration, identification and avoidance of triggers, and proper bathing practices to support overall skin health. In addition, topical corticosteroids are strongly recommended as the first-line anti-inflammatory agents, followed by topical calcineurin inhibitors and topical PDE-4 inhibitors.
For more moderate-to-severe cases, representing approximately 40% of adults and one-third of children with AD in the US,10,11 advanced treatment with phototherapy or a systemic therapy may be required. Both the AAD and JTF recommend dupilumab (an IL-4 and IL-13 inhibitor) and tralokinumab (an IL-13 inhibitor) as biologics for moderate-to-severe AD, followed by oral JAK inhibitors upadacitinib and abrocitinib for when biologics are not sufficient or appropriate. JAK inhibitor therapies require careful patient selection and monitoring due to potential adverse effects such as infections and thromboembolic events.
Lebrikizumab (an IL-13 inhibitor similar to tralokinumab) and nemolizumab (an IL-31 receptor inhibitor) were approved after the publication of the guidelines but are important additions with unique attributes. Additionally, phototherapy with narrowband ultraviolet B (NB-UVB) is recommended as a noninvasive option for moderate-to-severe disease, particularly for contraindications for systemic therapy or patients who prefer to avoid biologics. While cyclosporine is recommended for short-term use during severe flares, other systemic immunosuppressants, such as methotrexate, azathioprine, and mycophenolate mofetil, were conditionally recommended against in the JTF guideline documents given their numerous potential safety issues and relative lack of rigorous efficacy data. Most importantly, however, both the AAD and JTF guidelines emphasize tailoring therapies based on disease severity, patient preferences, comorbidities, and the risk-benefit profile of the treatments.
Choosing the right therapy for an individual with AD involves more than determining which agent is objectively “better.” Rather, a more nuanced approach is required, ideally guided by a shared decision-making process unique to each patient. To differentiate AD therapies effectively, it may be beneficial to assess them based on five key criteria: Efficacy (including onset, depth, durability, and reliability), Safety, Tolerability, Accessibility, and Remission/Remittive Effects—collectively referred to as the ESTAR framework.
Efficacy
A medication may excel in various metrics, such as affordability, a favorable safety profile, and ease of use, but the central question remains: How effective is it? While efficacy and potency graphs provide valuable insights into pharmacodynamics, the practical consideration prevails: Will it effectively address the patient’s condition? When evaluating medications, it is crucial to consider their effectiveness across diverse patient populations, recognizing the significant variability in individual responses.
Most therapies exhibit a wide range of effects, influenced by factors such as dose dependency and potential synergy with other treatments. These variables further shape their practical utility and overall efficacy, emphasizing the need for a comprehensive and adaptable approach to assessment. Broadly speaking, efficacy may be assessed through speed of onset, depth of effect, durability of effect, and reliability. Key questions to include are: What percentage of patients will experience some effect? What is the distribution of the effect, considering variability among individuals? These factors collectively form a nuanced framework for understanding therapeutic effectiveness. Unfortunately, clinicians rarely have sufficient data to answer such questions, let alone comparative evidence for more subtle distinctions across therapies. As such, these remain more subjective, experiential, and descriptive concepts rather than clearly defined objective metrics.
As an example, among the most rapid-acting therapies for AD are the oral JAK inhibitors. These agents demonstrate rapid onset of action, with significant improvements in symptoms, such as itch, seen as early as 1 to 2 days after initiation of treatment. This quick effect makes them particularly attractive for severe or refractory cases, where immediate symptom relief is of highest concern to the patient. The oral JAK inhibitors also have some of the best efficacy data in terms of achieving clinical endpoints, such as improvement in the Eczema Area and Severity Index (EASI) score, an aspect we might refer to as “depth” of effect, as measured across therapies using a Bayesian network meta-analysis approach.12
Safety
Safety, rooted in the principle of nonmaleficence, one of medicine’s foundational pillars, remains a key consideration, and often a decisive factor, in evaluating therapies. When assessing the safety profiles of AD therapies, both short- and long-term considerations are crucial. Frustratingly, many unanswered questions about safety remain, including vehement disagreements about how it is framed, sometimes going against a substantial amount of evidence, as in the case of the topical calcineurin inhibitors. These agents continue to have boxed warnings that specifically mention malignancy (skin and lymphoma), which can be tremendously off-putting for patients and families despite compelling evidence that this may no longer be warranted.13,14
Ruxolitinib (a JAK1/JAK2 inhibitor) cream provides a localized option with reduced systemic risks, though concerns about rare but potential systemic absorption and associated risks remain, represented by the boxed warning on this treatment as well. Discussing the actual safety risk with a patient can be extremely challenging, as these remain somewhat theoretical and class-based concerns, ignoring the specific drug, route of administration, and personalized attributes of the individual patient. This was illustrated recently by a disagreement between the AAD and JTF guidelines around topical ruxolitinib; the AAD guideline strongly recommends it, while the JTF conditionally suggests against topical ruxolitinib. Importantly, despite reviewing the same evidence, the JTF cites the safety profile and noted: “The panel inferred that most patients with mild-to-moderate AD would prefer to avoid the uncertain increase in death, cancer, thrombosis, and serious infections, particularly when there are multiple safer treatment options with larger certain benefits and higher certainty for safety.”
Notably, both guidelines conditionally recommend against systemic corticosteroids given their numerous safety concerns, especially in the longer term. Conventional immunosuppressants, such as cyclosporine and methotrexate, are associated with significant risks, including nephrotoxicity and hepatotoxicity, limiting their use primarily to severe, refractory cases and short-term flares. Biologics generally demonstrate more favorable safety profiles, with far fewer safety signals than conventional immunosuppressants.15,16
Systemic JAK inhibitors also carry a boxed warning for serious risks, including thromboembolism, malignancy, and serious infections, necessitating careful patient selection and laboratory monitoring. Phototherapy with NB-UVB is generally safe but can cause burns and, with prolonged use, an increased risk of photodamage.3,8 The expanding treatment landscape underscores the importance of individualized risk assessment and patient preferences, weighing safety as part of this larger framework to optimize outcomes in AD management.
Tolerability
Tolerability, distinct from safety, plays a critical role in treatment adherence and overall patient satisfaction. While tolerability issues, such as injection site pain or stinging/burning with application, may not constitute true safety concerns, they can significantly influence a patient’s willingness to initiate or continue treatment.
For example, crisaborole, despite its relatively favorable safety profile compared to topical corticosteroids, is reported to cause application site pain in 4 to 31% of patients, affecting its overall tolerability.17,18 Similarly, data from biologics like dupilumab indicate a favorable safety profile but highlight specific tolerability issues such as conjunctivitis, which has been reported in 9 to 22% of patients.8,19 Topical ruxolitinib has minimal tolerability issues but can cause mild application site reactions, and topical tapinarof, while possessing a very reassuring safety profile, has associated follicular events in approximately 10% of patients.20 Systemic therapies like JAK inhibitors, while highly effective, can carry tolerability concerns such as gastrointestinal upset and headache.3,8,9 Such experiences underscore that even mild adverse effects can influence adherence and treatment selection.
Given these considerations, a nuanced understanding of tolerability is essential for shared decision-making. Patients should be informed about potential adverse effects, their likelihood, and their impact on treatment experience to align therapy choices with individual preferences and lifestyle factors, ensuring better adherence and outcomes.
Accessibility
The cost of a drug is an important factor in understanding its accessibility but remains only part of a web that continues to complicate getting a medication in the American healthcare system. There is often a significant gap between the listed price of a medication and what patients ultimately pay, influenced by factors such as insurance coverage, pharmacy benefit managers, and healthcare provider policies. For many patients, affordability depends on navigating the intricate processes of insurance coverage and prior authorizations, which have become increasingly arduous with the introduction of biologics and other high-cost therapies.
Accessibility, therefore, goes beyond cost alone and includes the practical challenges of obtaining a medication. For example, a 60g tube of the generic calcineurin inhibitor cream pimecrolimus is listed for as much as $399 on GoodRx.com, a commonly used tool for drug pricing information.21 In contrast, while dupilumab is listed as up to $4,000 for two doses on the same site, patients with insurance coverage may pay nothing for the latter and possibly the full $399 for the former, making dupilumab surprisingly accessible for a newer, powerful systemic agent.21 While this example illustrates one potential scenario, the actual out-of-pocket cost for patients can vary substantially due to factors such as insurance coverage, deductibles, availability of manufacturer assistance programs, and regional pricing differences. Indeed, newer branded medications like roflumilast, though significantly pricier than older generics, are paradoxically often more readily accessible due to specialty pharmacies, manufacturer discount programs, and coupon offers.22
The time and effort required for prior authorization, combined with uncertainties about approval outcomes, often add additional barriers for patients and clinicians. These challenges are particularly evident in the age of biologics, where high upfront costs and coverage variability can hinder access even for insured patients. Given the rapid changes in drug pricing, regional differences, and the impact of individual insurance plans, the accessibility and affordability of AD treatments are highly individualized. Patients and healthcare providers must navigate this complex landscape to identify the most cost-effective and appropriate treatment options on a case-by-case basis.
Remission/remittive Effects
Achieving sustained remission is a key goal in the management of AD, particularly for patients with moderate-to-severe disease. Remission is admittedly somewhat difficult to define as it has a number of potential meanings. Perhaps the most basic definition is a period of minimal or no disease activity. However, while a patient is on the same dose of a medication, we could classify this simply as a durable response.
Ideally, remission would refer to maintained improvement without pharmacotherapy, or complete cessation of the use of a medication. Decreasing the dose, the frequency of use, or the use of adjunctive therapies such as topical medications is also meaningful and, in our definition, constitutes types of remission. In addition to alleviating symptoms of active disease, therapies with remittive effects may extend the time between flares, reduce the overall burden of treatment, and enhance patient quality of life. Patients in remission from AD may exhibit unique patterns of inflammation, offering valuable insights into the biological pathways underpinning sustained disease control.23
Some systemic therapies have demonstrated a remittive effect, potentially altering the course of AD by addressing subclinical inflammation that may be missed with topical agents.24 Emerging evidence suggests that while dupilumab does not have an approved extended dosing interval, many patients can maintain disease control at longer intervals, with some even achieving sustained remission even after complete discontinuation of the medication.25
Tralokinumab, lebrikizumab, and now nemolizumab all feature extended dosing intervals as part of their approved dosing regimens for patients who achieve adequate clinical response. Tralokinumab and lebrikizumab increase the spacing from every two weeks to monthly dosing, while nemolizumab increases from monthly dosing to every 8 weeks dosing, all while maintaining improvement in the vast majority of treated patients.26-28
Furthermore, the gradual discontinuation of adjunctive treatments, such as topical corticosteroids or calcineurin inhibitors, in patients on systemic therapies can indicate something beyond durable disease control and shed light on achieving remission through reduced reliance on combination therapies.29 While the definition of remission may still be debated, it is evident that these outcomes hold significant value for both patients and clinicians, demonstrating that improvements initially achieved with more frequent dosing can often be sustained at far less than that in many patients.
Complicating this landscape, however, is the inherently relapsing-remitting nature of AD. Some patients may achieve transient, spontaneous remission,30 reflecting the waxing and waning disease trajectory characterized by cycles of escalation and quiescence. Consequently, reduced treatment schedules or discontinuation of therapy might be feasible during periods of low disease activity, although this remains a subject of ongoing investigation.
Beyond this, the concept of disease modification is increasingly discussed, perhaps prompted by advancements in the OX40 pathway, where research suggests that longer-term remission and disease modification may be possible with regulatory T cell-targeted immunomodulatory therapy.31 Understanding these dynamics is crucial as new therapies emerge; these strategies not only alleviate the treatment burden on patients but also mitigate the long-term risks associated with prolonged medication use, contributing to an improved quality of life and more sustainable disease management.
Practical Implications
Combining the ESTAR framework with shared decision-making, prioritizing patient preferences and values, is essential for balancing immediate symptom relief with sustainable long-term disease control. While there are no fixed metrics for these parameters, clinicians can form individualized assessments of treatments to guide their considerations.
We present a radar chart as an example of a useful visual aid for comparing medications, mapping various attributes on an arbitrary scale of 0 to 5 (Figure 1). This figure illustrates our conceptualization of a comparison between nemolizumab and upadacitinib for the treatment of AD. It is important to recognize that interpretations of these parameters may vary among clinicians. Engaging in such evaluations and sharing them with patients can facilitate discussions and support treatment decisions, particularly as new therapeutic options continue to emerge and become available.
References
Share:
Recent Articles:
Lawrence Eichenfield, MD: Insights from the TRuE-AD3 Pediatric Atopic Dermatitis Trial
Tristan Hasbargen, PA-C: Setting Expectations for a Long-Term Approach to Repigmentation
David Rosmarin, MD: Efficacy of Ruxolitinib Cream in Progressive and Stable Vitiligo
Ted Lain, MD, MBA: Reviewing Results of the THRIVE-AA1 Trial in Severe Alopecia Areata
Leon Kircik, MD: Maximizing Bioavailability and Patient Outcomes With Oral Acne Therapy
Categories:
Recent Articles:
Christopher Bunick, MD, PhD: Providing Rapid Itch Relief for Patients With Atopic Dermatitis
Letter to the Editor: March 2026
The Impact of Dupilumab on Psychological Wellbeing in Moderate-to-Severe Atopic Dermatitis Patients: A Phase IV Clinical Trial
Expanding the Topical Therapeutic Landscape for Atopic Dermatitis: A Systematic Review
A Multicenter Trial Evaluating the Safety and Efficacy of a Serum Containing Plant Adaptogens in Patients With Mild-to-Moderate, Persistent Centrofacial Erythema Associated With Rosacea