J Clin Aesthet Dermatol. 2026;19(1):17–26.
by Beth A. Childs, MS, and Joseph F. Merola, MD, MMSc
Ms. Childs and Dr. Merola are with the Department of Dermatology and Department of Medicine, Division of Rheumatology at the University of Texas Southwestern Medical Center in Dallas, Texas.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Merola is a consultant and/or investigator for AbbVie, Amgen, AstraZeneca, Biogen, Boehringer Inhelheim, Bristol Myers Squibb, Dermavant, Incyte, Janssen, LEO Pharma, Lilly, Moonlake, Novartis, Pfizer, Sanofi-Regeneron, Sun Pharma, and UCB. Ms. Childs has no conflicts to declare.
ABSTRACT: Objective: We aim to review current treatment strategies for cutaneous lupus erythematosus (CLE) and highlight emerging therapies, evolving outcome measures, and practical considerations that may inform future management of this clinically heterogeneous disease. Methods: We conducted a narrative review of the literature using PubMed to identify relevant clinical trials, observational studies, and mechanistic investigations related to CLE treatment. Emphasis was placed on studies published within the past 15 years. Expert opinion from dermatology and rheumatology was incorporated to contextualize evolving therapies and their application in clinical practice. Results: Current management of CLE follows a stepwise framework, with antimalarials and immunosuppressants forming the foundation of systemic therapy. However, many patients experience refractory disease, especially in chronic or hypertrophic subtypes. Advances in the understanding of CLE pathogenesis have led to the development of targeted biologics and small molecule agents that modulate Type I interferon signaling, B- and T-cell activation, and inflammatory cytokine pathways. Novel tools such as Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI-A) and the Investigator Global Assessment for Cutaneous Lupus (CLA-IGA-R) are being validated to standardize disease assessment, while RNA tape sampling and real-world electronic health record (EHR)-based algorithms are enhancing research precision. Emerging therapies, such as belimumab, anifrolumab, litifilimab, deucravacitinib, and enpatoran, demonstrate promise in both clinical trials and real-world settings. Limitations: As a narrative review, this study is limited by the lack of systematic inclusion criteria and formal quality assessment of individual studies. Conclusion: Ongoing therapeutic innovation, guided by mechanistic insights and strengthened by the development of standardized outcome measures, is transforming the CLE landscape and advancing the goal of precision-based, durable disease control. Keywords: Cutaneous lupus, systemic lupus erythematosus, screening, rheumatology, quality improvement, biologics
Introduction
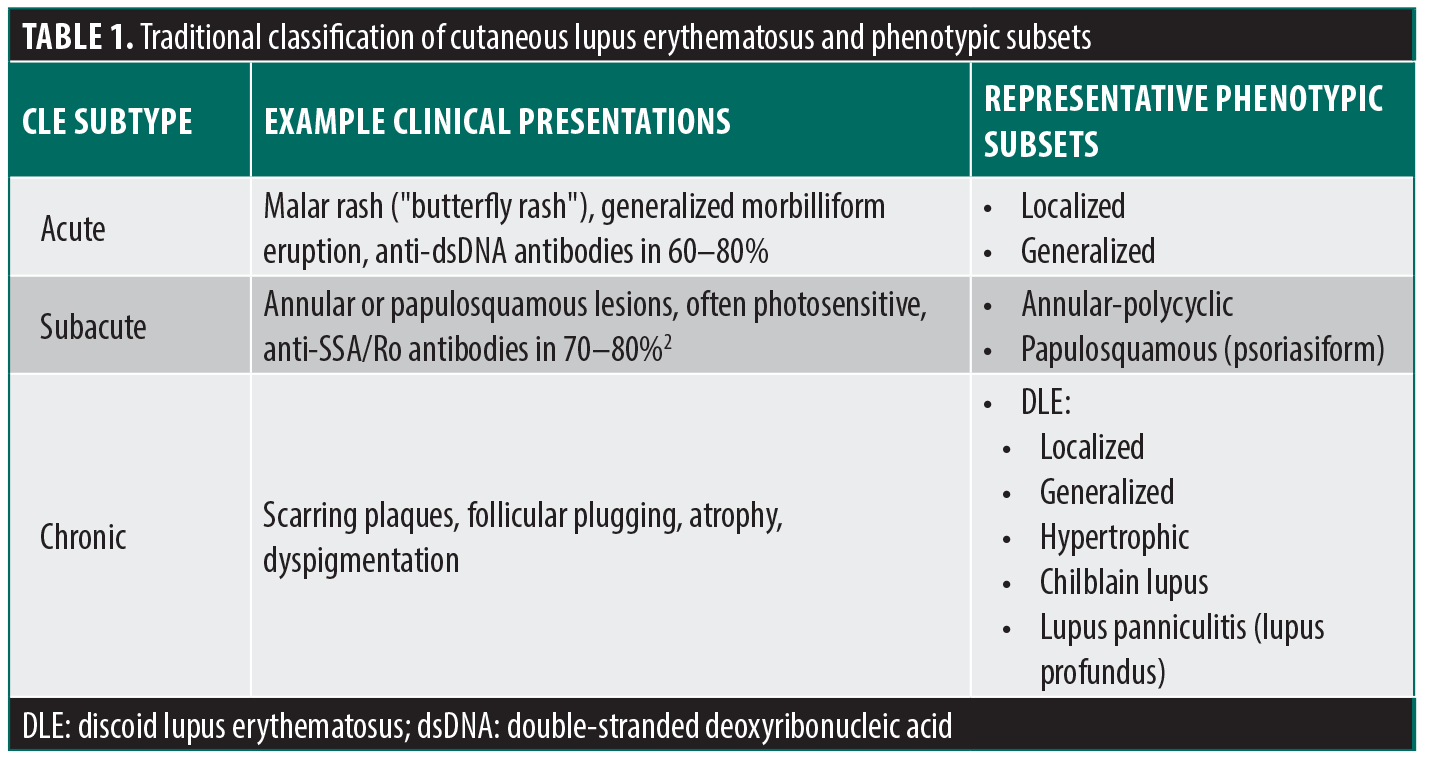
Cutaneous lupus erythematosus (CLE) is a chronic autoimmune skin disease that encompasses a heterogeneous array of clinical subtypes classified as acute, subacute, or chronic. Acute CLE (ACLE) includes manifestations such as malar erythema; subacute CLE (SCLE) is characterized by annular or papulosquamous lesions; and chronic CLE (CCLE) comprises localized, generalized, and hypertrophic forms of discoid lupus erythematosus (DLE) (Table 1).1,2
Despite incremental therapeutic advances, CLE remains a difficult disease to manage. Many patients continue to experience persistent or relapsing disease activity, and current treatment options are limited by teratogenicity, systemic toxicity, and tolerability concerns. These limitations contribute to substantial morbidity, particularly in generalized and hypertrophic DLE subtypes that are often recalcitrant to treatment. In some cases, permanent scarring and disfigurement may further compromise quality of life.3
While clinical guidelines, such as those issued by the British Association of Dermatologists (BAD) in 2021, offer recommendations for first- through third-line therapies, they often fall short in guiding the integration of newly emerging therapies into practice.4 Advances in our understanding of CLE pathogenesis have paved the way for targeted biologic and small molecule therapies designed to modulate specific immune pathways. However, practical frameworks are needed to help clinicians apply these novel and often off-label therapies in real-world care.
This narrative review synthesizes current CLE treatment strategies while highlighting the evolving therapeutic landscape. We focus on emerging biologics and small molecule agents, discuss gaps in existing management frameworks, and consider evolving outcome measures that may better capture clinical benefit in future trials.
Methods
Herein, we provide a relevant literature review and practical update on therapeutic approaches as well as a glimpse into emerging therapies that are certain to change the landscape of CLE treatment and positively impact the lives of patients living with CLE. Additional considerations include an evolving understanding of drug-induced SCLE (DI-SCLE), the potential for biomarker-driven therapy, targeted use of novel interventions (eg, Janus kinase [JAK] inhibitors), and complications such as calcinosis cutis.
Our synthesis draws upon PubMed-indexed literature from the past 15 years, with emphasis on clinical trials, mechanistic studies, and real-world evidence. Expert insights from dermatology and rheumatology helped contextualize emerging therapies within current clinical practice and inform perspectives on evolving standards of care.
Results
Current treatment guidelines. General management. Current treatment guidelines for CLE, using the BAD guidelines as a model, emphasize a stepwise approach beginning with general management strategies such as photoprotection, smoking cessation, vitamin D supplementation, evaluation for drug-induced CLE (DI-CLE) (especially in SCLE) and screening for systemic lupus erythematosus (SLE), often in collaboration with rheumatology. For limited or localized disease, potent topical corticosteroids remain as first-line treatment, with topical calcineurin inhibitors as steroid-sparing options, especially for sensitive areas. Intralesional corticosteroids may be used for persistent, isolated lesions. Systemic therapy is used for more extensive, disseminated, and/or refractory disease, with antimalarials (especially hydroxychloroquine [HCQ]) as the backbone of therapy. Antimalarials are considered first-line agents for several reasons including efficacy, relatively favorable safety and tolerability profiles, low cost, and additional benefits to SLE prevention and treatment. Alternative antimalarial agents, often used in HCQ-resistant disease, include quinacrine and chloroquine.4 Some studies suggest that chloroquine can be effective in a subset of patients who have failed HCQ. In one cohort, all 3 patients initiated on chloroquine after HCQ failure demonstrated clinical response, including one who had also failed HCQ-quinacrine combination therapy.5 Additional reports have described similar benefit, supporting chloroquine as a viable second-line antimalarial option in select cases.5,6
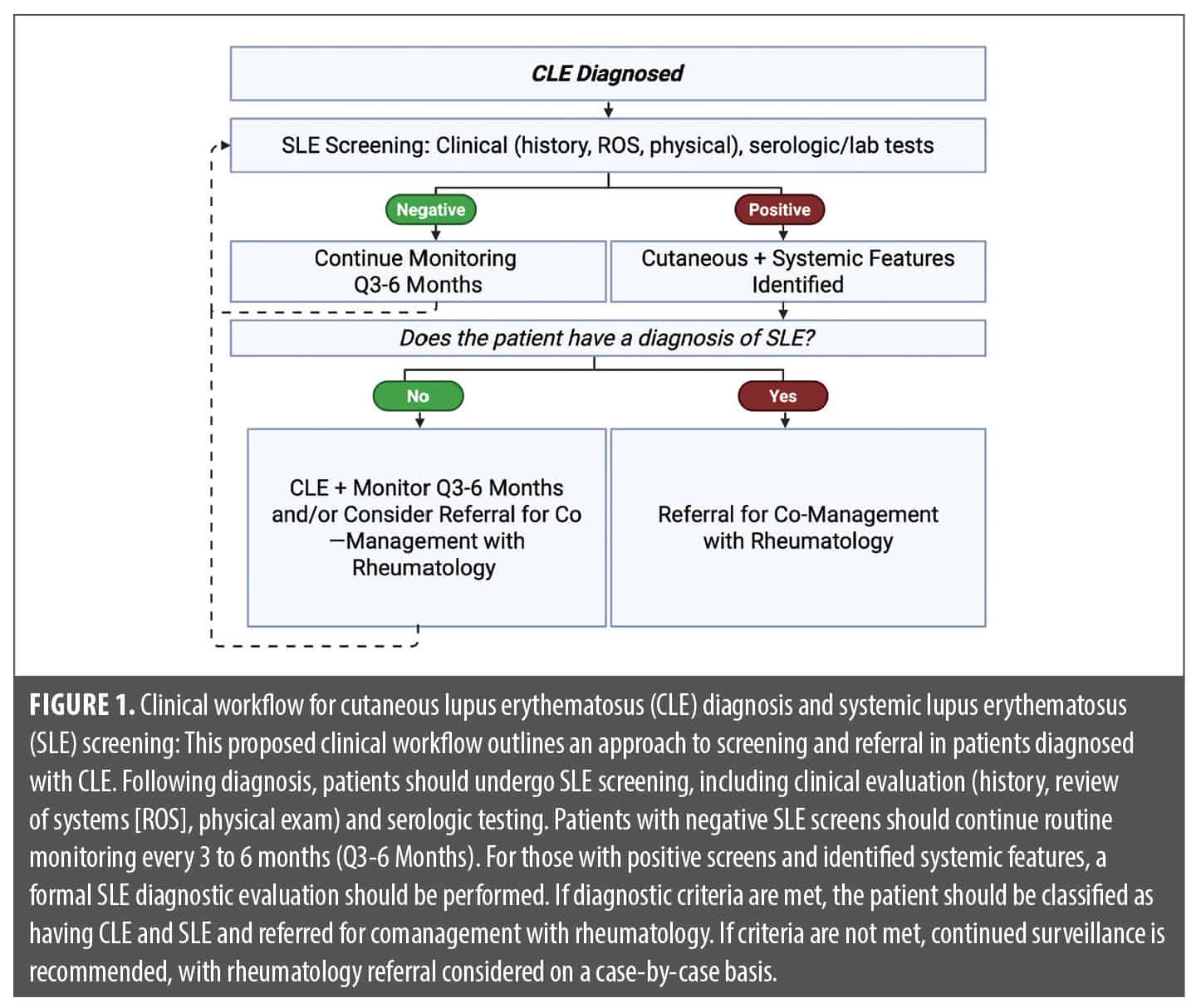
Short courses of systemic corticosteroids may be used for severe flares, although less commonly and typically with less success than when utilized for SLE end-organ disease flares. If antimalarials fail and/or in severe disease, second-line agents such as methotrexate (MTX), mycophenolate mofetil (MMF), dapsone, and acitretin have historically been considered next. In refractory cases, third-line options typically include thalidomide, lenalidomide, belimumab, and intravenous immunoglobulin (IVIG), with mixed case series supporting use of anifrolumab, rituximab, deucravacitinib, litifilimab, plasmapheresis or photopheresis, and baricitinib.4,7-12 The shifting therapeutic landscape of emerging agents is already impacting current real-world use, as further discussed in subsequent sections. The comanagement of patients with a rheumatology specialist is advised among patients who screen positive for SLE or who have evolving features of SLE (Figure 1).
HCQ monitoring. HCQ blood-level monitoring is a valuable tool to assess adherence and guide therapeutic dosing. Approximately 10% of patients may have undetectable HCQ levels, indicating potential non-adherence and contributing to treatment failure. Additionally, concerns about retinal toxicity have led to conservative dosing guidelines from the American Academy of Ophthalmology. However, this has raised concerns about underdosing and subtherapeutic levels. Blood-level monitoring offers a strategy to ensure patients receive effective doses while minimizing long-term ocular risk, ultimately supporting more personalized and effective use of HCQ in CLE.13,14
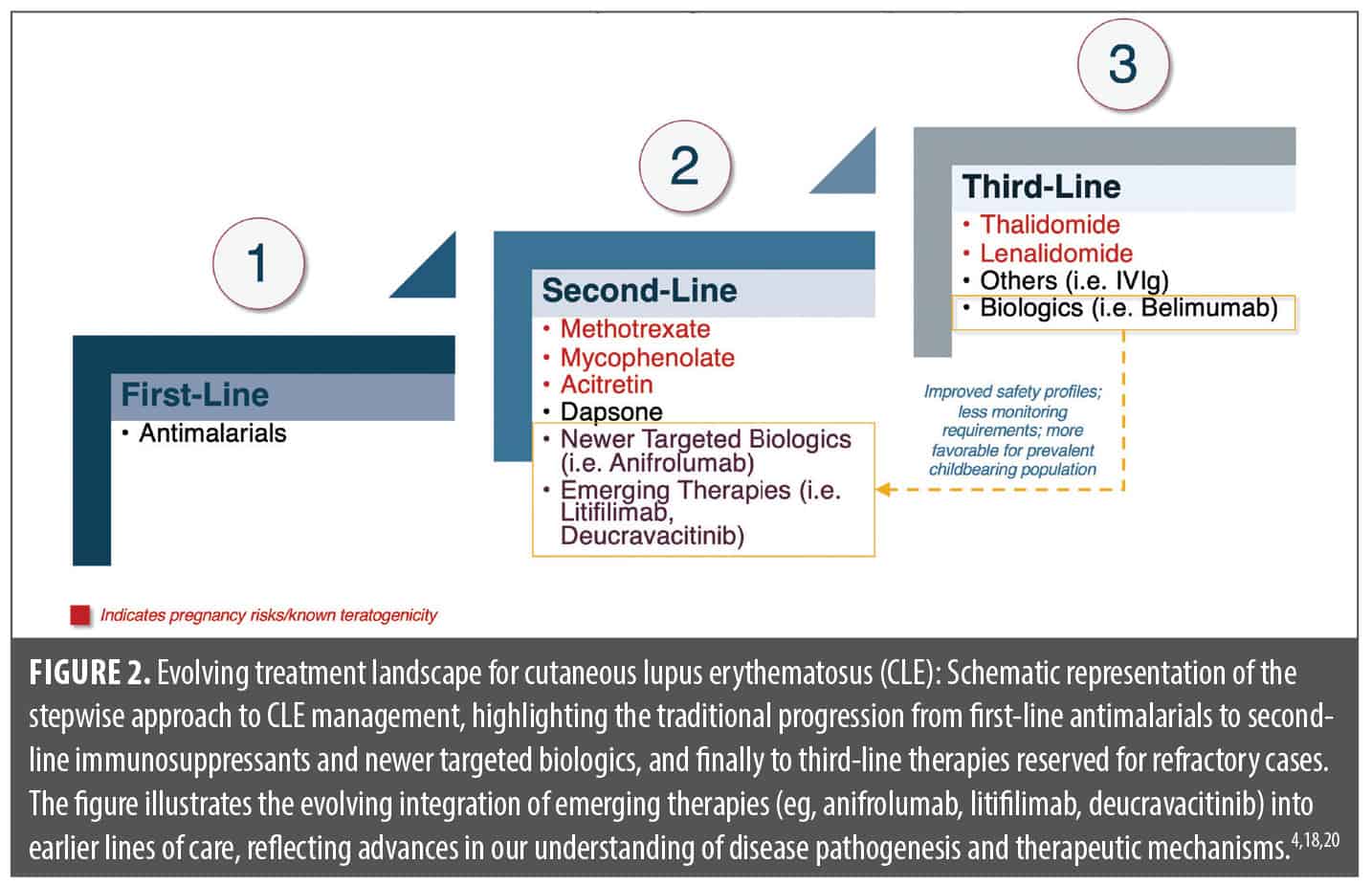
Emerging treatment paradigm. The treatment paradigm for CLE is undergoing a significant transformation driven by advances in immunologic understanding and drug development. Although still considered most typically after failure of traditional oral agents such as MTX, MMF, azathioprine, and acitretin due to their novelty and limited comparative data, novel targeted biologics are increasingly adopted as second-line options in clinical practice with promising efficacy and safety profiles. Agents such as anifrolumab (US Food and Drug Administration [FDA]-approved for SLE), deucravacitinib (FDA-approved for psoriasis and in trials for CLE/SLE, with emerging off-label use in case series), and litiflimab (not FDA approved, currently in late-phase trials) are increasingly being used as second-line options, often replacing or complementing traditional immunosuppressants such as MTX, MMF, dapsone, and acitretin (Figure 2).9,15-17
This shift is particularly relevant for women of childbearing potential, in whom traditional agents—such as methotrexate, mycophenolate, acitretin, thalidomide, and lenalidomide—may be contraindicated due to known teratogenicity. This is especially important given that SLE is at least six times more prevalent in women than in men, with the highest incidence occurring among women of childbearing age.16 In these scenarios, biologics with more favorable reproductive safety profiles, such as belimumab and anifrolumab, are often preferred, particularly as new safety data unfolds.17,18 Agents such as IVIG, thalidomide, and lenalidomide are typically reserved for highly refractory cases.
This evolving approach reflects a broader movement toward safer, more targeted therapies in CLE, mirroring movements in other immune-mediated dermatoses. Increasingly, clinicians should consider not only disease subtype and severity but also safety, tolerability, comorbid conditions, and the feasibility of drug monitoring. As these agents increasingly enter clinical use, integrating treatment decisions with broader systemic considerations becomes essential, especially in patients with CLE who exhibit or develop features of SLE. To support practical implementation, we include Table 2, aligning CLE-directed therapies with their corresponding roles in SLE end-organ management, including arthritis, nephritis, interstitial lung disease, and hypercoagulability.8,11,19,20 This approach reflects the growing need for interdisciplinary care and therapeutic decision-making incorporating not only cutaneous severity but also systemic involvement, reproductive safety, comorbid conditions, and long-term feasibility.
Although promising, these newer therapies also introduce challenges related to access, long-term safety, and clinical integration. High costs, insurance coverage limitations, and restricted distribution pathways (eg, intravenous [IV]-only or specialty pharmacy access) may hinder widespread use. Additionally, as most pivotal trials have limited duration and selective inclusion criteria, long-term safety and durability of response in diverse patient populations remain incompletely understood. Clinicians must also navigate how best to incorporate these agents into existing treatment algorithms, particularly in patients with overlapping systemic disease or partial response to traditional immunosuppressants.
Novel developments in tracking disease activity. Importance of outcome measures. Historically, the lack of standardized, skin-specific outcome measures has limited therapeutic progress in CLE and created uncertainty for regulatory approval. Recent efforts by groups such as International Dermatology Outcome Measures (IDEOM), Outcome Measures in Rheumatology (OMERACT), the Lupus Research Alliance, and Monash University (TRM-SLE) have led to the development of consensus-driven tools, including a proposed working core outcome set, to evaluate disease activity more consistently.21,22
The Cutaneous Lupus Erythematosus Disease Area and Severity Index (CLASI), particularly the activity component (CLASI-A), and the Investigator Global Assessment for Cutaneous Lupus (CLA-IGA and the revised CLA-IGA-R) have emerged as leading instruments for registrational trials.21 A multistage literature review and expert-led process culminated in the proposal of a working core outcome set for CLE. This domain set includes: (1) skin-specific disease activity, (2) skin-specific disease damage, (3) Investigator’s Global Assessment of disease activity, (4) symptoms (including itch, pain, and photosensitivity), (5) health-related quality of life (HRQOL), and (6) patient global assessment of disease activity.21,22
At the 5th International Conference on Cutaneous Lupus Erythematosus, experts reached consensus on a core domain set for clinical trials, with strong support for including both CLASI and CLA-IGA-R as skin-specific outcome measures, despite varied preferences regarding optimal thresholds.21 The validation of CLA-IGA-R represents a key advance toward more consistent, clinician-friendly assessments.23 These tools are essential to improving clinical trial design, aligning with regulatory expectations, and ultimately bringing effective therapies to patients. Recent consensus supports the CLASI as the registrational primary endpoint for trials.21
Capturing disease activity in CLE with RNA tape sampling. Novel biomarker-based approaches such as ribonucleic acid (RNA) tape sampling offer promising tools for non-invasive assessment of disease activity in CLE. This technique captures gene expression signatures directly from lesional skin, enabling longitudinal monitoring of interferon (IFN)-driven and CLE-associated transcriptional programs.24 By providing molecular characterization of disease activity over time, RNA tape sampling may enhance diagnostic accuracy, facilitate earlier detection of flares, and eventually serve as a complementary outcome measure in clinical trials.
DLE classification criteria: a step toward standardized subtype identification. Efforts to establish subtype-specific diagnostic criteria have led to the development of a validated classification algorithm for DLE. This scoring system incorporates clinical features such as atrophic scarring (3 points), location in the conchal bowl (2 points), predilection for the head and neck (2 points), dyspigmentation (1 point), follicular hyperkeratosis or plugging (1 point), and erythematous-violaceous hue (1 point). A total score of 5 or more classifies a case as DLE with 84.1% sensitivity and 75.9% specificity, while a more stringent threshold of 7 or more yields 73.9% sensitivity and 92.9% specificity.25 Adoption of these criteria may support more accurate and consistent subtype classification in both clinical and research settings.
EHR-based algorithms for CLE identification: real-world research. As interest grows in leveraging electronic health records (EHRs) and real-world data for CLE research, algorithm development has become essential to accurately identify patients across large datasets.21,26 A recent study from Brigham and Women’s Hospital evaluated the positive predictive value (PPV) of various EHR-based case definitions. Requiring at least three CLE-related diagnostic codes, with at least one assigned by a dermatologist, yielded the highest PPV.26 Findings from this study underscore the value of dermatology-coded diagnoses and provide a validated framework for identifying CLE cohorts in claims-based and EHR-linked research, opening new opportunities for real-word studies and insights.27
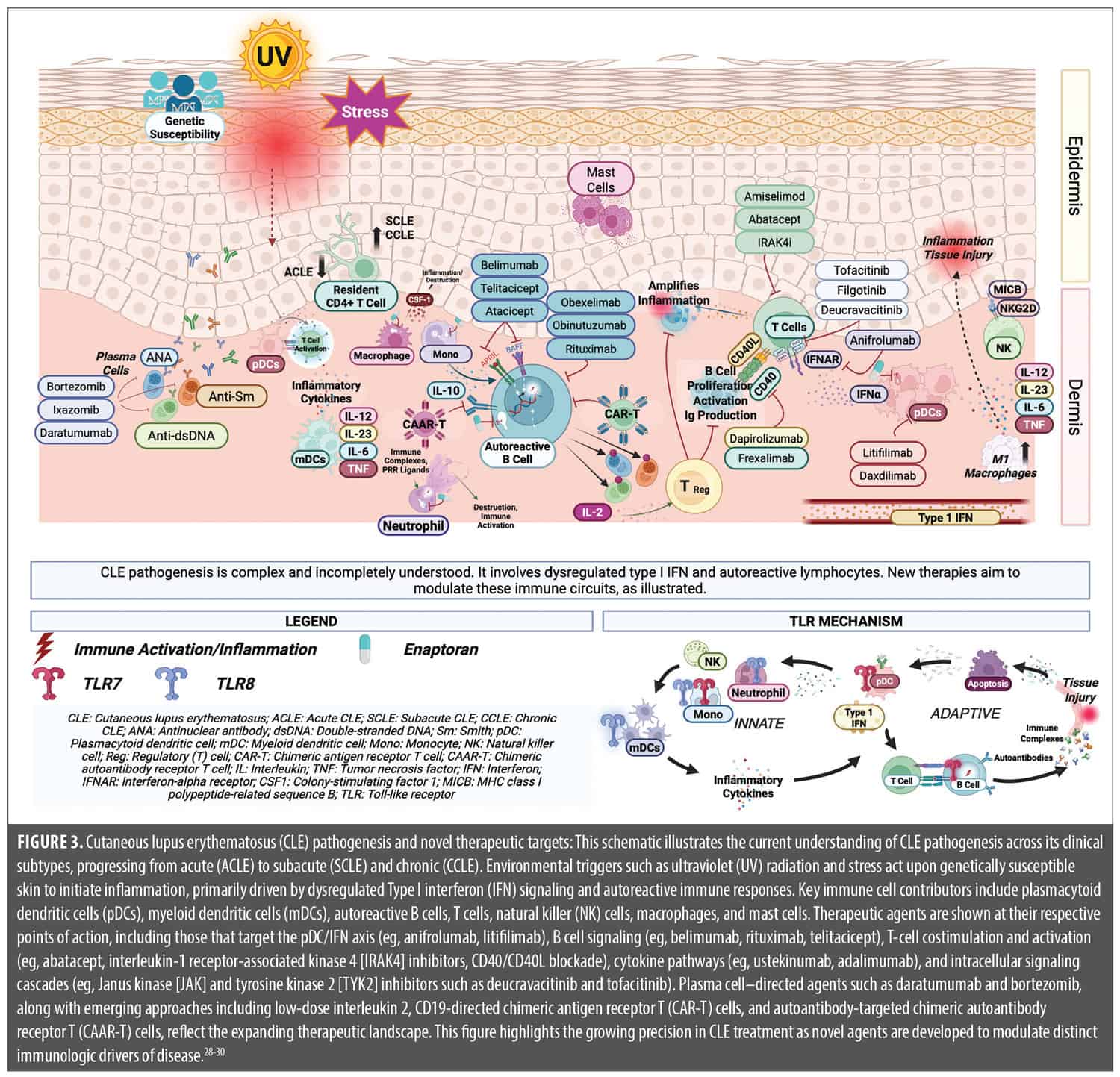
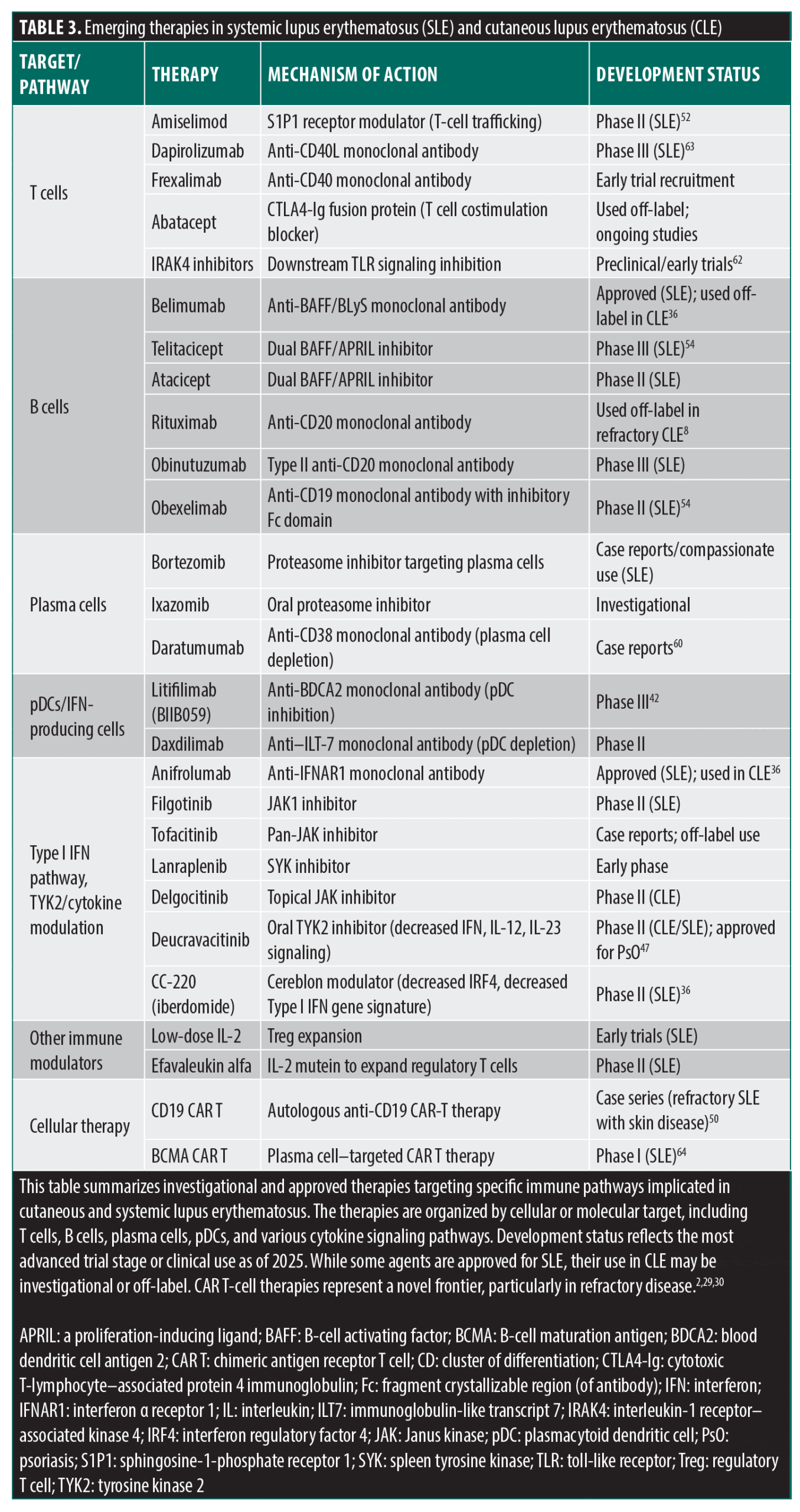
Novel and pipeline therapies. The therapeutic landscape of CLE is transforming, driven by advances in our mechanistic understanding of disease pathogenesis. Nonspecific effects and long-term toxicity of conventional immunosuppressants underscore the need for more selective, targeted interventions. Recent and ongoing clinical trials have yielded promising agents that modulate critical immune pathways implicated in SLE and CLE, such as Type I IFN signaling, B-cell activation, and T-cell costimulation. This section highlights key therapies at the forefront of this paradigm shift, including recently approved agents such as belimumab and anifrolumab, and late-stage investigational drugs such as litifilimab and deucravacitinib. Emerging immunotherapeutic approaches such as chimeric antigen receptor T-cell (CAR T) and chimeric autoantibody receptor T-cell (CAAR T) therapy, though currently under investigation primarily in SLE and lupus nephritis, may have future implications for select CLE subsets as mechanistic understanding evolves (Figure 3).28-30 These therapies reflect a growing movement toward personalized, pathway-specific treatment strategies in CLE.
Belimumab. Belimumab is a monoclonal antibody targeting B-lymphocyte stimulator (BLyS), approved for the treatment of SLE. Although not specifically assessed in CLE, it has been used off-label in select cases. The pivotal trials were not designed with validated skin-specific outcome measures, limiting conclusions about its efficacy in CLE. Post hoc analyses of the British Isles Lupus Assessment Group (BILAG) index suggest modest benefit in mucocutaneous disease, particularly at higher doses.31 Additionally, real-world data describing patients with refractory CLE across acute, subacute, and chronic subtypes, with and without SLE, suggest that many experience at least partial clinical benefit from belimumab.32,33 While not routinely used as a primary agent for CLE, belimumab may have a role in refractory cases, particularly in patients with concomitant systemic disease.
Anifrolumab. Anifrolumab, a monoclonal antibody targeting the Type I IFN receptor, represents a promising advancement in CLE treatment. Unlike previous SLE trials that lacked skin-specific endpoints, the TULIP-1 and TULIP-2 Phase III studies incorporated the CLASI-A to assess skin response. Pooled analyses demonstrated that among patients with baseline CLASI-A scores ≥10, 60–70% achieved at least a 50% reduction in disease activity after one year—substantially outperforming placebo.34 These data offer high-level evidence supporting a pathogenesis-driven therapy specifically for CLE that modulates its underlying Type I IFN activity. Clinical benefit has also been observed in severe, treatment-refractory cases, including a recent case report describing near-complete skin clearance in a patient with SCLE unresponsive to conventional agents such as methotrexate and thalidomide.35,36 Recent case series have also reported promising outcomes in patients with refractory DLE.37,38 In one prospective, multicenter study of patients with biopsy-proven CLE unresponsive to at least three systemic therapies, including belimumab, all patients achieved a 50% reduction in CLASI-A (CLASI-A-50) at Week 16, highlighting the potential utility of anifrolumab even in highly treatment-resistant disease.39 Although anifrolumab is currently available only through IV administration, Phase III trials of subcutaneous (SC) dosing are underway, which may expand accessibility.40 Notably, while generally well tolerated, real-world use has been associated with serious infections, disease flares, and reports of end-organ involvement rebound with discontinuation.41 These findings underscore the therapeutic potential of IFN blockade in CLE, particularly in subacute and chronic forms, and highlight the importance of incorporating validated skin-specific outcome measures in clinical trials.
Litifilimab. Litifilimab (formerly BIIB059) is a monoclonal antibody targeting blood dendritic cell antigen 2 (BDCA2), a receptor selectively expressed on plasmacytoid dendritic cells (pDCs), which are key producers of Type I IFN in CLE. By inhibiting pDC activation, litifilimab suppresses upstream drivers of Type I IFN production and downstream proinflammatory cytokines central to CLE pathogenesis.10 A Phase II randomized controlled trial (LILAC) demonstrated significant clinical benefit; patients receiving litifilimab showed clear reductions in disease activity compared to placebo, with a favorable safety and tolerability profile.42,43 Phase III trials are ongoing, focusing on patients with DLE and SCLE with or without systemic lupus, and are expected to further define its role as a targeted immunomodulator in skin-limited disease.
Deucravacitinib. Deucravacitinib, an oral selective tyrosine kinase 2 (TYK2) inhibitor currently approved for psoriasis, is under investigation for a range of connective tissue diseases, including SLE and CLE. By modulating the TYK2 signaling axis, deucravacitinib reduces Type I IFN, interleukin (IL)-12, and IL-23 activity—cytokines implicated in CLE pathogenesis.44 In the Phase II PAISLEY trial for active SLE, post hoc analyses showed that deucravacitinib led to marked improvements in skin disease as measured by the CLASI-A. Notably, a substantial proportion of patients with baseline skin involvement achieved high thresholds of response, with approximately 60% achieving CLASI-50 and almost 25% achieving CLASI-100 at Week 48.15
Building on this, the dedicated PAISLEY CLE trial evaluated deucravacitinib 3mg and 6mg twice daily versus placebo in patients with DLE and/or SCLE, with or without concurrent SLE.45 Among 74 randomized patients with moderate-to-severe disease (CLASI ≥8), those treated with deucravacitinib achieved significantly greater mean percentage reductions in CLASI-A scores at Week 16 (–47.5% for 3mg and –50.0% for 6mg vs. 28.4% with placebo), meeting the primary endpoint. Rates of CLASI 50 response were significantly higher in both deucravacitinib arms (56.7% and 52.3%) compared to placebo (19.0%), and numerically greater improvements were observed as early as Week 4 and sustained through Week 52. Skin pain, a patient-reported outcome, also improved with deucravacitinib.46
Importantly, deucravacitinib was well-tolerated with no treatment discontinuations due to adverse events, and no serious safety signals emerged. These data underscore its potential as a well-tolerated oral alternative to traditional immunosuppressants and injectable biologics in CLE, supporting ongoing Phase III trials (POETYK SLE: NCT05617677, NCT05620407).46,47
A recent comparative review further underscored the potential of deucravacitinib, suggesting it may offer superior CLASI improvements compared to both anifrolumab and litifilimab.47 Thus, deucravacitinib may constitute a compelling oral therapeutic option in CLE, particularly for patients seeking alternatives to traditional and injectable agents.
Enpatoran. Enpatoran is a novel oral small molecule inhibitor of toll-like receptors 7 and 8 (TLR7/8), key innate immune sensors implicated in the pathogenesis of both CLE and SLE. By targeting these receptors, enpatoran is designed to disrupt Type I IFN-driven inflammation at its upstream source. In the Phase II WILLOW trial (NCT05162586), enpatoran demonstrated a significant dose-dependent reduction in cutaneous disease activity among patients with CLE or mild SLE and active skin involvement (CLASI-A≥8), achieving the primary endpoint at Week 16 (p=0.0002). Up to 91.3% of patients receiving enpatoran achieved CLASI-50, and up to 60.9% achieved CLASI-70, compared to 38.5% and 11.5% in the placebo arm, respectively.48
Enpatoran was generally well-tolerated, although higher doses were associated with increased rates of treatment-emergent adverse events, primarily infections.48 Thus, enpatoran is poised to become a promising oral therapeutic candidate for CLE, particularly for patients with moderate-to-severe skin disease who are unresponsive to current therapies.
CAR T- and CAAR T-cell therapy. CAR T therapy, traditionally developed for hematologic malignancies, is now gaining momentum as a potential treatment for autoimmune diseases. In SLE with severe systemic or renal involvement, CAR T therapy has shown promise in targeting autoreactive B cells and plasma cells, which are central to autoantibody production and immune complex–mediated tissue damage.49 Most investigations have focused on anti-cluster of differentiation (CD) 19 and anti–B-cell maturation antigen (BCMA) CAR T therapy constructs, with early-phase trials and case series demonstrating durable clinical remission in patients with severe, treatment-refractory disease.50 Recent reviews highlight the transformative potential of CAR T therapy in autoimmune disease, emphasizing its ability to induce sustained immunologic reprogramming.51 While CLE has not been a primary target in these investigations, patients with systemic disease and prominent skin involvement may also experience cutaneous improvement as part of a broader clinical remission.
Emerging innovations such as CAAR T offer a more targeted approach. CAAR T cells are engineered to selectively recognize and eliminate B cells that produce specific pathogenic antibodies without affecting the broader B-cell repertoire. Although still in early development, CAAR T therapy may hold particular promise for patients with refractory CLE in the context of systemic involvement.51 Ongoing studies will be critical in defining the long-term efficacy, safety, and durability of response, but early findings suggest CAR T and CAAR T therapies could represent a paradigm-shifting option for a subset of patients with lupus for whom conventional therapies remain inadequate.
Other innovative approaches. Beyond the therapies discussed in prior sections, a broad arsenal of investigational agents is emerging in SLE and CLE, reflecting the increasing focus on precision immunomodulation. These therapies target diverse cellular and cytokine pathways implicated in CLE pathogenesis and may expand future treatment options.
Several agents modulate the activity of T cells, B cells, or plasma cells. T-cell–directed therapies include amiselimod (a sphingosine-1-phosphate receptor modulator), dapirolizumab (anti-CD40L), frexalimab (anti-CD40), and efavaleukin alfa (a low-dose IL-2 mutein that expands regulatory T cells).52 B-cell–targeting therapies beyond belimumab and rituximab include telitacicept (a dual B-cell activating factor [BAFF]/ a proliferation-inducing ligand [APRIL] inhibitor), atacicept (another dual BAFF/APRIL inhibitor) and obinutuzumab (a Type II anti-CD20 monoclonal antibody).36,53,54 Plasma cell–depleting agents such as bortezomib and ixazomib have shown potential in refractory lupus, though their use remains largely investigational. Additional modulation of pDCs is being explored with daxdilimab, which depletes pDCs via
anti–IL-T7 targeting.2
Therapies borrowed from other skin conditions such as eczema and psoriasis that disrupt pathogenic cytokine signaling include JAK and spleen tyrosine kinase (SYK) inhibitors such as filgotinib (JAK1), tofacitinib (pan-JAK), lanraplenib (SYK), delgocitinib (topical JAK), R333 (topical JAK3/SYK), and GSK246264 (JAK1/TYK2).36,55-57 While not all have been studied specifically in CLE, and some have had mixed results, their mechanisms target relevant inflammatory pathways. Agents such as low-dose IL-2, which plays an immunoregulatory role via regulatory T-cell expansion, also hold promise.57,58
A number of novel immunomodulators are in development, including CC-220 (iberdomide), a cereblon modulator that reduces IRF4 and Type I IFN activity; daratumumab, an anti-CD38 monoclonal antibody targeting plasma cells; and abatacept (cytotoxic T-lymphocyte-associated protein 4 immunoglobulin [CTLA4-Ig]), which modulates T-cell costimulation.59-61 Interleukin-1 receptor-associated kinase 4 (IRAK4) inhibitors, anti–IL-6 receptor antibodies, and obexelimab (an anti-CD19 monoclonal antibody with an inhibitory fragment crystallizable [Fc] region domain) are also under investigation.62 Additional agents targeting sphingosine-1-phosphate receptor 1 (S1P1) signaling may offer new strategies to modulate lymphocyte trafficking.30,58
These investigational therapies, as summarized in Table 3, reflect a shift toward mechanism-driven treatment in CLE, targeting underlying immune dysregulation to improve symptomatic control and patient outcomes.
Special considerations. DI-SCLE. DI-SCLE represents a subset of CLE that is important to consider, particularly in patients with new-onset, widespread, or atypical skin lesions.65 Numerous medications have been implicated in the development of SCLE, with distinct patterns across drug classes. Cardiovascular agents such as hydrochlorothiazide, calcium channel blockers, angiotensin-converting enzyme (ACE) inhibitors, beta blockers, and statins are among the most commonly reported culprits. Other frequently associated drug classes include proton pump inhibitors (omeprazole, pantoprazole, lansoprazole), antifungals (terbinafine), oncologic agents (capecitabine, paclitaxel, docetaxel, tamoxifen, anastrozole, uracil-tegafur, leuprolide), tumor necrosis factor α (TNF-α) inhibitors, immune checkpoint inhibitors, and various others including leflunomide, lamotrigine, bupropion, ranitidine, and acebutolol.66-68
Clinically, DI-SCLE often presents with features that distinguish it from idiopathic SCLE. It is more likely to exhibit a widespread distribution (odds ratio [OR]: 66.1), bullous or erythema multiforme–like morphology (OR: 65), and vasculitic changes (OR: 32).69 In contrast to idiopathic SCLE, DI-SCLE is often transient, with anti-Ro/SSA antibodies disappearing after clinical resolution in approximately 73% of cases. However, tissue eosinophilia, mucin deposition, and the nature of the inflammatory infiltrate do not reliably differentiate DI-SCLE from idiopathic SCLE.70
Given these clinical distinctions, it is critical to maintain a high index of suspicion for drug-induced disease, particularly in patients presenting with new or atypical SCLE in the context of recent medication exposure. Prompt recognition and withdrawal of the offending agent can lead to complete remission and prevent unnecessary escalation of immunosuppressive therapy.
Biomarker-guided therapy and JAK inhibition in myogenic and IFN-driven lupus. As our understanding of CLE pathogenesis deepens, biomarker-driven approaches are emerging to guide therapeutic selection, particularly in genetically defined or IFN-driven subtypes. Familial chilblain lupus, an early-onset form of cutaneous lupus linked to mutations in the TREX1 gene, exemplifies the therapeutic potential of this strategy. These patients typically exhibit elevated Type I IFN signatures, making JAK inhibition a reasonable targeted intervention. Case series support successful administration of baricitinib, 4mg daily, in patients with TREX1-associated familial chilblain lupus with marked clinical improvement.71 Similarly, isolated reports suggest that refractory chilblain lesions in patients with systemic lupus have responded favorably to tofacitinib (11mg daily), reinforcing the link between interferon signaling and disease activity in CLE.72
These cases underscore a broader trend toward individualized treatment based on genetic or molecular profiling. As transcriptomic and genomic tools become more integrated into clinical care, therapies like JAK inhibitors may be increasingly deployed in a precision medicine framework, particularly in patients with high IFN signatures or poor response to conventional therapies.
Calcinosis cutis: new solutions to a challenging problem. Calcinosis cutis, though uncommon in CLE, presents a formidable treatment challenge when it occurs, particularly in association with lupus panniculitis. Characterized by the deposition of insoluble calcium salts in skin and subcutaneous tissue, calcinosis may cause pain, ulceration, and secondary infection, significantly impairing quality of life. To date, there are no consistently effective treatments, and management remains largely supportive. However, small case series and anecdotal reports have suggested potential benefit from intralesional sodium thiosulfate injections, a treatment more commonly employed in calciphylaxis.73-75 In the context of lupus panniculitis–associated calcinosis, this approach has shown success in softening lesions and reducing discomfort.76
Despite these isolated advances, the treatment of calcinosis cutis remains an unmet need in CLE. Future studies aimed at elucidating the underlying pathophysiology of dystrophic calcification in lupus could help identify novel targets and improve therapeutic outcomes for this debilitating complication.
Conclusion
CLE remains a clinically heterogeneous and therapeutically challenging disease, complicated by variable phenotypes, limited approved treatments, and a lack of skin-specific outcome measures. However, a new era of targeted therapy is rapidly emerging, informed by advances in immunopathogenesis, biomarker discovery, and translational research. From Type I IFN blockade and TYK2 inhibition to cellular therapies and precision-guided immunomodulation, novel agents are reshaping the therapeutic landscape of CLE. Parallel efforts to standardize clinical trial endpoints and leverage real-world data are critical to accelerating drug development and ensuring regulatory clarity. As the field moves toward mechanism-based treatment and personalized care, dermatologists will play a central role in translating these innovations into improved outcomes for patients living with CLE.
References
- Gilliam JN, Sontheimer RD. Distinctive cutaneous subsets in the spectrum of lupus erythematosus. J Am Acad Dermatol. 1981;4(4):471-475.
- Wenzel J. Cutaneous lupus erythematosus: new insights into pathogenesis and therapeutic strategies. Nat Rev Rheumatol. 2019;15(9):519-532.
- Zhang AJ, Ezeh N, Childs B, et al. Cutaneous lupus matters: independent and additive quality-of-life burden shown by 2022 WLFGI Survey. J Eur Acad Dermatol Venereol. 2025;39(10):e908-e911.
- O’Kane D, McCourt C, Meggitt S, et al. British Association of Dermatologists guidelines for the management of people with cutaneous lupus erythematosus 2021. Br J Dermatol. 2021;185(6):1112-1123.
- Chang AY, Piette EW, Foering KP, Tenhave TR, Okawa J, Werth VP. Response to antimalarial agents in cutaneous lupus erythematosus: a prospective analysis. Arch Dermatol. 2011;147(11):1261-1267.
- Chasset F, Bouaziz JD, Costedoat-Chalumeau N, Frances C, Arnaud L. Efficacy and comparison of antimalarials in cutaneous lupus erythematosus subtypes: a systematic review and meta-analysis. Br J Dermatol. 2017;177(1):188-196.
- Bao A, Petri MA, Fava A, Kang J. Case series of anifrolumab for treatment of cutaneous lupus erythematosus and lupus-related mucocutaneous manifestations in patients with SLE. Lupus Sci Med. 2023;10(2):e001007.
- Quelhas da Costa R, Aguirre-Alastuey ME, Isenberg DA, Saracino AM. Assessment of of response to B-cell depletion using rituximab in cutaneous lupus erythematosus. JAMA Dermatol. 2018;154(12):1432-1440.
- Zhang A, Gaffney RG, Merola JF. Treatment of recalcitrant lupus erythematosus tumidus with deucravacitinib. JAAD Case Rep. 2024;45:110-112.
- Cho SK, Vazquez T, Werth VP. Litifilimab (BIIB059), a promising investigational drug for cutaneous lupus erythematosus. Expert Opin Investig Drugs. 2023;32(5):345-353.
- Frankel HC, Sharon VR, Vleugels RA, Merola JF, Qureshi AA. Lower-dose thalidomide therapy effectively treats cutaneous lupus erythematosus but is limited by neuropathic toxicity. Int J Dermatol. 2013;52(11):1407-1409.
- Morand EF, Vital EM, Petri M, et al. Baricitinib for systemic lupus erythematosus: a double-blind, randomised, placebo-controlled, phase III trial (SLE-BRAVE-I). Lancet. 2023;401(10381):1001-1010.
- Chasset F, Arnaud L, Costedoat-Chalumeau N, Zahr N, Bessis D, Frances C. The effect of increasing the dose of hydroxychloroquine (HCQ) in patients with refractory cutaneous lupus erythematosus (CLE): an open-label prospective pilot study. J Am Acad Dermatol. 2016;74(4):693-9.e3.
- Francès C, Cosnes A, Duhaut P, et al. Low blood concentration of hydroxychloroquine in patients with refractory cutaneous lupus erythematosus: a French multicenter prospective study. Arch Dermatol. 2012;148(4):479-484.
- Morand E, Pike M, Merrill JT, et al. Deucravacitinib, a tyrosine kinase 2 inhibitor, in systemic lupus erythematosus: a phase II, randomized, double-blind, placebo-controlled trial. Arthritis Rheumatol. 2023;75(3):242-252.
- Feldman CH, Hiraki LT, Liu J, et al. Epidemiology and sociodemographics of systemic lupus erythematosus and lupus nephritis among US adults with Medicaid coverage, 2000-2004. Arthritis Rheum. 2013;65(3):753-763.
- Liu L, Ma X, Liu J, et al. Role of belimumab in recurrent spontaneous abortions amongst patients with lymphocyte dysfunction: a retrospective case-control study. BMC Pregnancy Childbirth. 2025;25(1):463.
- Correia S, Katus U, Huda S, et al. AB1125 real-world safety of anifrolumab in patients with SLE: a multinational program of secondary database post authorization safety studies. Ann Rheum Dis. 2024;83(suppl 1):1894-1895.
- Richter P, Cardoneanu A, Dima N, et al. Interstitial lung disease in systemic lupus erythematosus and systemic sclerosis: how can we manage the challenge? Int J Mol Sci. 2023;24(11):9388.
- Katarzyna PB, Wiktor S, Ewa D, Piotr L. Current treatment of systemic lupus erythematosus: a clinician’s perspective. Rheumatol Int. 2023;43(8):1395-1407.
- Zhang AJ, Perez-Chada LM, Werth VP, Merola JF. Expert consensus achieved on a working core outcome set for cutaneous lupus erythematosus research in survey following the 5th International Conference on Cutaneous Lupus Erythematosus (ICCLE). Lupus Sci Med. 2024;11(1):e001165.
- Guo LN, Perez-Chada LM, Borucki R, Nambudiri VE, Werth VP, Merola JF. Development of a working core outcome set for cutaneous lupus erythematosus: a practical approach to an urgent unmet need. Lupus Sci Med. 2021;8(1):e000529.
- Merola JF, Zhang AJ, Childs BA, et al. Inter-rater and intra-rater reliability of the Cutaneous Lupus Activity-Investigator Global Assessment-Revised Instrument. J Invest Dermatol. 2025;145(8):2085-2088.e2.
- Merola JF, Wang W, Wager CG, et al. RNA tape sampling in cutaneous lupus erythematosus discriminates affected from unaffected and healthy volunteer skin. Lupus Sci Med. 2021;8(1):e000428.
- Elman SA, Joyce C, Braudis K, et al. Creation and validation of classification criteria for discoid lupus erythematosus. JAMA Dermatol. 2020;156(8):901-906.
- Guo LN, Said JT, Woodbury MJ, Nambudiri VE, Merola JF. Development and validation of algorithms to identify individuals with cutaneous lupus from healthcare databases. J Cutan Med Surg. 2025;29(2):131-136.
- Guo LN, Said J, Nambudiri VE, Merola JF. Development and validation of algorithms to identify cutaneous lupus patients using diagnostic codes and prescription data. Abtract presented at: American College of Rheumatology Convergence 2022; November 10-14, 2022; Philadelphia, PA. Abstract 0138.
- Ramaswamy M, Tummala R, Streicher K, Nogueira da Costa AN, Brohawn PZ. The pathogenesis, molecular mechanisms, and therapeutic potential of the interferon pathway in systemic lupus erythematosus and other autoimmune diseases. International J Mol Sci. 2021;22(20):11286.
- Soto JA, Melo-González F, Riedel CA, Bueno SM, Kalergis AM. Modulation of of immune cells as a therapy for cutaneous lupus erythematosus. Int J Mol Sci. 2022;23(18):10706.
- Liossis SN, Staveri C. What’s new in the treatment of systemic lupus erythematosus. Front Med (Lausanne). 2021;8:655100.
- Manzi S, Sanchez-Guerrero J, Merrill JT, et al. Effects of belimumab, a B lymphocyte stimulator-specific inhibitor, on disease activity across multiple organ domains in patients with systemic lupus erythematosus: combined results from two phase III trials. Ann Rheum Dis. 2012;71(11):1833-1838.
- Salle R, Chasset F, Kottler D, et al. Belimumab for refractory manifestations of cutaneous lupus: a multicenter, retrospective observational study of 16 patients. J Am Acad Dermatol. 2020;83(6):1816-1819.
- Zen M, Gatto M, Depascale R, et al. Early and late response and glucocorticoid-sparing effect of belimumab in patients with systemic lupus erythematosus with joint and skin manifestations: results from the Belimumab in Real Life Setting Study-Joint and Skin (BeRLiSS-JS). J Pers Med. 2023;13(4):691.
- Morand EF, Furie RA, Bruce IN, et al. Efficacy of anifrolumab across organ domains in patients with moderate-to-severe systemic lupus erythematosus: a post-hoc analysis of pooled data from the TULIP-1 and TULIP-2 trials. Lancet Rheumatol. 2022;4(4):e282-e292.
- Gaffney RG, Merola JF. Rapid response of refractory subacute cutaneous lupus after single dose anifrolumab. JAAD Case Rep. 2024;44:71-73.
- Xie L, Lopes Almeida Gomes L, Stone CJ, Faden DF, Werth VP. An update on clinical trials for cutaneous lupus erythematosus. J Dermatol. 2024;51(7):885-894.
- Aljohani R. Anifrolumab for refractory discoid lupus: two case reports of successful outcomes in Saudi Arabia. Medicine (Baltimore). 2025;104(20):e42518.
- Hulin M, Le Seac’h A, Jaume L, et al. Progressive repigmentation of hypopigmented lesions in discoid lupus erythematosus with anifrolumab: a report of two cases. Lupus. 2025;34(7):761-763.
- Chasset F, Jaume L, Mathian A, et al; EMSED (Etude des maladies systéiques en dermatologie). Rapid efficacy of anifrolumab in refractory cutaneous lupus erythematosus. J Am Acad Dermatol. 2023;89(1):171-173.
- Tummala R, Rouse T, Berglind A, Santiago L. Safety, tolerability and pharmacokinetics of subcutaneous and intravenous anifrolumab in healthy volunteers. Lupus Sci Med. 2018;5:e000252.
- Chatham WW, Furie R, Saxena A, et al. Long-term safety and efficacy of anifrolumab in adults with systemic lupus erythematosus: results of a phase II open-label extension study. Arthritis Rheumatol. 2021;73(5):816-825.
- Werth VP, Furie RA, Romero-Diaz J, et al; LILAC Trial Investigators. Trial of anti-BDCA2 antibody litifilimab for cutaneous lupus erythematosus. N Engl J Med. 2022;387(4):321-331.
- Werth VP, Furie RA, Romero-Diaz J, et al. Part B of the LILAC study of litifilimab for cutaneous lupus erythematosus: a plain language summary. Immunotherapy. 2025;17(3):161-173.
- Hile GA, Werth VP. Understanding the role of type I interferons in cutaneous lupus and dermatomyositis: toward better therapeutics. Arthritis Rheumatol. 2025;77(1):1-11.
- Bosworth T. Phase II trial supports TYK2 inhibitor for cutaneous lupus, paving way for pivotal trials. WebMD, LLC. Published April 1, 2025. Accessed June 26, 2025. https://www.medscape.com/viewarticle/phase-2-trial-supports-tyk2-inhibitor-cutaneous-lupus-paving-2025a10007sp
- Merola JF, Gottlieb AB, Aranow C, et al. Efficacy and safety of oral deucravacitinib in patients with cutaneous manifestations of lupus erythematosus: results from PAISLEY CLE, a global, randomized, placebo-controlled, phase II trial. Ann Rheum Dis. 2025;84(suppl 1):940-941.
- Bokor LA, Martyin K, Krebs M, et al. Deucravacitinib shows superior efficacy and safety in cutaneous lupus erythematosus compared to various biologics and small molecules – a systematic review and meta-analysis. Autoimmun Rev. 2025;24(3):103723.
- Pearson D, Morand E, Wenzel J, et al. Randomized, placebo-controlled phase II study of enpatoran, a small molecule toll-like receptor 7/8 inhibitor, in cutaneous lupus erythematosus: results from cohort A. J Rheum. 2025;52(suppl 1):11.
- Uppin V, Gibbons H, Troje M, Feinberg D, Webber BR, Moriarity BS, Parameswaran R. CAR-T cell targeting three receptors on autoreactive B cells for systemic lupus erythematosus therapy. J Autoimmun. 2025;151:103369.
- Mackensen A, Müller F, Mougiakakos D, et al. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nat Med. 2022;28(10):2124-2132.
- Zhou J, Lei B, Shi F, et al. CAR T-cell therapy for systemic lupus erythematosus: current status and future perspectives. Front Immunol. 2024;15:1476859.
- Tanaka Y, Kondo K, Ichibori A, et al. Amiselimod, a sphingosine 1-phosphate receptor-1 modulator, for systemic lupus erythematosus: a multicenter, open-label exploratory study. Lupus. 2020;29(14):1902-1913.
- Ma X, Fu X, Cui B, Lin H. Telitacicept for recalcitrant cutaneous manifestations of systemic lupus erythematosus: a case report and review of the literature. Tohoku J Exp Med. 2022;258(3):219-223.
- Merrill JT, Guthridge J, Smith M, et al. Obexelimab in systemic lupus erythematosus with exploration of response based on gene pathway co-expression patterns: a double-blind, randomized, placebo-controlled, phase II trial. Arthritis Rheumatol. 2023;75(12):2185-2194.
- Maruyama A, Katoh N. Subacute cutaneous lupus erythematosus successfully treated with topical delgocitinib. J Dermatol. 2023;50(3):e110-e111.
- Werth VP, Fleischmann R, Robern M, et al. Filgotinib or lanraplenib in moderate to severe cutaneous lupus erythematosus: a phase II, randomized, double-blind, placebo-controlled study. Rheumatology (Oxford). 2022;61(6):2413-2423.
- Presto JK, Okon LG, Feng R, et al. Computerized planimetry to assess clinical responsiveness in a phase II randomized trial of topical R333 for discoid lupus erythematosus. Br J Dermatol. 2018;178(6):1308-1314.
- Niebel D, de Vos L, Fetter T, Brägelmann C, Wenzel J. Cutaneous lupus erythematosus: an update on pathogenesis and future therapeutic directions. Am J Clin Dermatol. 2023;24(4):521-540.
- Serrano-Benavente B, Bello N, Nieto-González J, et al. AB0406 experience from abatacept use in systemic lupus erythematosus: effectiveness and safety: table 1. Ann Rheum Dis 2016;75(suppl 2):1045.
- Ostendorf L, Burns M, Durek P, et al. Targeting CD38 with daratumumab in refractory systemic lupus erythematosus. N Engl J Med. 2020;383(12):1149-1155.
- Werth VP, Merrill JT, Furie R, et al. Effect of iberdomide on cutaneous manifestations in systemic lupus erythematosus: a randomized phase II clinical trial. J Am Acad Dermatol. 2025;92(3):435-443.
- Dudhgaonkar S, Ranade S, Nagar J, et al. Selective IRAK4 inhibition attenuates disease in murine lupus models and demonstrates steroid sparing activity. J Immunol. 2017;198(3):1308-1319.
- Askanase A, Clarke A, Gladman DD, et al. Improvements observed in skin and joint manifestations of systemic lupus erythematosus with dapirolizumab pegol treatment: results from a phase III trial. J Rheum. 2025;52(suppl 1):10-11.
- Wang W, He S, Zhang W, et al. BCMA-CD19 compound CAR T cells for systemic lupus erythematosus: a phase I open-label clinical trial. Ann Rheum Dis. 2024;83(10):1304-1314.
- Marzano AV, Vezzoli P, Crosti C. Drug-induced lupus: an update on its dermatologic aspects. Lupus. 2009;18(11):935-940.
- Lowe GC, Henderson CL, Grau RH, Hansen CB, Sontheimer RD. A systematic review of drug-induced subacute cutaneous lupus erythematosus. Br J Dermatol. 2011;164(3):465-72.
- Cabanillas M, Suárez-Amor O, Ramírez-Santos A, et al. Lamotrigine induced subacute cutaneous lupus erythematous. Dermatol Online J. 2012;18(8):12.
- Merola JF. Drug-induced lupus. In: Connor RF, Pisetsky DS, Callen JP, Case SM, eds. UpToDate. Wolters Kluwer; 2025.
- Marzano AV, Lazzari R, Polloni I, Crosti C, Fabbri P, Cugno M. Drug-induced subacute cutaneous lupus erythematosus: evidence for differences from its idiopathic counterpart. Br J Dermatol. 2011;165(2):335-341.
- Hillesheim PB, Bahrami S, Jeffy BG, Callen JP. Tissue eosinophilia: not an indicator of drug-induced subacute cutaneous lupus erythematosus. Arch Dermatol. 2012;148(2):190-193.
- Zimmermann N, Wolf C, Schwenke R, et al. Assessment of clinical response to Janus kinase inhibition in patients with familial chilblain lupus and TREX1 mutation. JAMA Dermatol. 2019;155(3):342-346.
- Elman SA, Mazori DR, Merola JF. Tofacitinib for refractory chilblain lupus erythematosus. Int J Dermatol. 2022;61(4):e156-e157.
- Benandi K, Sieving D, Bumgardner A, Wolf K. Undiluted 25% intralesional sodium thiosulfate in the management of dystrophic calcinosis cutis. JAAD Case Rep. 2024;52:8-10.
- Le Q, Robinson A, Stevens W, Li J. Recalcitrant digital calcinosis cutis successfully treated with intralesional sodium thiosulfate under digital nerve blockade. BMJ Case Rep. 2022;15(4):e248707.
- Tubau C, Cubiró X, Amat-Samaranch V, Garcia-Melendo C, Puig L, Roé-Crespo E. Clinical and ultrasonography follow-up of five cases of calcinosis cutis successfully treated with intralesional sodium thiosulfate. J Ultrasound. 2022;25(4):995-1003.
- Gunasekera NS, Maniar LEG, Lezcano C, Laga AC, Merola JF. Intralesional sodium thiosulfate treatment for calcinosis cutis in the setting of lupus panniculitis. JAMA Dermatol. 2017;153(9):944-945.