J Clin Aesthet Dermatol. 2026;19(3):47–49.
by Eingun James Song, MD, FAAD, and Abarajithan Chandrasekaran, BS
Dr. Song is with Frontier Dermatology, Mill Creek, Washington. Mr. Chandrasekaran is with the University of Arizona, College of Medicine – Phoenix, Phoenix, Arizona.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Song reports relationships with AbbVie, Amgen, Arcutis, Bristol Myers Squibb, Boehringer Ingelheim, Dermavant, Incyte, Janssen, Lilly, Novartis, Pfizer, Sanofi & Regeneron, Sun Pharma, and UCB. Mr. Chandrasekaran has no relevant conflicts of interest.
ABSTRACT: Cutaneous lichenoid drug eruptions (LDE) are a group of relatively uncommon adverse drug reactions characterized by widespread erythematous to violaceous scaly papules that can be difficult to differentiate from idiopathic lichen planus. Their increasing prevalence with the use of biologics, tyrosine kinase inhibitors, and immune checkpoint inhibitors has become of particular interest in dermatology. More commonly used medications, including antibiotics, antihypertensives, and nonsteroidal anti-inflammatory drugs, have also been reported to cause LDEs. While the exact pathophysiology of LDE remains to be fully elucidated, certain medications are believed to increase the antigenicity of skin proteins, thereby triggering a T-cell–mediated response via CD8+ cytotoxic T lymphocytes that target basal keratinocytes, while increased interferon γ signaling further amplifies the inflammatory process. Herein, we report a case of LDE induced by an antihistamine (cetirizine) successfully treated with both upadacitinib and dupilumab after failure of systemic corticosteroids. Keywords: Lichenoid drug eruption, adverse drug reaction, lichen planus, immune checkpoint inhibitors, Janus kinase inhibitors
Introduction
Cutaneous lichenoid drug eruptions (LDE) are a group of relatively uncommon adverse drug reactions (ADR) characterized by widespread erythematous to violaceous scaly papules that can be difficult to differentiate from idiopathic lichen planus.1,2 The long latency period after drug exposure (mean: 15.7 weeks) and failure to resolve even after drug cessation add to the diagnostic challenge.1 Moreover, the rise of new therapies such as biologics, tyrosine kinase inhibitors, and immune checkpoint inhibitors (ICIs) has appeared to increase the prevalence of drug-induced lichenoid reactions.3 Other classes of medications that have been implicated include antibiotics, antihypertensives, diuretics, antimalarials, and nonsteroidal anti-inflammatory drugs (NSAIDs).4
Among the many culprit medications, antihistamines are rarely reported to cause LDEs, with only a few published case reports.4,5 Treatment of LDEs involves discontinuation of the offending drug and symptomatic treatment until resolution of the rash.1 Current treatments for lichen planus often lead to suboptimal results.6 Targeted therapies such as Janus kinase (JAK) inhibitors and biologics have been reported in the literature and are attractive due to potentially superior efficacy and safety.7,8 Herein, we present a case of cetirizine-induced LDE in a 40-year-old female patient who failed systemic corticosteroids but was successfully treated with sequential upadacitinib and dupilumab therapy. The patient provided verbal informed consent and photo consent.
Case Presentation
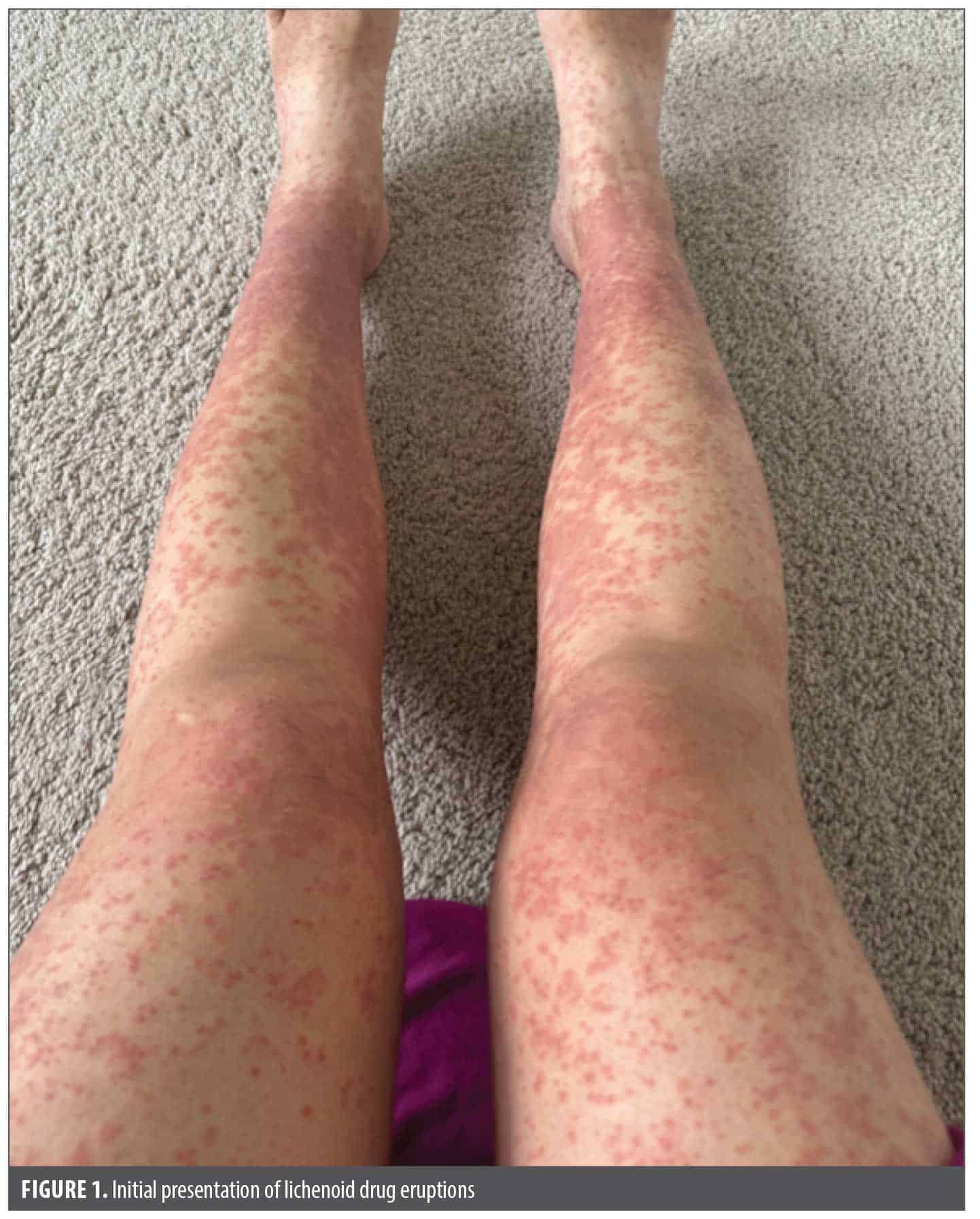
A 40-year-old Asian female patient with a history of mild atopic dermatitis presented with a pruritic rash on the arms and trunk that had progressed over the last few years and was only partially responsive to topical corticosteroids. Patch testing (North American 80 Comprehensive Series) was notable for positivity to fragrance, Balsam of Peru, limonene, colophony, and nickel. The patient reported some improvement with allergen avoidance, but the rash never fully cleared. Approximately 9 months later, she reported a widespread pruritic lichenoid eruption (Figure 1) that looked different from her previous rash. The patient denied any recent illnesses and was not taking any medications other than over-the-counter cetirizine 10 mg daily, which she recently started for allergies, though she had taken it in the past without issue.
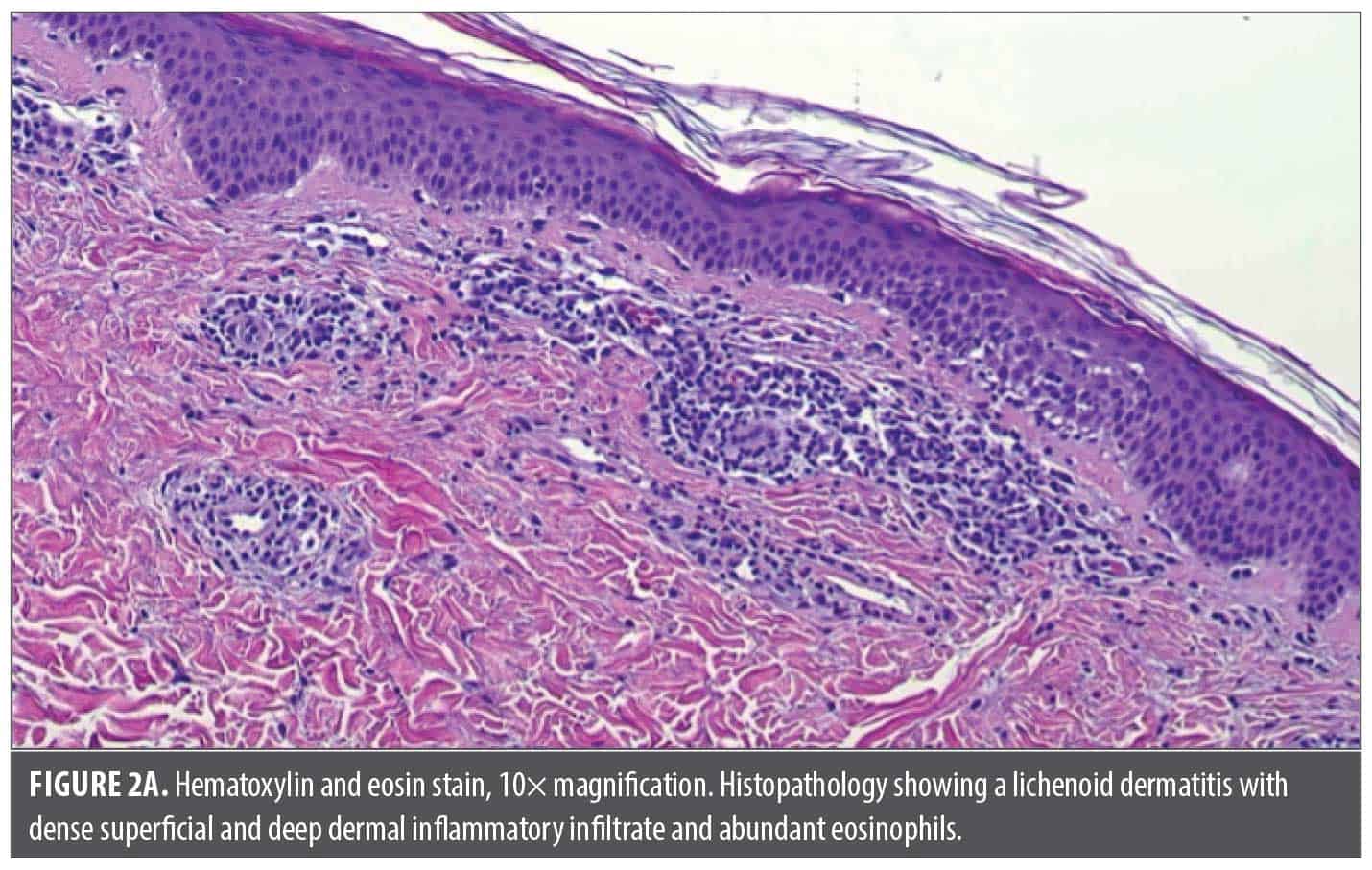
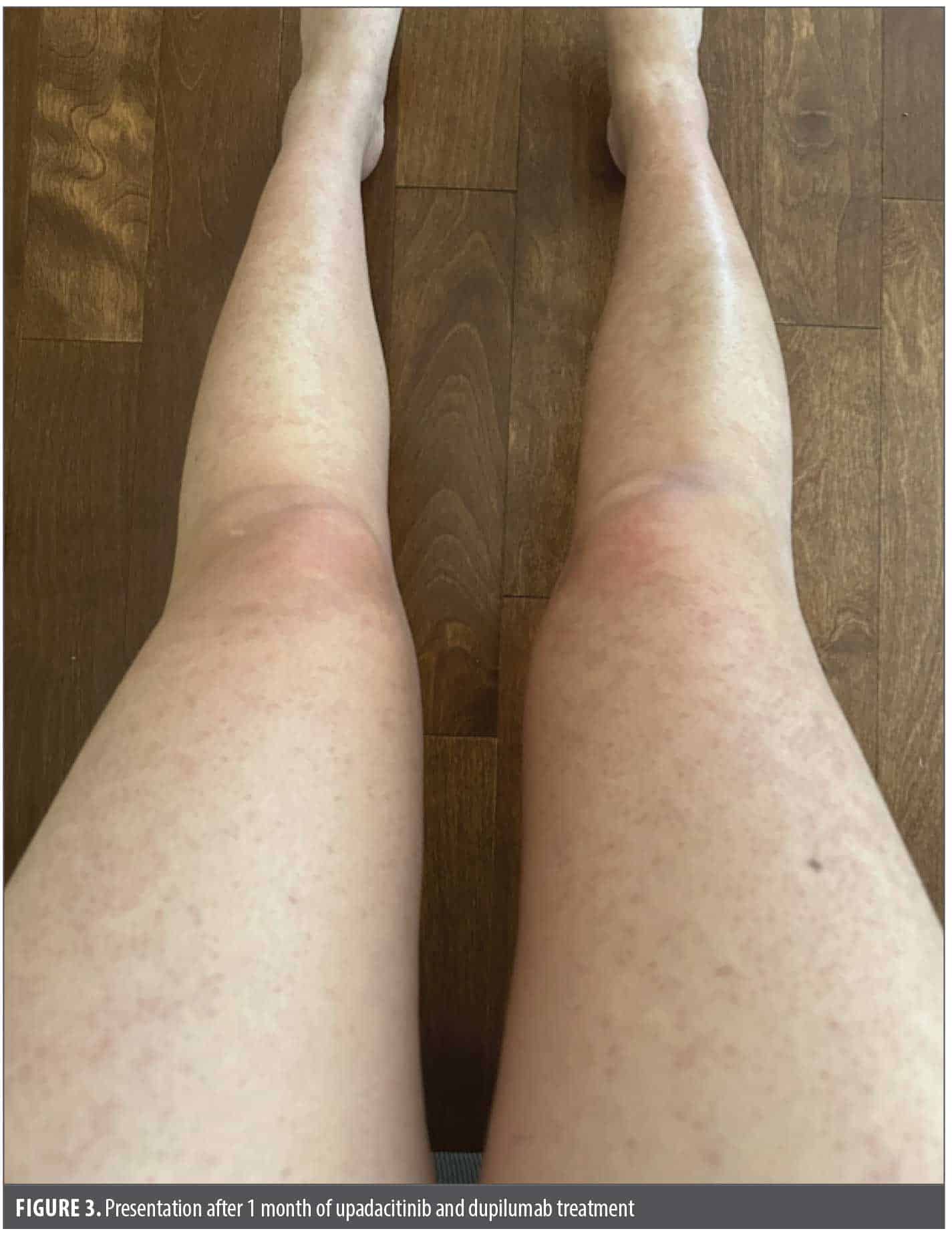
Punch biopsy of the most representative areas showed a lichenoid dermatitis with an abundance of eosinophils, highly suggestive of a drug-induced lichenoid eruption (Figure 2). Cetirizine was discontinued, and the patient was started on prednisone 60 mg daily along with clobetasol cream, 0.05%, twice daily. After 11 days of prednisone treatment, the patient reported marginal improvements in her rash and itch and had difficulty sleeping because of the corticosteroids. At this point, the patient was started on samples of upadacitinib 15 mg daily (after baseline laboratory evaluation) and quickly tapered off prednisone. After 14 days of upadacitinib treatment, she reported her itch had improved from a 6 out of 10 (while on prednisone) to a 1 out of 10, but she still had extensive skin involvement. This prompted a dose escalation of upadacitinib to 30 mg daily. At her 3-week follow-up, her itch remained under good control, but she still had active skin disease. Dupilumab (600-mg loading dose, then 300 mg every 2 weeks) was added, and at her 4-week follow-up, her skin was nearly completely clear except for mild postinflammatory erythema (Figure 3). The patient was subsequently tapered off upadacitinib and dupilumab over 2 additional months and her skin has remained clear.
Discussion
LDEs are becoming an important entity for dermatologists, given their increasing prevalence with the use of ICIs. However, as our case demonstrated, LDEs can occur even with over-the-counter antihistamines. While it is difficult to establish causality, the time course, histopathology, clinical presentation, and absence of any other potential culprits all supported the likelihood that cetirizine was the most likely cause. Although the exact pathogenesis of LDE has yet to be fully elucidated, studies1 have shown that certain medications can alter skin proteins, increase their antigenicity, and thereby trigger a T-cell–mediated response via CD8+ cytotoxic T lymphocytes that target basal keratinocytes, while cytokine dysregulation (increased interferon γ signaling) results in further amplification of the inflammatory loop.1
While most LDEs can be treated with medications typically used for idiopathic lichen planus, there is still a large unmet need for highly effective and safe therapies. Not only was our patient an inadequate responder to high-dose systemic corticosteroids, but she was also unable to tolerate them due to severe sleep disruption. This is particularly relevant in the setting of ICI-induced LDE, given that cutaneous eruptions are often a positive prognostic sign of treatment response, and early use of systemic corticosteroids is associated with poorer clinical outcomes, highlighting the need for nonsteroidal alternatives.9
Several case reports have demonstrated the utility of dupilumab in generalized lichen planus.10,11 Furthermore, dupilumab is now part of the National Comprehensive Cancer Network (NCCN) Guidelines for treating LDEs from ICIs.12 JAK inhibitors have also shown efficacy even in refractory cutaneous, follicular, nail, and mucosal lichen planus, although there are limited data on LDEs.13 There are also reports of combination dupilumab and JAK inhibitor use for refractory cases of atopic dermatitis.14 Given the better overall safety profile of JAK inhibitors compared to traditional immunosuppressants,15 it can be argued that JAK inhibitors and biologics should supplant existing legacy medications, particularly systemic corticosteroids, for LDE treatment.
Admittedly, drug access can be an issue, and patients often have to rely on medication samples. In the author’s (E.J.S.) experience, patients have been able to receive off-label treatment for lichenoid dermatosis in certain cases after a letter of medical necessity and peer-to-peer appeals.
References
- Maul JT, Guillet C, Oschmann A, et al. Cutaneous lichenoid drug eruptions: a narrative review evaluating demographics, clinical features and culprit medications. J Eur Acad Dermatol Venereol. 2023;37(5):965–975.
- Lage D, Juliano PB, Metze K, de Souza EM, Cintra ML. Lichen planus and lichenoid drug-induced eruption: a histological and immunohistochemical study. Int J Dermatol. 2012;51(10):1199–1205.
- Cheraghlou S, Levy LL. Fixed drug eruptions, bullous drug eruptions, and lichenoid drug eruptions. Clin Dermatol. 2020;38(6):679–692.
- Gopal S, Gnanasegaran S, Raj GM, Murugesan S, Adhimoolam M. Cetirizine-induced fixed drug eruption. J Res Pharm Pract. 2018;7(2):111–114.
- Suryana K. Lichenoid reaction caused by antihistamines and corticosteroids. J Asthma Allergy. 2020;13:205–211.
- Boch K, Langan EA, Kridin K, Zillikens D, Ludwig RJ, Bieber K. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Xia M, Zhang J, Tian X, Chen Z, Liang J, Liu Y. Successful treatment of ustekinumab-associated lichenoid drug eruption with upadacitinib: a case report. Clin Cosmet Investig Dermatol. 2025;18:801–804.
- Trum NA, Zain J, Abad C, Rosen ST, Querfeld C. Dupilumab as a therapy option for treatment refractory mogamulizumab-associated rash. JAAD Case Rep. 2021;14:37–42.
- Goodman RS, Johnson DB, Balko JM. Corticosteroids and cancer immunotherapy. Clin Cancer Res. 2023;29(14):2580–2587.
- Kazemi S, Murphrey M, Hawkes JE. Rapid resolution of widespread cutaneous lichen planus and generalized pruritus in an elderly patient following treatment with dupilumab. JAAD Case Rep. 2022;30:108–110.
- Read MH, Swain E, Lovell KK, Feldman SR. Refractory lichen planus: success with dupilumab. Clin Exp Dermatol. 2025;50(6):1204–1205.
- Thompson JA, Schneider BJ, Brahmer J, et al. NCCN Guidelines® Insights: management of immunotherapy-related toxicities, version 2.2024. J Natl Compr Canc Netw. 2024;22(9):582–592.
- Didona D, Caposiena Caro RD, Calabrese L, et al. Use of JAK inhibitors in lichen planus: an update. Medicina (Kaunas). 2025;61(6):1056.
- Shahriari N, Strober B, Shahriari M. JAK-inhibitors as rescue therapy in dupilumab-refractory severe atopic dermatitis: a case series of 6 patients. JAAD Case Rep. 2023;33:81–83.
- Daniele S, Bunick C. JAK inhibitor safety compared to traditional systemic immunosuppressive therapies. J Drugs Dermatol. 2022;21(12):1298–1303.