J Clin Aesthet Dermatol. 2026;19(2):33–42.
by Gabriella Resnick, BA; Megan Khajeh-Afzaly, BS; Faraz Yousefian, DO; Abbas Raza, PhD; and Naiem T. Issa, MD, PhD
Ms. Resnick is with Georgetown University School of Medicine in Washington, District of Columbia. Ms. Khajeh-Afzaly is with Edward Via College of Osteopathic Medicine in Blacksburg, Virginia. Dr. Yousefian is with the University of Incarnate Word in San Antonio, Texas. Dr. Raza is with the University of Minnesota, Cosmetic Science & Product Innovation, Center for Drug Design in Minneapolis, Minnesota. Dr. Issa is with Forefront Dermatology in Vienna, Virginia, the University of Miami Miller School of Medicine, Dr. Phillip Frost Department of Dermatology and Cutaneous Surgery in Miami, Florida, and the Department of Dermatology at George Washington University School of Medicine and Health Sciences in Washington, District of Columbia.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors declare no conflict of interest related to the content of this article.
ABSTRACT: Objective: This systematic review aims to critically assess the literature on the mechanisms of action, clinical uses, formulation strategies, and adverse effects of self-tanning agents, with a focus on dihydroxyacetone (DHA), melanotan, forskolin, and carotenoids. Methods: A systematic literature review was conducted following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. Studies were screened for relevance to the mechanisms of action, clinical applications, chemical formulations, or adverse effects of self-tanning molecules. A total of 68 peer-reviewed studies were included. Results: DHA induces pigmentation via the Maillard reaction and has demonstrated additional dermatologic applications, including use in vitiligo and erythropoietic protoporphyria and as a potential topical antifungal. Concerns persist about DHA-related cytotoxicity, genotoxicity, and systemic absorption. Unregulated melanotan I and II use has caused serious adverse effects, including rhabdomyolysis, renal infarction, and priapism. While forskolin stimulates melanin production independently of melanocortin receptors and has demonstrated efficacy in animal models, carotenoids, when ingested orally, accumulate in skin and subcutaneous fat, creating a yellow-orange hue. Both agents remain underresearched in human populations. Limitations: Limitations include lack of standardized reporting across included studies, variability in study outcomes, and limited long-term safety data. Conclusion: Sunless tanning agents offer UV-free alternatives for cosmetic pigmentation but are not without risk. While DHA and melanotan remain the dominant agents in current use, forskolin and carotenoids offer alternative pathways for pigmentation and photoprotection. Further clinical studies are necessary to evaluate long-term safety, efficacy across skin types, and formulation optimization. Regulatory frameworks and dermatologic guidance must evolve to reflect the expanding landscape of sunless tanning modalities. Keywords: Self-tanners, tanning, dihydroxyacetone, melanotan, forskolin, beta carotene
Over the past 20 years, the popularity of sunless tanners has increased significantly. As awareness of skin cancer has grown, many individuals have shifted toward sunless tanning to achieve the cosmetic effects of a tan without the risks associated with ultraviolet (UV) exposure. This shift is particularly important given that skin cancer is the most common form of cancer in the United States (US), with an estimated $8.1 billion spent annually on treatment.1
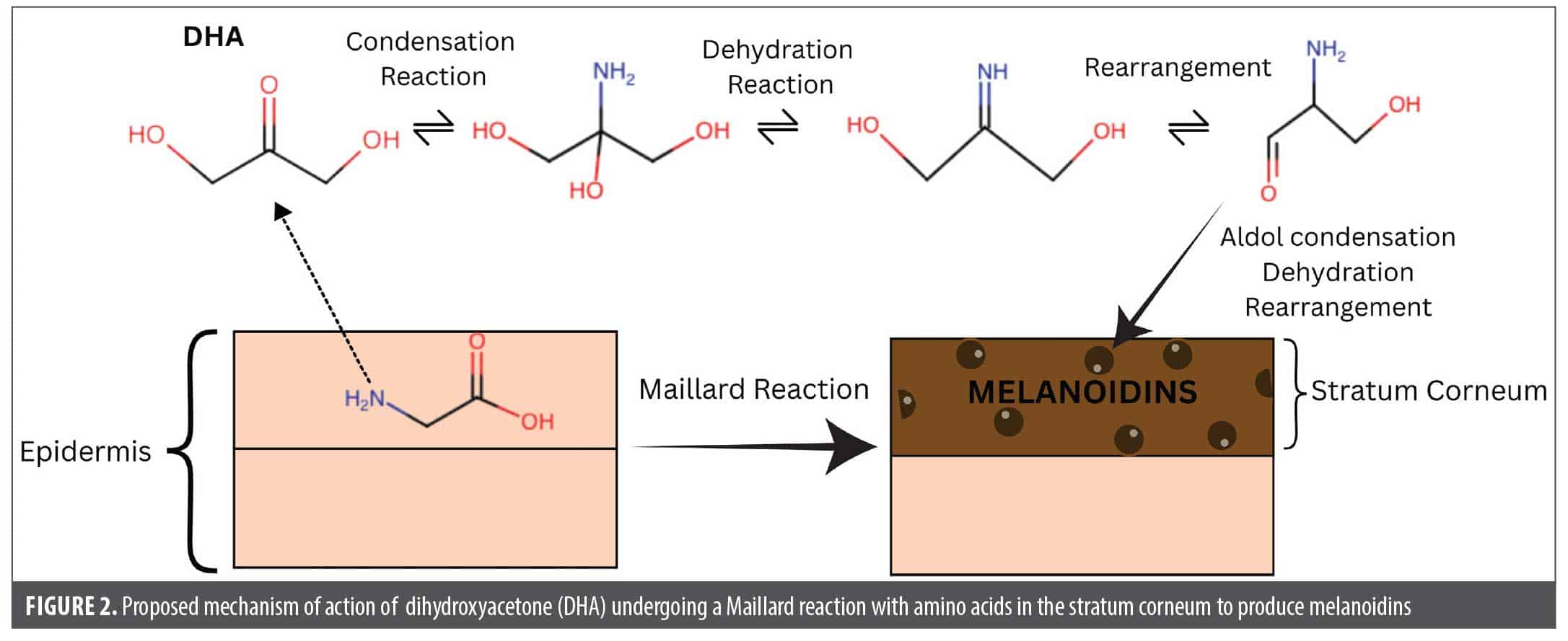
Despite the well-documented risks of UV tanning, a strong societal preference for tanned skin persists. As the sunless tanning industry has expanded, dihydroxyacetone (DHA), a highly reactive, simple 3-carbon sugar, has become the primary active ingredient in most products. DHA was discovered as a skin-browning agent in the 1920s, when healthcare providers noticed localized skin darkening in children being treated orally with DHA for glycogen storage disease.2 Upon application to the skin, DHA undergoes a Maillard reaction: a nonenzymatic chemical reaction between the carbonyl group of a reducing sugar and the free amino group of amino acids or proteins, forming brown-colored polymers known as melanoidins.3 In the context of DHA-containing self-tanners, DHA reacts with free amino acids in the stratum corneum, producing melanoidins and tanning the skin.
There has been little improvement in the longevity of these treatments. Results typically last a few days before reapplication is required. In response to this limitation, there has been a rise in the illicit sale and use of melanotan I and melanotan II synthetic tanning agents.4 Melanotan I and II are synthetic analogs of α-melanocyte-stimulating hormone (α-MSH) and act as nonselective agonists of various melanocortin receptors (MCRs).5 MCRs are a family of five G protein-coupled receptors (GPCRs) expressed in diverse tissues, that have diverse physiological roles including pigmentation, energy homeostasis, feeding, exocrine function, and steroidogenesis.6 Because these compounds are not approved by the US Food and Drug Administration (FDA) for use as tanning agents, there is no formal oversight of their production or distribution.
Other compounds with perceived natural origins and unique modes of action are under investigation. Forskolin, a plant-derived molecule, stimulates melanin production via a melanogenesis pathway independent of UV-exposure and melanocortin receptors in animal models.7 Carotenoids accumulate in the skin and impart a yellow-orange hue, producing the appearance of a tan without affecting existing melanin levels. The extent of these molecules’ efficacy and safety in the context of sunless tanning remains less established.
Given the widespread use of sunless tanning products and the growing safety concerns, a comprehensive evaluation is necessary. This systematic review aims to critically assess the mechanisms of action, usage trends, efficacy, and safety profiles of DHA-based products, synthetic melanocortin analogs, and less conventional agents such as forskolin and carotenoids. By integrating current evidence, we seek to inform clinical practice, guide public health messaging, and identify key areas where further research and regulation are warranted.
Methods
A systematic review of the published literature was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.8 The search strategies incorporated a combination of database-specific subject headings and keywords that represented the concepts of sunless tanning methods.
Studies were included if they met the following criteria: (1) reported data on the mechanism of action (MOA), usage, or adverse effects of self-tanning molecules; (2) written in English; and (3) published in a peer-reviewed journal. There was no restriction on the publication date. Exclusion criteria included studies that were unpublished/abstract only or book chapters and were not relevant to the topic of the review.
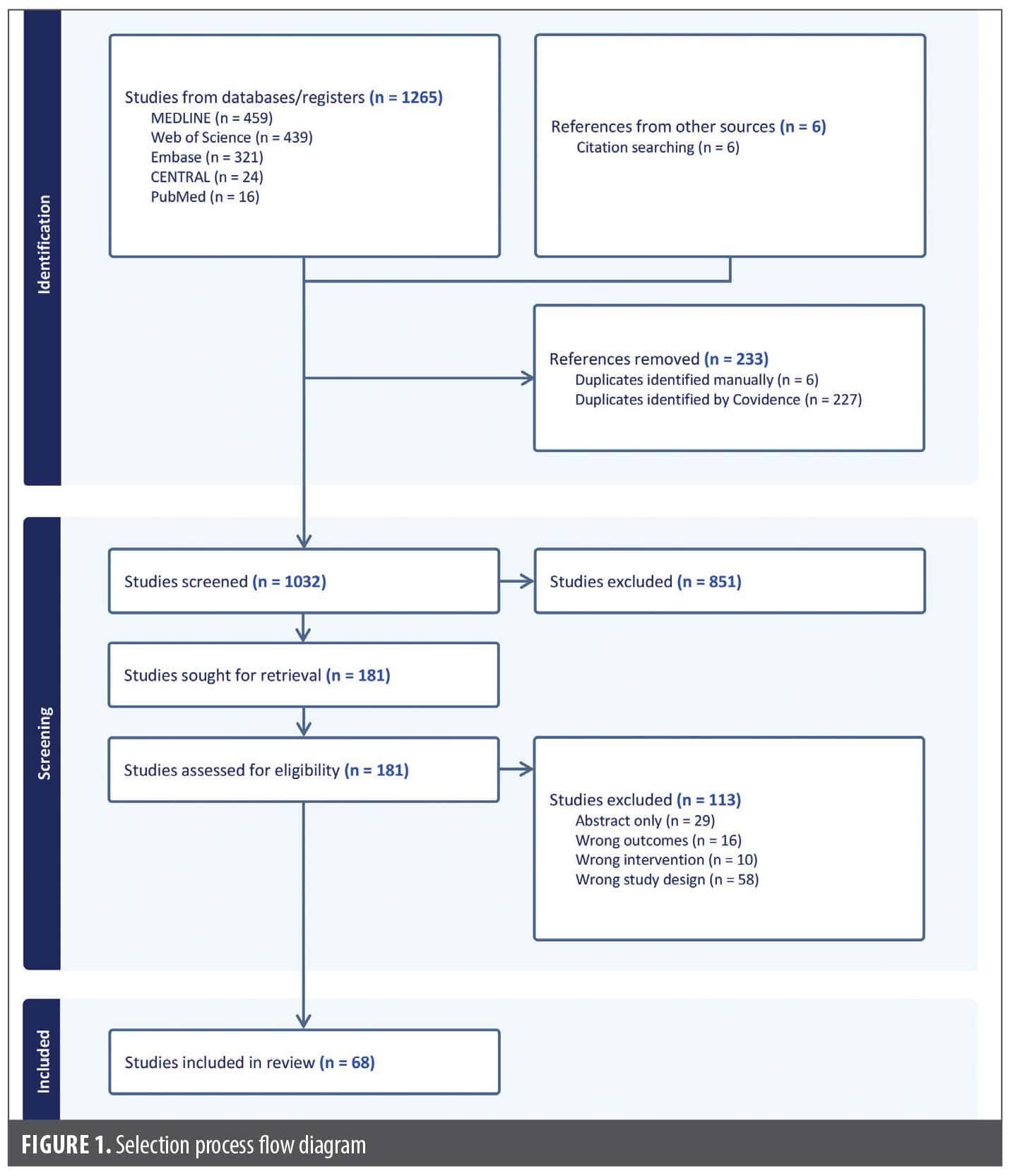
Titles and abstracts were screened for relevance by two investigators (G.F.R. and M.K.). The reference lists of the full-text articles were additionally analyzed for further relevant studies. Data extraction was performed independently by two investigators (G.F.R. and M.K.) to minimize bias. A total of 68 studies meeting the predetermined criteria were included in the analysis (Figure 1).
Results
Use of self-tanners overview. Frequency of sunless tanner use. Several studies with a large number of participants have been conducted to investigate the prevalence of sunless tanner use and associated sun protection practices. In a 2012 study analyzing the sun-protective behaviors of Australian female adolescents, 34.5% of respondents reported use of fake tanning products in the past 12 months.9 Another study conducted among female college students in the US found that 87% of respondents used sunless tanning products, including bronzers, lotions, and spray-on tans.10 Tella et al11 evaluated the artificial tanning behaviors of 713 French teenagers, with 55.9% of respondents declaring that it was important to be tanned during the summer, 9.9% having used tanning creams, and 1% having used tanning pills.
In a 2023 study that analyzed sunless tanning use data from a 2015 survey, 17.7% of women reported sunless tanning, with lotions used more frequently (15.3%) than spray tanning (6.8%).1 Women aged 18 to 29 years had a significantly higher prevalence of sunless tanning (21.2%) compared with women aged 30 to 39 years (16.5%) and 40 to 49 years (14.9%) (p=0.004).1
Sun-protection behaviors. Williams et al9 found that female sunless tanning users were less likely to report wearing a hat, upper body protective clothing, or lower body protective clothing and unsurprisingly experienced more frequent sunburns. No difference was found between fake tan users and nonusers in the use of sunscreen, shade-seeking behavior, wearing sunglasses, and avoidance of peak UV hours.9 While Tella et al11 also found that sunless tanning users practiced less sun-protective behaviors, Russo et al10 found that sunless tanner users were more aware of the negative effects of sun tanning compared to nonusers.
Seidenberg et al1 found that female sunless tanner users had mixed data on engaging in sun-protective behaviors. While women reporting a sunburn in the past year had a significantly higher prevalence of lotion tanning (18.2% vs. 11.3%; p<0.001), women who reported ever having a full-body skin exam had a significantly higher prevalence of lotion tanning (21.0% vs.13.5%; p<0.001). The authors found no significant association between wearing sunscreen and using tanning lotions.1
Allergens in self-tanners. Many self-tanner manufacturers do not participate in the Contact Allergen Management Program database.12 Buonomo and Hylwa13 analyzed 262 sunless tanning products (STPs) in 2021, identifying a total of 36 different allergens, with all 262 STPs containing at least two American Contact Dermatitis Society (ACDS) allergens. Nearly all products (98.9%) contained at least 1 fragrance, with the most common being linalool, limonene, citronellol, coumarin, and benzyl alcohol; a maximum of 12 fragrances was observed in a single product. Additional common ingredients included phenoxyethanol (67.2%), tocopherol (65.7%), and sorbic acid derivatives (56.1%).13 Spray tans contained the fewest allergens per product (5.3), while lotions contained the most (7.1).13 Rigali et al12 analyzed a total of 220 products in 2025, finding a total of 32 different allergens across all products. They found that 73% of products had fragrance/parfum present, with other frequently found fragrance molecules being limonene (40%), linalool (32%), and geraniol (23%). Interestingly, 22 products (10%) had “fragrance-free” on the labeling, yet only 17 were truly fragrance-free upon ingredient review.12
Dihydroxyacetone (DHA). Mechanism of action. DHA is a simple three-carbon saccharide commonly used as the active ingredient in sunless tanning products. After being approved in the late 1970s for topical use in cosmetics at concentrations up to 20%, to date, it is the only legal tanning agent approved by the FDA.14 Upon application to the skin, DHA undergoes a Maillard reaction: the carbonyl group of DHA reacts with amino acids such as arginine, histidine, and lysine in the stratum corneum to produce brown-black chromophores, known as melanoidins, which provide a brown coloration (Figure 2).3 DHA is rapidly absorbed in cells and tissues through an unknown transporter and then converted to dihydroxyacetone phosphate by triose kinase/flavin mononucleotide (FMN) cyclase, after which it can then be incorporated into many different metabolic pathways, including glycolysis.15
Uses of DHA. Beyond its cosmetic use for skin tanning, DHA has been investigated for a range of dermatological applications, including as a treatment adjunct for vitiligo, visible light shielding for erythropoietic protoporphyria (EPP), and antifungal activity.
In the 1960s, Maibach and Kligman16 analyzed sunless tanner with varying DHA concentrations as a treatment for vitiligo, finding that 5% DHA concentration was satisfactory for White patients, while up to 30% concentration was necessary for Black patients. In 2008, Rajatanavin et al17 explored DHA cream (concentrations 3.5%, 4.2%, 5%, and 6%) as a treatment for vitiligo; most patients (89.9%) reported moderate satisfaction with the 6% DHA cream, and concentrations of 5% or higher achieved better color matching for individuals with darker skin color. Hsu18 reported a case of a 52-year-old Hispanic woman with recalcitrant vitiligo that affected the face, trunk, and extremities who achieved near-complete cosmetic coverage using DHA sunless tanner, reporting that her vitiligo was unrecognizable in person.
Heerfordt et al3 assessed the protection factor of DHA against visible light in patients with EPP, a rare genetic disorder that causes extreme hypersensitivity to UV-A radiation. DHA has been reported to absorb some UV radiation, with a sun protection factor of 1.3 to 2.2 (p<0.001), thereby offering theoretical protection against EPP. Nineteen patients underwent weekly DHA applications during the springs and summers of 2018 through 2021, with the primary outcome of self-reported effect on photosensitivity. Of these patients, 68% reported reduced photosensitivity, and 84% noted good cosmetic results. Three years after the study, 13 patients continued DHA use and endorsed benefit.3
Stopiglia et al20 demonstrated in vitro antifungal activity of DHA against Candida strains and dermatophytes at the same concentrations used in artificial sunless tanning lotions (25-100 mg mL-1), with the fungicidal activity varying from 1.6 to 50 mg mL-1. DHA is a potential low-toxicity topical antifungal agent that may be used because of its penetration of and quick reaction in the stratum corneum, yielding minimal concern over systemic absorption.20
Formulations. Numerous strategies have been explored to enhance the stability, efficacy, and cosmetic outcome of DHA-based products. Bobin et al21 demonstrated that sulfur-containing amino acid derivatives such as methionine sulfoxide (MSO) could serve as adjuvants to DHA. Topical application of MSO prior to DHA enhanced the rate, intensity, and durability of the resulting color compared to DHA alone.21 This approach permits the use of lower DHA concentrations; however, due to their separate stability profiles, MSO and DHA require dual-compartment packaging.21
Sun et al14 isolated three colored compounds from DHA reactions, with arginine (A1), histidine (H1), and lysine (L1) responsible for the browning effect in sunless tanning products. They found that all share a common chromophore responsible for yellow coloration.14 The color intensity, measured via color dilution factor (CDF), was highest with histidine (CDF:864), followed by lysine (CDF:816), and lowest with arginine (CDF:174).14 Histidine and lysine reacted faster with DHA than arginine due to lower energy barriers, which explains their high tanning efficacy.14
Skin hydration and environmental humidity also modulate DHA efficacy. Nguyen and Kochevar22 reported a biphasic effect of humidity on pigmentation: skin coloration increased with humidity levels up to 75% but decreased at higher levels, particularly under occlusion, suggesting that optimal humidity levels are crucial for tanning. Although DHA reacted more at pH 7 than pH 5, vehicle type (gel vs. lotion) had minimal effect, suggesting that environmental and epidermal conditions may outweigh vehicle composition in determining tanning outcomes.22 Skin acid mantle is typically more acidic, which may explain the lower DHA reaction efficiency at pH 7.23
The chemical instability of DHA remains a formulation challenge. Pantini et al24 found that perfluoropolyether phosphate improves DHA stability and provides acidic buffering, maintaining an optimal pH for storage and application. To optimize DHA delivery and prolong its effect, Solé et al25 investigated chitosan-based hydrogels as release systems. DHA interacted noncovalently with chitosan, delaying hydrogel gelation and allowing for a slower, more sustained release compared to L-carnitine and nonloaded controls. The interaction between DHA and chitosan slows crosslinking by competing for amino groups.25 Gelation time for nonloaded controls was 52 minutes, compared to L-carnitine at 56 minutes and DHA-loaded at 162 minutes.25 Overall, these hydrogels are proposed as promising biocompatible carriers for DHA in topical cosmetic delivery systems.
Further kinetic analysis by Sun et al26 confirmed that temperature had the strongest effect on color intensity, followed by reaction time and pH. Histidine and lysine produced significantly more pigment than arginine under identical conditions, and a Box-protective lysine (Boc-Lys), which preserves the ε-NH2 group, yielded deeper pigmentation than unprotected lysine, implicating the ε-amino group of lysine as a key reactive site.26
Adverse Effects. While contact dermatitis from sunless tanner components is common, rare causes of allergic contact dermatitis (ACD) to DHA itself have been reported. Bovenschen et al2 reported a woman developing ACD 24 to 48 hours after applying three DHA-containing sunless tanners. Though fragrance and cobalt chloride may have contributed, DHA was implicated. Human case reports often cannot isolate DHA as the sole allergen, so the most direct evidence of DHA allergy comes from animal studies. In one study, repeated topical 5% DHA application in hairless dogs led to irritant contact dermatitis, with histopathological findings of epidermal thickening, degeneration, and inflammatory changes, while vehicle-only controls showed no such effects, indicating DHA as the causative agent.27Additionally, DHA-containing sunless tanners have been implicated in dermoscopic lesion changes. Gyllencreutz et al28 published a pilot study of whether DHA lead to changes in pigmented skin lesions that would stimulate clinically relevant dermoscopic changes. In facial pigmented skin lesions, equivocal lesions were registered by two independent evaluators significantly more often after DHA use than before DHA use (42% vs. 12%, p=0.021; and 69% vs. 19%, p=0.001, respectively). Follicular pigmentation mimicking lentigo maligna was seen significantly more often after DHA use than before (81% vs. 12%, p<0.001; and 69% vs. 15%, p<0.001, respectively), prompting biopsy recommendation.28 No significant changes were seen in nevi on the body.28
At concentrations relevant to consumer use, DHA has been shown to exert cytotoxic, genotoxic, and metabolic effects across various human and animal cell types. Smith et al29 demonstrated that DHA is cytotoxic to melanoma cells through reaction oxygen species (ROS) generation, mitochondrial dysfunction, G2/M cell cycle phase arrest, and apoptotic cell death. DHA significantly inhibited colony formation in melanoma cells at concentrations as low as 1.9 mM.29 G2/M cell cycle phase arrest was observed within 24 hours of exposure to 5 mM DHA, with no recovery from G2/M arrest seen by 72 hours.29 Apoptosis occurred after 72 hours of DHA exposure.29 A mild increase in senescence markers was observed at 24 and 48 hours, suggesting early cellular stress response preceding death.29 Similar cell cycle disruptions and apoptotic responses were reported in human keratinocytes (HaCaT), where DHA induced chromatin condensation, cytoplasmic budding, and cell detachment in a time- and dose-dependent manner.30 Marked cell death was observed after 24 hours of exposure to
≥25 mM DHA, while short-term exposure (3 hours) to ≤50 mM caused no immediate toxicity.30 These effects were attenuated by antioxidant pretreatment, implicating ROS as a mechanistic driver.30 Concentrations used in the study (25-100 mM) were even lower than those found in commercial tanning lotions (≥330 mM). Perer et al31 confirmed the upregulation of stress-responsive genes (HMOX1, HSPA6, CRYAB, XRCC2) and advanced glycation end products (AGEs) following DHA exposure. DHA at concentrations ≤20 mM did not impair viability, whereas higher doses (≥40 mM) led to significant cell death.31 Acute DHA exposure (defined as ≤50 mM; 1hr in phosphate-buffered saline followed by 24 hrs in growth medium) also triggered early stress signaling pathways, including p38 MAPK activation, Hsp27 phosphorylation, and eIF2α phosphorylation.31 Beyond skin models, DHA has been shown to be cytotoxic to liver (HepG3), cardiac (Ac16), and bronchial epithelial (BEAS-2B) cells at concentrations as low as 1.5 mM to 7.5 mM.32 Contrary to early skin model studies, Hernandez et al32 found only low or inconsistent levels of ROS and AGEs in systemic models, suggesting that DNA damage is not primarily ROS-mediated and instead may occur through mechanisms such as glycation or crosslinking of DNA repair proteins.
Inhalation exposure to DHA is increasingly relevant given its widespread use in spray tanning and e-cigarette flavorants. In mice, aerosolized DHA exposure (6 hours/day for 5 days) caused pulmonary fibrosis, elevated bronchoalveolar lavage markers, and histologic evidence of lung injury.33 In a human in vitro airway model, repeated DHA aerosol exposure for 5 weeks (mimicking spray tanning behavior) transiently and irreversibly reduced ciliary beating frequency and mucin secretion, while the barrier function was maintained and no apoptosis occurred.34 DHA also causes mitochondrial dysfunction and metabolic reprogramming in hepatic and cardiac cells. Hernandez et al15 found that the 50% and 90% inhibitory concentration values in HepG3 liver cells were around 1.5 mM and 4.6 to 6.25 mM, respectively, which are within exposure range for chronic vapers or frequent spray tanners. DHA exposure suppressed mTOR signaling and induced autophagic flux, reflecting disruption of nutrient sensing pathways.15 DHA also caused replication stress, apoptosis, autophagy, and lysosomal stress, suggesting a hybrid cell death mechanism.15 In rat cardiomyocytes (H9c2), low-dose DHA (2 mM) paradoxically enhanced short-term cell viability and proliferation, though it still caused significant stress and long-term metabolic disruption. Conversely, cytotoxic effects were observed with a DHA dose greater than 5 mM.32 Belfeur et al35 confirmed the metabolic impact of DHA, noting alterations in lactate and triose phosphate levels, mitochondrial dysfunction, and gene expression changes in exposed cells. Collectively, these data highlight the capacity of DHA to disrupt glycolysis, redox hemolysis, and mitochondrial integrity across diverse cell types.
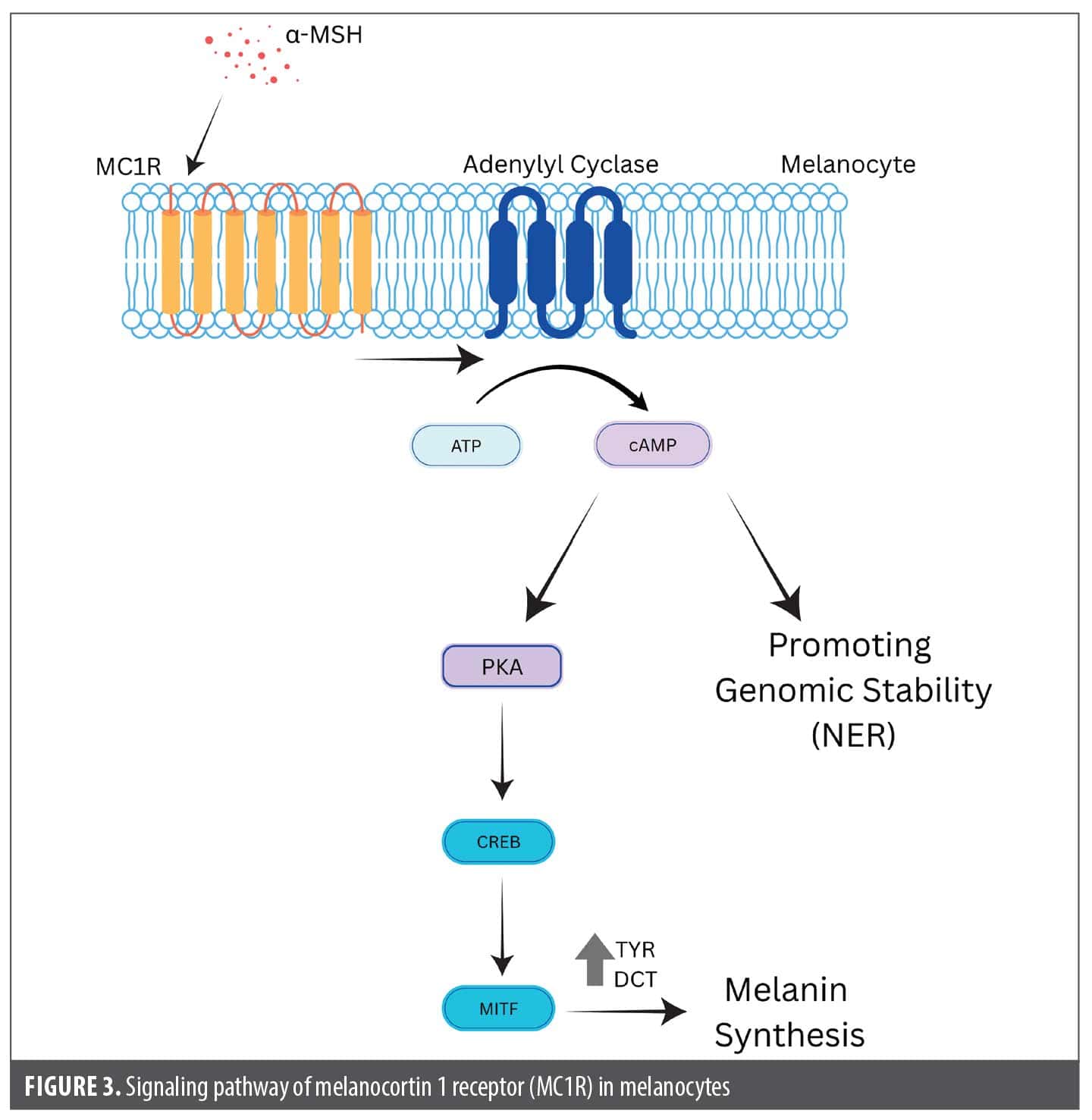
Melanotan. Mechanism of action. Melanotan is a synthetic analog of α-MSH. It acts as a nonselective agonist of one of several MCRs. When acting on the melanocortin 1 receptor (MC1R), melanotan stimulates melanin production and tyrosinase activity, leading to melanocyte proliferation and skin hyperpigmentation (Figure 3).36 Notably, as MC1R is not expressed by melanocyte stem cells, melanotan cannot have an effect on melanocyte differentiation.37 Beyond regulating pigmentation, α-MSH also acts on various other MCRs, playing a role in processes such as appetite regulation and sexual stimulation. Melanotan I (also known as afamelanotide) acts on MC1R and has increased resistance to enzymatic breakdown, whereas melanotan II is a shorter cyclic variant that increases pigmentation at a lower cumulative dose and is less specific and more lipophilic, thereby crossing the blood-brain barrier more easily, leading to more widespread adverse effects.5 Both melanotan I and II are unregulated compounds sold illegally online as a means to stimulate a tan.5
Uses. While traditionally administered via subcutaneous injection, a recent surge in popularity of nasal spray formulations, in particular melanocortin II, has been promoted through social media platforms. These products, marked for their ability to induce rapid skin pigmentation, are absorbed through nasal mucosa, are distrubted systemically and act on MCRs across the body.38 Survey data suggest that most users access melanotan through unregulated online vendors, with 77% of respondents reporting it was “very easy” or “easy” to purchase melanotan.5 Despite the lack of long-term safety data and the presence of unidentified impurities (4.1%-5.9%) in commercially available products, the majority of respondents (93%) indicated that they planned to continue using melanotan, stating that it increased their self-perceived attractiveness and overall quality of life.5 Afamelanotide) is approved by the FDA for EPP.39 Afamelanotide is also used off label for vitiligo.40
Adverse effects. Melanotan analogs are associated with a range of adverse effects, including serious dermatologic, vascular, and systemic complications. Multiple case reports have linked melanotan use to the development of new or changing pigmented lesions, raising concern about its potential melanocytic stimulatory effects. A case of oral mucosal malignant melanoma was reported in a young woman following one month of melanotan II nasal spray use, administered as two puffs into each nostril twice daily.36 Within three weeks of starting the spray, she developed gingival pigmentation and a rapidly growing anterior maxillary lesion that was ultimately diagnosed as pT4 melanoma with alveolar bone invasion.36 Other reports have described melanoma in situ (MMIS) arising from preexisting nevi shortly after melanotan II injection, with patients often concurrently using tanning beds to augment the pigmentary response.41,42 These cases suggest that melanotan may accelerate melanocytic activity in nevi, although causality remains unproven. Conversely, some studies show that there is no link between melanotan and melanoma, so chronic activation of MC1R leading to melanoma remains a topic of debate.43 In fact, MC1R activation in melanocytes themselves has been shown to activate antioxidant, DNA repair, and survival pathways.44
Rare but serious complications include priapism, attributed to melanotan II activity on MC3R and MC4R receptors involved in erectile function. One patient required emergency aspiration of 700 mL of cavernosal blood and intracavernosal phenylephrine after experiencing a prolonged 22-hour erection shortly after the subcutaneous injection of melanotan.45 While this adverse effect has been explored therapeutically in erectile dysfunction trials, its occurrence in unregulated cosmetic use poses significant health risks.
Further reported systemic complications include renal infarction and rhabdomyolysis. A 45-year-old man presented to the hospital with right-sided abdominal pain, vomiting, and increased urination. Upon questioning, he was found to have injected a total of 27 mg of melanotan II subcutaneously over the last six months.46 A computed tomography (CT) scan showed right-sided renal infarction affecting 50% of the kidney, with the findings attributed to melanotan.46 Melanotan stimulates receptors involved in vascular tone and inflammation, leading to reduced renal blood flow and thrombosis via sympathetic stimulation. In another case, a 39-year-old man who had injected 6 mg of melanotan II subcutaneously presented to the hospital two hours after injection with tachycardia, diffuse body aches, a sensation of anxiety, and sweating.47 Upon admission, the patient’s creatine was 2.25 mg/dL, troponin was 0.23 ng/mL, and creatine phosphokinase (CPK) was 1760 IU/L, which rose to 17,773 IU/L 12 hours later. Urinalysis revealed myoglobinuria from rhabdomyolysis.47
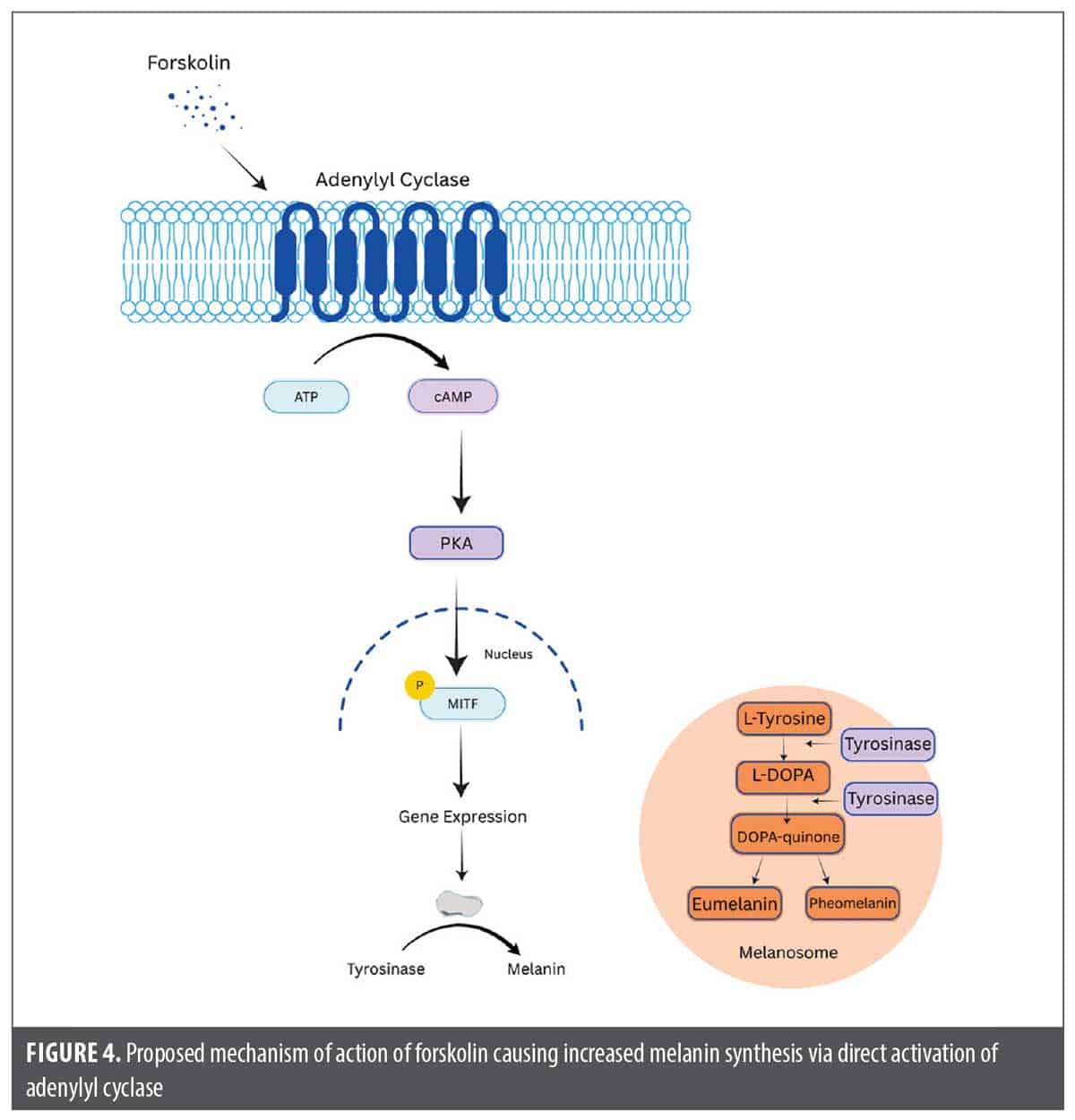
Forskolin. Mechanism of action. Forskolin is a plant-derived compound isolated from the root tissue of Plectranthus barbatus, with demonstrated skin-permeable properties. It directly activates adenylyl cyclase in epidermal cells, leading to increased intracellular cyclic adenosine monophosphate (cAMP) levels, which in turn stimulate melanogenesis in melanocytes independent of MC1R function. The increase in cAMP activates downstream signaling cascades, primarily through protein kinase A (PKA), and upregulates the expression of melanogenic enzymes, resulting in increased eumelanin synthesis and visible skin darkening (Figure 4). This mechanism has been demonstrated in animal models, including MC1R-defective mice, where topical forskolin application led to persistent epidermal melanization.48
Uses. Topical forskolin application has been shown to induce melanogenesis in MC1R-deficient mice, resulting in persistent tanning and increased UV resistance.48 This suggests potential application for fair-skinned individuals who are unable to tan naturally, thereby reducing the incidence of UV-induced skin cancers. Additionally, forskolin promotes epidermal thickening and enhances DNA repair through mechanisms independent of pigmentation.49 This indicates that forskolin may also provide benefits for individuals with impaired pigmentation or even those lacking melanocytes entirely, such as patients with vitiligo. Oral ingestion of forskolin has been found to lead to weight loss and increased bone and lean body mass.50 No topical forskolin currently exists for human use, likely due to formulation challenges, though some studies have explored nano-emulsions as effective carriers for topical administration.51
Adverse effects. Spry et al48 demonstrated that chronic topical application of forskolin in MC1R-deficient mice led to epidermal thickening, attributed to the accumulation of nucleated keratinocytes, and increased melanocyte presence. Importantly, no signs of toxicity or adverse histologic changes were observed, indicating that thickening was part of a healthy adaptive response. Scott et al49 showed that forskolin also induced epidermal thickening in both melanocyte-rich and melanocyte-deficient skin models. Together, these findings suggest that forskolin may enhance UV protection through both pigment-dependent and pigment-independent mechanisms.
Oral forskolin can treat cardiovascular disorders, hypertension, and gastrointestinal (GI) symptoms. While clinical studies generally administer 50 mg of pure forskolin twice daily, most oral supplements provide 250-500 mg Coleus forskolii extract (CFE) standardized to 10% forskolin taken once or twice daily. In a survey assessing the safety of CFE, a popular weight‑loss supplement in Japan, 10.5% of users reported adverse events, with 92% of those experiencing GI symptoms.52 The primary GI symptom was diarrhea, which was significantly associated with the amount of CFE consumed (p=0.005).52 Oral ingestion of forskolin stimulates chloride and water secretion into the intestinal lumen, reduces water reabsorption, and causes decreased smooth muscle relaxation and cAMP modulation in the intestines, contributing to diarrhea. Forskolin also causes hypotension due to its vasodilatory properties, which may cause dizziness, lightheadedness, headaches, and reflex tachycardia. In a study analyzing the effects of Coleus forskohlii on blood pressure, a 10% to 13% decrease in systolic blood pressure and 8% to 11% decrease in diastolic blood pressure (p<0.001) was found.53 Additionally, its effects on vascular tone and platelet function may increase bleeding risk, warranting caution in patients with bleeding disorders or those on blood thinners. These antihypertensive and anticoagulant effects may cause drug interactions with antihypertensives and anticoagulants. By activating adenylyl cyclase, forskolin bypasses the need for thyroid-stimulating hormone (TSH) stimulation and directly influences thyroid hormone secretion. Forskolin stimulated the secretion of thyroid hormones thyroxine (T4) and triiodothyronine (T3) from perfused dog thyroid lobes significantly more than TSH, suggesting that forskolin activates adenylate cyclase more rapidly, leading to enhanced hormone release.54 Human data on the effects of forskolin on thyroid hormones remains limited.
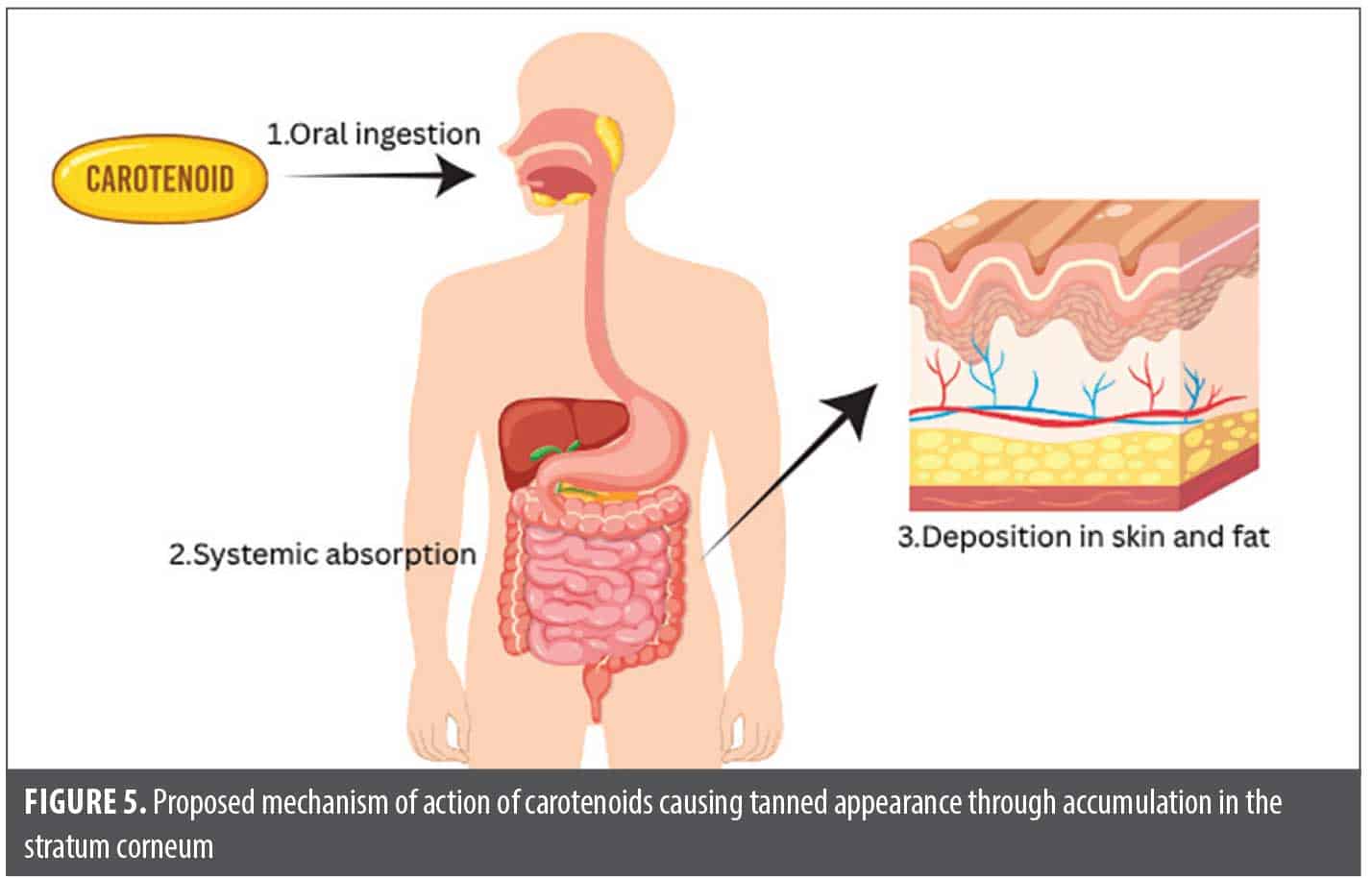
Carotenoids. Mechanism of action. Orally administered carotenoids are absorbed in the small intestine, packaged into chylomicrons, and transported via lipoproteins to peripheral tissues, most notably the skin.55 Within the epidermis, they function as potent antioxidants by neutralizing ROS generated by UV exposure.56 This antioxidant activity reduces UV-induced oxidative stress, thereby decreasing lipid peroxidation, DNA damage, and inflammatory response. Simultaneously, carotenoids accumulate in the stratum corneum, producing a gradual yellow-orange hue that becomes visible over time, particularly in low-melanin and sun protected areas such as the palms or inner arms (Figure 5).57 This coloration process occurs independently of melanocyte stimulation.58
Uses. Oral tanning pills marketed online typically rely on carotenoids to provide photoprotection and enhance skin pigmentation. The most common compounds used in these formulations include canthaxanthin, beta carotene, lycopene, lutein, and zeaxanthin.55 Canthaxanthin is FDA approved as a food dye but not as a tanning agent due to safety risks, as a large dose is required for tanning effects. Beta carotene, through its antioxidant activity, can enhance the skin’s defense against UV-induced oxidative stress when administered orally.56 Dietary supplementation reduces sensitivity to UV-induced erythema and may contribute to long-term protection against photoaging.56 Coetzee and Perrett reported that oral carotenoid supplementation led to dermal accumulation and a gradual yellow-orange hue.57 In their 8-week study among Black participants, a statistically significant increase in pigmentation was observed only on the palm (p<0.05), likely due to reduced melanin interference in that area.57 In contrast, darker, sun-exposed areas showed minimal change, likely due to
melanin masking.
Adverse Effects. An 84-year-old woman who had taken oral canthaxanthin for 10 years developed yellow-gold birefringent crystal deposits around the macula visible on a fundoscopic exam.59 While most cases of canthaxanthin-induced retinopathy are asymptomatic, this patient had significant visual loss that gradually improved after discontinuing the supplement, highlighting that high-dose canthaxanthin can cause retinal crystal accumulation and visual impairment. Furthermore, beta carotene is significantly associated with an increased risk of cardiovascular mortality (odds ratio: 1.10).60 Long-term, high-dose beta carotene supplementation (20–30 mg/day) increases lung cancer mortality by approximately 20% in people who smoke and individuals with prior asbestos exposure, while its photoprotective benefits only become reliable after 6 to 12 weeks at ≥12 mg/day.56 Lastly, carotenoid deposition in the skin can produce a yellow-orange hue that may not match natural melanin tones, sometimes resulting in an uneven appearance.58
Discussion
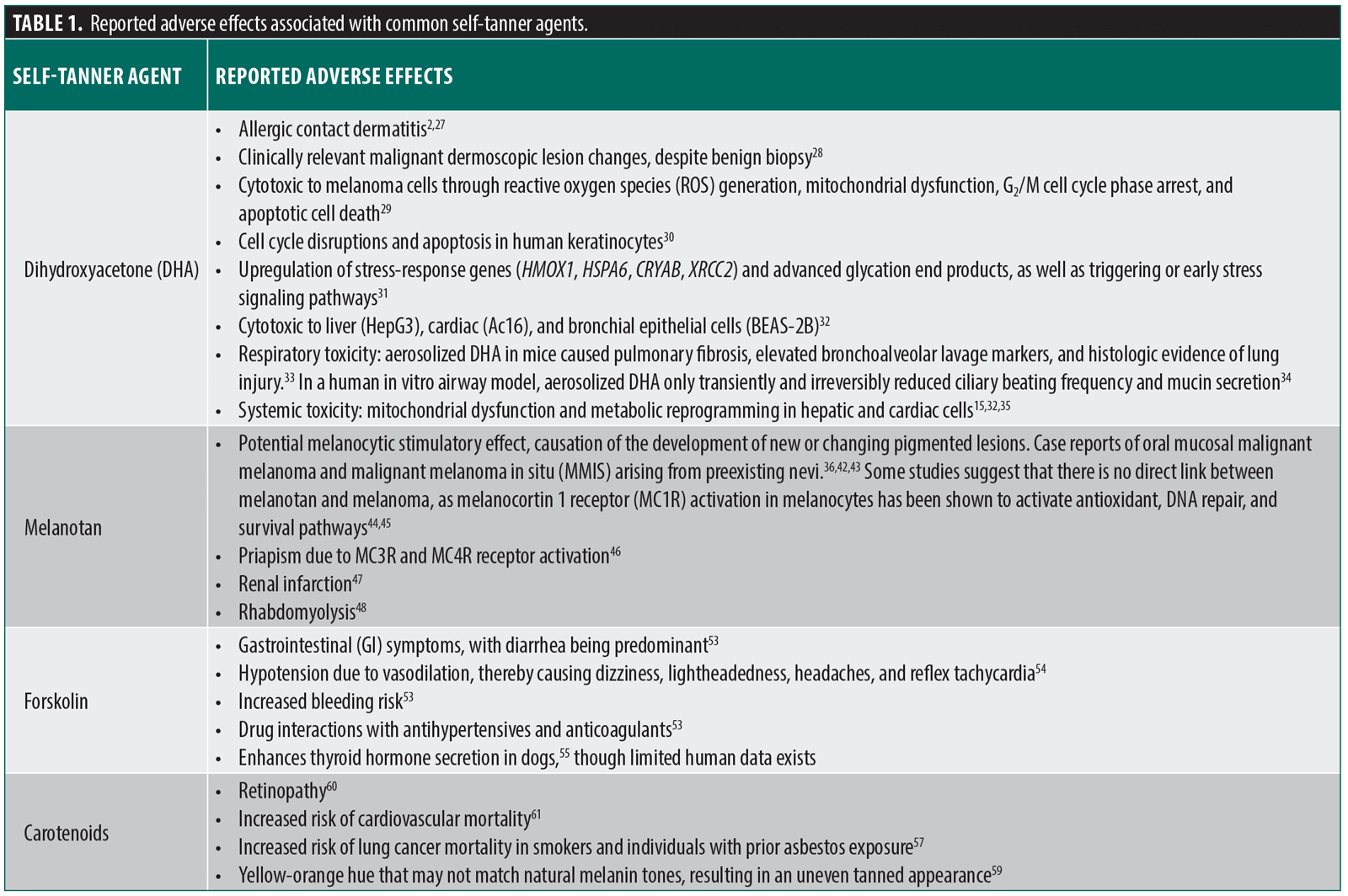
This systematic review highlights the expanding role of sunless tanning agents in cosmetic dermatology while emphasizing the growing need for targeted research, regulatory oversight, and clinician awareness. Although these products are often marketed as safer alternatives to UV tanning, emerging data raise questions regarding their long-term safety and systemic effects (Table 1). In addition to established tanning agents such as DHA and melanotan analogs, emerging compounds such as forskolin and carotenoids offer alternative pathways for skin melanin modulation and photoprotection.
Future research needs. Population-level data on sunless tanning behaviors are outdated, with the most recent national survey using data from 2015. With the evolving landscape of aesthetic practices, particularly among adolescent and social media users, up-to-date epidemiologic data are needed to better understand use patterns and risk behaviors. Additionally, while studies have highlighted the presence of common allergens in sunless tanners, it remains unclear whether product reformulations have meaningfully reduced allergen content.
Although DHA has been approved by the FDA for topical use, recent findings suggest that systemic exposure is not negligible. Up to 30% of applied DHA may reach viable epidermal layers, and approximately 0.5% may enter systemic circulation, particularly when applied in high concentrations or via aerosolized spray tans. These exposure routes, including inhalation and mucosal contact, are not currently addressed by regulatory frameworks and may pose risks to internal organs. Clinical studies are needed to evaluate whether these in vitro findings translate to measurable adverse outcomes in humans, particularly with chronic use.
To bridge the gap between preclinical findings and clinical application, future research on forskolin and carotenoids should prioritize human safety and tolerability to establish optimal dosing and formulation. Efficacy trials across diverse skin types are essential to confirm the melanogenesis-stimulating effects of forskolin and the skin pigmentation and photoprotective effects of carotenoids. Pathway studies using human skin models can validate mechanisms observed in animals, while long-term studies should assess safety of chronic use. Reports on carotenoid levels in human skin remain limited, and many available studies are outdated or focus solely on beta carotene.
Regulatory considerations and safety concerns. The continued use of unregulated agents like melanotan I and II is particularly concerning. These products are readily available online and often falsely labeled as being intended “for research use only” or for “licensed researchers,” despite being widely marketed to and purchased by the general public. Survey data reveal that the vast majority of users find these products easy to obtain and intend to continue using them regardless of potential adverse effects. The combination of melanocytic stimulation, systemic sympathomimetic activity, and vascular dysregulation associated with melanotan, particularly in the context of unstandardized dosing and formulations and their promotion via social media platforms, highlights the need for stricter regulation and greater public health messaging. Regulatory bodies should consider targeted enforcement against false advertising and implement clearer labeling and import restrictions for products containing synthetic melanocortin analogs.
Studies have shown the cytotoxic effects of DHA, which sometimes occurred even with DHA concentrations lower than those in commercial tanning lotions. With the growing popularity of sunless tanners, not only is increased research necessary into these cytotoxic effects but also increased awareness of such effects. A role for dermatologists exists in this space. While dermatologists denounce UV tanning and may offer sunless tanners as an alternative, patients often leave confused as to which sunless tanner they should use; they resort to social media for recommendations, where they may be exposed to toxic tanning methods such as melanotan. Dermatologists should be educated on the adverse effects of various sunless tanners and on brands of sunless tanners so that they can recommend specific products to patients.
Clinical implications. The widespread appeal of tanning reflects a cultural preference for tanned skin and an increasing reliance on appearance-enhancing pharmacologic interventions. Sunless tanning products have been embraced as part of this aesthetic trend, yet this review highlights the tradeoffs between cosmetic benefits and medical risks. Dermatologists are in a unique position to address this tension through patient education, as they can instruct on safer tanning practices while remaining vigilant for adverse effects tied to both regulated and unregulated agents.
Many different sunless tanners have shown the potential to cause benign lesions to falsely appear malignant.28 Dermatologists should proactively inquire about sunless tanning use when encountering unclear dermoscopic findings or unexplained systemic symptoms to avoid unnecessary biopsies and treatment.
ACD remains an important, underrecognized adverse effect of sunless tanning products. While DHA has been implicated in isolated cases of ACD, most allergic reactions are likely attributable to other components in product formulations, particularly fragrances, preservatives, and emulsifiers. Dermatologists should consider patch testing in patients who present with dermatitis of uncertain etiology and inquire about the use of sunless tanners. When ACD is suspected, testing should include both DHA (if available) and commonly implicated ingredients. Incorporating a focused history of cosmetic use, including sunless tanners, into routine dermatologic evaluation may help uncover otherwise overlooked causes of ACD and improve patient outcomes through allergen avoidance.
Conclusion
Sunless tanning products occupy a growing trend in cosmetic dermatology, offering aesthetic benefits without the known carcinogenic effects of UV radiation. However, emerging clinical data on DHA challenge the long-standing perception of its safety, particularly when used at high concentrations or through inhalation exposure. Furthermore, the rise of unregulated melanotan analogs presents additional dermatologic and systemic risks that are poorly understood. While these two continue to be the main avenues to sunless tanning, the emerging industry for molecules such as forskolin and carotenoids remains widely unknown and can present new ways to offer photoprotection and skin color development.
This review highlights the dual need for innovation in formulation science to enhance the safety and efficacy of DHA-based products and for increased regulatory scrutiny and public education surrounding unapproved agents such as melanotan. Additionally, short- and long-term effects of emerging molecules such as forskolin and carotenoids are crucial to develop a comprehensive understanding of their safety profile and cosmetic potential in the growing sunless tanning market. Dermatologists should remain informed about these trends to better counsel patients and identify potential adverse effects early. Future research should prioritize long-term clinical safety studies, improved delivery vehicles, and targeted public health strategies to ensure that the benefits of sunless tanning do not come at the expense of patient safety.
References
- Seidenberg AB, Julian AK, Hartman AM, Holman DM. Sunless and indoor tanning among US non-Hispanic White women ages 18-49 years. J Dermatol Nurses Assoc. 2023;15(3):123-132.
- Bovenschen HJ, Körver JEM, van der Valk PGM. Contact dermatitis to self-tanning products. Contact Dermatitis. 2009;60(5):290-291.
- Heerfordt IM, Philipsen PA, Lerche CM, Wulf HC. Protection against visible light by dihydroxyacetone in erythropoietic protoporphyria. Photodiagnosis Photodyn Ther. 2023;41(103302):103302.
- Mallory CW, Lopategui DM, Cordon BH. Melanotan tanning injection: a rare cause of priapism. Sex Med. 2021;9(1):100298.
- Callaghan DJ III. A glimpse into the underground market of melanotan. Dermatol Online J. 2018;24(5):13030/qt2gz9f9jk.
- Ramachandrappa S, Gorrigan RJ, Clark AJL, Chan LF. The melanocortin receptors and their accessory proteins. Front Endocrinol (Lausanne). 2013;4:9.
- Amaro-Ortiz A, Yan B, D’Orazio JA. Ultraviolet radiation, aging and the skin: prevention of damage by topical cAMP manipulation. Molecules. 2014;19(5):6202-6219.
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
- Williams M, Jones SC, Caputi P, Iverson D. Do Australian adolescent female fake tan (sunless tan) users practice better sun-protection behaviours than non-users? Health Educ J. 2012;71(6):654-661.
- Russo VA, Van Acker MM, Vander Wal JS, Sinha AA. Patterns of use of sunless tanning product alternatives to indoor tanning among female college students. Arch Dermatol. 2012;148(7):855-857.
- Tella E, Beauchet A, Vouldoukis I, et al. French teenagers and artificial tanning: Teenagers and artificial tanning. J Eur Acad Dermatol Venereol. 2013;27(3):e428-e432.
- Rigali S, Liszewski W. Contact allergens in self tanner products. Dermatitis. 2025;36(1):e107-e108.
- Buonomo M, Hylwa S. Allergenic ingredients in sunless tanning products. Dermatitis. 2021;32(6):e136-e138.
- Sun Y, Zhang P, Wang X, Al-Zahrani FAM, de Leeuw NH, Lin L. Deciphering key coloured compounds from sunless tanning reactions. Dyes Pigm. 2022;204:110448.
- Hernandez A, Sonavane M, Smith KR, Seiger J, Migaud ME, Gassman NR. Dihydroxyacetone suppresses mTOR nutrient signaling and induces mitochondrial stress in liver cells. PLoS One. 2022;17(12):e0278516.
- Maibach HI, Kligman AM. Dihydroxyacetone: a suntansimulating agent. Arch Dermatol. 1960;82(4):505-507.
- Rajatanavin N, Suwanachote S, Kulkollakarn S. Dihydroxyacetone: a safe camouflaging option in vitiligo. Int J Dermatol. 2008;47(4):402-406.
- Hsu S. Camouflaging vitiligo with dihydroxyacetone. Dermatol Online J. 2008;14(8):23.
- Faurschou A, Janjua NR, Wulf HC. Sun protection effect of dihydroxyacetone. Arch Dermatol. 2004;140(7):886-887.
- Stopiglia CDO, Vieira FJ, Mondadori AG, Oppe TP, Scroferneker ML. In vitro antifungal activity of dihydroxyacetone against causative agents of dermatomycosis. Mycopathologia. 2011;171(4): 267-271.
- Bobin M, Martini M, Cotte J. Effects of color adjuvants on the tanning effect of dihydroxyacetone. J Cosmet Sci. 1984;35(5):265-272.
- Nguyen BC, Kochevar IE. Factors influencing sunless tanning with dihydroxyacetone. Br J Dermatol. 2003;149(2):332-340.
- Brooks SG, Mahmoud RH, Lin RR, Fluhr JW, Yosipovitch G. The skin acid mantle: an update on skin pH. J Invest Dermatol. 2025;145(3):509-521.
- Pantini G, Ingoglia R, Brunetta F, Brunetta A. Sunless tanning products containing dihydroxyacetone in combination with a perfluoropolyether phosphate. Int J Cosmet Sci. 2007;29(3):201-209.
- Solé I, Vílchez S, Miras J, Montanyà N, García-Celma MJ, Esquena J. DHA and l-carnitine loaded chitosan hydrogels as delivery systems for topical applications. Colloids Surf A Physicochem Eng Asp. 2017;525:85-92.
- Sun Y, Lee S, Lin L. Comparison of color development kinetics of tanning reactions of dihydroxyacetone with free and protected basic amino acids. ACS Omega. 2022;7(49):45510 45517.
- Kimura T. Contact dermatitis caused by sunless tanning treatment with dihydroxyacetone in hairless descendants of Mexican hairless dogs. Environ Toxicol. 2009;24(5):506-512.
- Gyllencreutz JD, Boström KB, Terstappen K. Does it look like melanoma? A pilot study of the effect of sunless tanning on dermoscopy of pigmented skin lesions. Br J Dermatol. 2013;168(4):867-870.
- Smith KR, Granberry M, Tan MCB, Daniel CL, Gassman NR. Dihydroxyacetone induces G2/M arrest and apoptotic cell death in A375P melanoma cells. Environ Toxicol. 2018;33(3):333-342.
- Petersen AB, Wulf HC, Gniadecki R, Gajkowska B. Dihydroxyacetone, the active browning ingredient in sunless tanning lotions, induces DNA damage, cell-cycle block and apoptosis in cultured HaCaT keratinocytes. Mutat Res. 2004;560(2):173-186.
- Perer J, Jandova J, Fimbres J, et al. The sunless tanning agent dihydroxyacetone induces stress response gene expression and signaling in cultured human keratinocytes and reconstructed epidermis. Redox Biol. 2020;36:101594.
- Hernandez A, Hedlich-Dwyer J, Hussain S, et al. Acute exposure to dihydroxyacetone promotes genotoxicity and chromosomal instability in lung, cardiac, and liver cell models. Toxicol Sci. 2024;201(1):85-102.
- Hernandez A, Levi H, Juncos JXM, et al. Inhalation exposure to dihydroxyacetone promotes lung injury and pulmonary fibrosis in A/J mice. Toxicol Rep. 2025;14:101878.
- Wang Y, Wu Q, Muskhelishvili L, Davis K, Bryant M, Cao X. Assessing the respiratory toxicity of dihydroxyacetone using an in vitro human airway epithelial tissue model. Toxicol In Vitro. 2019;59: 78-86.
- Belfleur L, Sonavane M, Hernandez A, Gassman NR, Migaud ME. Solution chemistry of dihydroxyacetone and synthesis of monomeric dihydroxyacetone. Chem Res Toxicol. 2022;35(4):616-625.
- Yassin Alsabbagh A, Bhujel N, Singh RP. Melanotan II nasal spray: a possible risk factor for oral mucosal malignant melanoma? Int J Oral Maxillofac Surg. 2025;54(9):806-809.
- Dong D, Jiang M, Xu X, et al. The effects of NB-UVB on the hair follicle-derived neural crest stem cells differentiating into melanocyte lineage in vitro. J Dermatol Sci. 2012;66(1):20-28.
- Newton J, Reinschmidt A, Van Gorp R, Sonstegard A. Nasal tanning sprays: illuminating the risks of a popular TikTok trend. Cutis. 2024;113(1):E3-E4.
- Center for Drug Evaluation, Research. Scenesse (afamelanotide) is FDA-approved for EPP. U.S. Food and Drug Administration. Revised August 2024. Accessed July 8, 2025. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/210797s007lbl.pdf
- Passeron T. Indications and limitations of afamelanotide for treating vitiligo. JAMA Dermatol. 2015;151(3):349-350.
- Ong S, Bowling J. Melanotan-associated melanoma in situ. Australas J Dermatol. 2012;53(4):301-302.
- Langan EA, Ramlogan D, Jamieson LA, Rhodes LE. Change in moles linked to use of unlicensed “sun tan jab.” BMJ. 2009;338:b277.
- Arisi M, Rossi M, Rovati C, et al. Clinical and dermoscopic changes of acquired melanocytic nevi of patients treated with afamelanotide. Photochem Photobiol Sci. 2021;20(2):315 320.
- Abdel-Malek ZA, Swope VB, Starner RJ, Koikov L, Cassidy P, Leachman S. Melanocortins and the melanocortin 1 receptor, moving translationally towards melanoma prevention. Arch Biochem Biophys. 2014;563:4-12.
- Dreyer BA, Amer T, Fraser M. Melanotan-induced priapism: a hard-earned tan. BMJ Case Rep. 2019;12(2):e227644.
- Peters B, Hadimeri H, Wahlberg R, Afghahi H. Melanotan II: a possible cause of renal infarction: review of the literature and case report. CEN Case Rep. 2020;9(2):159-161.
- Nelson ME, Bryant SM, Aks SE. Melanotan II injection resulting in systemic toxicity and rhabdomyolysis. Clin Toxicol (Phila). 2012;50(10):1169-1173.
- Spry ML, Vanover JC, Scott T, et al. Prolonged treatment of fair-skinned mice with topical forskolin causes persistent tanning and UV protection. Pigment Cell Melanoma Res. 2009;22(2):219-229.
- Scott TL, Christian PA, Kesler MV, et al. Pigment-independent cAMP-mediated epidermal thickening protects against cutaneous UV injury by keratinocyte proliferation. Exp Dermatol. 2012;21(10):771-777.
- Godard MP, Johnson BA, Richmond SR. Body composition and hormonal adaptations associated with forskolin consumption in overweight and obese men. Obes Res. 2005;13(8):1335-1343.
- Miastkowska M, Sikora E, Lasoń E, et al. Nano-emulsions as vehicles for topical delivery of forskolin. Acta Biochim Pol. 2017;64(4):713-718.
- Nishijima C, Chiba T, Sato Y, Umegaki K. Nationwide online survey enables the reevaluation of the safety of Coleus forskohlii extract intake based on the adverse event frequencies. Nutrients. 2019;11(4):866.
- Jagtap M, Chandola HM, Ravishankar B. Clinical efficacy of Coleus forskohlii (Willd.) Briq. (Makandi) in hypertension of geriatric population. Ayu. 2011;32(1):59-65.
- Laurberg P. Forskolin stimulation of thyroid secretion of T4 and T3. FEBS Lett. 1984;170(2):273-276.
- Scarmo S, Cartmel B, Lin H, et al. Significant correlations of dermal total carotenoids and dermal lycopene with their respective plasma levels in healthy adults. Arch Biochem Biophys. 2010;504(1):34-39.
- Stahl W, Sies H. β-carotene and other carotenoids in protection from sunlight. Am J Clin Nutr. 2012;96(5):1179S-1184S.
- Coetzee V, Perrett DI. Effect of beta carotene supplementation on African skin. J Biomed Opt. 2014;19(2):025004.
- Whitehead RD, Re D, Xiao D, Ozakinci G, Perrett DI. You are what you eat: within-subject increases in fruit and vegetable consumption confer beneficial skin-color changes. PLoS One. 2012;7(3):e32988.
- Beaulieu RA, Warwar RE, Buerk BM. Canthaxanthin retinopathy with visual loss: a case report and review. Case Rep Ophthalmol Med. 2013;2013:140901.
- O’Connor EA, Evans CV, Ivlev I, et al. Vitamin and mineral supplements for the primary prevention of cardiovascular disease and cancer: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2022;327(23):2334-2347.