J Clin Aesthet Dermatol. 2024;17(1):41–44.
J Clin Aesthet Dermatol. 2024;17(1):41–44.
by Ally-Khan Somani, MD, PhD; Sherrif F. Ibrahim, PhD, MD; Michael Tassavor, MD; Jane Yoo MD; and
Aaron S. Farberg, MD
Dr. Somani is with the Department of Dermatology at Indiana’s University School of Medicine in Indianapolis, Indiana. Dr. Ibrahim is with Rochester Dermatologic Surgery in Victor, New York.
Dr. Tassavor is with Medical Dermatology and Cosmetic Surgery Centers in New York, New York. Dr. Yoo is with the Icahn School of Medicine at Mount Sinai in New York, New York. Dr. Farberg is with Baylor Scott and White Health System in Dallas, Texas.
FUNDING: Funding was provided by Castle Biosciences, Inc.
DISCLOSURES: Dr. Farberg is an advisor for Castle Biosciences, Inc. All other authors report no conflicts of interest relevant to the article.
ABSTRACT: Objective. Adjuvant radiation therapy (ART) is often recommended for high-risk cSCC patients but carries significant costs and risks. This study aims to determine if utilizing the 40-GEP test to guide ART can reduce healthcare costs in cSCC management.
Methods. Medical claims data with new diagnoses of cSCC for the 12 months ending June 2022 in the Medicare (≥65 years) population (source: IQVIA claims database) were obtained and normalized to the general population for missingness. CPT codes associated with radiation therapy within one-year post diagnosis were used to establish adjuvant RT use (defined as ‘ART’). Average weighted direct costs for four major ART modalities were calculated from published studies and (IQVIA). Sensitivity analysis was used to assess the financial impact of ART treatment using varying distributions of 40-GEP Class results.
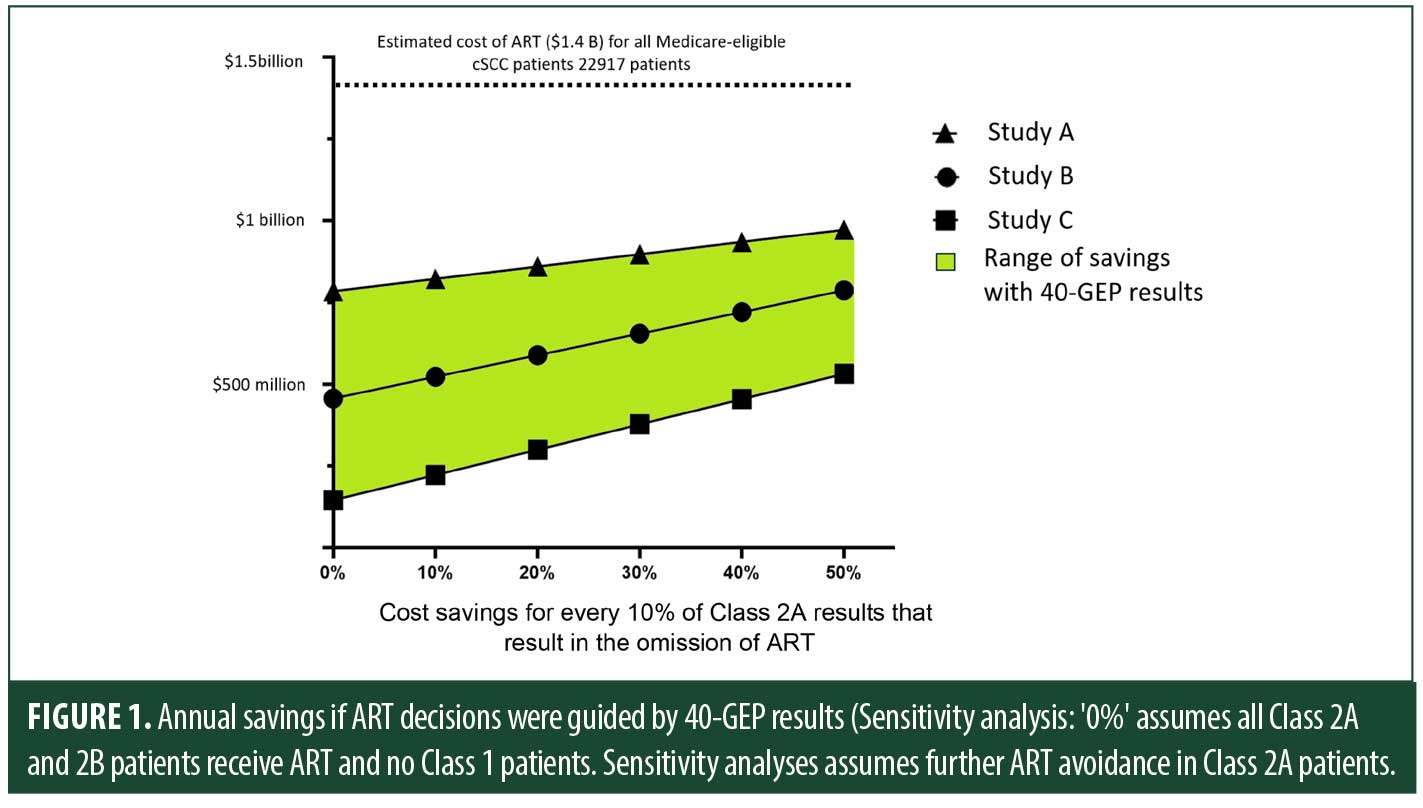
Results. Normalized medical claims data identified 22,917 Medicare-eligible cSCC patients who received ART within the United States. The weighted average direct cost for ART, which includes the four most used CPT code-defined modalities (IGRT, IMRT, IMPT, and XRT), was $60,693 per patient, amounting to an annual projected ART cost of $1.4 billion. Using the distribution of 40-GEP results from published studies, utilization of a 40-GEP test result to avoid ART in these patients could save up to $972 million in Medicare-eligible population. Sensitivity analysis shows, depending upon the distribution of the 40-GEP results, that for every 10% of Class 2A test results omitting ART, an extra $38-66 million in annual savings is expected
Limitations. Potential limitations include a need for more comprehensive patient information and the cost of ART-related complications.
Conclusion. Utilizing the 40-GEP test results to guide ART decision-making would result in material savings to Medicare.
Keywords: 40-Gene Expression Profile (40-GEP), Cutaneous Squamous Cell Carcinoma (cSCC), Adjuvant radiation therapy (ART), Cost, Medicare
Studies have shown that ART is effective in reducing metastasis as well as local recurrence.1–3 As such, NCCN guidelines for cSCC recommend consideration of adjuvant radiation therapy (ART) in patients with localized high or very high risk cSCC with poor prognostic features.4 However, patients categorized as high or very high risk represent a broad range of actual metastatic risk leading to over-treatment as well as under-treatment; the majority of patients who may be considered for ART are metastasis-free without any additional intervention.5 ART is reported to benefit subsets of high risk cSCC patients; however, this subset has not been well-defined by clinical and pathological features alone, resulting in broad recommendations for its usage by national societies and guidelines.3,4
ART is also associated with clinically significant complications, ranging from short-term adverse events of radiation dermatitis, hair loss, nausea, and vomiting, as well as more long term adverse events such as lymphedema and dementia.6 Furthermore, guidelines discourage subsequent radiation therapy in the prior radiation field due to the increased risk of radiation induced secondary cancers and long-term sequelae.4 Thus, there is an unmet clinical challenge to identify those who truly benefit from ART and to optimize the balance of complications, treatment outcomes, and healthcare costs for cSCC patients.
The 40-GEP test is validated to improve upon current clinicopathologic-based risk prediction staging systems, providing more precise risk stratification for metastasis (regional/nodal and distant) in patients with high-risk cSCC.7,8 The 40-GEP test categorizes patients into three distinct risk levels for metastatic potential: low risk (Class 1), moderate risk (Class 2A), or high risk (Class 2B). 40-GEP classification is objective and independent of the clinicopathologic staging systems, including NCCN, BWH, and AJCC, as well as individual clinicopathologic risk factors.7 Retrospective and prospective clinical utility studies have shown that the 40-GEP test can direct personalized patient management decisions such as follow-up care, surveillance imaging, and ART use.9–12 The 40-GEP test significantly stratified the risk of nodal or distant metastasis in an ART-eligible population, particularly between Class 1, Class 2A and Class 2B, indicating that ART could potentially be safely avoided for Class 1 patients, and should be strongly considered for Class 2B patients based on metastatic risk.13 Furthermore, a recent multi-center study showed that patients with a Class 2B result had improved outcomes if treated with ART, while a similar benefit was not observed for Class 1 or Class 2A cases. Most importantly, in the same study, other than the 40-GEP Class 2B result, no other risk factors or staging identified a subset of patients substantially benefiting from ART.14 In light of recent evidence, this study aims to determine if utilizing the 40-GEP results to guide ART can reduce costs for cSCC management among a Medicare-eligible patient population.
Methods
cSCC patients in the 12-month period ending May 2022 were identified in the IQVIA database using established ICD-10 codes. Claims data included radiation therapy CPT codes within one year post-diagnosis that were used to establish adjuvant RT (defined as “ART”). Within the same cohort, four primary CPT code-defined radiation therapy modalities, namely image-guided radiation therapy (IGRT), intensity-modulated radiation therapy (IMRT), intensity modulated proton therapy (IMPT), and radiation therapy (XRT) were identified. Cost for each of these modalities were calculated from published literature, and the weighted average cost for these modalities was calculated based on the IQVIA claims database. Data were filtered for Medicare-eligible patients (>65 years old) and normalized to national cSCC incidence (1.8 million diagnoses per year).15–17 The absence of standardized patient selection criteria for ART and a wide range of medical practices can result in diverse outcomes, affecting the distribution of 40-GEP results. Therefore, we conducted sensitivity analyses in three distinct scenarios representing frequencies observed in retrospective studies and prospective clinical orders for ART-eligible patients. Study A18 reflects prospective clinical orders with a 40-GEP result composition of 70.4 percent for Class 1, 27.1 percent for Class 2A, and 2.5 percent for Class 2B in the Medicare and ART-eligible population. Similarly, Study B7 represents the clinical validation study, with Class 1 -46.8 percent, Class 2A- 47.5 percent, Class 2B- 5.7 percent, while Study C14 represents distribution observed from a high risk cSCC study population who received ART (Cohort B: Class 1-24.4%, Class 2A-55.6%, Class 2B-20%) to encompass the financial implications of forgoing ART treatment in a comprehensive 40-GEP result composition.
Results
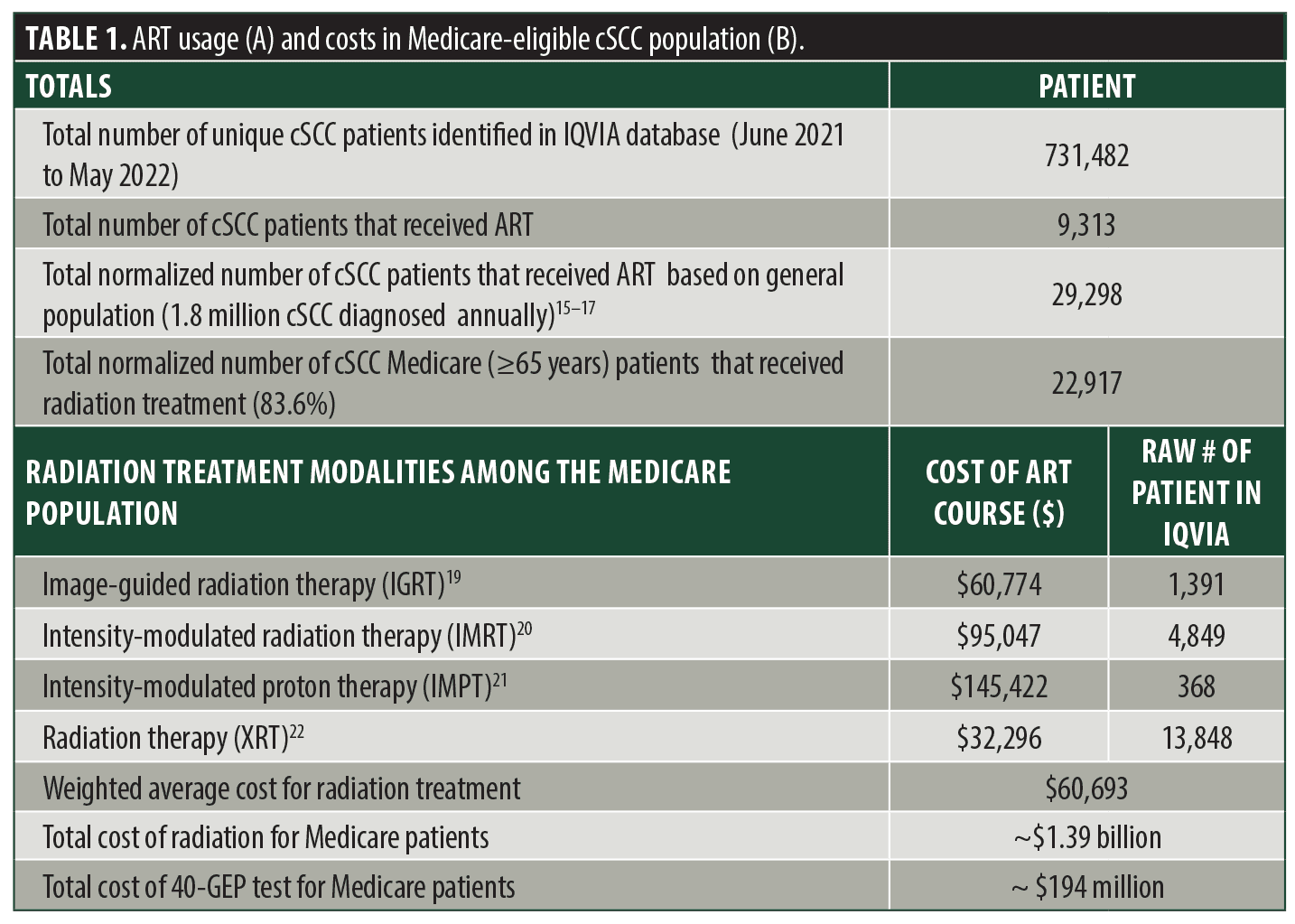
The IQVIA database identified 11,906 patients who had received ART (Table 1A). Given ~1.8 million patients with cSCC are diagnosed annually, the normalized number of cSCC patients that received ART was 29,298.15–17 The total normalized number of cSCC Medicare (≥65 years) patients that received radiation treatment based on IQVIA data resulted in 22,917 Medicare patients with cSCC receiving ART. Based on four radiation treatment modalities, the weighted average cost for radiation treatment for cSCC was $60,693 per patient, detailed in Table 1B. The total direct annual cost for radiation therapy for Medicare patients and the cost of performing the 40-GEP were estimated at ~$1.4 billion and ~$195 million, respectively.
In Study A, avoiding ART in 40-GEP Class 1 results among the ART-eligible Medicare population can generate substantial annual cost savings of ~$784 million in the Medicare Trust fund. Additionally, avoidance of ART in patients that receiving a Class 2A result would result in further savings. Avoidance of ART in 10 percent of patients receiving a Class 2A result in the same study would result in an additional $38 million savings, and can add up to ~$972 million in savings if the avoidance of ART is implemented in ~50 percent of Class 2A patients (Figure 1). Similarly, in Study B, avoidance of ART only in Class 1 patients can result in ~$456 million in savings. In Study C, which comprises high-risk cSCC patients who received ART, conservatively avoiding ART only in 40-GEP Class 1 patients could save roughly $145 million annually for the Medicare Trust fund and an additional $77 million in savings if 10 percent of Class 2A patients avoided radiation, indicating significant savings in different 40-GEP results composition.
Discussion
cSCC presents a clinical challenge due to its risk of metastasis and the broad guidelines-driven recommendation for ART in a subset of “high-risk” patients. Identifying patients who can potentially benefit from ART is crucial to balance improvement in outcomes, impact of complications, and costs for both. A recent study showed that patients with a 40-GEP Class 2B test result substantially benefited from ART compared to Class 1 or Class 2A, or to those identified as high risk by any other risk factors or staging systems.14 Building upon that finding, this study demonstrates that guiding ART treatment using 40-GEP results can lead to substantial Medicare healthcare savings ranging from $144 million to ~$972 million. This cost reduction is primarily attributed to avoiding direct costs of unnecessary ART in patients with low-risk 40-GEP profiles and the judicious selection of patient treatment based on molecular risk assessment.
The overall complication of ART rate is high and ranges from minor radiation-related bruises to level 3-4 adverse events, and is especially pronounced in the aging Medicare population. Thus, besides the clinicopathological features, clinicians consider patient preferences, social situations, including the ability to frequent hospital visits, health conditions, performance status, and remaining lifespan.6 Integrating the objective 40-GEP tools in the management of ART eligible high risk cSCC can identify those who may safely avoid ART who would, therefore, have reduced complications, a net improvement in health outcomes, and reduced cost in the Medicare population.
Potential limitations include a need for more comprehensive patient information and the cost of ART-related complications rather than comprehensive clinical outcomes.
Conclusion
This study underscores the economic and health benefits of using the 40-GEP test to guide adjuvant radiation therapy decisions for ART-eligible cSCC patients. By reducing unnecessary treatments and associated complications, we can achieve a more cost-effective and resource-efficient approach to cSCC management while providing personalized care based on objective molecular risk profiles. In the clinical setting, these findings underscore the value for the 40-GEP test to significantly reduce healthcare costs within the Medicare system associated with cSCC management.
References
- Ruiz ES, Kus KJB, Smile TD, et al. Adjuvant radiation following clear margin resection of high T-stage cutaneous squamous cell carcinoma halves the risk of local and locoregional recurrence: A dual-center retrospective study. J Am Acad Dermatol. 2022;87(1):87–94.
- Harris BN, Pipkorn P, Nguyen KNB, et al. Association of Adjuvant Radiation Therapy With Survival in Patients With Advanced Cutaneous Squamous Cell Carcinoma of the Head and Neck. JAMA Otolaryngology–Head & Neck Surgery. 2019;145(2):153–158.
- Likhacheva A, Awan M, Barker CA, et al. Definitive and Postoperative Radiation Therapy for Basal and Squamous Cell Cancers of the Skin: Executive Summary of an American Society for Radiation Oncology Clinical Practice Guideline. Pract Radiat Oncol. 2020;10(1):8–20.
- NCCN. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Version 1.2023 — March 10, 2023 Continue Squamous Cell Skin Cancer. https://www.nccn.org/professionals/physician_gls/pdf/squamous.pdf.
- Farberg AS, Fitzgerald AL, Ibrahim SF, et al. Current Methods and Caveats to Risk Factor Assessment in Cutaneous Squamous Cell Carcinoma (cSCC): A Narrative Review. Dermatol Ther (Heidelb). 2022;12(2):267–284.
- Majeed H, Gupta V. Adverse Effects of Radiation Therapy. In: StatPearls. StatPearls Publishing; 2023. Accessed October 1, 2023.http://www.ncbi.nlm.nih.gov/books/NBK563259/
- Ibrahim SF, Kasprzak JM, Hall MA, et al. Enhanced metastatic risk assessment in cutaneous squamous cell carcinoma with the 40-gene expression profile test. Future Oncology. 2022;18(7):833–847.
- Wysong A, Newman JG, Covington KR, et al. Validation of a 40-gene expression profile test to predict metastatic risk in localized high-risk cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2021;84(2):361–369.
- Farberg AS, Hall MA, Douglas L, et al. Integrating gene expression profiling into NCCN high-risk cutaneous squamous cell carcinoma management recommendations: impact on patient management. Curr Med Res Opin. 2020;36(8):1301–1307.
- Singh G, Tolkachjov SN, Farberg AS. Incorporation of the 40-Gene Expression Profile (40- 229 GEP) Test to Improve Treatment Decisions in High-Risk Cutaneous Squamous Cell Carcinoma (cSCC) Patients: Case Series and Algorithm. Clin Cosmet Investig Dermatol. 2023;16:925–935.
- Saleeby E, Bielinski K, Fitzgerald A, et al. A Prospective, Multi-Center Clinical Utility Study Demonstrates That the 40-Gene Expression Profile (40-GEP) Test Impacts Clinical Management for Medicare-Eligible Patients with High-Risk Cutaneous Squamous Cell Carcinoma (cSCC). SKIN The Journal of Cutaneous Medicine. 2022;6(6):482–496.
- Hooper PB, Farberg AS, Fitzgerald AL, et al. Real-World Evidence Shows Clinicians Appropriately Use the Prognostic 40-Gene Expression Profile (40-GEP) Test for High-Risk Cutaneous Squamous Cell Carcinoma (cSCC) Patients. Cancer Invest. 2022;40(10):911–922.
- Koyfman SA, Wysong A, Arron S, et al. Improved risk stratification in an adjuvant radiation therapy (ART) eligible cutaneous squamous cell carcinoma (cSCC) patient population by integration of the 40-gene expression profile prognostic test (40-GEP). JCO. 2021;39(15_suppl):e21589–e21589.
- Arron ST, Canueto J, Siegel JJ, et al. Association of a 40-Gene Expression Profile With Risk of Metastatic Disease Progression of Cutaneous Squamous Cell Carcinoma (cSCC) and Benefit of Adjuvant Radiation Therapy. Fall Clinical Dermatology Conference, 2023.
- Garrett GL, Lowenstein SE, Singer JP, et al. Trends of skin cancer mortality after transplantation in the United States: 1987 to 2013. J Am Acad Dermatol. 2016;75(1):106–112.
- Karia PS, Han J, Schmults CD. Cutaneous squamous cell carcinoma: estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J Am Acad Dermatol. 2013;68(6):957–966.
- Lukowiak TM, Aizman L, Perz A, et al. Association of Age, Sex, Race, and Geographic Region With Variation of the Ratio of Basal Cell to Cutaneous Squamous Cell Carcinomas in the United States. JAMA Dermatol. 2020;156(11):1192–1198.
- Singh G, Prasai A, Fitzgerald AL, et al. Clinicians’ use of the 40-gene expression profile (40-GEP) test for high-risk cutaneous squamous cell carcinoma (cSCC) patients is consistent with intended use parameters. In: AAD; 2023.
- Hsia TC, Tu CY, Fang HY, et al. Cost and effectiveness of image guided radiotherapy for non-operated localized lung cancer: a population-based propensity score-matched analysis. J Thorac Dis. 2015;7(9):1643–1649.
- Razfar A, Mundi J, Grogan T, et al. IMRT for head and neck cancer: Cost implications. Am J Otolaryngol. 2016;37(6):479–483.
- Thaker NG, Boyce-Fappiano D, Ning MS, et al. Activity-Based Costing of Intensity Modulated Proton versus Photon Therapy for Oropharyngeal Cancer. Int J Part Ther. 2021;8(1):374–382.
- Staackmann C, Schild Se, Rades D. Palliative Radiotherapy for Cutaneous Squamous Cell Carcinoma of the Head-and-Neck Region. In Vivo. 2021;35(4):2283–2288.