J Clin Aesthet Dermatol. 2025;18(11):21–23.
by Joshua Burshtein, MD, and Todd Schlesinger, MD
Dr. Burshtein is with the Department of Dermatology at the University of Illinois-Chicago in Chicago, Illinois. Dr. Schlesinger is with the Clinical Research Center of the Carolinas in Charleston, South Carolina, and the Department of Dermatology at George Washington University School of Medicine and Health Sciences in Washington, District of Columbia.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Burshtein declares no conflicts of interest relevant to the content of this article. Dr. Schlesinger serves as a consultant, investigator, speaker, and/or advisor for AbbVie, Almirall, Allergan, ASLAN, Arcutis, Biofrontera, Beiersdorf, Benev, Bristol-Myers Squibb, Castle Biosciences, Dermsquared, ExoCoBio, Galderma, Incyte, Janssen, LEO, Lilly, L’Oreal, Medicus, Novartis, Pfizer, RBC Consultants, Regeneron, Sanofi, Sun Pharma, Takeda, UCB, and Verrica. He receives a salary from Avant Health.
ABSTRACT: Cutaneous squamous cell carcinoma (cSCC) is the second most common skin cancer worldwide. Treatments of locally advanced cSCC (laCSCC) and metastatic cSCC (mCSCC) have been evolving with the introduction of immunotherapies, which target the programmed cell death 1 (PD-1)/programmed cell death ligand 1 (PD-L1) pathway. Recently, cosibelimab was the first PD-L1 antibody approved by the United States Food and Drug Administration for the treatment of patients with laCSCC or mCSCC who are not candidates for curative surgery or radiation. Cosibelimab not only offers blockade of PD-L1 but also induces antibody-dependent cellular cytotoxicity. In its pivotal clinical trial, the objective response rate for patients with mCSCC treated with cosibelimab was 47.4%. Due to the different mechanisms of action, differences have been noted in the safety between PD-1 inhibitors and PD-L1 inhibitors, with PD-L1 inhibitors having a lower occurrence of Grade 3 or higher treatment-related adverse events, including immune-related adverse events. Additional real-world studies are needed to further evaluate its efficacy and safety. Cosibelimab is a novel immune checkpoint inhibitor and represents a promising therapeutic option for patients with advanced cSCC. Keywords: Advanced squamous cell carcinoma, locally advanced cSCC, metastatic cSCC, immunotherapy, PD-1, PD-L1
Introduction
Cutaneous squamous cell carcinoma (cSCC) is the second most common skin cancer in the world.1,2 Treatment of locally advanced cSCC (laCSCC) and metastatic cSCC (mCSCC) have been evolving with the introduction of immunotherapies which target the programmed cell death 1 (PD-1)/programmed cell death ligand 1 (PD-L1) pathway (also known as checkpoint inhibitors). Cemiplimab is a high-affinity PD-1 antibody and was the first therapy approved by the United States Food and Drug Administration (FDA) for patients with laCSCC and mCSCC who are not eligible for curative surgery or radiation.3 Pembrolizumab is another PD-1 antibody that is FDA-approved for the treatment of laCSCC and recurrent or metastatic cSCC not curable by surgery or radiation.4 A multidisciplinary expert consensus panel recommended that immunotherapy should be considered as a first-line therapy in patients with laCSCC and mCSCC.5 Recently, cosibelimab was the first PD-L1 antibody FDA-approved for the treatment of patients with laCSCC or mCSCC who are not candidates for curative surgery or radiation.6
Efficacy
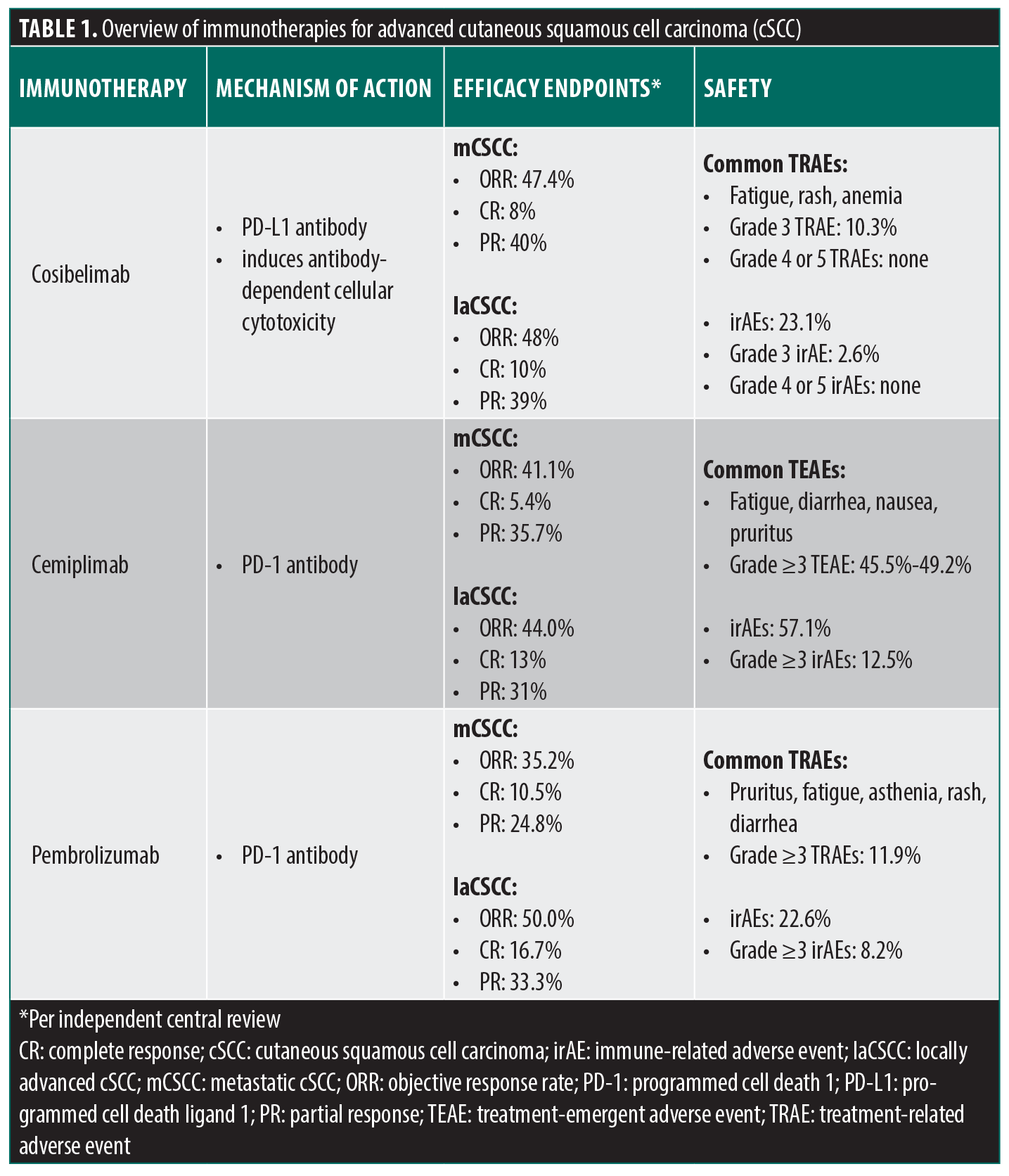
Cosibelimab not only offers blockade of PD-L1, but also induces antibody-dependent cellular cytotoxicity (ADCC), a dual mechanism that may help overcome resistance seen with other checkpoint inhibitors.7 This is due to the functional Fc domain, which activates natural killer cells and leads to direct lysis of tumor cells.7 Cosibelimab received FDA-approval in December 2024, based on results from a Phase I clinical trial of 78 patients with mCSCC.8 In this study, objective response rate (ORR), assessed by independent central review (ICR) according to Response Evaluation Criteria in Solid Tumors (RECIST) criteria8, was 47.4% with a median follow-up of 15.4 months. Of these patients, 8% had a complete response and 40% had a partial response. Median duration of response was not reached but response was ongoing in 73% of patients. The Kaplan-Meier estimated probabilities of sustained response at 6, 12, and 24 months were 88.9%, 73.0%, and 73.0%, respectively. Notably, in the Phase I trial, 60.3% of patients underwent prior surgery for their malignancy and 65.4% underwent radiation. The majority (91.0%) did not receive prior systemic therapy for cSCC.8 The FDA-approved dose of cosibelimab is a 1200mg infusion over 60 minutes every three weeks until disease progression or unacceptable toxicity.9
Of 31 patients with laCSCC, ORR was 48% with 10% achieving complete response and 39% achieving partial response.9 For those who responded to therapy, the median duration of response was 17.7 months, with 87% having at least 6 months of response and 20% having at least 12 months.9 In the long-term analysis, with a median follow-up duration of 29.3 months for those with mCSCC and 24.1 months for those with laCSCC, the ORR per ICR was 50.0% and 54.8%, respectively.10,11 Among patients with mCSCC, 12.8% achieved a complete response, while 25.8% of those with laCSCC experienced a complete response.10,11 The probability of maintaining response at 24 months was 72.1% for mCSCC and 80.2% for laCSCC.10,11
Though there have not been any head-to-head clinical trials, the efficacy of cosibelimab is consistent with that of other immunotherapies for cSCC (Table 1). In the Phase II clinical trial of patients with mCSCC treated with cemiplimab, combined ORR for mCSCC with both forms of treatment (fixed dosing and weight-based dosing) was 45.3% per ICR.12 The estimate for duration of response was 95% for fixed dosing at 8 months and 88.9% for weight-based dosing at 12 months.12 In patients with laCSCC receiving cemiplimab with weight-based dosing every two weeks, the ORR was 44% as assessed by ICR and 53% by investigator assessment.13 A complete response was observed in 13% of patients, while 31% achieved a partial response, and at 12 months, an estimated 87.8% of patients maintained their response.13 The FDA-approved dose of cemiplimab is 350mg every three weeks until disease progression, unacceptable toxicity, or up to 24 months.3
For those with cSCC treated with pembrolizumab, ORR for both laCSCC and mCSCC combined was 40.3%.14 For those with laCSCC, ORR was 50.0%, with complete response in 16.7% and partial response in 33.3%. Most (84.1%) of responses were estimated to last at least 12 months. For mCSCC, ORR was 35.2%, and of these, 10.5% achieved complete response, 24.8% achieved partial response, and 77.8% of responses were estimated to last at least 12 months.14
Safety
Due to the different mechanisms of action, differences in the safety between PD-1 inhibitors and PD-L1 inhibitors have been noted, with PD-L1 inhibitors having lower occurrence of Grade 3 or higher treatment-related adverse events (TRAEs), including immune-related adverse events (irAEs).13,16 This is hypothesized to be due to the fact that while PD-1 antibodies block interaction of T cells with PD-L1 and PD-L2, PD-L1 antibodies are specific to PD-L1 and preserve PD-L2 signaling.7,8 In the Phase I trial for cosibelimab, common TRAEs were fatigue (26.9%), rash (16.7%), and anemia (15.4%).7 Only 10.3% of patients had a Grade 3 TRAE and there were no Grade 4 or 5 TRAEs.7 IrAEs were recorded in 23.1% of patients with 2.6% having Grade 3 AEs and no Grade 4 or 5 AEs. There were 11.5% of patients who discontinued therapy due to an AE though only 2.6% were deemed related to cosibelimab.7 In the Phase I clinical trial, three patients experienced AEs that led to death, and all were considered unrelated to cosibelimab.7 In the long-term analysis, the most common treatment-emergent AEs were consistent with those previously reported.10,11 There were 27.6% who experienced irAEs, with only 3.6% being Grade 3 and no Grade ≥4 irAEs.10,11
Though there have not been any head-to-head clinical trials, other immunotherapies for cSCC have slightly higher reported AEs. For cemiplimab, the long-term analysis of the Phase II trial found that TEAEs Grade ≥3 occurred in 45.5 to 49.2% of patients.17 Of these, 31.1 to 34.5% were considered serious and 7.3% of patients discontinued treatment due to a TEAE.17 In the Phase II trial for pembrolizumab, 69.2% had at least one TRAE and 11.9% experienced TRAEs Grade ≥3. Only 8.8% of patients discontinued treatment due to any TRAE.14
Future Considerations
Additional real-world studies are needed to further evaluate the efficacy and safety of cosibelimab. Currently, data on its use in immunocompromised patients are limited. However, given its mechanism of action targeting ADCC, cosibelimab may offer potential benefit in this population, warranting further investigation.
Conclusion
Cosibelimab represents a promising therapeutic option for patients with advanced cSCC. As a novel immune checkpoint inhibitor, it exerts its effects through PD-L1 blockade and potential ADCC, distinguishing it from the current immunotherapy landscape. Clinical trial data have demonstrated ORRs comparable to established agents such as pembrolizumab and cemiplimab. Its favorable safety profile, including a lower incidence of high-grade irAEs, underscores its potential as a viable and well-tolerated treatment for cSCC.
References
- Dessinioti C, Pitoulias M, Stratigos AJ. Epidemiology of advanced cutaneous squamous cell carcinoma. J Eur Acad Dermatol Venereol. 2022;36(1):39-50.
- Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78(3):560-578.
- LIBTAYO (cemiplimab-rwlc). Package insert. Regeneron; 2024. Accessed February 1, 2025. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761097s014lbl.pdf
- KEYTRUDA® (pembrolizumab). Package insert. Merck; 2025. Accessed February 1, 2025. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/125514s096lbl.pdf
- Rabinowits G, Migden MR, Schlesinger TE, et al. Evidence-based consensus recommendations for the evolving treatment of patients with high-risk and advanced cutaneous squamous cell carcinoma. JID Innov. 2021;1(4):100045.
- Lee A. Cosibelimab: first approval. Drugs. 2025;85(5):695-698.
- Idris OA, Westgate D, Saadaie Jahromi B, et al. PD-L1 inhibitor cosibelimab for cutaneous squamous cell carcinoma: comprehensive evaluation of efficacy, mechanism, and clinical trial insights. Biomedicines. 2025;13(4):889.
- Clingan P, Ladwa R, Brungs D, et al. Efficacy and safety of cosibelimab, an anti-PD-L1 antibody, in metastatic cutaneous squamous cell carcinoma. J Immunother Cancer. 2023;11(10):e007637.
- UNLOXCYT (cosibelimab-ipdl): Prescribing information. Checkpoint Therapeutics. 2024. Accessed July 1, 2025. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/761297s000lbl.pdf.
- Muñoz-Couselo E, Montaudie H, Berciano Guerrero MA, et al. 1136P Cosibelimab in advanced cutaneous squamous cell carcinoma (cSCC): longer-term efficacy and safety results from pivotal study. Ann Oncol. 2024;35:S745-S746.
- Ruiz ES, Muñoz-Couselo E, Montaudié H, et al. Efficacy and safety of cosibelimab in advanced cutaneous squamous cell carcinoma: Results from a Pivotal Open-label Study with a median follow-up of ≥2 years. J Am Acad Dermatol. Published online September 29, 2025.
- Rischin D, Migden MR, Lim AM, et al. Phase II study of cemiplimab in patients with metastatic cutaneous squamous cell carcinoma: primary analysis of fixed-dosing, long-term outcome of weight-based dosing. J Immunother Cancer. 2020;8(1):e000775.
- Migden MR, Khushalani NI, Chang ALS, et al. Cemiplimab in locally advanced cutaneous squamous cell carcinoma: results from an open-label, Phase II, single-arm trial. Lancet Oncol. 2020;21(2):294-305.
- Hughes BGM, Munoz-Couselo E, Mortier L, et al. Pembrolizumab for locally advanced and recurrent/metastatic cutaneous squamous cell carcinoma (KEYNOTE-629 study): an open-label, nonrandomized, multicenter, Phase II trial. Ann Oncol. 2021;32(10):1276-1285.
- Wang Y, Zhou S, Yang F, et al. Treatment-related adverse events of PD-1 and PD-L1 inhibitors in clinical trials: a systematic review and meta-analysis. JAMA Oncol. 2019;5(7):1008-1019.
- Baxi S, Yang A, Gennarelli RL, et al. Immune-related adverse events for anti-PD-1 and anti-PD-L1 drugs: systematic review and meta-analysis. BMJ. 2018;360:k793.
- Hughes BGM, Guminski A, Bowyer S, et al. A Phase II open-label study of cemiplimab in patients with advanced cutaneous squamous cell carcinoma (EMPOWER-CSCC-1): final long-term analysis of groups 1, 2, and 3, and primary analysis of fixed-dose treatment group 6. J Am Acad Dermatol. 2025;92(1):68-77.