J Clin Aesthet Dermatol. 2024;17(2):32–42.
J Clin Aesthet Dermatol. 2024;17(2):32–42.
by Blanca Estupiñan MD; Adam Souchik, MD; Alexandra Kiszluk, BS; and Shraddha Desai, MD
Drs. Estupiñan, Souchik, and Desai are with the Division of Dermatology at Loyola University Medical Center in Maywood, Illinois. Ms. Kiszluk is with the Stritch School of Medicine at Loyola University Chicago in Maywood, Illinois. Additionally, Dr. Desai is with Duly Health and Care Dermatology in Naperville, Illinois and the Division of Dermatology at Rush University in Chicago, Illinois.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors report no conflicts of interest relevant to this article.
ABSTRACT: Objective. Our aim was to review the current and emerging dermatological applications of the novel thermomechanical fractional injury (TMFI) device, Tixel® (Novoxel, Netanya, Israel).
Methods. A systematic review of PubMed using the search terms of “Tixel”, “thermomechanical fractional”, [“thermomechanical ablation” and “skin”], and [“thermomechanical ablation” and “dermatology”].
Results. Thirty-six articles matched our inquiry. Fifteen articles did not meet inclusion criteria. Of the remaining 21 articles, eight were related to device-assisted drug delivery, seven related to photoaging, and seven related to scientific/ preclinical exploration. Preclinical studies have shown ablative and non-ablative microchannel formation similar to that of CO₂ laser but without charring, with clinical studies demonstrating efficacy for a wide range of applications including rhytides, hypertrophic scarring, infantile hemangiomas, and acne/rosacea. The treatment is well tolerated with minimal discomfort and downtime, showing promise for pain-averse and pediatric populations. Few adverse events have been reported, with a high degree of safety demonstrated in all Fitzpatrick types.
Limitations. Heterogeneous result reporting among studies. Limited number of randomized controlled trials.
Conclusion. Tixel® is an emerging TMFI device with a wide range of current and potential applications, including device-assisted drug delivery and treatment of rhytides, photoaging, and scars among other conditions. The device has both ablative and non-ablative settings and has been safely used in all Fitzpatrick skin types. Larger and randomized controlled trials are needed to compare this device to current standard of care treatments.
Keywords: thermomechanical, fractional ablation, Tixel, rhytides, device-assisted drug delivery.
Tixel (Novoxel, Netanya, Israel) is a thermomechanical fractional injury (TMFI) device that gained U.S. Food and Drug Administration (FDA) approval in February 2021 for procedures necessitating ablation and resurfacing of the skin.1 It features non-ablative and ablative settings and has gained traction in recent years for a variety of dermatologic conditions. The device has a ceramic base that is heated to 400°C which transfers heat to 81 (9×9) blunt, titanium tips in a 10x10mm array that do not pierce the skin.2 The tips are applied with pulse durations of 5-18ms at a protrusion depth of 100-1000µm. This creates discrete coagulation zones in the target tissue. The device does not have a target chromophore and is theoretically safe in all skin types.
This review aims to highlight therapeutic uses, including off-label indications, of this new, emerging thermomechanical fractional device. We also reviewed studies that investigated the device’s ability to increase drug delivery. Given the device’s recent approval, it is important to note that there is a lack of randomized controlled trials, and therefore, a systematic review following PRISMA guidelines was conducted with limitations. The purpose of this review is to assess current uses and safety of Tixel and to highlight the need for additional research.
Methods
A comprehensive search of PubMed of was conducted. The search terms “Tixel”, “thermomechanical ablation”, [“thermomechanical ablation” and “skin”], and [“thermomechanical ablation” and “dermatology”] were used to identify studies from the years 2009 to 2023. The last search was conducted March 30th, 2023. Inclusion criteria included: 1) English language, 2) use of thermomechanical ablative fractional device (Tixel), 3) original articles, and 4) relevance to dermatology or skin properties. For clinical studies, case series, prospective trials of any size, and retrospective chart reviews were included. Conference abstracts, single case reports, commentaries, and reviews of studies were excluded. Pre-clinical trials must include in-vivo or ex-vivo portions of the study. Outcomes of interest included objective grading scales, physician-reported and patient-reported outcomes, safety data, and pre-clinical data with visualization of findings (i.e. histology, optical coherence tomography).
All articles were independently reviewed by at least two authors to ensure eligibility criteria were met. Discrepancies were discussed between authors. Each author collected data independently from the divided articles and input data into a shared spreadsheet for review by co-authors. This review aims to examine the skin effects of a thermomechanical ablation fractional device (Tixel) and its current uses. Clinical studies were assessed on effectiveness and safety of Tixel by subjective and objective measures, while pre-clinical studies were assessed on visualization of tissue responses to treatment.
Results
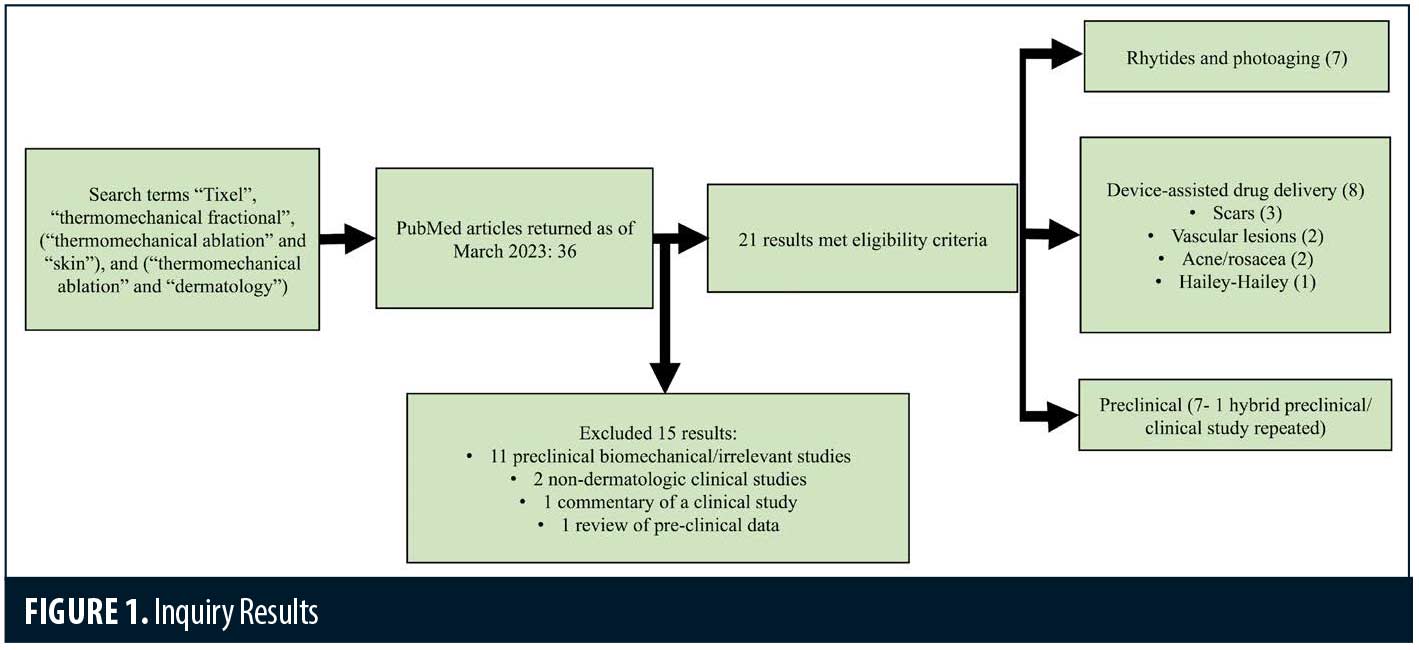
The database inquiries yielded 36 unique articles. Screening of articles by title and abstract eliminated 11 preclinical biomechanical/irrelevant studies, two non-dermatologic clinical studies, one commentary of a clinical study and one review of preclinical data. A total of 21 articles met eligibility criteria (Figure 1).
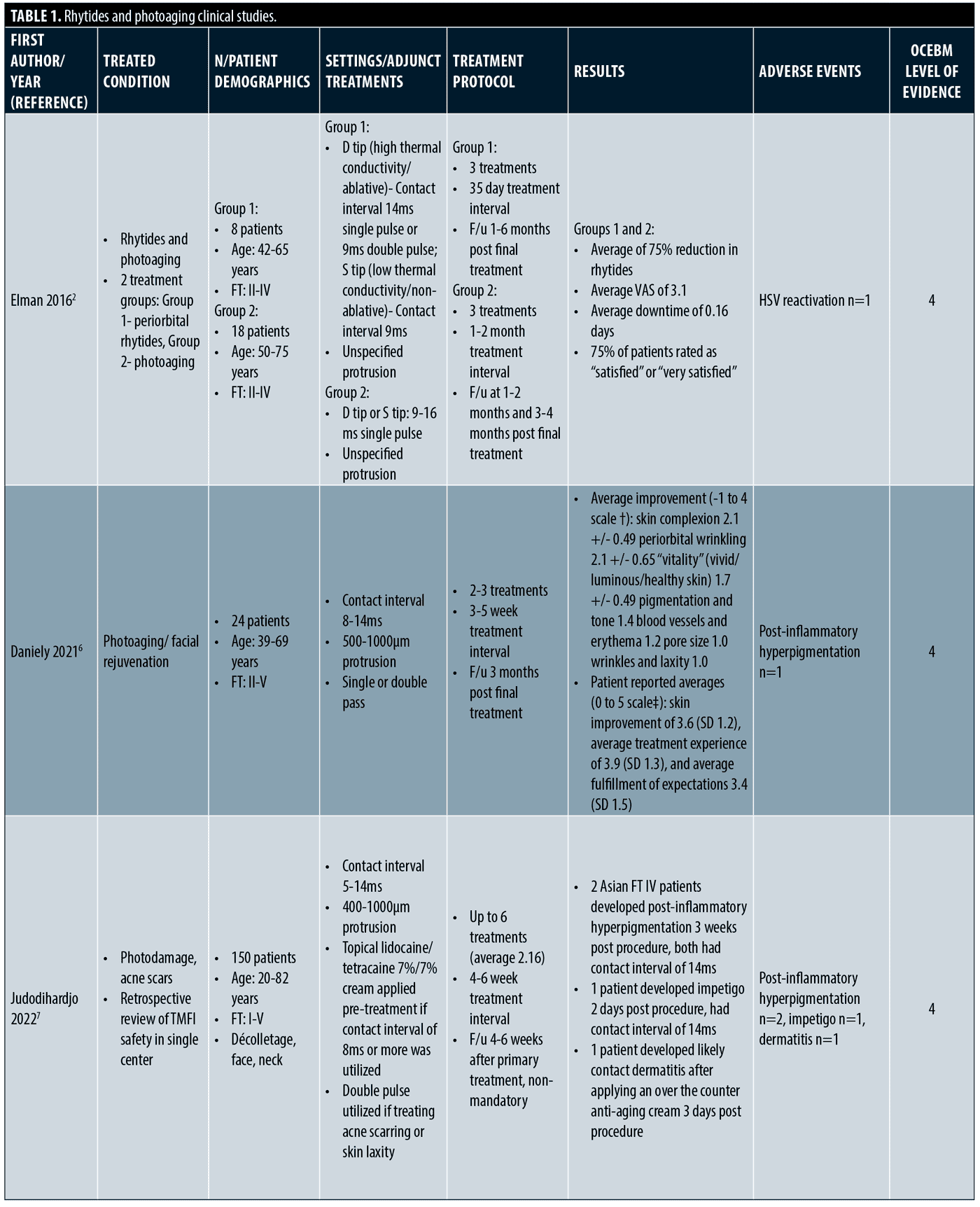
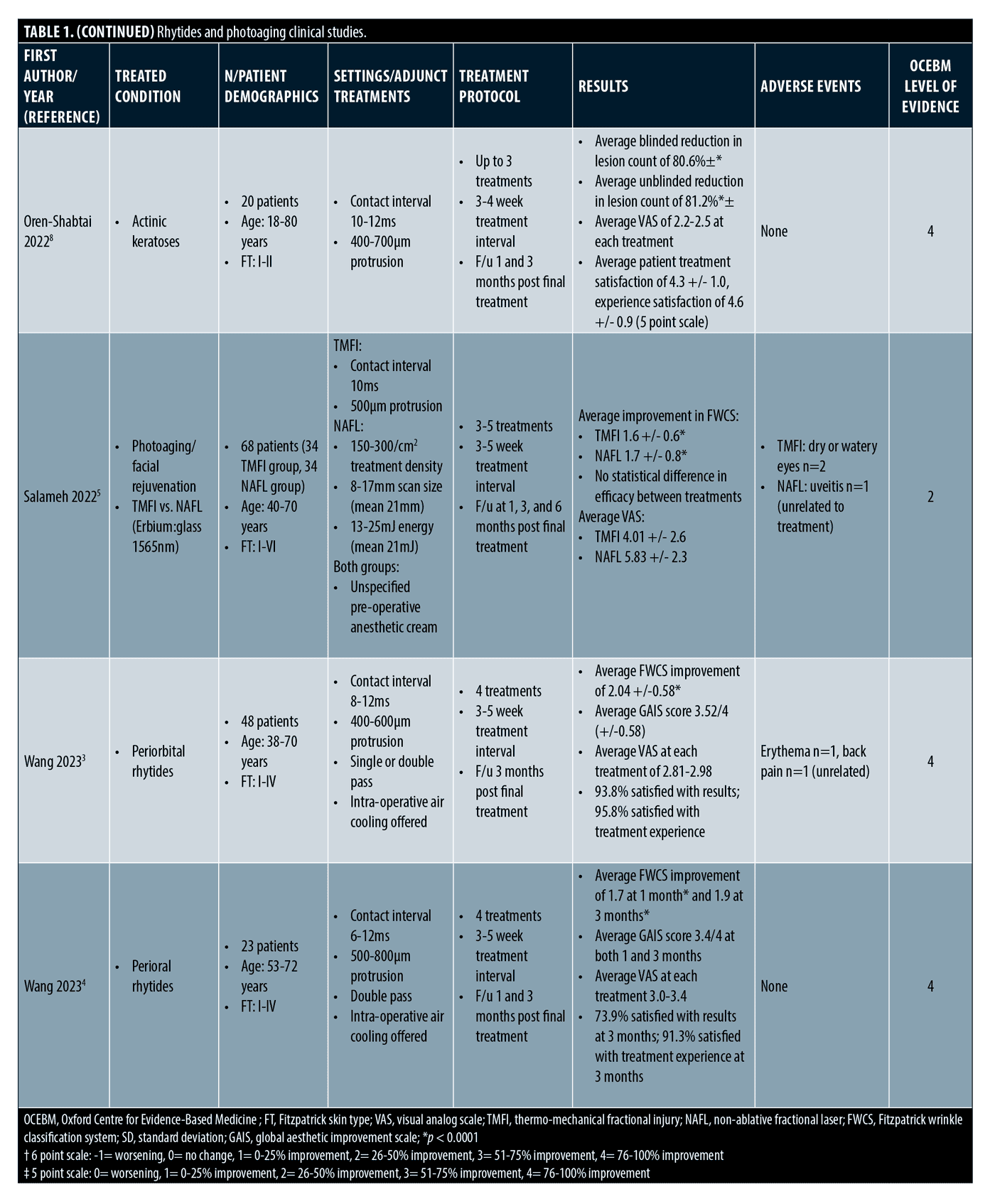
Rhytides and photoaging. The majority of clinical trials focus on rhytides and photoaging. (Table 1) A total of 303 patients were treated with Tixel for rhytides and rejuvenation, with an additional 20 receiving treatment for facial and scalp actinic keratoses. Treatment areas included the scalp, full face, periorbital, perioral, neck, and décolletage. Fitzpatrick Skin Types I to V were treated. Treatment settings ranged from 6 to 16ms pulse duration and protrusion depth ranged from 400 to 1,000µm. Most studies delivered 2 to 4 treatments with final follow up ranging from 1 to 6 months.
The first published clinical study treated 10 subjects for periorbital and perioral rhytides and another 23 subjects for mild to moderate photodamage.2 After three treatments, all patients had improvement of photodamage and complexion, and about three quarters of patients had improvement in wrinkles, some having improvement after one treatment. Erythema resolved within three days for most patients, but four to six days if more aggressive settings were used. Microcrusting appeared for some patients at day five. All subjects agreed that no anesthesia was necessary for the treatments. Downtime ranged from 0 to 1 day for over 90 percent of patients.
More recently, a clinical trial with 48 subjects found significant improvement of moderate to severe periorbital rhytides after treating above and below the eyebrow and lower eyelid.3 Both investigators and three blinded reviewers reviewed final outcomes. All participants responded to treatment with 95.9 percent having a 51 percent or greater improvement on the Global Aesthetic Improvement Scale (GAIS), over half of whom had a 76 to 100 percent improvement. Fitzpatrick Wrinkle Classification System (FWCS) scores demonstrated a significant improvement of rhytides. Patients reported a low level of pain at 2 to 3 out of 10 without topical anesthetic and only optional intraoperative air cooling and post-procedural cold packs offered. Expected erythema and edema lasted three days or less, and subjects were able to return to normal activities in one day or less. The same authors conducted a prospective trial on perioral rhytides in 23 subjects.4 After four treatments, there was a statistically significant improvement in rhytides and nearly 70 percent improved 76 percent to 100 percent on the GAIS. All patients responded to treatment. Similar to the prior study, only optional air cooling was offered and patients reported a low level of pain, with an average Visual Analogue Scale (VAS) score of 3.1.
A prospective randomized controlled study comparing Tixel to non-ablative fractional (NAFL) 1565nm erbium: glass fiber laser included 68 patients with mild to moderate periorbital rhytides.5 Three blinded physicians rated outcomes and found a moderate improvement in both treatment arms. Both treatments had a statistically significant improvement in rhytides and there was not a significant difference between the groups. With pre-operative topical anesthetic, the VAS score average was significantly lower in the Tixel treatment arm (p<0.05). Microcrusting was present in 52 percent of Tixel and 16 percent of NAFL subjects. There was no difference in resolution of erythema, edema, and downtown time. This study suggests that Tixel is safe and comparable to NAFL in the treatment of periorbital rhytides but is better tolerated.
One group retrospectively reviewed 24 patient cases, including six with Fitzpatrick Skin Type IV+, who underwent two to three Tixel treatments at about one month intervals.6 By rating photographs, reviewers appreciated improvements in rhytides, pore size, pigmentation, erythema, laxity, and overall complexion. Erythema lasting 3 to 6 days was present in three patients. One Fitzpatrick Type III patient who received higher settings with a protrusion depth of 800µm and 14ms pulse duration with more passes had hyperpigmentation, which resolved with bleaching agents. Average downtime was less than two days. A larger retrospective study assessing safety and tolerability of Tixel included 150 patients with photoaging or acne scarring.7 Areas treated included the face, including periorbital and perioral areas, neck, and décolletage. The majority of treatments had higher settings with protrusion of 700 and 1,000µm and pulse durations of 14ms. Of note, 39 Fitzpatrick Type IV and 16 Fitzpatrick Type V subjects received 10-14ms pulse durations. Out of the 327 total treatments, there were only two cases of hyperpigmentation, both in Asian patients with Fitzpatrick Skin Type IV. They had higher settings with pulse duration of 14ms and protrusion of 700 or 1,000µm. Fortunately, the pigmentation self-resolved in three months without intervention.
Tixel was also used to treat scalp and facial actinic keratoses with mild to moderate thickness in a prospective trial of 20 patients.8 There was a decreased lesion burden after 2 to 3 treatments as evaluated by blinded and unblinded evaluators. At final follow up three months after treatment, 75 percent of patients had 76 to 100 percent improvement. Regarding side effects, redness, edema, and scabs were noted for 0 to 2 days and there were no adverse events. The treatment was well tolerated with a low level of pain and subjects were able to return to activities in two days or less.
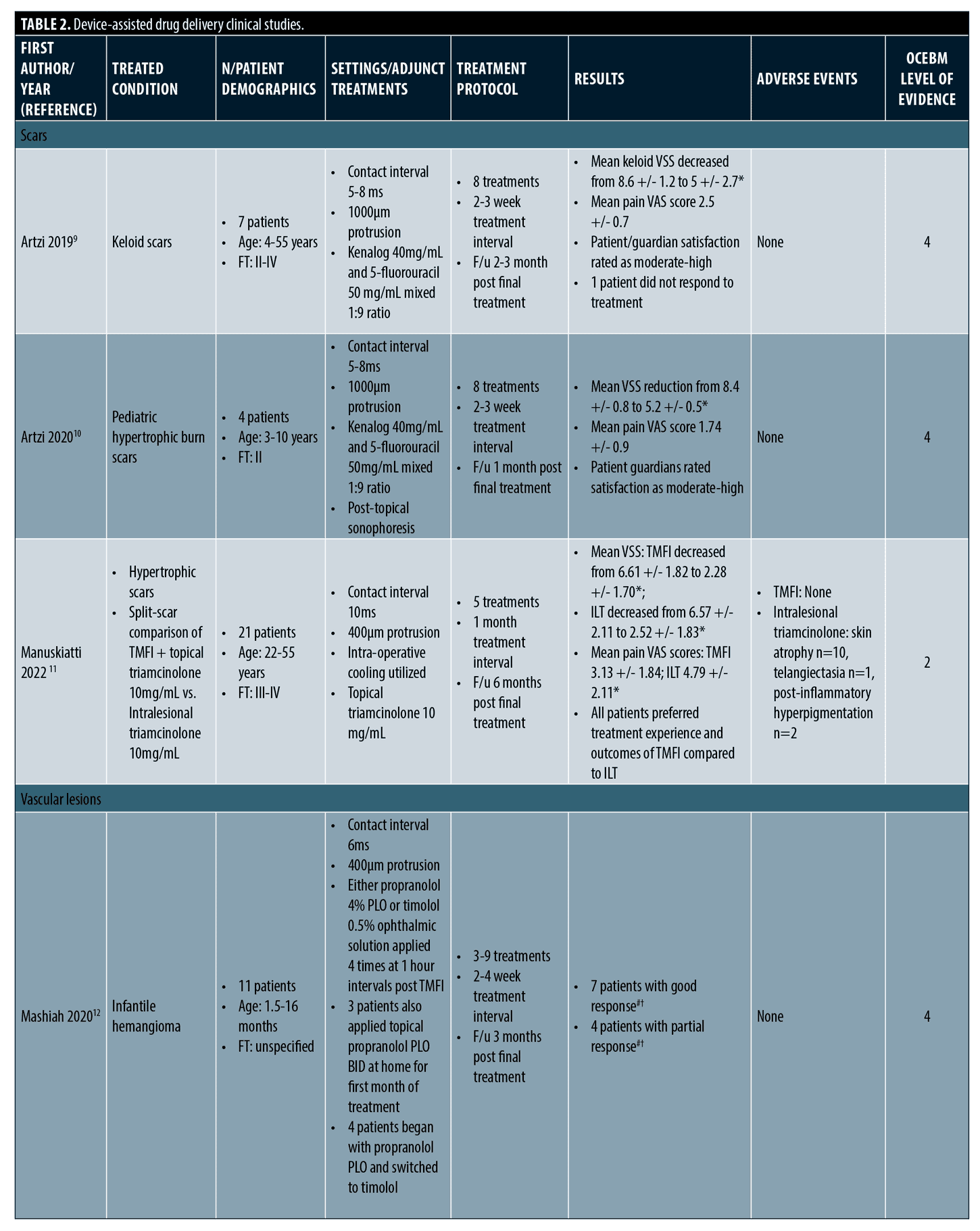
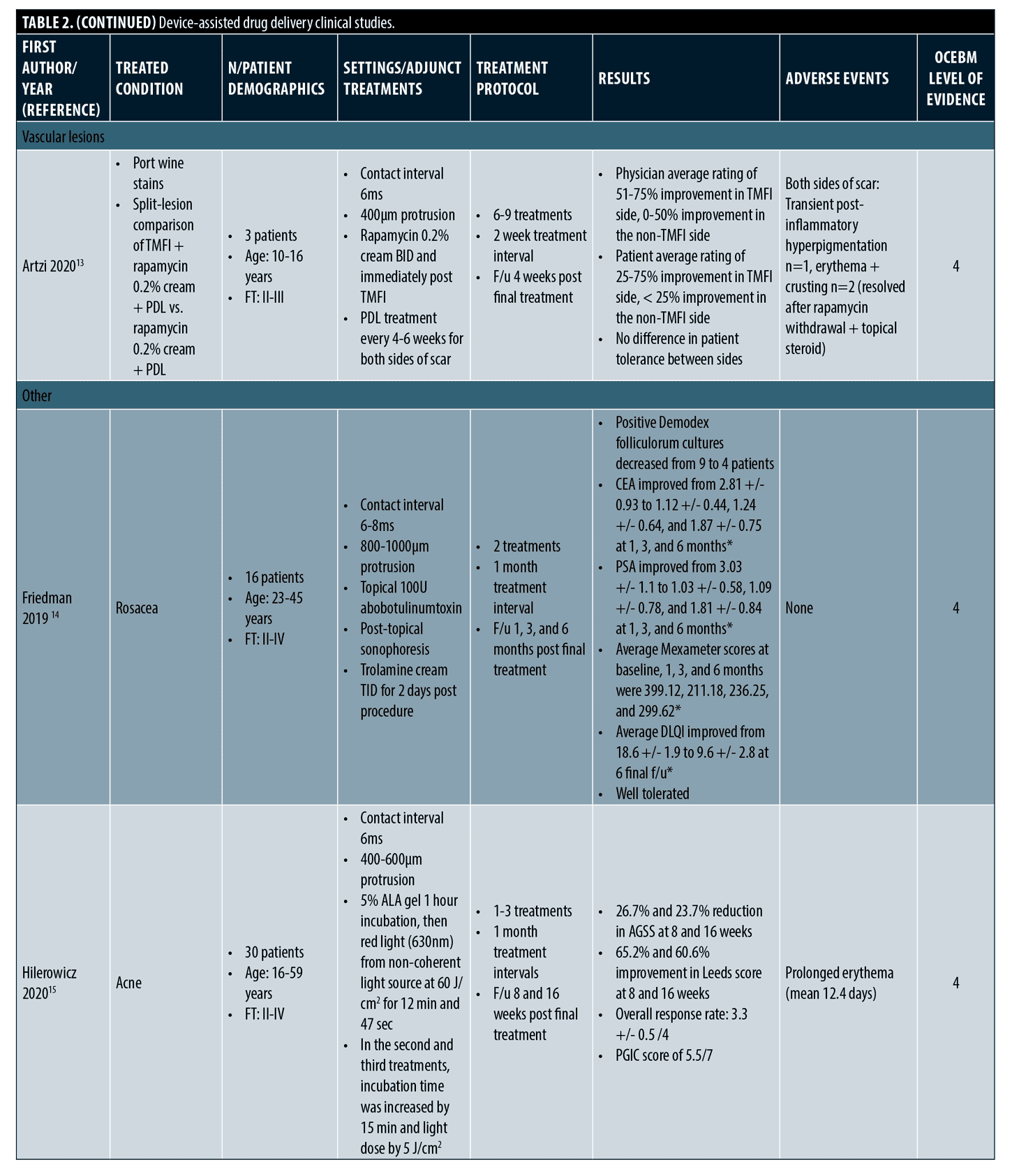
Device-assisted drug delivery. Study design, treatment protocols, and key findings for device-assisted drug delivery studies are summarized in Table 2.
Scars. Regarding treatment of scars, treatment sites included the trunk and extremities and there were no adverse events, including in Fitzpatrick Type IV patients. Typically, a protrusion of 1,000µm and pulse duration of 5-10ms was utilized. Treatments ranged from 5 to 8 sessions over 2 to 4 week intervals. Final follow up ranged from 1 to 6 months. Two studies on hypertrophic and keloid scars including a pooled total of 11 patients, aged as young as three years old, found Tixel-assisted drug delivery of 1:9 triamcinolone 40mg/mL: 5-fluorouracil 50mg/mL to be effective in improving scar quality.9,10 Mean Vancouver Scar Scale (VSS) scores improved significantly and patients (or their guardians) were highly satisfied with the treatment experience. There was one non-responder and no adverse events occurred. One split-scar study comparing Tixel-assisted triamcinolone 10mg/mL delivery to standard intralesional triamcinolone found comparable outcomes in scar improvements.11 The Tixel treatment arm was better tolerated and preferred by patients and did not result in adverse events as seen with the intralesional steroid. Further split-scar studies are needed to compare Tixel to standard of care scar treatments.
Vascular lesions. A retrospective study included 11 patients with infantile hemangiomas on the head, neck, and extremities that were treated with Tixel-assisted delivery of timolol 0.5% eye drops and/or a propranolol 4% gel.12 Tixel was used with a protrusion of 400µm and pulse duration of 6ms and delivered over a series of 4 to 9 treatments. Based on grading of size regression, color lightening, flattening, and deep component reduction, all patients had “good” or “partial” response to treatment. None had “poor” or “no response”. Of note, a large 20 x 6cm and other substantially thick hemangiomas had a “good” response to treatment. No recurrence of lesions or systemic or local adverse events occurred. The authors noted that with an average of 5.5 treatments and total duration of 17 weeks, the addition of Tixel-assisted drug delivery provides a shorter treatment course compared to the standard 5 to 7.5 months of beta-blocker monotherapy.
In one case series, three pediatric patients with port wine stains, who had insufficient response to pulsed dye laser treatments, underwent Tixel-assisted drug delivery of rapamycin.13 Half of each lesion received 595-nm pulsed dye laser, Tixel 2 to 14 days later, and topical rapamycin 0.2% cream immediately after Tixel and twice daily. The other half was treated with pulsed dye laser and rapamycin 0.2% cream only. Investigators found that the Tixel-treated halves had a higher clearance rate and patients were more satisfied with the outcome. There was no difference in side effects between treatment arms.
Acne and rosacea. For treatment of resistant rosacea facial flushing, 16 patients underwent Tixel-assisted abobotulinumtoxin A delivery. (14) Patients underwent two treatments of Tixel at 800-1,000µm protrusion with 6-8ms pulse durations, followed by topical application of 100 units of botulinum toxin diluted in saline, applied with sonophoresis. Both clinician and patient assessments of erythema at baseline were around a “moderate” grading, which improved to an “almost none” to “mild” grading at 1, 3, and 6 months post-treatment. This effect was also demonstrated with Mexameter measurements, which significantly improved as well. Additionally, Demodex cultures were significantly reduced and quality of life scores significantly improved. The treatment was well tolerated.
As an adjunct to photodynamic therapy for acne, Tixel-assisted drug delivery of aminolevulinic acid 5% gel was used on 30 patients, including pediatric patients and Fitzpatrick Types II-IV.15 Treatment was delivered with a protrusion of 400-600µm and 6ms pulse duration. Next, the gel was applied on the face under occlusion for one hour and then exposed to 630nm red light. After an average of about two treatments, the burden of inflammatory lesions, overall lesions and erythema improved and sustained benefit was appreciated at 16 weeks after the final treatment. Patients reported high satisfaction and low VAS pain scores (average 3.3), though half of patients had prolonged erythema lasting almost two weeks. Average downtime was less than one week.
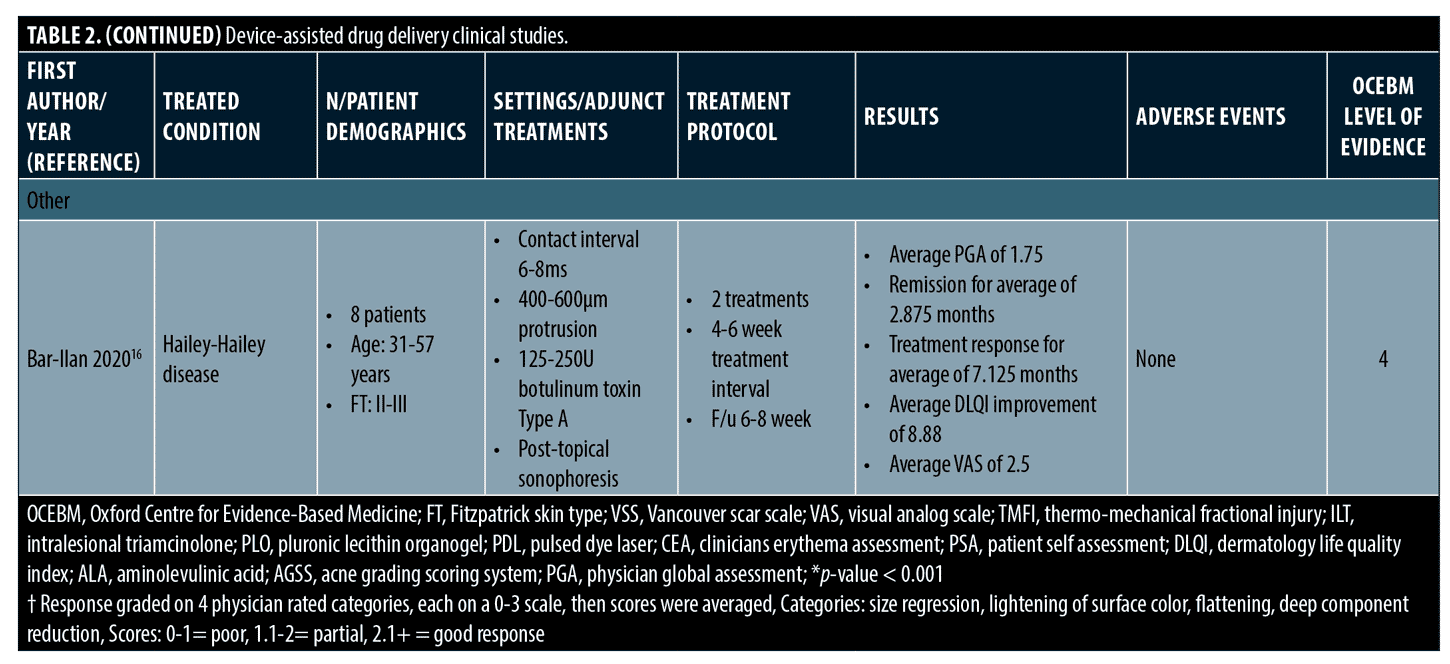
Hailey-Hailey disease. Given botulinum toxin’s effects on decreasing sweat production and potential resultant decrease in microorganism overgrowth, one group studied Tixel-assisted botulinum toxin delivery in eight patients with biopsy-confirmed Hailey-Hailey disease.16 After Tixel treatment, 125-250 units of botulinum toxin A diluted in saline was applied with sonophoresis. Findings were promising with 7/8 patients having a good or partial response at 6 to 8 weeks after treatment and significantly improved Physician Global Assessment (PGA) and Dermatology Life Quality Index (DLQI) scores. Recurrence occurred an average of seven months and up to one year after treatment and all treatment sites showed similar effectiveness. There were no systemic or local side effects.
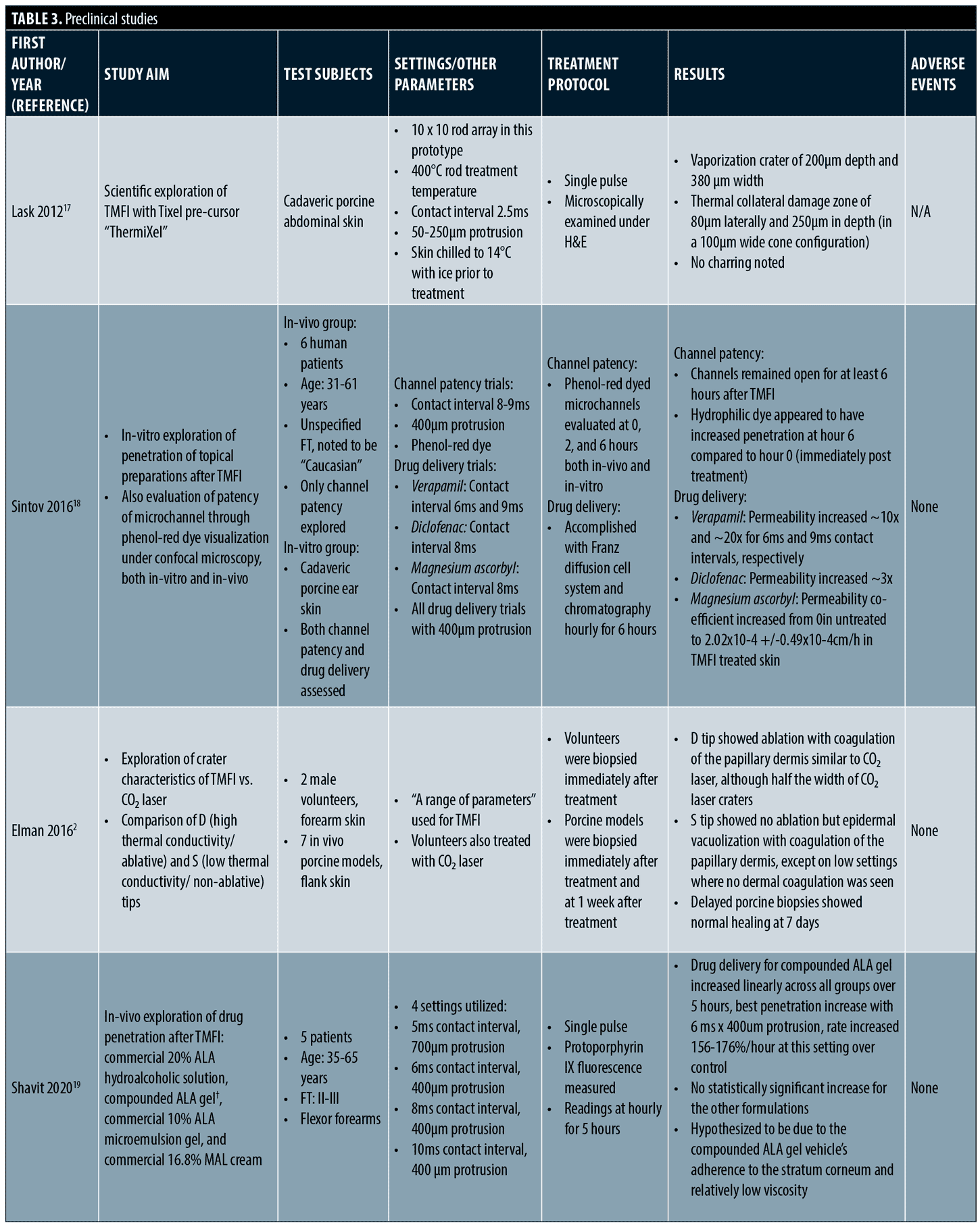
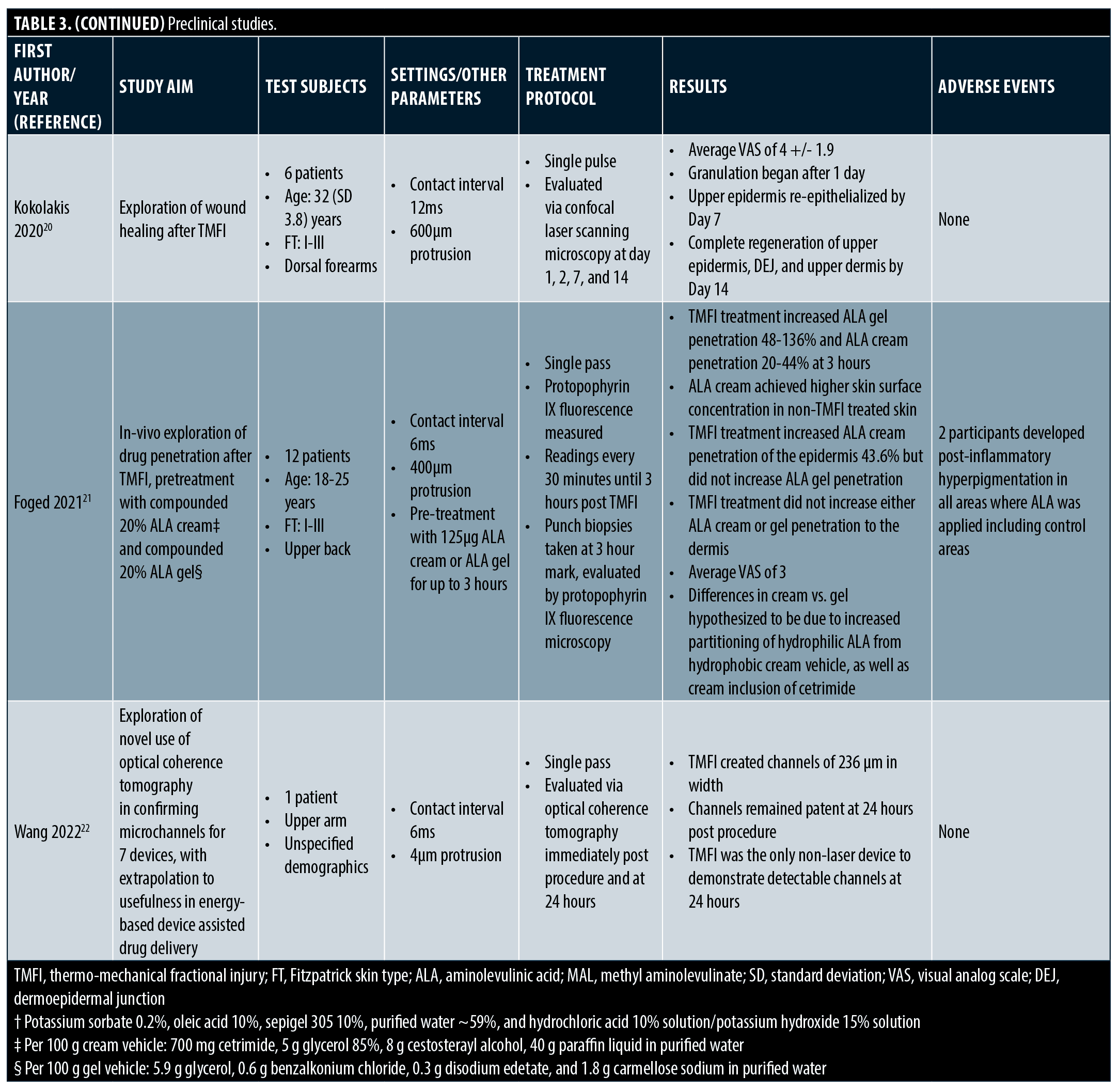
Preclinical studies. Preclinical and proof of concept studies have demonstrated mechanistic justification for clinical findings. (Table 3) Multiple studies have shown TMFI ablation zones ranging from 100-380 microns in width and 100 to 180 microns in depth, with coagulation zones roughly 460 microns in width and 250 microns in depth, depending on settings used. Ablative settings cause vaporization of the stratum corneum. However, on non-ablative settings, the stratum corneum becomes desiccated and “cracked”. These changes last at least 24 hours. Due to these phenomena, drug delivery is enhanced with both ablative and non-ablative settings, with increasing levels of drug penetration demonstrated up to six hours post-TMFI. The concomitant dehydration of tissue in the coagulation zone enhances the penetration of hydrophilic compounds, as osmotic flux into the skin is increased. Microchannels occupy roughly 2 percent of the treatment area, allowing for rapid scar-free healing, which has been demonstrated to be complete via confocal laser scanning microscopy after two weeks.2,17–22
Conclusion
With versatile settings, Tixel has been utilized for a variety of cosmetic and medical dermatologic indications. Tixel-assisted drug delivery studies typically used pulse durations under 10 ms, with all but one study utilizing 5-8 ms. For protrusion, scars were treated with 400 or 1000 µm, rosacea with 800-1000 µm, and vascular lesions, acne and Hailey-Hailey disease with 400 µm. Aesthetic studies included a wider range of settings, considering sensitive areas such as the periorbital region were treated, but pulse durations of up to 14 ms and protrusions of 400-1000 µm were used. Longer pulse durations result in more thermal transfer and deeper dermal coagulation, resulting in more neocollagenesis. Thus pulse durations of >10 ms may be more suited for rhytides, while shorter pulse durations are best for drug delivery.
Some authors have postulated that double passes with moderate settings (i.e. 600 µm, 12 ms) have an estimated treatment density of 15% which may increase downtime and pose a higher risk of adverse events, while another study found hyperpigmentation to occur after a 14 ms pulse duration and protrusion depth of 700 µm and higher were utilized. (6,7) To be cautious, more aggressive settings (i.e. pulse durations >10ms), especially with multiple passes should be used with caution in higher Fitzpatrick Skin Types given the risk of hyperpigmentation. Of note, the device does not have a target chromophore, and thus may be overall safer than some lasers in skin of color.
Given the challenge of both dynamic and static wrinkles in the periorbital and perioral regions, it is promising that multiple trials using Tixel have found clinically meaningful improvement. In addition, the perioral area has been associated with persistent dyschromia, which did not occur in any study.23 Both the thin, delicate periorbital skin and neck, containing less pilosebaceous units, carry a higher risk of scarring, which was also not reported in any study.24,25
All studies exhibited a low level of pain, most without topical anesthesia, which is not only of benefit to the patient but also shortens clinic visits. Aside from the study on photodynamic therapy for acne, downtime was typically less than two days with many patients returning to activities in less than one day. Transient side effects included erythema, edema, and microcrusting and only rare cases of hyperpigmentation were reported. The device features self-sterilizing tips, thus eliminating the cost of tip replacements as seen with other energy-based devices. Despite a logical concern of soft tissue infection, there was only one case of mild impetigo reported.
When comparing Tixel to other well-established energy-based devices in the dermatologist’s armamentarium, the ablative capacity of the device is comparable to the fractional carbon dioxide laser in regards to improvement of rhytides and photoaging, but with significantly less pain, side effects, and downtime. Non-ablative settings are similar to the 1565nm erbium: glass laser and likely similar to 1550nm models as well, but treatment discomfort is reduced. The compact design, versatility and safety of the device combined with positive patient reviews and tolerability make Tixel a favorable option.
In general, there is a need for larger studies and randomized, controlled blinded trials. Limitations of this review include variability in grading scales, making comparisons difficult, as well as studies with smaller sample sizes and retrospective analyses. Tixel-assisted drug delivery has been proven useful for several medical conditions. Future studies can explore the use of other topicals such as cosmeceuticals in enhancing skin quality, minoxidil or 5-alpha reductase inhibitors for alopecia, and imiquimod or 5-fluorouracil for actinic damage. Other aesthetic directions could include hand rejuvenation, skin laxity, and Mohs surgery scarring.
In summary, Tixel is a novel energy-based device with promising results for a variety of dermatologic conditions as demonstrated by the studies included in this review.
References
- United States Food and Drug Administration. February 25, 2021. https://www.accessdata.fda.gov/cdrh_docs/pdf20/K202988.pdf. Accessed April 10, 2023.
- Elman M, Fournier N, Barnéon G, et al. Fractional treatment of aging skin with Tixel, a clinical and histological evaluation. J Cosmet Laser Ther. 2016;18(1):31–37.
- Wang JV, Bajaj S, Orbuch D, et al. Safety and Efficacy of a Thermomechanical Fractional Injury Device for Periorbital Rhytides. Dermatol Surg. 2023;49(4):374–377.
- Wang JV, Bajaj S, Steuer A, et al. Prospective Evaluation of the Safety and Efficacy of Thermomechanical Fractional Injury for Perioral Rhytides [published online ahead of print, 2023 Mar 16]. Dermatol Surg. 2023;10.1097/DSS.0000000000003762.
- Salameh F, Daniely D, Kauvar A, et al. Treatment of periorbital wrinkles using thermo-mechanical fractional injury therapy versus fractional non-ablative 1565 nm laser: A comparative prospective, randomized, double-arm, controlled study. Lasers Surg Med. 2022;54(1):46–53.
- Daniely D, Judodihardjo H, Rajpar SF, et al. Thermo-Mechanical Fractional Injury Therapy for Facial Skin Rejuvenation in Skin Types II to V: A Retrospective Double-Center Chart Review. Lasers Surg Med. 2021;53(9):1152–1157.
- Judodihardjo H, Rajpar S. Retrospective study on the safety and tolerability of clinical treatments with a novel Thermomechanical Ablation device on 150 patients. J Cosmet Dermatol. 2022;21(4):1477–1481.
- Oren-Shabtai M, Sloutsky N, Lapidoth M, et al. Efficacy and safety of a thermal fractional skin rejuvenation system (Tixel) for the treatment of facial and/or scalp actinic keratoses. Lasers Med Sci. 2022;37(7):2899–2905.
- Artzi O, Koren A, Niv R, et al. The Scar Bane, Without the Pain: A New Approach in the Treatment of Elevated Scars: Thermomechanical Delivery of Topical Triamcinolone Acetonide and 5-Fluorouracil. Dermatol Ther (Heidelb). 2019;9(2):321–326.
- Artzi O, Koren A, Niv R, et al. A new approach in the treatment of pediatric hypertrophic burn scars: Tixel-associated topical triamcinolone acetonide and 5-fluorouracil delivery. J Cosmet Dermatol. 2020;19(1):131–134.
- Manuskiatti W, Yan C, Artzi O, et al. Efficacy and safety of thermomechanical fractional injury-assisted corticosteroid delivery versus intralesional corticosteroid injection for the treatment of hypertrophic scars: A randomized split-scar trial. Lasers Surg Med. 2022;54(4):483–489.
- Mashiah J, Bar-Ilan E, Koren A, et al. Enhanced Percutaneous Delivery of Beta-Blockers Using Thermal Resurfacing Drug Delivery System for Topical Treatment of Infantile Hemangiomas. Dermatology. 2020;236(6):565–570.
- Artzi O, Mehrabi JN, Heyman L, et al. Treatment of port wine stain with Tixel-induced rapamycin delivery following pulsed dye laser application. Dermatol Ther. 2020;33(1):e13172.
- Friedman O, Koren A, Niv R, et al. The toxic edge-A novel treatment for refractory erythema and flushing of rosacea. Lasers Surg Med. 2019;51(4):325–331.
- Hilerowicz Y, Friedman O, Zur E, et al. Thermomechanical Ablation-Assisted Photodynamic Therapy for the Treatment of Acne Vulgaris. A Retrospective Chart Review of 30 Patients. Lasers Surg Med. 2020;52(10):966–970.
- Bar-Ilan E, Koren A, Shehadeh W, et al. An enhanced transcutaneous delivery of botulinum toxin for the treatment of Hailey-Hailey disease. Dermatol Ther. 2020;33(1):e13184.
- Lask G, Elman M, Fournier N, et al. Fractional vaporization of tissue with an oscillatory array of high temperature rods: part I: ex vivo study. J Cosmet Laser Ther. 2012;14(5):218–223.
- Sintov AC, Hofmann MA. A novel thermo-mechanical system enhanced transdermal delivery of hydrophilic active agents by fractional ablation. Int J Pharm. 2016;511(2):821–830.
- Shavit R, Dierickx C. A New Method for Percutaneous Drug Delivery by Thermo-Mechanical Fractional Injury. Lasers Surg Med. 2020;52(1):61–69.
- Kokolakis G, von Grawert L, Ulrich M, et al. Wound Healing Process After Thermomechanical Skin Ablation. Lasers Surg Med. 2020;52(8):730-734.
- Foged C, Haedersdal M, Bik L, et al. Thermo-Mechanical Fractional Injury Enhances Skin Surface- and Epidermis- Protoporphyrin IX Fluorescence: Comparison of 5-Aminolevulinic Acid in Cream and Gel Vehicles. Lasers Surg Med. 2021;53(5):622–629.
- Wang JV, Mehrabi JN, Zachary CB, et al. Evaluation of Device-Based Cutaneous Channels Using Optical Coherence Tomography: Impact for Topical Drug Delivery. Dermatol Surg. 2022;48(1):120–125.
- Weniger FG, Weidman AA, Barrero Castedo CE. Full-Field Erbium:YAG Laser Resurfacing: Complications and Suggested Safety Parameters. Aesthet Surg J. 2020;40(6):NP374–NP385.
- Love LP, Farrior EH. Periocular anatomy and aging. Facial Plast Surg Clin North Am. 2010;18(3):411–417.
- Sturm A, Shokri T, Ducic Y. Nonsurgical Rejuvenation of the Neck. Clin Plast Surg. 2023;50(3):497–507.