J Clin Aesthet Dermatol. 2026;19(1):30–33.
by Aaron Cheng, BA, BS; Ambri Qeleshi, BA, BS; Andrew Kosa, BS; and David G. Cotter, MD, PhD
Mr. Cheng is with the Long School of Medicine at the University of Texas Health San Antonio in San Antonio, Texas. Mr. Qeleshi and Mr. Kosa are with the University of Nevada, Las Vegas in Las Vegas, Nevada. Dr. Cotter is with Las Vegas Dermatology and the University of Nevada, Las Vegas School of Medicine in Las Vegas, Nevada.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no conflicts of interest relevant to the contents of this article.
ABSTRACT: Atopic dermatitis (AD) is the most common chronic inflammatory skin condition and typically presents with pruritus, xerosis, eczematous lesions, and lichenification. Treatment options vary depending on severity; however, core management involves trigger avoidance, daily skin care, and anti-inflammatory therapies. Dupilumab, a monoclonal antibody that blocks interleukin-4 (IL-4) receptor α, is approved for the treatment of moderate-to-severe AD in patients aged 6 months or older. While this approval represents a significant advancement in AD therapy, the full extent of its side effect profile continues to emerge. Herein, we report two cases of refractory AD in which dupilumab therapy was limited by conjunctivitis and paradoxical worsening of cutaneous symptoms. We present these cases to describe the presentation, clinical progression, and management of dupilumab-associated adverse reactions in AD treatment, thereby contributing to a deeper understanding of its side effect profile. Notably, these cases demonstrate the successful use of nemolizumab as a therapeutic alternative in the setting of dupilumab intolerance. Keywords: Atopic dermatitis, biologics, adverse drug reactions
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin condition affecting approximately 15% to 30% of children and 2% to 10% of adults in developed countries.1 The pathophysiology is complex and multifactorial. Decreased levels of ceramides in the stratum corneum contribute to a dysfunctional skin barrier, making it more susceptible to xerosis, environmental irritants, and allergens consistent with classic findings of AD.2 Irritant and allergen penetration into the skin prompts an aberrant inflammatory response via an overactive helper T (Th) cell
2 response characterized by increased interleukin (IL) 4, IL-13, IL-31, and OX40 signaling, among activation of non-Th2 pathomechanisms, including Th17 and Th22 pathways, in certain patients.3 Accordingly, treatment options include trigger avoidance, daily skin care to maintain a proper skin barrier, and prescription topical and systemic anti-inflammatory therapies. The Food and Drug Administration–approved biologic dupilumab, a monoclonal antibody that mitigates IL-4 and IL-13 via blockade of IL-4 receptor α, has shown significant efficacy in reducing disease burden.3 However, dupilumab has been associated with several adverse effects, including conjunctivitis and paradoxical worsening of skin inflammation.3 Herein, we demonstrate the clinical improvement of dupilumab-associated facial erythema, conjunctivitis, and arthralgia symptoms after transitioning to nemolizumab therapy.
Case 1
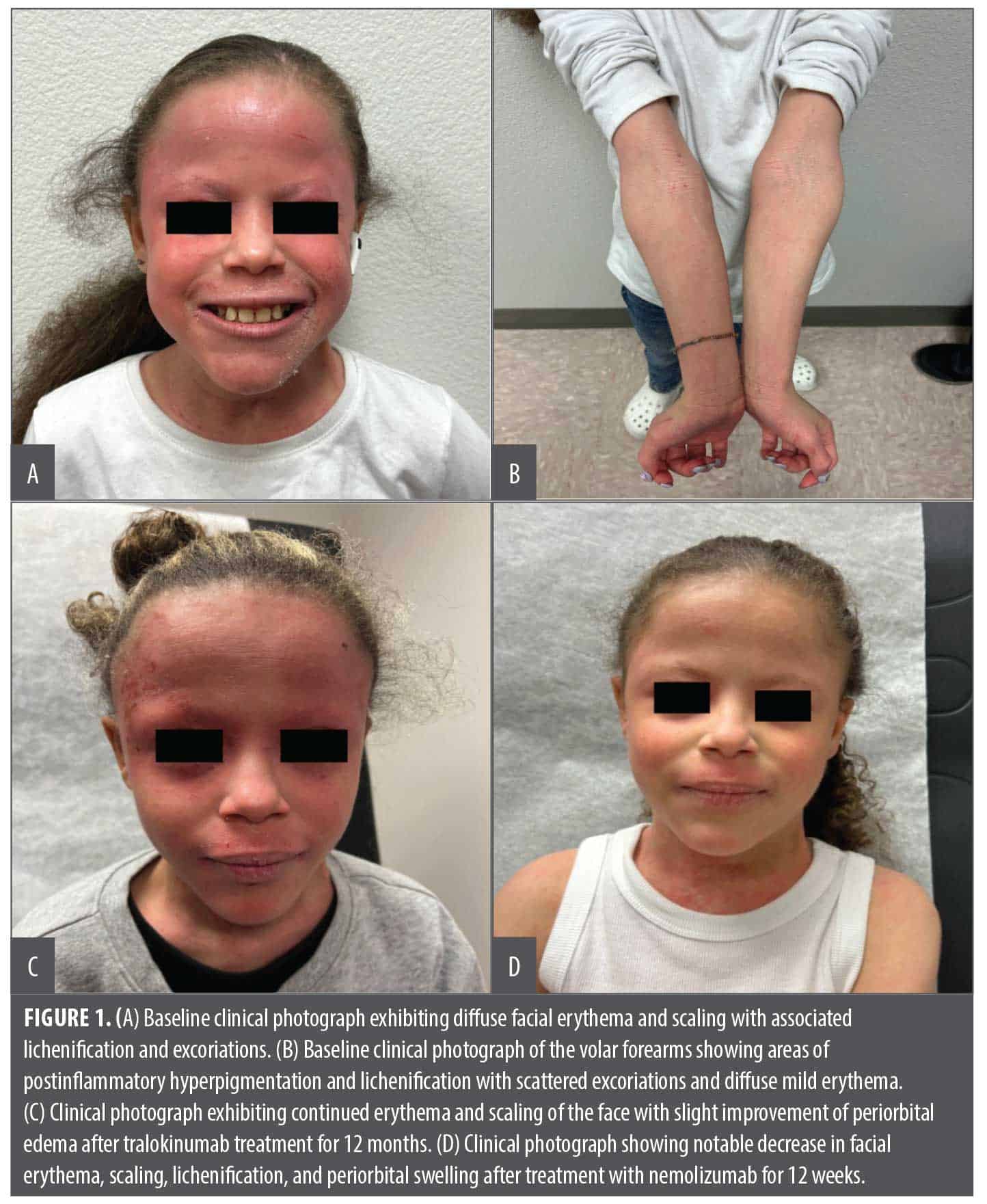
An 11-year-old female patient with a history of AD, currently managed with as-needed cetirizine and over-the-counter moisturizers, presented with severe AD (body surface area [BSA]: 40%, itch numeric rating scale [NRS]: 10, and Investigator’s Global Assessment [IGA]: severe) with notable facial involvement (Figure 1A). Previous treatment with dupilumab for six months provided some symptomatic relief; however, dupilumab was discontinued due to severe conjunctivitis and worsening facial erythema. On physical examination, diffuse facial erythema and scaling, most prominent over the forehead, periorbital region, and cheeks, with associated lichenification and excoriations, were observed, consistent with moderate-to-severe AD (Figure 1A). Additionally, examination of the volar forearms showed areas of postinflammatory hyperpigmentation and lichenification with scattered excoriations and diffuse mild erythema bilaterally (Figure 1B).
Due to the extensive involvement and severity of the disease, the patient was started on tralokinumab with a loading dose of 300mg administered subcutaneously, followed by a 150-mg dose two weeks later and a maintenance regimen of 150mg every two weeks. Over the year, the patient reported intermittent flares of her symptoms despite initial improvement (BSA: 10–20%, NRS: 3–5, and IGA from almost clear to focally severe). Her flaring facial dermatitis prompted patch testing, which revealed a compositae allergy consistent with airborne allergic contact dermatitis. Recurrent conjunctivitis and worsening facial dermatitis prompted discontinuation of tralokinumab after one year (Figure 1C). She was switched to nemolizumab with a 60-mg loading dose and a maintenance dose of 30mg every four weeks. She exhibited rapid improvement (itch NRS 2 by Week 4) and marked improvement in her facial erythema, scaling, lichenification, and periorbital swelling after 12 weeks on nemolizumab (Figure 1D). At her last follow-up after six months of nemolizumab, she has not experienced recurrence of conjunctivitis or worsening dermatitis.
Case 2
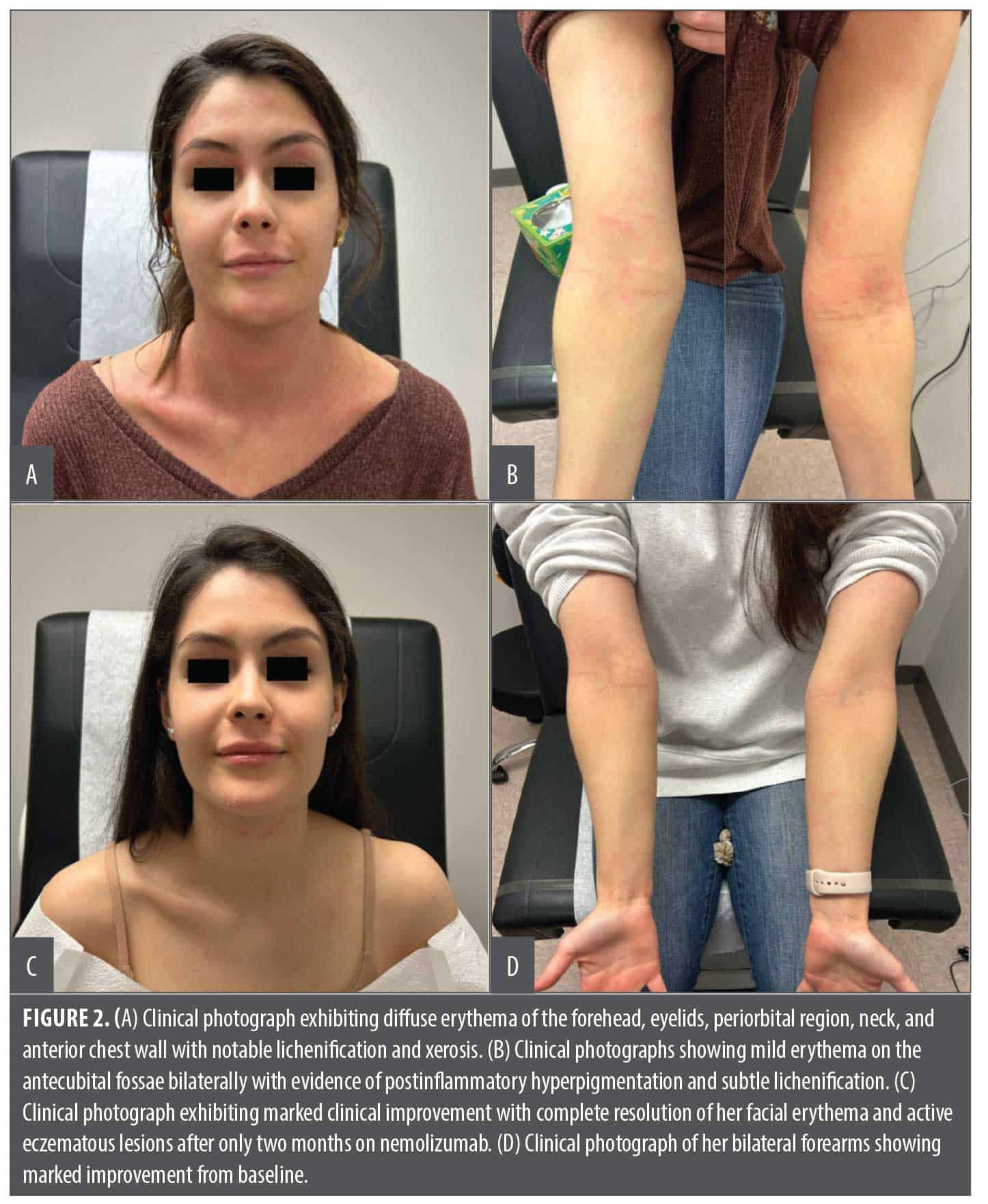
A 22-year-old female patient with a past medical history of AD since childhood, managed on dupilumab for the past six months, presented to the clinic with worsening erythema and pruritus. On physical examination, diffuse erythema of the forehead, eyelids, periorbital region, neck, and anterior chest wall was observed with notable lichenification and xerosis (Figure 2A). Additionally, mild erythema was noted on the antecubital fossae bilaterally with evidence of postinflammatory hyperpigmentation and subtle lichenification (Figure 2B). The patient reported that dupilumab injections appeared to exacerbate her AD with worsening facial and neck erythema as well as new-onset diffuse joint pain. Despite prior temporary improvement with topical and oral corticosteroids, these therapies had since
lost their efficacy and no longer provided symptomatic relief.
Upon presentation, dupilumab was discontinued, and the patient was started on nemolizumab with a 60-mg loading dose and a maintenance dose of 30mg every four weeks. At her one-month follow-up, her pruritus had significantly decreased to 2 out of 10 on a 0 to 10 numeric rating scale, with 10 representing worst imaginable pruritus. At her two-month follow-up, she reported complete resolution of her symptoms. On physical examination, the patient demonstrated marked clinical improvement with complete resolution of her facial erythema and active eczematous lesions (Figure 2C). Her bilateral forearms showed a significant reduction in erythema and minimal residual lichenification consistent with a robust therapeutic response to nemolizumab (Figure 2D). At the last available follow-up, after seven months of nemolizumab treatment, she continued to do well and did not experience any adverse effects other than mild, intermittent headaches.
Discussion
AD is a chronic inflammatory skin condition caused by abnormalities in the skin barrier, immune system dysregulation, and environmental influences.2,3 AD can be debilitating for patients, as it significantly impacts their quality of life (QOL). It commonly results in debilitating pruritus and erythema, leading to sleep disturbances, psychosocial distress, and impaired daily functioning.4 In more severe cases, AD can lead to skin discoloration, oozing and crusting, and lichenification of the affected areas, all of which can further negatively impact patient QOL.4
Treatment options such as topical corticosteroids are limited by long-term side effects, including skin atrophy, striae, rosacea, and telangiectasias.5 Targeted biologics have transformed the treatment landscape of AD.6,7 Biologics, such as dupilumab, tralokinumab, lebrikizumab, and nemolizumab, offer improved disease control with minimal local and systemic side effects. When compared to topical corticosteroids, they lead to greater disease control, especially when combined with other complementary therapies. Dupilumab was the first FDA-approved biologic for adults with moderate-to-severe AD whose disease was not adequately controlled with topical medications.7 Since then, its approval has been subsequently expanded to the pediatric population, offering a highly effective treatment option for children as young as six months of age.8
Although biologics are generally well tolerated, several adverse events have been reported. The most common reported adverse events are injection-site reactions, which can be attributed to adjuvants or excipients that activate the innate immune system and cause acute hypersensitivity reactions.9 Conjunctivitis, nasopharyngitis, and bacterial and herpetic skin infections have also been reported with the use of dupilumab.9 Additionally, dupilumab as monotherapy may be insufficient to treat AD in some cases, making adjunctive or alternative therapy necessary.10
Informed consent and photoconsent was received for both patients. In our first case, dupilumab therapy was limited by adverse events and paradoxical worsening of cutaneous symptoms. It highlights a pediatric patient with extensive AD managed with dupilumab who initially improved but later developed severe conjunctivitis, leading to discontinuation. This case was further complicated by a failed trial of tralokinumab that resulted in similar ocular symptoms and intermittent flares despite adequate dosing and initial response. However, treatment with nemolizumab provided rapid improvement with marked reduction in pruritus and inflammation. Notably, the patient initially expressed hesitation regarding starting another biologic due to prior injection site reactions. However, because the patient had an indwelling ventriculoperitoneal shunt, an oral Janus kinase (JAK) inhibitor was declined due to concern for the risk of infection. Thus, she reluctantly initiated nemolizumab, despite her fear of injection site pain. After her first injection, she reported with laughter that the nemolizumab “injection was painless.” This may be attributed to its low injection volume of approximately 0.49mL, the smallest among currently available biologics for AD.
In our second case, the patient experienced worsening facial and neck erythema and persistent joint pain following treatment with dupilumab. While common adverse effects of dupilumab include conjunctivitis and injection-site reactions, arthralgias are seen in about 1% to 2% of people on dupilumab.11 Joint pain while on dupilumab is most often seen when starting the medication and usually resolves after cessation of dupilumab.11 The combination of erythema, arthralgia, and persistent pruritus despite dupilumab treatment seen in our case prompted a change to nemolizumab. Notably, the patient reported significant improvement following the transition to nemolizumab, suggesting that IL-4 and/or IL-13 blockade contributed to her symptoms. With nemolizumab, she experienced a rapid decrease in pruritus and resolution of joint pain and facial and neck erythema.
Conclusion
Overall, these cases highlight the unique adverse events associated with dupilumab and convey the importance of personalized treatment in managing AD. While some biologics, such as dupilumab, have revolutionized how we treat AD by providing robust skin clearance and symptom control, they are not universally effective and may exacerbate symptoms or trigger uncommon side effects in some patients. Our cases demonstrate the utility of alternative biologic treatments for AD when challenged with severe adverse reactions and decreased tolerability. Nemolizumab can be a safe alternative after dupilumab adverse events, highlighting need for individualized biologic therapy in AD.
References
- Archer CB. Atopic dermatitis. Medicine. 2021;49(6):370-373.
- Imokawa G, Abe A, Jin K, Higaki Y, Kawashima M, Hidano A. Decreased level of ceramides in stratum corneum of atopic dermatitis: an etiologic factor in atopic dry skin? J Invest Dermatol. 1991;96(4):523-526.
- Facheris P, Jeffery J, Del Duca E, Guttman-Yassky E. The translational revolution in atopic dermatitis: the paradigm shift from pathogenesis to treatment. Cell Mol Immunol. 2023;20(5):448-474.
- Larney C, Foley P, Wu J, Daniel BS. Real world side-effects of dupilumab: a narrative review. Australas J Dermatol. 2025;66(6):e324-e334.
- Artusa S, Mazzuca G, Piacentini G, et al. Paediatric atopic dermatitis: the unexpected impact on life with a specific look at the molecular level. Int J Mol Sci. 2024;25(9):4778.
- Kleinman E, Laborada J, Metterle L, Eichenfield LF. What’s new in topicals for atopic dermatitis? Am J Clin Dermatol. 2022;23(5):595-603.
- Seegräber M, Srour J, Walter A, Knop M, Wollenberg A. Dupilumab for treatment of atopic dermatitis. Expert Rev Clin Pharmacol. 2018;11(5):467-474.
- Cork MJ, Thaçi D, Eichenfield LF, et al. Dupilumab safety and efficacy in a phase III open-label extension trial in children 6-11 years of age with severe atopic dermatitis. Dermatol Ther (Heidelb). 2023;13(11):2697-2719.
- Phan M, Linfante A, Kroger K. Ulcerative injection-site reaction associated with dupilumab therapy. JAAD Case Rep. 2020;6(12):1350-1352.
- Shahriari N, Strober B, Shahriari M. JAK-inhibitors as rescue therapy in dupilumab-refractory severe atopic dermatitis: a case series of 6 patients. JAAD Case Rep. 2023;33:81-83.
- See Tow HX, Yew YW. Joint pain and inflammatory arthropathy after dupilumab use in atopic dermatitis: a systematic review and meta-analysis. Dermatitis. Published online May 28, 2025.