J Clin Aesthet Dermatol. 2026;19(2):47–49.
by Kyle Machynia, MPAS, PA-C, and Jason E. Hawkes, MD, MS, FAAD
Mr. Machynia is with the University of Nevada, Reno School of Medicine in Reno, Nevada. Dr. Hawkes is with the Oregon Medical Research Center and Oregon Health and Science University in Portland, Oregon.
FUNDING: No funding was provided for this article.
DISCLOSURES: Mr. Machynia is an honorary speaker for AbbVie, Galderma, Johnson&Johnson, and Sanofi Regeneron, and has served on advisory boards for AbbVie, Bausch, Blueprint Medicines, Bristol Myers Squibb, Castle Biosciences, Galderma, Johnson & Johnson, LEO Pharma, Ortho Dermatologics, and Sanofi Regeneron. Dr. Hawkes is an advisor/consultant/speaker for AbbVie, Apogee, Arcutis, BI, Blueprint Medicines, Bristol Myers Squibb, Dermavant, Galderma, Janssen, LEO Pharma, Lilly, Novartis, Oruka, Regeneron, Sanofi, Sun Pharma, Takeda, and UCB, and is a clinical investigator for AbbVie, Acelyrin, Allakos, Alumis, Amgen, Apogee, Celldex, Concert Pharmaceuticals, Incyte, Janssen, LEO Pharma, Lilly, Novartis, Oruka, Pfizer, Sanofi, Sun Pharma, Takeda, and UCB.
ABSTRACT: Lichen planus (LP) is an idiopathic, immune-mediated inflammatory disorder commonly characterized by pruritic, violaceous papules and plaques that often involve the extremities. Although a variety of topical and systemic therapies are used, no targeted treatments are currently approved by the US Food and Drug Administration for this indication, and management of refractory disease remains challenging. Tyrosine kinase 2 (TYK2) inhibitors selectively modulate several cytokine signaling pathways, including interleukin (IL)-12, IL-23/IL-17, and type I interferon signaling, which are implicated in the pathogenesis of LP. Here, we report a case of biopsy-confirmed, treatment-refractory, cutaneous LP that achieved near-complete resolution after 2 months of therapy with the oral TYK2 inhibitor, deucravacitinib. Keywords: Lichen planus, deucravacitinib, TYK2 inhibition, Janus kinase signaling, pruritus
Introduction
Lichen planus (LP) is an idiopathic, heterogeneous, immune-mediated lymphocytic inflammatory disease involving the skin, nails, hair follicles, and/or mucous membranes.1 Classic LP is characterized by pruritic, violaceous papules that predominantly affect the extremities.1,2 Currently, there are no universally accepted effective treatments for LP, and no novel or targeted therapies have been approved by the US Food and Drug Administration or the European Medicines Agency for this condition.2 Therefore, broad-acting immunosuppressant or off-label targeted therapies are commonly used in the management of severe or refractory disease. LP management typically employs high-potency topical corticosteroids, alone or in combination with systemic corticosteroids, topical or systemic steroid-sparing immunomodulators, oral calcineurin inhibitors, methotrexate (MTX), oral retinoids, oral antibiotics, or phototherapy.1-3 Treatment efficacy is difficult to assess due to reliance on limited case series and anecdotal evidence,3 and no single scoring system has achieved universal acceptance.
While the immunopathogenesis of LP remains unclear, interferon (IFN)-γ has been identified as a predominant cytokine driving the condition, making its signaling pathway a potential therapeutic target.4,5 Novel Janus kinase (JAK) family (eg, JAK1, JAK2, JAK3, and tyrosine kinase [TYK] 2) inhibitors are of interest for the potential treatment of LP due to their ability to simultaneously block IFN-γ as well as other immune signaling pathways such as helper T cell (TH) 2 and TH17 signaling.4-6 Deucravacitinib inhibits TYK2 and disrupts type I IFNs, interleukin (IL)-12, and IL-23/IL-17 immune signaling, all of which have been implicated in LP.5-7 TYK2 inhibitors also provide a relatively robust safety profile due to their increased selectivity compared to broader-acting JAK inhibitors.8 Herein, we present a case of cutaneous LP with near complete resolution following 2 months of treatment with deucravacitinib.
Case Report
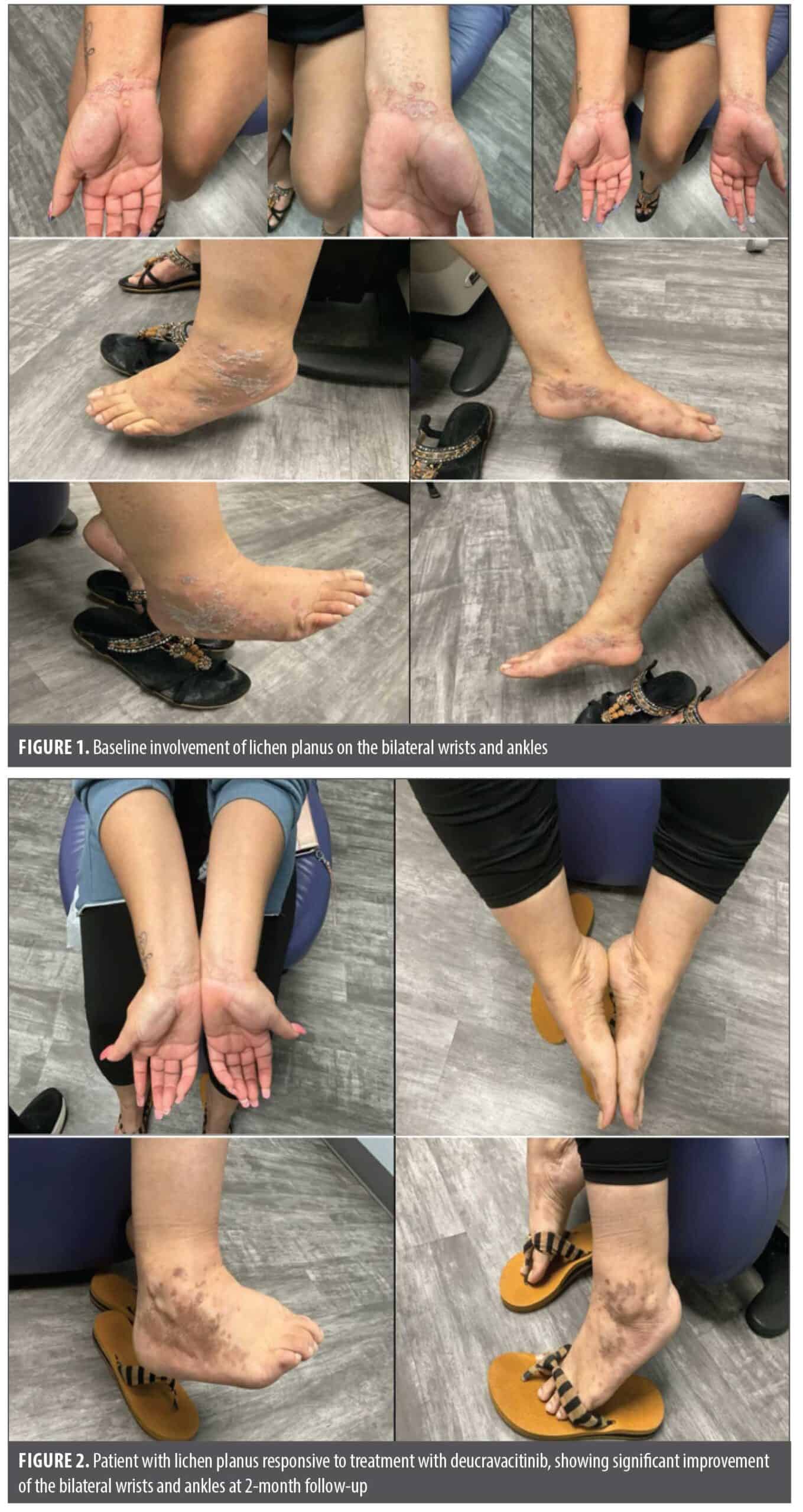
A 26-year-old woman presented with a 4-month history of biopsy-confirmed LP with minimal response to medium-potency topical corticosteroids. She complained of significant pruritus of the affected skin that resulted in difficulty sleeping and maintaining focus at work. On physical exam, she presented with pruritic (Worst Pruritus Numeric Rating Scale [WP-NRS] of 8), violaceous, flat-topped papules and plaques on the bilateral wrists and ankles (Figure 1). Serologies for HIV-1, HIV-2, hepatitis B, and hepatitis C were negative. She was initially treated with oral metronidazole 500mg twice-daily along with a single intramuscular triamcinolone dose of 40mg, and twice-daily topical triamcinolone cream, 0.1%; however, this did not significantly improve her skin lesions or pruritus after 6 weeks of treatment. Conventional therapy with MTX, mycophenolate mofetil, oral retinoids, and off-label JAK1 inhibitors was offered, but the patient initially declined. After further discussion, the patient was prescribed deucravacitinib (6mg once daily) and discontinued all other treatments. At her 2-month follow-up, she reported significant improvement of her pruritus (WP-NRS of 8 to 2) and the lichenoid lesions had largely resolved with postinflammatory hyperpigmented macules and patches (Figure 2). She discontinued deucravacitinib at follow-up due to mild acneiform eruption on the trunk, which later resolved with no further treatment.
Discussion
Despite the availability of broad-acting immunosuppressant therapies commonly used to treat LP, additional targeted treatments to disrupt key immune pathways driving this inflammatory condition are needed. The expanding role of JAK inhibition across various inflammatory dermatoses has prompted investigation into underlying mechanisms, with recent research implicating JAK/signal transducer and activator of transcription (STAT) signaling in the pathogenesis of LP. A systematic review of 56 patients with LP treated with JAK inhibitors reported complete resolution in 100% of those receiving upadacitinib, 25% with baricitinib, 16.7% with ruxolitinib, and 10% with tofacitinib.6 The unique binding properties of deucravacitinib confer high selectivity for TYK2, enabling inhibition of IL-12 family cytokine receptors (IL-12, IL-23) and type I IFNs, which are implicated in LP pathogenesis. In contrast, cytokine receptors for JAK1/JAK2, JAK1/JAK3, or JAK2/JAK2 signaling remain unaffected. This TYK2-selective profile of decravactinib may reduce the risk of off-target and serious adverse events seen with less selective JAK inhibitors which act more broadly on the immune response.8 Successful use of deucravacitinib has been reported in both hypertrophic and oral forms of LP.9,10 IL-4 receptor α blockade with dupilumab has also demonstrated rapid clearance of cutaneous LP and pruritus in one case report.11 Although these agents target distinct pathways, their efficacy underscores the cytokine-driven nature of LP and supports the potential for pathway-specific immunomodulation in its management.
Transcriptomic analysis from one study indicated that both type I and type II IFNs contribute substantially to LP pathogenesis, with JAK inhibition reversing the expression profile of IFN-regulated genes.12 Notably, elevated dermal IL-12 and serum IL-23 concentrations have been reported with cutaneous LP.13 Larger, placebo-controlled clinical trials with extended monitoring for safety are needed to better define the efficacy and safety of TYK2 inhibition in LP.
Our case adds to the growing evidence supporting deucravacitinib as a potential option for refractory LP and may help inform the rationale for its formal evaluation. This report is affected by the lack of standardized LP severity scoring and the narrow scope for broader clinical extrapolation. However, the sustained clinical improvement and favorable long-term tolerability observed in our patient underscore the need for larger prospective or retrospective studies to further clarify its role in clinical practice.
References
- Le Cleach L, Chosidow O. Clinical practice. Lichen planus. N Engl J Med. 2012;366(8):723-732.
- Ioannides D, Vakirlis E, Kemeny L, et al. European S1 guidelines on the management of lichen planus: a cooperation of the European Dermatology Forum with the European Academy of Dermatology and Venereology. J Eur Acad Dermatol Venereol. 2020;34(7):1403-1414.
- Nussbaum D, Kalen J, Zahn J, Friedman A. Offering an off-label therapeutic algorithm for lichen planus. J Drugs Dermatol. 2022;21(4):444-446.
- Wenzel J, Scheler M, Proelss J, Bieber T, Tüting T. Type I interferon-associated cytotoxic inflammation in lichen planus. J Cutan Pathol. 2006;33(10):672-678.
- Shao S, Tsoi LC, Sarkar MK, et al. IFN-γ enhances cell-mediated cytotoxicity against keratinocytes via JAK2/STAT1 in lichen planus. Sci Transl Med. 2019;11(511):eaav7561.
- Abduelmula A, Bagit A, Mufti A, Yeung KCY, Yeung J. The use of Janus kinase inhibitors for lichen planus: an evidence-based review. J Cutan Med Surg. 2023;27(3):271-276.
- Vičić M, Hlača N, Kaštelan M, Brajac I, Sotošek V, Prpić Massari L. Comprehensive insight into lichen planus immunopathogenesis. Int J Mol Sci. 2023;24(3):3038.
- Catlett IM, Aras U, Hansen L, et al. First-in-human study of deucravacitinib: a selective, potent, allosteric small-molecule inhibitor of tyrosine kinase 2. Clin Transl Sci. 2023;16(1):151-164.
- Sood S, Yadav G. Treatment of hypertrophic lichen planus using deucravacitinib. JAAD Case Rep. 2025;51:38-40.
- Stolte KN, Mesas-Fernández A, Meier K, et al. TYK2 inhibition with deucravacitinib ameliorates erosive oral lichen planus. Exp Dermatol. 2024;33(4):e15080.
- Kazemi S, Murphrey M, Hawkes JE. Rapid resolution of widespread cutaneous lichen planus and generalized pruritus in an elderly patient following treatment with dupilumab. JAAD Case Rep. 2022;30:108-110.
- Brumfiel CM, Patel MH, Severson KJ, et al. Ruxolitinib cream in the treatment of cutaneous lichen planus: a prospective, open-label study. J Invest Dermatol. 2022;142(8):2109-2116.e4.
- Mardani M, Mofidi H, Dastgheib L, Ranjbar S, Hamidizadeh N. Elevated serum interleukin-23 levels in patients with oral and cutaneous lichen planus. Mediators Inflamm. 2021;2021:5578568.