J Clin Aesthet Dermatol. 2026;19(3–4 Suppl 1):S12–S18.
by Chantel K. Hillestad, MSN, DCNP, FNP-C
Ms. Hillestad is with Trinity Health, Minot, North Dakota.
Funding: No funding was provided for this article.
Disclosures: The author has no relevant conflicts of interest.
ABSTRACT: An 80-year-old White male individual presented with a rapidly growing lesion on his left forearm that had been increasing in size for the past 2 months. The patient resides in a northern rural state with limited access to dermatology care. The shave biopsy performed indicated Merkel cell carcinoma (MCC). The patient was referred to oncology and surgical oncology. The patient underwent a wide local excision, then adjuvant immunotherapy with pembrolizumab. The patient presented 2 years later with a rapidly growing lesion on the left forearm in the location of the prior surgical scar, and this was found to be recurrent MCC. Two months later, a new lesion developed approximately 7 cm from the most recent surgical incision; this was also found to be MCC. MCC is a rare form of skin cancer that is the most fatal, is more invasive than other types of skin cancer, and has a high rate of mortality. Diagnosis is confirmed by biopsy. Currently, no screening tests are in place regarding MCC or recurrent MCC. Treatment often involves wide local surgical excision and radiation therapy. Adjuvant immunotherapy and targeted therapies are promising treatment options and are continuing to evolve. Patients diagnosed with MCC in rural areas have been found to have decreased life expectancy compared to urban patients. Rural patients often encounter extraneous challenges involving travel, limited access to care, and financial hardship compared to urban patients. Treating rural patients can pose challenges to the clinician involving access to care barriers, nonadherence, patient inducement, and adhering to clinical guidelines to achieve the best outcome. Keywords: Merkel cell carcinoma, Merkel cell polyomavirus, positron emission tomography (PET) scan, sentinel lymph node biopsy, adjuvant immunotherapy, adjuvant chemotherapy, adjuvant radiation therapy, receptor tyrosine kinase inhibitors
Introduction
The most fatal skin cancer is Merkel cell carcinoma (MCC).1 MCC has a mortality rate of 30%.2 MCC is a rare invasive skin cancer that has a high risk of recurrence and metastases.1 In 1972, MCC was first described as “trabecular carcinoma of the skin” by Cyril Toker. The name was later changed to MCC due to close resemblance to Merkel cells.3 Public knowledge of MCC is limited due to low incidence and a lack of awareness. Misdiagnosis can occur as MCC can resemble other skin dermatoses or skin cancers, and MCC is often asymptomatic, leading to delayed treatment.
An increased incidence of locally staged MCC has been found in patients residing in rural areas compared to patients in urban areas.4 The United States (US) Census Bureau defines rural as populations under 50,000 people.5 Currently, 19% of the total US population resides in rural areas.5 Rural populations in the US also encompass 97% of the total land area compared to urban centers.5 Though rural patients often present with locally staged MCC initially, it has been found that these patients have worse outcomes compared to urban patients.3 Rural patients have also been found to be less likely to wear sunscreen, are less likely to undergo skin cancer screening exams, and experience longer intervals for dermatology appointments due to less access to dermatology care.6–8 These circumstances often lead to advanced disease before diagnosis.6–8
The unfortunate passing of famed singer Jimmy Buffett in 2023 widened exposure to MCC. Google and Wikipedia online searches for MCC spiked after the singer passed.9 MCC most often affects White male individuals, aging adults, individuals with immunosuppression, and those with a prior history of other malignancies.10
Worldwide, cases of MCC are on the rise, with the highest occurrences in Australia and New Zealand.11 It is suspected that incidence in the US is on the rise due to an aging population and increased ultraviolet (UV) exposure. It is predicted that 3,200 new cases of MCC will be diagnosed in the US in 2025, compared to 2,488 cases in 2013.12 Advances in pathological stains might be a factor in the increased incidence of MCC. Cytokeratin 20 (CK20) immunohistochemistry staining emerged in the 1990s and is now a gold-standard diagnostic marker, which has improved MCC detection.13 Risk factors include UV radiation exposure, Merkel cell polyomavirus infection (MCPyV), and germline variants associated with cancer predisposition.13–15 Elderly male individuals residing in rural areas have been found to have an increased incidence of MCC, and this might be related to persistent UV exposure from occupations in the farming industry.
Clinical tumors are typically located on the head and neck, followed by the extremities and buttocks.14 Presentation is often noted with an asymptomatic violaceous, pink, or red firm dome-shaped papule that might illustrate signs of ulceration.14 Differential diagnoses can include amelanotic melanoma, basal cell carcinoma, squamous cell carcinoma, pyogenic granuloma, abscess, lymphoma, and angiosarcoma.14,15
The pathogenesis of MCC remains controversial.15 Hypotheses include development from Merkel cells in the basal layer, immature multipotent stems cells that involve neuroendocrine features during malignant transformation, or development from keratinocytes; it has also been debated that MCC tumors might not have a single cell or origin. MCPyV has been detected in up to 80% of MCC cases.2,3,14,15 MCPyV has been found to be part of the human skin virome.16 It has been detected in normal healthy skin and has been found in other skin tumors, including squamous cell carcinoma, follicultropic mycosis fungoides, and chronic lymphocytic leukemia.16,17 It has been suggested that MCPyV contributes to oncogenic effects after coalescing with the host genome and undergoing mutations.15–18 Compromised immune surveillance might enhance the carcinogenic potential of MCPyV. It is suspected that specific mutations from environmental toxins, such as UV exposure, contribute to the transformation of MCPyV from a benign virus to carcinogenic transformation.3,14,16,17,19
MCPyV-negative MCC cases are not fully understood but are suspected to involve somatic mutations in tumor suppressor genes such as RB1 and TP53. It has been hypothesized that epigenetic alterations involving deoxyribonucleic acid (DNA) methylation and microribonucleic acids (miRNAs) evolves to aberrant expression and ultimately to the action of oncogenes.18 It is presumed that a substantial frequency of DNA mutations are generated by UV damage.3,16
Diagnosis of MCC is completed by histopathological and immunohistochemical examination of specimens from skin tissue or other organs. Features of MCC commonly involve perinuclear dot-like staining with CK20 and the detection of MCPyV.20 The National Comprehensive Cancer Network (NCCN), recommends an appropriate immunopanel for preliminary workup involving CK20 and thyroid transcription factor 1 (TTF-1).20,21 MCC cases can be negative for both CK20 and MCPyV.19
Staging of MCC is crucial when determining treatment. MCC is staged by a system developed by the American Joint Committee on Cancer (AJCC). This system involves tumor size, nodal involvement and the presence of distant metastasis.14
Once a biopsy report confirms MCC, patients should undergo full-body positron emission tomography (PET) or computed tomography (CT) of the chest, abdomen, and pelvis with contrast. If the primary lesion is on the head or neck, CT of the head and neck with contrast should also be completed.14,20,21 Evaluation of lymph nodes should be completed for all patients.14,21 If the patient does not appear to have lymph node involvement noted on exam or imaging, a sentinel lymph node biopsy (SLNB) is advised.14,15,21 Standard treatment involves wide local surgical excision, which can be very intricate, as often lesions are located on the head and neck. Adjuvant radiation therapy often follows wide local surgical incision in cases with a positive SLNB and in cases where the SLNB is negative but risks factors for nodal recurrence are present; anatomical location or immunosuppression are linked to an additional probability of reccurrence.14,15,21,22
Treatment of MCC with immunotherapy continues to evolve. In 2017, the US Food and Drug Administration (FDA) approved avelumab for treatment of advanced MCC.23 In 2018, pembrolizumab was approved for treating locally advanced or metastatic MCC.23 These anti-programmed cell death 1 and programmed death-ligand 1 antibodies function to restore active T-cell response against the MCC tumor.11 The role of adjuvant chemotherapy after resected MCC remains controversial. In a retrospective study involving 6,908 cases of MCC in the US National Cancer Database, adjuvant chemotherapy did not improve the overall survival (OS) in patients with local or nodal disease.3
Case Report
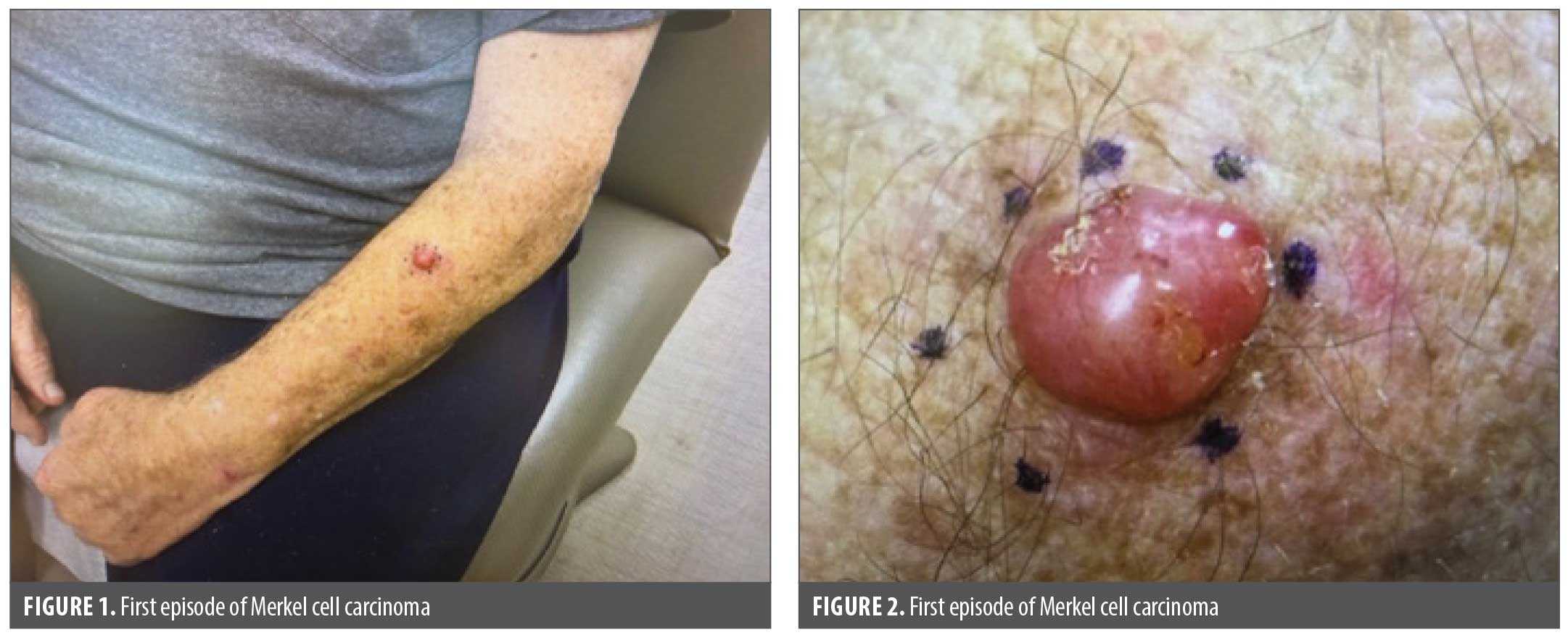
In April 2023 an 80-year-old White widowed male patient presented to clinic with concern of a rapidly growing red lesion to his left forearm over a 2-month period. The patient resides in a rural community 90 miles from the closest dermatology provider. The patient denied any bleeding, pain, or itching. The patient explained that the lesion would catch on shirt sleeves and became irritated and inflamed. The patient had a history of several basal cell carcinomas and squamous cell carcinoma. Additional medical history involved aortic stenosis, chronic anticoagulation, stage IV chronic kidney disease, congestive heart failure, atrial fibrillation, type 2 diabetes, sleep apnea, obesity, chronic normocytic anemia, and bilateral knee replacements. A mobile, pink-to-pearly, dry, scaly papule that measured approximately 1.5 cm in circumference was noted (Figures 1 and 2). No axillary lymphadenopathy noted. Shave biopsy was completed and indicated MCPyV negativity.
The patient was referred to oncology and oncology surgery. The patient underwent wide local excision by an oncology surgeon. The patient underwent a PET scan that illustrated mildly fluorodeoxyglucose avid left external iliac chain lymph node measuring 1.9 × 1.4 cm that was nonspecific and might have been reactive for metastases. Due to concern of advanced disease with SLNB report pending, pembrolizumab was initiated. SLNB was completed and found to be negative. Radiation therapy was discussed with the patient. The patient had concerns regarding adherence to radiation therapy, including living 90 miles from the closest radiation facility, having limited access to transportation, and concern for subzero winter temperatures and hazardous winter road conditions. The patient also had mobility concerns for making numerous appointments due to severe osteoarthritis to bilateral knees and often ambulated with a walker. The patient received infusions of pembrolizumab every 3 weeks. The patient completed 7 infusions of pembrolizumab. Unfortunately, before his eighth infusion, the patient was hospitalized with pneumonia and exacerbation of chronic heart failure, and this led to respiratory failure, ending his 6-month course of pembrolizumab. The patient was hesitant to restart pembrolizumab because he felt that this was the cause of his hospital admission with respiratory failure. Surveillance PET scans were completed every 6 months for 18 months, which never illustrated any malignant concerns. The patient continued regular dermatology follow-ups every 3 to 4 months.
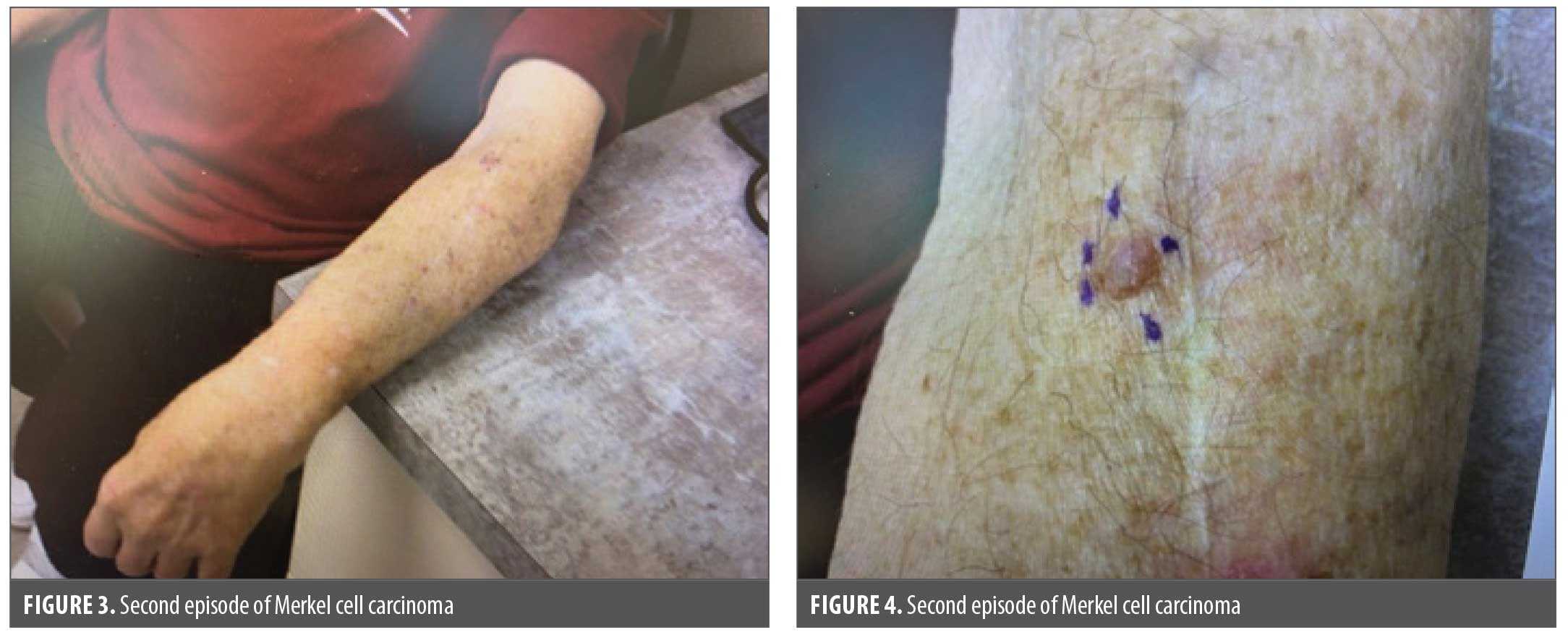
In April 2025, the patient contacted the dermatology office to report a rapidly growing lesion on his left forearm. On exam, a red-to-pink papule topped with straw crusts just millimeters from the prior surgical incision for MCC was noted (Figures 3 and 4). The patient denied any systemic symptoms, pain, or weight loss. No lymphadenopathy was noted on exam. Shave biopsy was completed and indicated recurrent MCC. The presence or absence of MCPyV was not noted on pathology report. The prior oncology surgeon was contacted and completed surgical incision. SLNB was performed, and negative results were noted. The patient followed up with oncology and underwent a PET scan. No further signs of involvement were noted on PET scan. The patient was hesitant to restart pembrolizumab due to his previous hospitalization with respiratory failure. The patient started erythropoietin. The oncologist advised starting pembrolizumab after radiation therapy. The patient was referred to a radiation oncologist. A 10-to-15 fraction regime was planned for the patient.
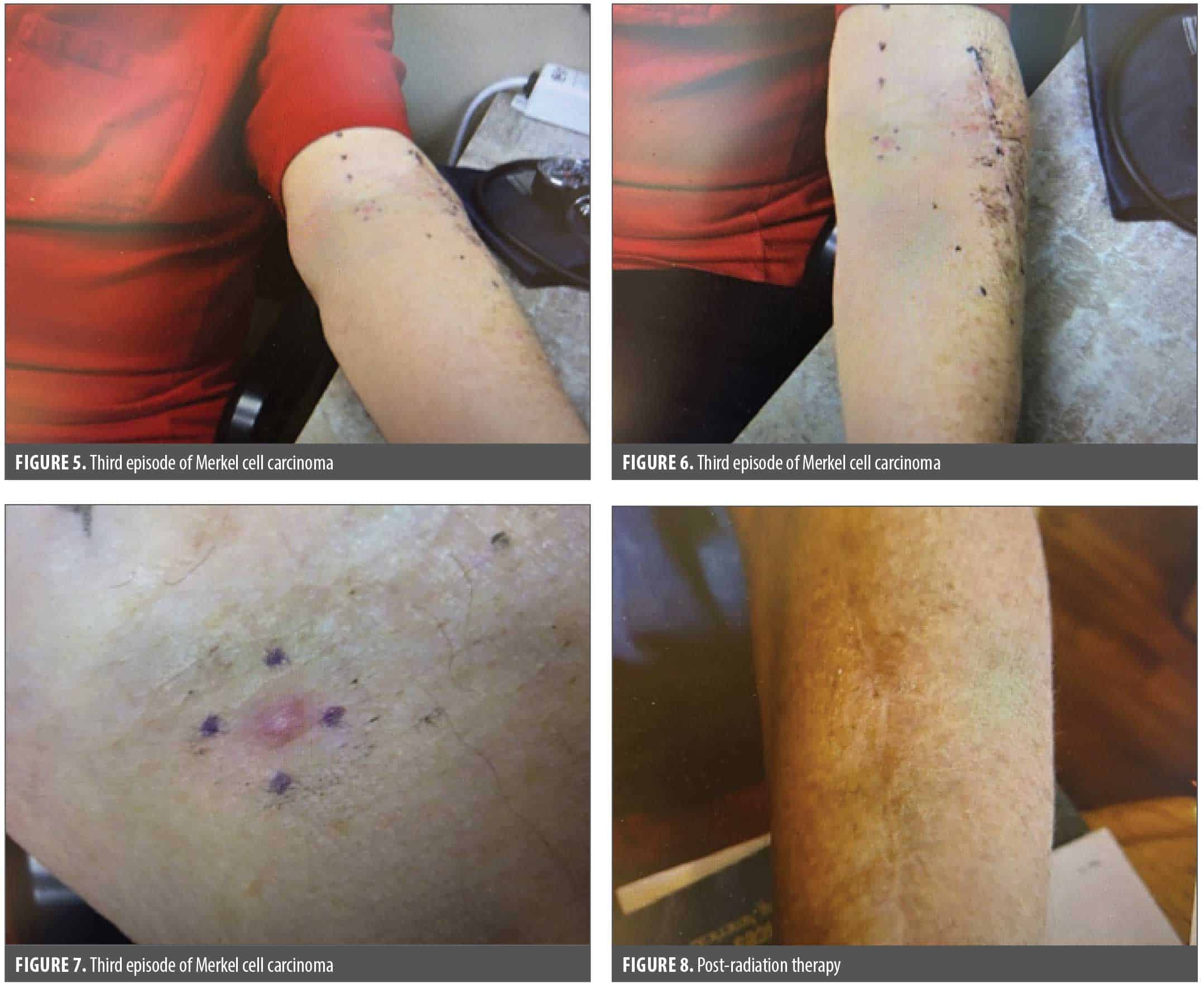
During a visit with the radiation oncologist, a pink papule was noted on exam to the left forearm just medial to the most recent surgical incision. The patient explained that he noticed this lesion in the last couple days. The radiation oncologist called the dermatology office asking if the patient could be seen on the same day for evaluation and potential biopsy. The patient was evaluated, and a 5 mm pearly papule was noted approximately 7 cm medial from the most recent surgical incision on the left forearm (Figures 5–7). No axillary lymphadenopathy or any other exam findings were noted. The pathology report indicated MCC once again. A PET scan was completed, and the patient was referred to oncology surgery for excision and SLNB. The oncology surgeon consulted with a nationally recognized premier cancer center. It was recommended that a third surgical excision be omitted, and adjuvant radiation therapy and treatment of the gross satellite recurrence be undertaken. The patient lived 90 miles away from the closest cancer center and was not willing to stay locally, so he consented to radiation therapy 3 times per week. The patient was set up with a 10-fraction course. In the meantime, the patient underwent dental extractions in preparation for transcatheter aortic valve replacement (TAVR). Unfortunately, significant hemorrhage occurred, leading to emergency room evaluation. The patient underwent blood transfusions as hemoglobin depleted to 6 g/dL. The patient was managed by oral surgery. The patient also had a TAVR. The patient completed 11 of 12 planned hypofractionated external beam radiotherapy treatments prior to the patient’s TAVR intervention. The patient voiced some irritation to the skin on the left arm after his last radiation treatment, and this inflammation resolved on its own. The patient did not finish his final radiation treatment because he was undergoing his TAVR. Monthly follow-ups with oncology and dermatology were completed. Radiation oncology and dermatology also coordinated appointments with the patient to be held on the same days to avoid additional travel. The patient was also referred to a lymphedema-certified physical therapist/occupational therapist for exercise and massage therapy. PET scans were scheduled every 4 months. The patient responded well to radiation, as the presentation of the third MCC completely resolved with no additional satellite lesions (Figure 8).
Discussion
The recurrence rate of MCC is approximately 40%. The average time to MCC recurrence is 8 months, and 95% of recurrences occur within 36 months.22 The risk of distant metastatic disease is approximately 35%. Incidence of reccurrence within the first year increases with higher staged cases of MCC.22 The overall average 5-year survival rates of MCC involving all stages ranges from 30% to 64%, compared to approximately 93% in average malignant melanoma cases. The 5-year survival rate when diagnosed at stage IV is 14% for MCC compared to 29% for stage IV melanoma.1 Educating patients on recurrence and completing frequent screening exams is of high importance for successful outcomes.
It has been found that the OS of rural patients with MCC compared to urban patients is remarkably lower, having a mean survival time of 34 vs 47 months, respectively.4 A recent study by Stenback et al24 noted that the 5-year survival rate for patients with MCC in metropolitan areas was 51.3%, compared to 46% for patients with MCC in nonmetropolitan areas. The lower survival rates could be due to decreased access to dermatology and oncology care. One-fifth of the US population resides in rural areas, but only one-tenth of medical providers practice in rural settings.24 Only 3% of oncologists practice in rural areas, and approximately 70% of US counties do not have to access to an oncologist.25 Rural patients with MCC have also been found to have decreased access to transportation, decreased education levels, and decreased annual income compared to urban patients with MCC.24,26
The patient started pembrolizumab immediately after the first surgical excision. NCCN 2025 guidelines recommend pembrolizumab for primary and recurrent regional disease if curative surgery and/or radiation therapy are not attainable.21 Though the patient underwent excision, the travel for radiation therapy was not feasible, and pembrolizumab infusions were performed every 3 weeks. In a multicenter phase 2 trial, 50 patients with MCC with distant metastatic or locoregional disease disinclined to undergo surgery or radiation therapy with no prior systemic therapy treatment received pembrolizumab 2 mg/kg every 3 weeks. Study participants were followed for a median of 31.8 months. The objective response rate was 58%, with 30% of patients having a complete response to treatment and 28% having a partial response.27 This study has a median progression-free survival (PFS) rate of 16.8 months, and the 3-year PFS rate was 39.1%. The overall 3-year survival rate was 59.4%, and it was 89.5% for patients with complete or partial response.27 This study illustrated durable OS and PFS in patients with MCC with distant metastatic or locoregional disease nonreceptive to definitive surgery or radiation therapy.27
According to NCCN 2025 guidelines, patients with MCC with local and regional disease who received radiation therapy instead of surgery had poorer outcomes.21 Several studies illustrate that radiation after surgical interventions has been found to lower recurrence and improve survival compared to surgery alone.21 Other studies have also supported the effectiveness of radiation therapy to reducing nodal relapse in node-negative patients.20
A patient’s access to transportation is considered a social determinant of one’s health.25 Approximately 1.6 million rural households do not have access to a motorized vehicle.25 Patients residing in rural settings have limited access to oncology care. It is estimated that 13% of radiation oncologists practice in rural settings.26 Approximately 34.4% of rural patients reside at least an hour from a radiation oncologist.26 Longer travel distances for patients have been associated with failure for patients to receive recommended radiation therapy.25,26 Studies demonstrate that if rural patients have options to undergo surgery or radiation or chemotherapy, they most often pick the surgical intervention due to less travel.25 A study completed by Morris et al28 reported that rural residents were nearly 20% more likely to be nonadherent to radiation therapy than nonrural patients; rural residents often missed 10% of their radiation treatments and had an 11% higher rate of mortality compared to nonrural residents.
Currently, there is no specific screening test to detect MCC. Anyone has the potential to develop MCC. Yearly skin cancer screenings and monthly home skin checks should be completed. A greater awareness and consideration of MCC in primary care settings could result in earlier diagnosis, as MCC can be misdiagnosed as an epidermal inclusion cyst, pyogenic granuloma, or suspected nonmelanoma skin cancer.11 The perceived nonthreatening diagnoses can delay biopsy and referral to dermatology.
Determining a histopathological and immunohistochemical examination protocol for biopsies could increase the accuracy of diagnosis on biopsy specimens. There currently is not a gold-standard technique to detect MCPyV. Polymerase chain reaction–based techniques have been used, but concern has been raised regarding sensitivity.19 Studies have compared the use of MCPyV antibody clones and CK20 clones for the diagnosis of MCC. In a cross-sectional study by Yeni Erdem et al,19 CK20 clone Ks20.8 was found to have consistent performance and could be a preferred option for diagnosing MCC, and Ab3 clone of MCPyV appeared superior to CM2B4 for identifying MCPyV-positive cases.
Determining whether recurrence is higher in those with positive or negative SLNBs could be an area for further study. It has been found that those patients with negative SLNBs often have more favorable outcomes. Some data have illustrated that patients with positive SLNBs have a higher rate of recurrence, but other studies have shown no difference in recurrence whether the SNLB was positive or negative..29 The Mayo Clinic completed a study including 150 patients with MCC who underwent SLNB and found there was no incongruity in recurrence rates of those patients with positive or negative SLNB results regarding regional disease, but those patients with a positive SLNB were found to have a higher incidence of in-transit metastases.30 Further studies are needed to determine if all patients with MCC, whether with a positive or negative SLNB, would have less recurrence if aggressive adjuvant radiation or targeted therapies are initiated.
Evolving research continues with targeted therapies that treat definitive cellular mechanisms that have been transformed during tumor formation and metastasis. These targeted therapies might allow improved clinical response and improved tolerability of treatment for patients. Receptor tyrosine kinase inhibitors (TKIs) and somatostatin analogs (SSAs) have been found to decrease tumor proportion and improve response rates in patients with MCC. Targeted therapies could be promising treatment options for patients with recurrent MCC who have failed traditional therapies or were limited with meager alternatives and poor prognosis.31
The NCCN recommends that patients diagnosed with MCC complete a skin examination and regional lymph node exam every 1 to 3 months for the first year, every 3 to 6 months the year after, and then annually thereafter. Current guidelines suggest imaging as clinically indicated.13,21,23 Patients who are immunosuppressed presenting with more aggressive disease may undergo more frequent PET/CT exams for additional surveillance.13,21 Considerations for determining the imaging frequency in rural patients can involve access to radiology services, including distance to travel. If patients remain recurrence-free for over 5 years, annual screening might be recommended.3
The patient in this case report had an extensive medical history with numerous comorbidities. It is difficult to pinpoint whether pembrolizumab alone was a contributing factor to the patient’s hospital admission with pneumonia that led to a congestive heart failure exacerbation and respiratory failure. It would be interesting to know if, had the patient restarted pembrolizumab once stable after hospitalization, his MCC would have been recurrent or not. The patient’s PET scans while undergoing pembrolizumab did not demonstrate any disease recurrence.
Currently, there is limited research on surgical management with local recurrences. Presently, recurrent MCC cases are often approached with the initial wide local excision depending on anatomical location.32 Management of recurrent MCC cases, whether local, locally advanced, regional, or disseminated recurrence, can involve systemic therapy, radiation therapy, surgery, or a combination of interventions.21,32
As more treatment modalities become available for the treatment of MCC, no single treatment plan is tailored for every single patient. Medical providers must design treatment plans for patients to achieve the best possible outcomes in a holistic manner. Understanding a patient’s obstacles can increase adherence, patient motivation, and the patient’s rapport with medical providers. Telemedicine, travel programs, patient assistant lodging, and mobile radiation units could be methods to assist rural patients with barriers to medical treatment.
Conclusion
MCC is the most lethal of all skin cancers. Though advances in detection have taken place, the number of MCC cases is on the rise globally. Emerging treatments targeting specific pathways continue to evolve. It has been found that the overall death rates from MCC have declined since 2010. This decline is speculated to be the result of new emerging therapy options, such as immunotherapy.
Increased public awareness of MCC might heighten early evaluation and treatment. Educating primary care providers that MCC has a higher mortality rate than melanoma might expediate biopsy and referral to dermatology on MCC presentations. Though MCC is rare, educating patients on how to complete home surveillance exams, having family on the look-out, and creating a comfortable provider-patient relationship wherein patients are at ease notifying their provider immediately if any changes develop can expedite diagnosis and improve patient outcomes. This case report heightens the cognizance of MCC, demonstrates the expeditious reccurrence that MCC can have, and illuminates the innovativeness medical providers must demonstrate when managing patients with the challenges of treatment in the rural US.
Ethics Statement
The patient gave verbal and signed consent to review his medical history and use images for this educational article. An informed consent form was completed and signed by the patient.
References
- Tribble JT, Brownell I, Cahoon EK, et al. A comparative study of Merkel cell carcinoma and melanoma incidence and survival in the United States, 2000-2021. J Invest Dermatol. 2025;145(8):1979–1986.
- Uchi H. Merkel cell carcinoma: an update and immunotherapy. Front Oncol. 2018;8:48.
- Becker JC, Stang A, DeCaprio JA, et al. Merkel cell carcinoma. Nat Rev Dis Primers. 2017;3:17077.
- Brunk D. Elevated mortality seen in Merkel cell patients from rural areas. MDedge. 17 Dec 2021. Accessed 4 Oct 2025. https://www.mdedge.com/familymedicine/article/250046/nonmelanoma-skin-cancer/elevated-mortality-seen-merkel-cell-patients
- America Counts Staff. What is rural America? United States Census Bureau. 9 Aug 2017. Accessed 1 Oct 2025. https://www.census.gov/library/stories/2017/08/rural-america.html
- New review highlights gaps in skin cancer care for older adults, rural residents, and others. Huntsman Cancer Institute | University of Utah Health. 16 Jul 2025. Accessed 4 Oct 2025. https://healthcare.utah.edu/huntsmancancerinstitute/news/2025/07/new-review-highlights-gaps-skin-cancer-care-older-adults-rural-residents-and-others
- Cunningham SA, Yu R, Shete S. Differences in sun protection behaviors between rural and urban communities in Texas. J Rural Health. 2019;35(2):155–166.
- Zahnd WE, Goldfarb J, Scaife SL, Francis ML. Rural-urban differences in behaviors to prevent skin cancer: an analysis of the Health Information National Trends Survey. J Am Acad Dermatol. 2010;62(6):950–956.
- Jimmy Buffett’s death from Merkel cell carcinoma was associated with an increase in online search activity related to skin cancer. Merkelcell.org. 30 Jun 2024. Accessed 1 Jul 2025. https://merkelcell.org/news-and-publications/2024/jimmy-buffetts-death-from-merkel-cell-carcinoma-was-associated-with-an-increase-in-online-search-activity-related-to-skin-cancer-2/
- Siqueira SOM, Campos-do-Carmo G, Dos Santos ALS, et al. Merkel cell carcinoma: epidemiology, clinical features, diagnosis and treatment of a rare disease. An Bras Dermatol. 2023;98(3):277–286.
- Mohsen ST, Price EL, Chan AW, et al. Incidence, mortality and survival of Merkel cell carcinoma: a systematic review of population-based studies. Br J Dermatol. 2024;190(6):811–824.
- Paulson KG, Park SY, Vandeven NA, et al. Merkel cell carcinoma: current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78(3):457–463.
- Bichakjian CK, Olencki T, Aasi SZ, et al. Merkel Cell Carcinoma, Version 1.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16(6):742–774.
- Brady M, Spiker AM. Merkel cell carcinoma of the skin. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025-. Updated 17 Jul 2023. https://www.ncbi.nlm.nih.gov/sites/books/NBK482329/
- Bolognia J, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018:2061–2064.
- Yang JF, You J. Merkel cell polyomavirus and associated Merkel cell carcinoma. Tumour Virus Res. 2022;13:200232.
- Silling S, Kreuter A, Gambichler T, et al. Epidemiology of Merkel cell polyomavirus infection and Merkel cell carcinoma. Cancers (Basel). 2022;14(24):6176.
- Erstad DJ, Cusack JC Jr. Mutational analysis of merkel cell carcinoma. Cancers (Basel). 2014;6(4):2116–2136.
- Yeni Erdem B, Baykal C, Ozluk Y, et al. Evaluating CK20 and MCPyV antibody clones in diagnosing Merkel cell carcinoma. Endocr Pathol. 2025;36(1):1
- Miller SJ, Alam M, Andersen J, et al. Merkel cell carcinoma. J Natl Compr Canc Netw. 2009;7(3):322–332.
- National Comprehensive Cancer Network. Merkel Cell Carcinoma Version 2.2025. 18 Apr 2025. Accessed 1 Jul 2025. https://merkelcell.org/wp-content/uploads/2025/06/NCCN_MCC_2025Guidelines.pdf
- McEvoy AM, Lachance K, Hippe DS, et al. Recurrence and mortality risk of Merkel cell carcinoma by cancer stage and time from diagnosis. JAMA Dermatol. 2022;158(4):382–389.
- NCI Staff. FDA approves pembrolizumab to treat Merkel cell carcinoma. National Cancer Institute. 4 Feb 2019. Accessed 23 Feb 2026. https://www.cancer.gov/news-events/cancer-currents-blog/2019/pembrolizumab-merkel-cell-carcinoma-fda-approval
- Steinback G, Chang J, Siscos M. 60972 Merkel cell carcinoma updated trends and overall survival rates considering urban vs rural. J Am Acad Dermatol. 2025;93(3 Suppl):AB64.
- Wercholuk AN, Parikh AA, Snyder RA. The road less traveled: transportation barriers to cancer care delivery in the rural patient population. JCO Oncol Pract. 2022;18(9): 652–662.
- Praeder R, Solberg T, Yorke AA. Underserved communities in the radiation therapy land of plenty – physicists’ perspective. J Appl Clin Med Phys. 2024;25(1):e14252.
- Nghiem P, Bhatia S, Lipson EJ, et al. Three-year survival, correlates and salvage therapies in patients receiving first-line pembrolizumab for advanced Merkel cell carcinoma. J Immunother Cancer. 2021;9(4):e002478.
- Morris BB, Hughes RT, Fields EC, et al. Sociodemographic and clinical factors associated with radiation treatment nonadherence and survival among rural and nonrural patients with cancer. Int J Radiat Oncol Biol Phys. 2022;116(1):28–38.
- Fields RC, Busam KJ, Chou JF, et al. Recurrence and survival in patients undergoing sentinel lymph node biopsy for Merkel cell carcinoma: analysis of 153 patients from a single institution. Ann Surg Oncol. 2011;18(9):2529–2537.
- Sims JR, Grotz TE, Pockaj BA, et al. Sentinel lymph node biopsy in Merkel cell carcinoma: the Mayo Clinic experience of 150 patients. Surg Oncol. 2018;27(1):11–17.
- Nammour HM, Madrigal K, Starling CT, Doan HQ. Advancing treatment options for Merkel cell carcinoma: a review of tumor-targeted therapies. Int J Mol Sci. 2024;25(20):11055.
- Park SY, Nghiem PT, Tai P, Silk AW. Treatment of recurrent and metastatic Merkel cell carcinoma. UpToDate. 29 Oct 2025. Accessed 1 Nov 2025. https://www.uptodate.com/contents/treatment-of-recurrent-and-metastatic-merkel-cell-carcinoma