 Dear Editor:
Dear Editor:
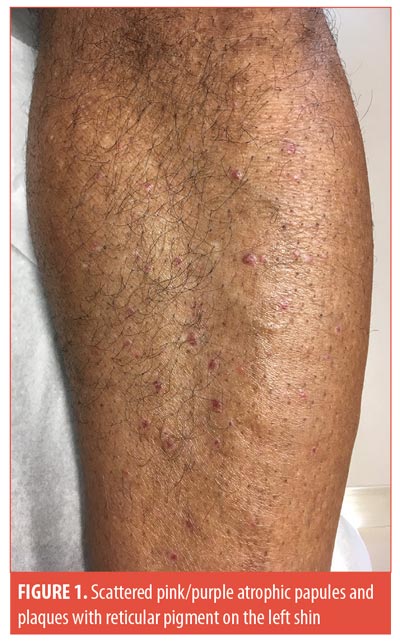
An African American man in his 50s presented with a one-year history of a pruritic rash on his anterior shins. His past medical history was significant for a recent diagnosis of diabetes. A physical examination revealed scattered, atrophic, pink/purple papules and plaques with reticular pigment on the bilateral anterior lower legs (Figure 1). No lesions were seen on oral examination. Punch biopsy showed an effaced and markedly thinned epidermis with overlying compact hyperkeratosis, a band-like lymphocytic infiltrate filling the papillary dermis, and numerous colloid bodies and melanophages along the dermal-epidermal junction, all consistent with atrophic lichen planus (LP). The patent was started on topical corticosteroids and showed an improvement in his symptoms at his four-month follow-up appointment.
Discussion. Typically, the clinical diagnosis of lichen planus is made when it presents as the class variant, characterized by purple, polygonal papules. However, many morphologic variants have been described and can make the diagnosis more challenging. Atrophic LP is one of the rarer variants of LP and typically affects the legs, with progressively enlarging papules that develop a characteristic central atrophy.1 Other reported sites included the axillae, glans penis, and trunk.2 Differential diagnosis includes subacute cutaneous lupus erythematosus, discoid lupus erythematosus, sarcoidosis, granuloma annulare, tinea, secondary syphilis, and mycosis fungoides. Due to the few reported cases, the etiology and pathogenesis remain poorly understood, but some cases are believed to be the end-stage lesion of other morphologic variants, such as hypertrophic LP.1
Many medications have been linked to drug-induced LP, including nonsteroidal anti-inflammatory agents, beta-blockers, antimalarials, methyl-dopa, and penicillamine.3 However, to our knowledge, no cases of drug-induced atrophic LP have been reported in the literature. Our patient was recently diagnosed with diabetes mellitus (DM), which has a long standing, controversial association with LP, as described in a recent meta-analysis that cited contradictory results.4 A major limitation of the studies analyzed was a lack of uniform method for confirming the diagnosis of DM. Only one study utilized hemoglobin A1C and found a statistically significant increase in A1C in patients with LP compared to controls.5 Routine screening for hepatitis C has been recommended for patients with recurrent LP, although this association is likely only significant in certain geographic areas.6 Treatment is identical to other cutaneous forms of LP. In localized cases, topical corticosteroids are considered first-line therapy, followed by narrowband ultraviolet B. There are numerous options for generalized or refractory cases, including systemic or intramuscular corticosteroids, plaquenil, dapsone, oral retinoids, or apremilast.
With regard,
Andrew J. Krispinsky, MD, and Catherine A. Ulman, MD
Affiliations. Drs. Krispinsky and Ulman are with the Division of Dermatology in the Department of Internal Medicine at The Ohio State University in Columbus, Ohio.
Disclosures. The author has no conflicts of interest relevant to the content of this article.
Correspondence. Catherine Ulman, MD; Email: catherine.ulman@osumc.edu
References
- Wagner G, Rose C, Sachse MM. Clinical variants of lichen planus. J Dtsch Dermatol Ges. 2013;11(4):
309–319. - Weston G, Payette M. Update on lichen planus and clinical variants. Int J Womens Dermatol. 2015;1(3):
140–149. - Thompson DF, Skaehill PA Drug-induced lichen planus. Pharmacother. 1994;14(5):561–571.
- Otero Rey EM, Yáñez-Busto A, Rosa Henriques IF, et al. Lichen planus and diabetes mellitus: systematic review and meta-analysis. Oral Dis. 2019;25(5):
1253–1264. - Seyhan M, Ozcan H, Sahin I, et al. High prevalence of glucose metabolism disturbance in patients with lichen planus. Diabetes Res Clin Pract. 2007;77(2):198–202.
- Lodi G, Giuliani M, Majorana A, et al. Lichen planus and hepatitis C virus: a multicenter study of patients with oral lesions and a systematic review. Br J Dermatol. 2004;151(6):1172–1181.
