J Clin Aesthet Dermatol. 2025;18(12):76–81.
by Promise Ufomadu, BSA; Bartley Joseph Gill, MD, PhD; Ikue Shimizu, MD; Ida Orengo, MD; and Theodore Rosen, MD
Mr. Ufomadu is with School of Medicine at Baylor College of Medicine in Houston, Texas. Dr. Gill is with Baylor College of Medicine and Memorial Hermann Health Systems in Houston, Texas.
Drs. Orengo, Rosen, and Shimizu are with the Department of Dermatology at Baylor College of Medicine in Houston, Texas.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no conflicts of interest relevant to the contents of this article.
ABSTRACT: Dermatologic infections, particularly fungal and viral, are globally prevalent and often lead patients to seek complementary and alternative medicines (CAMs). This review critically evaluates 17 studies investigating CAM efficacy in treating dermatologic infectious diseases, focusing on those evaluated in randomized controlled trials (RCTs). Tea tree oil shows modest benefit for fungal infections such as tinea pedis and onychomycosis, though studies are limited by poor blinding and dropout rates. For herpes simplex virus (HSV), propolis and Melissa officinalis demonstrate potential antiviral effects, but findings are limited by subjective outcomes and industry sponsorship. Green tea extract (polyphenon E) shows efficacy in treating genital warts, though adverse skin reactions and publication bias warrant caution. Other agents, including garlic-derived ajoene, honey mixtures, and podophyllin, show promise but lack robust RCT validation. While CAMs offer intriguing therapeutic avenues, rigorous trials with standardized outcomes are essential to guide evidence-based integration into dermatologic care and improve patient-centered treatment strategies. Keywords: Complementary and alternative medicine, herpes virus, HSV, fungal infections, condyloma, onychomycosis, botanical
Introduction
Dermatologic infections, including both fungal and viral etiologies, are prevalent worldwide, leading to significant chronic health burdens. In 2013, skin diseases ranked fourth among all nonfatal diseases globally, underscoring their prevalence and impact.1 Fungal infections, such as dermatophytosis and endemic mycoses, are particularly common in tropical regions due to favorable environmental conditions; thus, they affect an estimated 25% of the world population.2 Skin infections also impose a considerable economic burden, with substantial costs attributed to outpatient visits, hospitalizations, and long-term treatment regimens.3 Consequently, patients often rely on complementary and alternative medicines (CAMs) that might provide therapeutic relief. National surveys indicate widespread use of CAMs among the public, with roughly 30% to 50% of the adult population reporting use of some form of CAM therapy. There is also a greater than average use among certain subgroups, including women, older adults, non-White, and immigrant groups.4 Remarkably, 85% of patients in the National Health Interview Survey who reported having skin problems have also used some form of CAM.5 Furthermore, surveyed physicians overwhelmingly report that patients, not physicians, initiate discussions about CAM use, and while expressing skepticism about CAM efficacy, the substantial majority desire to learn more about CAM therapies to better counsel patients.6,7 The widespread use of CAMs among dermatology patients suggests patient needs may not be fully met by conventional dermatology. Comprehensive, objective knowledge of CAM therapies could help dermatologists connect better with patients and guide treatment plans with more sensitivity and potentially better success.8
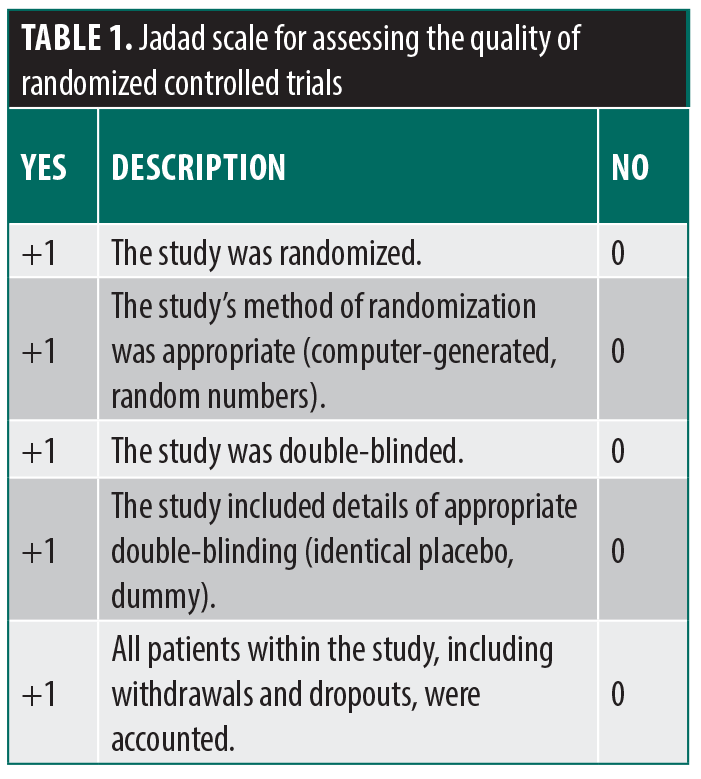
This review offers an overview of CAM therapies investigated for dermatologic infectious diseases, emphasizing viral and fungal infections and detailing only those evaluated in randomized controlled trials (RCTs) or other significant clinical studies. Where applicable, the quality of evidence for a CAM therapy will be assessed using the Jadad Scale to guide practitioners on potential efficacy (Table 1).9 Heightened scrutiny should help differentiate between scientifically proven CAM agents and those merely claimed effective by advocates.
Fungal Infections
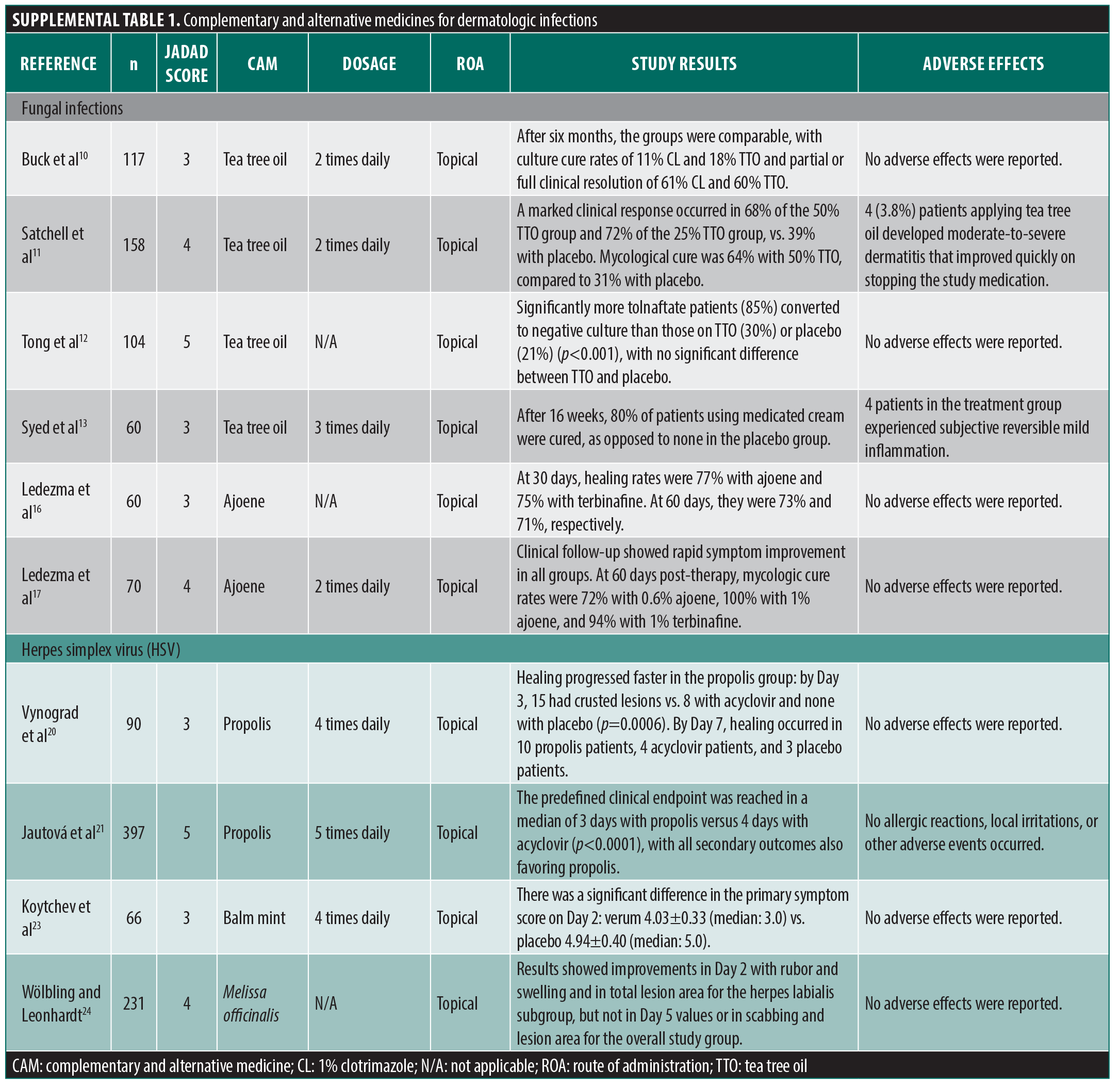
Tea tree oil (TTO) has been examined in RCTs for use in the treatment of onychomycosis and tinea pedis. The onychomycosis study compared 6-month treatment with 100% TTO and 1% clotrimazole solution, finding comparable mycologic culture and subjective clinical cure rates.10 Unfortunately, this study suffered from unclear blinding, no intent-to-treat analysis despite heavy loss to follow-up, and completely unspecified clinical outcome measures. One tinea pedis study by Satchell et al11 compared 4-week treatment response of two concentrations of TTO to a vehicle only control, finding significant improvement in subjective clinical assessment and skin scraping cure rates only in the active treatment groups. However, no information was provided on investigator blinding, the parameters of the clinical cure outcome were not provided, only intragroup comparisons were made, and an intent-to-treat analysis was not performed despite high dropouts in the TTO group. Notably, moderate-to-severe dermatitis was observed in some patients in the TTO group. Finally, Tong et al12 performed a 5-week study (n=104) examining treatment of tinea pedis with 10% TTO in sorbolene cream, 1% tolnaftate cream, or sorbolene vehicle only control; the study assessed mycological cure and improvement in a multicomponent clinical outcome score (scaling, inflammation, itching, burning). While the TTO group did show some statistically significant differences over vehicle in clinical improvements, mycological cure rate was equivalent but poor in the TTO and placebo groups (tolnaftate group was significantly greater than both) and complete cure rate (mycological + clinical response) was only significantly greater in the tolnaftate group compared to placebo. This study was overall well designed, though no intent-to-treat analysis was performed despite some dropouts, and there was a baseline significant difference in sex between the groups (but no difference in baseline clinical symptoms). Another study on TTO, subject to questionable statistical measures, also found this agent to be effective for managing fungal infections.13 Taking these studies together, we assign the quality of the evidence supporting the use of TTO for the treatment of fungal infections an average Jadad score of 3.75, due to issues with risk of bias, and poor design and noting a heightened potential for contact dermatitis with TTO use.
Many other agents have been suggested as effective CAM treatments for fungal infections, including honey, aloe vera, and lavender, but no RCT-level study yet exists to assess their effectiveness.14 One study demonstrated effectiveness of a topical gel containing ajoene, an organosulfur derived from garlic, in treating tinea pedis, but there was no control group for comparison.15 The authors performed two additional follow-up studies, where both trials compared ajoene-based treatment with terbinafine, and showed clinically improved findings that lacked proper significant analysis; adequate blinding was also in question.16,17 As a result, the average Jadad score for these two studies is 3.5. Two open studies have shown effectiveness in treating tinea infections with of a mixture of honey, olive oil and beeswax18 and an extract of snow gum (Eucalyptus pauciflora),19 but more robust controlled studies with these agents have yet to be performed.
Herpes Simplex Virus (HSV)
A few studies have examined the honeybee-derived substance propolis for treatment of herpes simplex virus (HSV) infections due to purported anti-inflammatory and virucidal effects of its constituent flavonoids.14 Vynograd et al20 compared ointments containing 3% propolis, 5% acyclovir, or vehicle only for the treatment of genital herpes, with subjective lesion healing as an outcome measured for participants classified by lesion stage at presentation (vesicular or ulcerated). Investigators found significantly improved healing rates in vesicular and combined lesions in the propolis group compared to the acyclovir group, and improved healing rates in all three measures (vesicular, ulcerated, and combined) compared to placebo. While the study was generally well designed, the authors noted that it was only a single-blind design, because the active treatment ointments varied substantially in appearance and odor, and the outcome measure relied only on intragroup measure of subjective healing with no less subjective intergroup comparisons (number of lesions, size, etc). A RCT prior to this study concluded that propolis ointment helped induce healing of outbreaks of HSV and herpes zoster significantly faster than a placebo ointment, but this study is unavailable in English for review.14 Another study on propolis also found this agent to be effective for managing HSV infections, albeit with frequent applications (5 times daily).21
A few RCTs have examined the use of balm mint/lemon balm (Melissa officinalis) in the treatment of herpes infections due to purported virustatic activity.22 One report, which contained no description of author blinding or placebo matching, compared the response of 1% balm mint cream to placebo for the treatment of herpes labialis.23 Measuring a multicomponent scoring system (complaints, blisters, affected area), investigators concluded efficacy based on a significant difference in scores between groups at Day 2. This conclusion is remarkably exaggerated, as all other measured outcomes, including disease scores at the three other timepoints and secondary outcomes like patient evaluation and blister number, showed no difference between groups (trending in favor of placebo). In a separate large study, Wölbling and Leonhardt24 examined patients with a variety of HSV infections and topical application of a commercial M. officinalis cream or vehicle placebo to lesions over 5 days. Authors concluded effectiveness but recorded mixed results: improvements in Day 2 rubor and swelling, but not Day 5 values, and improvement in herpes labialis subgroup total lesion area but no improvement in scabbing and lesion area of the entire study group. While the study was large with well-matched groups and intergroup comparisons, the process of investigator blinding was not well described; this is of increased importance with this study because it was sponsored by the manufacturer of the treatment lotion. Other agents that have purported effects on HSV infections include extracts of licorice and hibiscus, but these have yet to be fully tested in an RCT-level study. When assessing the quality of evidence for CAMs used to HSV, the average Jadad score assigned is 3.75 due to possible risks of bias and improper study design.
Warts
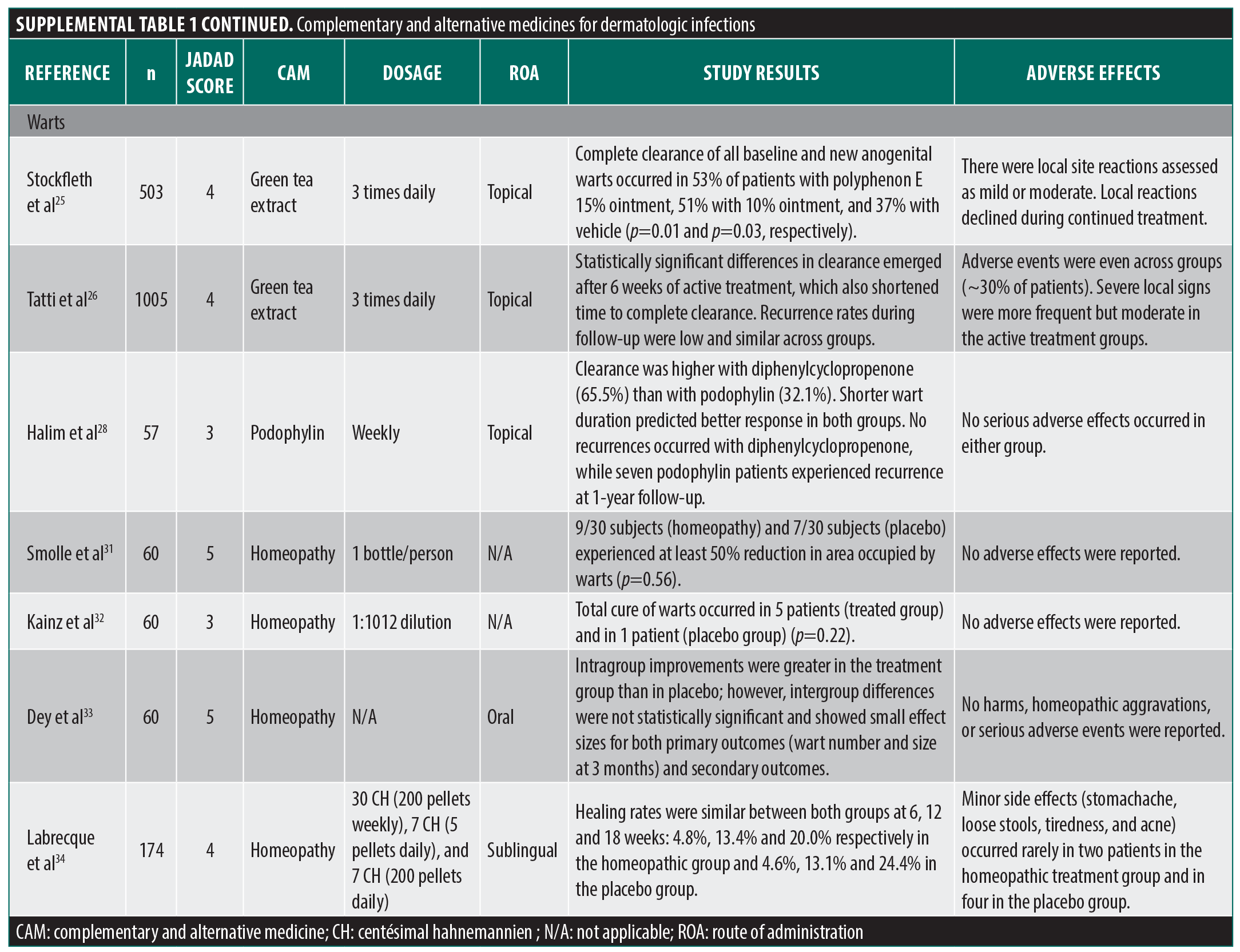
Several CAMs have been suggested as treatments for condyloma and verruca warts, including various herbal agents, green tea extract (GTE), homeopathy, and hypnosis. Two recent reports from the same group have examined the effectiveness of a commercial green tea extract cream (polyphenon E) in large RCTs for the treatment of genital and perianal warts.25,26 The investigators found increased wart clearance rates in both groups using the treatment ointment at 10% and 15% concentrations compared to the control (>50% vs. ~35% for vehicle only). Secondary outcomes showed significant improvement in median wart number and coverage area,25 significantly shorter time to complete clearance, and similar recurrence rates.26 Overall, these studies were well designed and properly performed; however, while the studies were labeled double-blind, no information was provided on blinding procedures and the authors were paid employees or consultants of the treatment cream manufacturer. Both of these issues may be deserving of heightened scrutiny given that the primary outcome was subjective clearance. Furthermore, the more objective outcomes were less impressive. While a difference was seen in median wart area and count (unsurprising, as they were reported as zero in treatment groups), no difference was seen in the average of these parameters, with averages perhaps being of greater relevance because this parameter would factor in a variability in response with standard deviations. Despite this, the average Jadad score for both studies is 4, due to risk of bias (publication bias, industry sponsor) and design flaw (blinding, outcome measures). It is also worth noting that unlike many other GTE studies, treatment groups in these studies did see a significantly higher incidence of adverse local skin reactions including erythema, edema, and erosion (no difference in systemic adverse events).
Another herbal agent of note in treating warts is podophyllin, derived from the root of the American mayapple (Podophyllum peltatum). Podophyllin has been widely reported as effective in treating condylomata acuminata, but one of the few RCTs available to fully assess effectiveness was not published in English.27 A recently published study comparing podophyllin to diphenylcyclopropenone found that the latter had a shorter wart duration and was significantly more effective than podophyllin with a lower recurrence at the 1-year follow-up28; however, we give this study a Jadad score of 3 due to lack of randomization details and proper blinding. Other herbal agents with purported effectiveness but no RCT published include nightshade (Solanaceae dulcamara), calotropis (Calotropis procera), celandine (Chelidonium majus), and thujone—an active constituent found in Thuja occidentalis herbs that has shown efficacy in case reports.29,30
Nonbotanical CAM agents that have been used in the treatment of warts include homeopathy and hypnosis. Multiple homeopathic preparations have been suggested for wart treatment by practitioners of homeopathic medicine. However, limited studies have confirmed their effectiveness, and a few well designed RCTs have found no improvement compared to placebo in common wart area reduction following serial application of several different homeopathic preparations.31-33 Labrecque et al34 also found no improvement in clearance of plantar warts with a mixture of three different preparations, suggesting that the extreme dilution of active agents in homeopathy ensures lack of effectiveness. In a different approach, a group of studies from the 1950s and 1960s claimed effectiveness of hypnotic suggestion on clearance of warts, but these results have not been reliably replicated in studies with robust methodology and appropriately stringent outcome measures.35
Conclusion
It is evident that, whether physician-guided or not, the use of CAM has become a widespread and notable component of many patients’ treatment regimens in dermatology. Driven by positive anecdotal evidence and significant in vitro findings, investigators have sought to transition many CAM agents into scientific medicine by conducting placebo-controlled, double-blind RCTs. Unfortunately, this effort has not been widely successful, as most studies concluding CAM benefit suffer major methodological problems that impact credibility, such as poor blinding, subjective outcomes, unreported baseline measures, lack of intergroup comparisons, and tendency to overstate positive findings while overlooking null results. Despite these reservations, some study results and the wealth of in vitro and murine data suggest many CAM agents warrant further rigorous investigation and consistent replication in well conducted RCTs. Moving forward, a pragmatic focus on comparing CAM treatments to conventional therapies, alone or integratively, would accurately reflect clinical use by patients and prove more useful for practitioners. In the interim, dermatologists should remain aware of demonstrated or purported CAM efficacy from published studies, while vigilantly monitoring for adverse effects that could impact conventional treatment plans. The widespread use of CAMs necessitates that physicians objectively evaluate their role to optimally guide patient interactions and improve outcomes through an informed, empathetic approach that integrates all therapeutic modalities.
References
- Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134(6):1527-1534.
- Martinez-Rossi NM, Peres NTA, Bitencourt TA, Martins MP, Rossi A. State-of-the-art dermatophyte infections: epidemiology aspects, pathophysiology, and resistance mechanisms. J Fungi (Basel). 2021;7(8):629.
- Dehkharghani S, Bible J, Chen JG, Feldman SR, Fleischer AB Jr. The economic burden of skin disease in the United States. J Am Acad Dermatol. 2003;48(4):592-599.
- Wootton J, Sparber A. Surveys of complementary and alternative medicine: part I. General trends and demographic groups. J Altern Complement Med. 2001;7(2):195-208.
- Fuhrmann T, Smith N, Tausk F. Use of complementary and alternative medicine among adults with skin disease: updated results from a national survey. J Am Acad Dermatol. 2010;63(6):1000-1005.
- Wahner-Roedler DL, Vincent A, Elkin PL, Loehrer LL, Cha SS, Bauer B. Physicians’ attitudes toward complementary and alternative medicine and their knowledge of specific therapies: a survey at an academic medical center. Evid Based Complement Alternat Med. 2006;3(4):495-501.
- Godin G, Beaulieu D, Touchette JS, Lambert LD, Dodin S. Intention to encourage complementary and alternative medicine among general practitioners and medical students. Behav Med. 2007;33(2):67-77.
- Ernst E. The usage of complementary therapies by dermatological patients: a systematic review. Br J Dermatol. 2000;142(5):857-861.
- Jadad A, Moore R, Carroll D, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1-12.
- Buck DS, Nidorf DM, Addino JG. Comparison of two topical preparations for the treatment of onychomycosis: Melaleuca alternifolia (tea tree) oil and clotrimazole. J Fam Pract. 1994;38(6):601-605.
- Satchell AC, Saurajen A, Bell C, Barnetson RSC. Treatment of interdigital tinea pedis with 25% and 50% tea tree oil solution: a randomized, placebo-controlled, blinded study. Australas J Dermatol. 2002;43(3):175-178.
- Tong MM, Altman PM, Barnetson RS. Tea tree oil in the treatment of tinea pedis. Australas J Dermatol. 1992;33(3):145-149.
- Syed TA, Qureshi ZA, Ali SM, Ahmad S, Ahmad SA. Treatment of toenail onychomycosis with 2% butenafine and 5% Melaleuca alternifolia (tea tree) oil in cream. Trop Med Int Health. 1999;4(4):284-287.
- Baumann L. Botanical ingredients in cosmeceuticals. J Drugs Dermatol. 2007;6(11):1084-1088.
- Ledezma E, De Sousa L, Jorquera A, et al. Efficacy of ajoene, an organosulphur derived from garlic, in the short-term therapy of tinea pedis. Mycoses. 1996;39(9-10):393-395.
- Ledezma E, López JC, Marin P, et al. Ajoene in the topical short-term treatment of tinea cruris and tinea corporis in humans. Randomized comparative study with terbinafine. Arzneimittelforschung. 1999;49(6):544-547.
- Ledezma E, Marcano K, Jorquera A, et al. Efficacy of ajoene in the treatment of tinea pedis: a double-blind and comparative study with terbinafine. J Am Acad Dermatol. 2000;43(5 Pt 1):829-832.
- Al-Waili NS. An alternative treatment for pityriasis versicolor, tinea cruris, tinea corporis and tinea faciei with topical application of honey, olive oil and beeswax mixture: an open pilot study. Complement Ther Med. 2004;12:45-47.
- Shahi SK, Shukla AC, Bajaj AK, et al. Broad spectrum herbal therapy against superficial fungal infections. Skin Pharmacol Appl Skin Physiol. 2000;13(1):60-64.
- Vynograd N, Vynograd I, Sosnowski Z. A comparative multi-centre study of the efficacy of propolis, acyclovir, and placebo in the treatment of genital herpes (HSV). Phytomedicine. 2000;7(1):1-6.
- Jautová J, Zelenková H, Drotarová K, Nejdková A, Grünwaldová B, Hladiková M. Lip creams with propolis special extract GH 2002 0.5% versus acyclovir 5.0% for herpes labialis (vesicular stage): randomized, controlled double-blind study. Wien Med Wochenschr. 2019;169(7-8):193-201.
- Dimitrova Z, Dimov B, Manolova N, et al. Antiherpes effect of Melissa officinalis L. extracts. Acta Microbiologica Bulgarica. 1993;29:65-72.
- Koytchev R, Alken RG, Dundarov S. Balm mint extract (Lo-701) for topical treatment of recurring herpes labialis. Phytomedicine. 1999;6(4):225-230.
- Wölbling RH, Leonhardt K. Local therapy of herpes simplex with dried extract from Melissa officinalis. Phytomedicine. 1994;1(1):25-31.
- Stockfleth E, Beti H, Orasan R, et al. Topical polyphenon E in the treatment of external genital and perianal warts: a randomized controlled trial. Brit J Dermatol. 2008;158(6):1329-1338.
- Tatti S, Stockfleth E, Beutner KR, et al. Polyphenon E: a new treatment for external anogenital warts. Brit J Dermatol. 2010;162(1):176-184.
- Reuter J, Merfort I, Schempp C. Botanicals in dermatology: an evidence-based review. Am J Clin Dermatol. 2010;11(4):247-267.
- Halim HM, Ahmed SFAE, Eyada MM. Effectiveness and safety of topical application of diphenylcyclopropenone versus podophyllin in treatment of genital warts. Int J STD AIDS. 2023;34(9):641-648.
- Joseph R, Pulimood SA, Abraham P, John GT. Successful treatment of verruca vulgaris with Thuja occidentalis in a renal allograft recipient. Indian J Nephrol. 2013;23(5):362-364.
- Perroud HM. The treatment of warts today. Article in German. Ther Umsch. 1989;46(2):102-106.
- Smolle J, Prause G, Kerl H. A double-blind, controlled clinical trial of homeopathy and an analysis of lunar phases and postoperative outcome. Arch Dermatol. 1998;134(11):1368-1370.
- Kainz JT, Kozel G, Haidvogl M, Smolle J. Homoeopathic versus placebo therapy of children with warts on the hands: a randomized, double-blind clinical trial. Dermatology. 1996;193(4):318-320.
- Dey S, Hashmi S, Saha S, et al. A randomized, double-blind, placebo-controlled, pilot trial of individualized homeopathic medicines for cutaneous warts. Homeopathy. 2021;110(3):149-159.
- Labrecque M, Audet D, Latulippe LG, Drouin J. Homeopathic treatment of plantar warts. CMAJ. 1992;146(10):1749-1753.
- Shenefelt PD. Hypnosis in dermatology. Arch Dermatol. 2000;136(3):393-399.