J Clin Aesthet Dermatol. 2026;19(2):50–54.
by Mark S. Nestor, MD, PhD; Aysham Chaudry, DO; Robert J. Vanaria, BS; Wilhelmina Lam, DO, MPH; Alexandra DeVries, BS; and Alec Lawson, BS
Dr. Nestor is with the Center for Clinical and Cosmetic Research in Aventura, Florida and the Department of Dermatology and Cutaneous Surgery and the Department of Surgery, Division of Plastic Surgery at the University of Miami, Miller School of Medicine in Miami, Florida. Drs. Chaudry and Lam are with the Center for Clinical and Cosmetic Research in Aventura, Florida. Mr. Vanaria is with the Center for Clinical and Cosmetic Research in Aventura, Florida and the Hackensack Meridian School of Medicine in Nutley, New Jersey. Ms. DeVries is with the Center for Clinical and Cosmetic Research in Aventura, Florida and the Rocky Vista University College of Osteopathic Medicine in Parker, Colorado. Mr. Lawson is with the Center for Clinical and Cosmetic Research in Aventura, Florida and the Arizona College of Osteopathic Medicine in Glendale, Arizona.
FUNDING: Dr. Nestor reports research grants from Biofrontera.
DISCLOSURES: Dr. Nestor is a consultant for Biofrontera. The remaining authors have no conflicts of interest relevant to the contents of this article.
ABSTRACT: BACKGROUND: Cutaneous squamous cell carcinoma in situ (isSCC) is a common skin cancer often arising on the face. Standard care involves excision or destruction, but less invasive therapies are becoming increasingly explored. Aminolevulinic acid photodynamic therapy (ALA-PDT) with red light has shown potential for treating isSCC. This study assesses the safety, tolerability, and efficacy of ALA-PDT with red light for facial isSCC. METHODS: Twenty patients with biopsy-confirmed facial isSCC (0.4-1.3cm) were treated. Lesions were prepared with a 4×4 gauze, followed by application of ALA gel 10% with 3-hour incubation. Red light therapy (13 minutes and 30 seconds; 37J/cm²) was then administered. Patients received two sessions 28±3 days apart. Eight weeks after the second treatment, lesions were excised for histopathologic analysis. The primary endpoint was complete histologic clearance, with clinical clearance as a secondary endpoint. Safety was evaluated through adverse events (AEs), local skin reactions (LSRs), and treatment-site pain. RESULTS: All 20 patients completed the study. Complete histologic clearance was achieved, and complete clinical clearance was observed in 100% (20 of 20) patients. No treatment-related AEs occurred. One unrelated case of contact dermatitis was reported. The most common LSR was moderate erythema, peaking after treatment and resolving over time. Mild scaling was also observed with similar resolution. Median posttreatment pain scores were 0 (range: 0-3) after ALA application and 3.5 (range: 0-10) after red light therapy. CONCLUSION: ALA-PDT with 10% gel and red light demonstrated excellent efficacy, safety, and tolerability, supporting its role as a promising noninvasive treatment for facial isSCC. Keywords: ALA-PDT, photodynamic therapy, red light, NMSC, SCC
Introduction
Squamous cell carcinoma in situ (isSCC), also referred to as Bowen’s disease, is a superficial skin cancer that can be a precursor to invasive squamous cell carcinoma (SCC), much like actinic keratoses (AKs), which are histologically on the same spectrum.1 While isSCC itself does not metastasize, it has the potential to progress into SCC, which is associated with a higher risk of morbidity and mortality and a less favorable prognosis than other forms of nonmelanoma skin cancers (NMSC).2 According to a recent study, the incidence of SCC, including isSCC, is now greater than the incidence of basal cell carcinoma (BCC).3 Traditionally, the risk of progression from isSCC to SCC has been estimated at 3% to 5%, though newer research suggests that the risk could be as high as 16.3%.4 The main risk factor for isSCC is ultraviolet (UV) radiation exposure, especially UV-A, with additional contributing factors such as older age, lighter skin types, exposure to carcinogens, chronic inflammation, infection with human papillomavirus, and immunosuppression.1,4
Timely treatment of isSCC is essential to reduce growth and the risk of progression to invasive SCC. Although surgical excision, including Mohs micrographic surgery (MMS), and destruction using electrodesiccation and curettage (ED&C) are considered the gold standard for treating SCC, there is currently no universally accepted treatment protocol for SCC.5,6,7 For cases of low-risk SCC or isSCC, the 2025 National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology recommends standard excision with 4- to 6-mm margins of clinically normal skin.8 Nonsurgical therapies for isSCC include radiation therapy, topical chemotherapeutic agents, electrocoagulation, cryotherapy, and photodynamic therapy (PDT). Systemic chemotherapy is typically reserved for more advanced cases of SCC.7
PDT is widely used in dermatologic and oncologic conditions and involves the application of a photosensitizer that, when activated by specific wavelengths of light, generates reactive oxygen species to destroy targeted cells.9 PDT is currently approved for the treatment of AKs on the scalp and face and is also used off label for Bowen’s disease due to the high affinity of its photosensitizers for actinically damaged skin, AKs, NMSCs, and the pilosebaceous unit. Studies have shown that PDT can be effective in treating warts, BCC, AKs, and isSCC, particularly in solid organ transplant recipients; however, much of the existing evidence is limited by small sample sizes and shallow tumor depths.10-14 The most commonly used photosensitizer in the United States (US) is 5-aminolevulinic acid (ALA) in the 10% gel and 20% solution formulations. ALA-PDT gel employs 10% ALA as a prodrug that is metabolized in the skin to protoporphyrin IX (PpIX), which can be activated by visible light wavelengths, most effectively red light near 630 nm. The 10% ALA gel was developed for use with red light and the 20% ALA solution was developed for use with blue light.15 In a randomized clinical trial, ALA gel 10% with blue light was equivalent to 20% ALA solution with blue light for clearing AKs on the face and scalp with significantly less local skin reactions.11,15,16
Aminolevulinic acid hydrochloride topical gel 10% (AMELUZ), a porphyrin precursor, used in combination with a red-light illumination device (RhodoLED-XL, Biofrontera, Inc.), is approved for both lesion- and field-directed PDT of mild-to-moderate AKs within a treatment area of up to 20cm2 on the face and scalp.17 Red light penetrates deeper than shorter wavelengths, allowing activation of PpIX in the lower layers of the epidermis, where atypical keratinocytes may reside.18,19 This clinical study investigated the use of ALA-PDT as a nonsurgical treatment option for isSCC on the face, where surgery may not be optimal due to anatomic, functional, or cosmetic considerations.
Methods
Study design and population. This was a single-center, investigator-initiated, open-label study conducted from August 2024 to June 2025. The study protocol was reviewed and approved by the US Institutional Review Board. The study was conducted in accordance with the principles outlined in the Declaration of Helsinki. All patients provided written informed consent and photoconsent prior to the initiation of study.
Patients enrolled in the study were aged 18 years or older with facial isSCC histologically confirmed no more than 6 months prior to screening. Only lesions measuring 0.4cm to 1.3cm in diameter were included. Lesions with histological features of nodular BCC, superficial BCC, invasive SCC and other non-SCC tumors, severe squamous metaplasia, or infiltrative desmoplastic or micronodular growth patterns were excluded. History of recurrence of the target isSCC or evidence of confounding skin conditions in the treatment area was also exclusionary.
Treatment protocol. Each patient received two sessions of ALA-PDT 28 ± 3 days apart.
Lesion preparation. Prior to ALA application, the lesion was gently abraded using 4×4 gauze to remove scaling or crust.
Application of investigational product. Following lesion preparation, 10% ALA gel was applied topically to the lesion and approximately 5mm of the surrounding area. The treatment area was covered with an occlusive bandage and incubated for 3 hours ± 15 minutes. During this time, patients were instructed to avoid direct exposure to sunlight or other high-intensity light sources.
Irradiation with red light. After the incubation period, PDT was administered using red light at a wavelength of approximately 635nm, delivering a total of 37J/cm2 for 13 minutes and 30 seconds at a distance of 5cm to 8cm. Patients were instructed to avoid direct exposure to sunlight or other high-intensity light sources for approximately 48 hours following treatment.
Assessments. Surgical excision of the lesion site was performed 8 weeks after the second treatment. The excised tissue was examined for complete histological clearance via 1-mm bread loafing by a blinded board-certified dermatopathologist.
The lesion was assessed for size and complete clinical clearance (CCC; no clinically visible lesion at the site) by the investigator at each visit. The lesion site was evaluated for local skin reactions (LSRs) and pigmentation changes by the investigator at each visit. LSRs, such as erythema, flaking/scaling, crusting, swelling, vesiculation/pustulation, and erosion/ulceration, were recorded on a scale of 0 (not present) to 4 (high severity). Pigmentation assessment (hypopigmentation, hyperpigmentation, and depigmentation) was performed on a scale of 0 (none) to 3 (severe).
Patients used a visual analogue scale (VAS) ranging from 0 (no pain) to 10 (worst pain possible) to rate pain within 15 minutes after each ALA application and red light irradiation.
Outcomes. The primary endpoint of this study was the proportion of patients achieving complete histological clearance of isSCC by the end of treatment. The secondary endpoints were the proportion of patients achieving CCC of isSCC, change in size of the treated lesion, and the cosmetic appearance of the treated lesion as assessed by the LSRs.
Results
Twenty patients with a median age of 72 years (range: 56-86 years) and Fitzpatrick skin type of II or III were enrolled. Of these patients, 65% (13) were male and 35% (7) were female. The locations of the isSCCs were as follows: 5 (25%) on the right cheek or temple, 1 (5%) on the right cutaneous lip, 11 (55%) on the forehead, and 3 (15%) on the left cheek or temple. Overall, 55% of lesions were located on the right side of the face, 30% were located on the left side of the face, and 15% were located on the midface. The mean lesion diameter was 0.60cm (range: 0.40-1.1cm; standard deviation [SD]: 0.20cm). The mean lesion width was 0.50cm (range: 0.4-1.0cm; SD: 0.15cm).
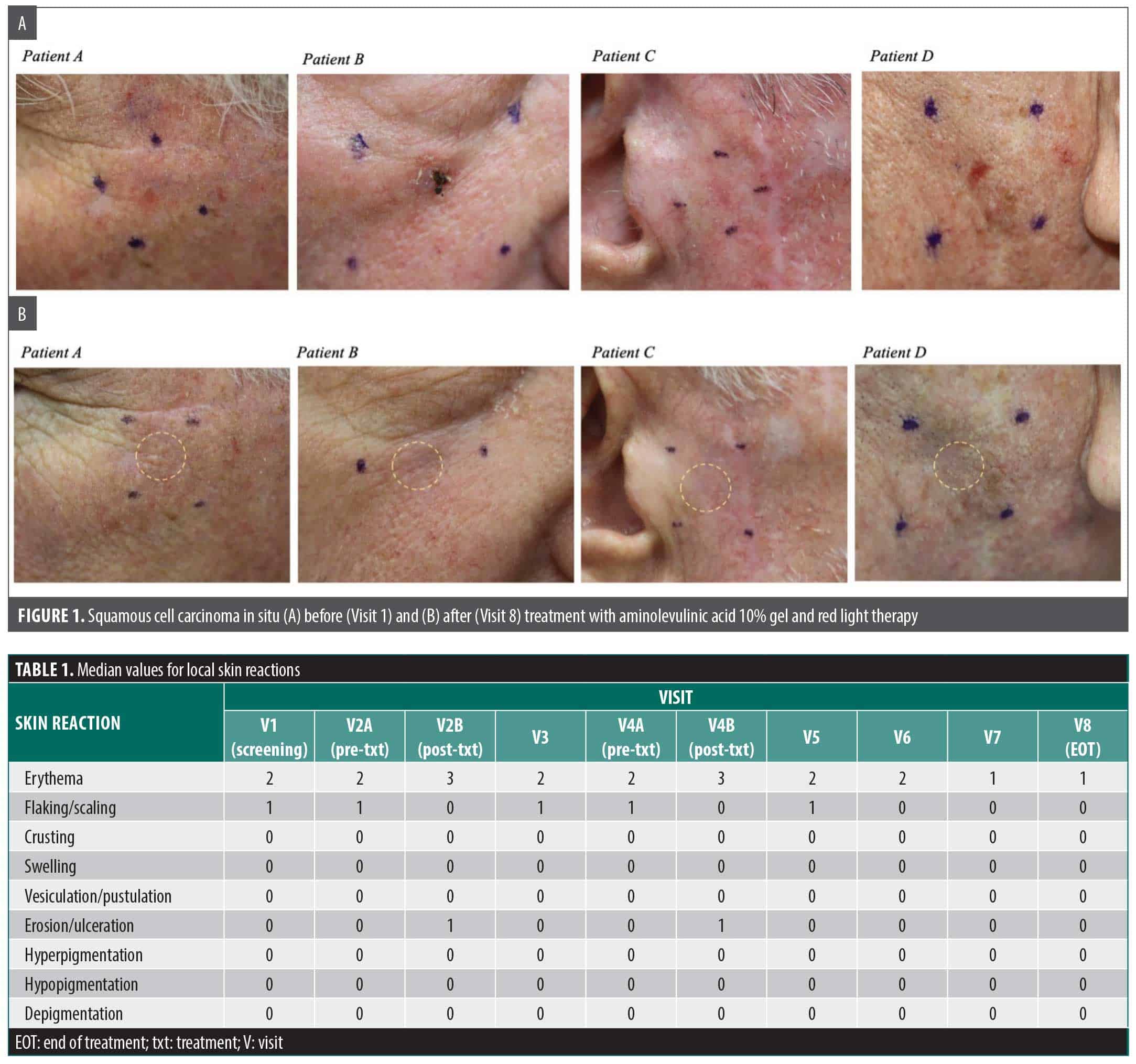
Efficacy. All patients (n=20) achieved complete histological clearance of isSCC by surgical excision or the end of treatment (EOT), which was 8 weeks after the second treatment. CCC was observed in all patients prior to the EOT (Figure 1). The median time until CCC was 62 days (range: 42-90 days; SD: 14 days).
Local skin reactions. Erythema was the most common LSR (n=20), primarily of moderate intensity (Table 1). Mild flaking or scaling was also observed in most patients (n=19). Both erythema and flaking or scaling worsened after treatment and improved over time. The scaling and flaking were resolved in all patients before the EOT. Rarely patients experienced mild erosion (n=1) that was unrelated to treatment and all patients experienced mild erosion at the lesion site during the treatment visit (n=20), which generally resolved by the next visit.
Pigmentation assessment. No patients exhibited any changes in pigmentation, including hyperpigmentation, hypopigmentation, or depigmentation, throughout the study.
Tolerability. The median immediate posttreatment pain score on the VAS was 0 (range: 0 to 3) within 15 minutes after ALA application and 3.5 (range: 0 to 10) within 15 minutes after red light therapy.
Safety. The majority (n=19) of patients did not experience any adverse events. One patient experienced mild contact dermatitis secondary to adhesive use around the surgical site 1 week after excision. This was unrelated to the investigational treatment and resolved within 1 week.
Discussion
ALA-PDT is approved for the treatment of isSCC in the European Union and has successfully been used off label in the US.20 While currently only approved in the US for the treatment of AKs, the efficacy of ALA-PDT for isSCC is well established, and our study adds to the growing literature.14,21
Surgical excision offers one of the highest clearance rates, at approximately 95%, in the treatment of SCC lesions.22 The clearance rate with MMS for SCC lesions is approximately 90% to 95% at the 5-year mark, depending on lesion morphology and history of recurrence.23 A large retrospective study showed up to a 97% clearance of isSCC lesions overall with MMS.24 Of note, this study found that isSCC required more MMS stages than invasive SCC to achieve tumor clearance.24 However, these invasive modalities pose a challenge, especially for individuals in whom surgical intervention may not be suitable, such as high-risk individuals, in cosmetically sensitive areas, or in areas with anatomical complexities.22 Topical chemotherapy agents, such as imiquimod and 5-fluorouracil, were found to have clearance rates of 81% and 78%, respectively.25 The clearance rate of isSCC with a noninvasive modality such as ED&C was approximately 70% in one study.26 Additionally, this treatment modality may not result in favorable cosmetic outcomes. As such, there is a need for a noninvasive treatment for isSCC that offers a favorable cosmesis, safety profile, and comparable clearance rates to surgical intervention. ALA-PDT is a promising treatment modality in these cases.
ALA-PDT has been shown to be most effective with longer incubation periods in treating isSCC. It has also been effective for lesions that are located on the face and are smaller in diameter.21,27 Our findings showed that a relatively short incubation period of 3 hours was effective in treating lesions up to 1.1cm in size. Use of red light at 635nm, which penetrates deeper than blue or shorter wavelength light, may have contributed to the observed higher efficacy, as it allows for greater activation of PpIX in deeper epidermal layers where atypical keratinocytes may reside.18,19 This is particularly relevant for isSCC, which involves the full thickness of the epidermis and requires deeper phototoxic activity for eradication.
Proper lesion preparation is essential for the effectiveness of ALA-PDT. Hyperkeratotic lesions can impair ALA absorption by reducing skin permeability.28 Additionally, increased melanin content acts as a competing chromophore to the light energy, which may diminish the penetration of the light. These factors may compromise the efficacy of ALA-PDT treatment.28 To optimize outcomes, gentle abrasion of the skin to exfoliate the stratum corneum is recommended prior to the ALA-PDT procedure.
ALA-PDT is also promising for field cancerization, which includes areas of skin at risk for both AKs and isSCC. As a field treatment for AKs and actinic damage in general, ALA-PDT demonstrates efficacy for clearance of both precancerous lesions and dysplastic cells or cellular atypia.15 This is particularly important as a recent study showed a significantly higher incidence of SCC (including isSCC) compared to BCC, warranting the need to address precancerous lesions.3 Patients with chronic actinic damage are at a higher risk for the development of skin cancers of any type.29 Thus, patients with biopsy-confirmed isSCC who are treated with ALA-PDT would effectively lower their risk of developing skin cancer in the treatment area.
Another major advantage of our results is the cosmetic appeal that patients seek when treating their cutaneous neoplasms. Traditional treatments, such as ED&C, MMS, or simple excisions, produce scars that patients may find unappealing. This is especially true of lesions on the face. Other treatments, such as superficial radiation therapy, may offer cosmetic appeal but at the cost of additional treatment sessions, which can be mitigated by ALA-PDT.30
Limitations of this study include a small sample size, a short follow-up period, smaller lesion size, and limited anatomical sites. Future studies should assess efficacy on larger lesions, shorter incubation times, and long-term recurrence.
Conclusion
ALA-PDT using red light appears to be a highly effective and well-tolerated treatment for facial isSCC, offering complete histologic and clinical clearance with minimal adverse effects and excellent cosmetic outcomes. These results support further investigation of red light ALA-PDT as an option for isSCC, particularly when surgical treatment poses functional or aesthetic challenges.
References
- Lee MM, Wick MM. Bowen’s disease. Clin Dermatol. 1993;11(1):43-46.
- Que SKT, Zwald FO, Schmults CD. Cutaneous squamous cell carcinoma: incidence, risk factors, diagnosis, and staging. J Am Acad Dermatol. 2018;78(2):237-247.
- Vanaria RJ, Chaudry A, Nestor MS. Nonmelanoma skin cancer in South Florida: a change in the relative incidence of basal and squamous cell carcinoma. J Clin Aesthetic Dermatol. 2025;18(11):12-15.
- Yamaguchi K, Matsunuma R, Kumeta T, et al. Concurrent Bowen’s disease of the nipple and breast cancer. Case Rep Oncol. 2020;13(3):1410-1414.
- Sharma A, Birnie AJ, Bordea C, et al. British Association of Dermatologists guidelines for the management of people with cutaneous squamous cell carcinoma in situ (Bowen disease) 2022. Br J Dermatol. 2023;188(2):186-194.
- Connolly SM, Baker DR, Coldiron BM, et al; Ad Hoc Task Force. AAD/ACMS/ASDSA/ASMS 2012 appropriate use criteria for Mohs micrographic surgery: a report of the American Academy of Dermatology, American College of Mohs Surgery, American Society for Dermatologic Surgery Association, and the American Society for Mohs Surgery. J Am Acad Dermatol. 2012;67(4):531-550.
- Alam M, Armstrong A, Baum C, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78(3):560-578.
- NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Squamous Cell Skin Cancer Version 2.2025. Published February 7, 2025. Accessed July 23, 2025. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1465.
- Allison RR, Moghissi K. Photodynamic therapy (PDT): PDT mechanisms. Clin Endosc. 2013;46(1):24-29.
- Wolff T, Chevinsky J. Behavioral counseling to prevent skin cancer. Am Fam Physician. 2018;98(2):105-106.
- Gold MH. Continuing medical education article-skin treatment: photodynamic therapy: indications and treatment. Aesthet Surg J. 2008;28(5):545-552; quiz 553-555.
- Lee PK, Kloser A. Current methods for photodynamic therapy in the US: comparison of MAL/PDT and ALA/PDT. J Drugs Dermatol. 2013;12(8):925-930.
- Keyal U, Bhatta AK, Zhang G, Wang XL. Present and future perspectives of photodynamic therapy for cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2019;80(3):765-773.
- Nestor MS, Han H, Ceci FM, Lawson A, Gade A. Evaluating the safety and efficacy of aminolevulinic acid 20% topical solution activated by pulsed dye laser and blue light in the treatment of facial cutaneous squamous cell carcinoma in situ. J Cosmet Dermatol. 2023;22(9):2471-2475.
- Nestor MS, Berman B, Patel J, Lawson A. Safety and efficacy of aminolevulinic acid 10% topical gel versus aminolevulinic acid 20% topical solution followed by blue-light photodynamic therapy for the treatment of actinic keratosis on the face and scalp: a randomized, double-blind study. J Clin Aesthetic Dermatol. 2019;12(3):32-38.
- Cohen DK, Lee PK. Photodynamic therapy for nonmelanoma skin cancers. Cancers. 2016;8(10):90.
- Ameluz. Important safety information. Accessed June 2, 2025. https://us.ameluz.com/important-safety-information
- Peng Q, Warloe T, Berg K, et al. 5-aminolevulinic acid-based photodynamic therapy. Clinical research and future challenges. Cancer. 1997;79(12):2282-2308.
- Agostinis P, Berg K, Cengel KA, et al. Photodynamic therapy of cancer: an update. CA Cancer J Clin. 2011;61(4):250-281.
- Zeitouni NC, Bhatia N, Ceilley RI, et al. Photodynamic therapy with 5-aminolevulinic acid 10% gel and red light for the treatment of actinic keratosis, nonmelanoma skin cancers, and acne: current evidence and best practices. J Clin Aesthetic Dermatol. 2021;14(10):E53-E65.
- Nestor MS, Chaudry A, Vanaria RJ, Bhupalam V. Evaluating the safety and efficacy of aminolevulinic acid 20% topical solution activated by blue light in the treatment of facial cutaneous squamous cell carcinoma in situ. J Drugs Dermatol. In Press.
- Moreno-Ramírez D, Silva-Clavería F, Fernández-Orland A, Eiris N, Ruiz de Casas A, Férrandiz L. Surgery for cutaneous squamous cell carcinoma and its limits in advanced disease. Dermatol Pract Concept. 2021;11(suppl 2):e2021167S.
- Rowe DE, Carroll RJ, Day CL Jr. Prognostic factors for local recurrence, metastasis, and survival rates in squamous cell carcinoma of the skin, ear, and lip. Implications for treatment modality selection. J Am Acad Dermatol. 1992;26(6):976-990.
- Kim Y, Khan AH, Papachristou C, Decker A, Lawrence N. Squamous cell carcinoma in situ achieves tumor clearance in more Mohs stages than invasive squamous cell carcinoma. Dermatol Surg. 2023;49(12):1104-1107.
- Petzold A, Wessely A, Steeb T, Berking C, Heppt MV. Efficacy of interventions for cutaneous squamous cell carcinoma in situ (Bowen’s disease): a systematic review and meta analysis of proportions. J Eur Acad Dermatol Venereol. 2025;39(3):543-554.
- Herron E, Hull C, Lobello S, Herron M. 40838 Outcomes of 3400 shave biopsies on the clearance of nonmelanoma skin cancer: benefits and limitations. J Am Acad Dermatol. 2023;89(3 suppl):AB69.
- Kibbi N, Zhang Y, Leffell DJ, Christensen SR. Photodynamic therapy for cutaneous squamous cell carcinoma in situ: impact of anatomic location, tumor diameter, and incubation time on effectiveness. J Am Acad Dermatol. 2020;82(5):1124-1130.
- Matsumoto Y, Akita Y. Safety guidelines for PDT of cutaneous lesions. Laser Ther. 2012;21(2):124-128.
- Guorgis G, Anderson CD, Lyth J, Falk M. Actinic keratosis diagnosis and increased risk of developing skin cancer: a 10-year cohort study of 17,651 patients in Sweden. Acta Derm Venereol. 2020;100(8):adv00128.
- Chaudry A, Vanaria RJ, Nestor MS. Radiation dermatitis: a comparative review of prevention and management. Dermatol Rev. 2025;6(4):e70043.