J Clin Aesthet Dermatol. 2026;19(1):50–55.
by Jacqueline Watchmaker, MD, FAAD, and Diane B. Nelson, RN, MPH
Dr. Watchmaker is with Southwest Skin Specialists in Scottsdale, Arizona. Ms. Nelson is a former employee of skinbetter science, a dermatological beauty brand of L’Oreal USA Inc.
FUNDING: This study was sponsored by skinbetter science, a Dermatological Beauty Brand of L’Oréal USA, Inc.
DISCLOSURES: Dr. Watchmaker was the study investigator, and Ms. Nelson was an employee of skinbetter science, a dermatological beauty brand of L’Oreal USA Inc.
ABSTRACT: Objective: We aimed to evaluate the safety and efficacy of a serum containing plant adaptogens (MYS) compared to a moisturizing lotion (ML) pre- and post-laser treatment. Methods: A double-blind, split-face controlled study enrolled women with mild-to-moderate photodamaged skin. Participants were randomized to twice-daily application of MYS and ML to one side of their face and ML only to the opposite side two weeks prior to and 10 days following a single, non-ablative fractional laser treatment. Tolerability and global skin healing were assessed immediately post-procedure and on Days 1, 2, 4, 7, and 10. Global skin quality (the total sum of scores for erythema, dullness, rough skin texture, pores, and uneven pigmentation) was calculated at baseline, Week 2 (pre-procedure/skincare only), and Day 10 (post-procedure). Results: Fifteen participants completed the study (mean age: 52 years). Significantly less dryness occurred on the MYS vs. the ML side of the face on Days 1 and 2 (p=0.05 and p=0.001, respectively), with less erythema on the MYS vs. the ML side (29% vs. 17%) on Day 2. Significant improvements occurred in global skin healing on the MYS vs. the ML side of the face on Days 1, 2, and 4 (p=0.04, p=0.003, and p=0.003, respectively) and in global skin quality at Week 2 and Day 10 (p=0.03 and p=0.001, respectively). All adverse events were temporary and related to the procedure. Conclusion: Twice-daily application of a serum containing plant adaptogens before and after non-ablative fractional laser treatment demonstrated significant reductions in dryness and erythema, and improvements in global skin healing and skin quality compared to ML. Keywords: Adaptogens, non-ablative fractional laser, skin homeostasis, global skin healing, skin quality, photodamage
Introduction
To reduce signs of photoaging, individuals are increasingly seeking minimally invasive treatment options such as laser resurfacing. According to the most recently published data, more than 3 million minimally invasive skin-resurfacing procedures were performed in 2023, encompassing ablative and non-ablative laser resurfacing, chemical peels, dermabrasion, and microdermabrasion.1
Skin resurfacing options include both ablative and non-ablative laser resurfacing. Ablative lasers vaporize the skin to stimulate collagenesis.2 Consequently, these procedures require many weeks to months for skin to fully recover. In contrast, non-ablative lasers deliver a controlled thermal injury, without vaporizing the skin.3 Fractionated non-ablative lasers create microscopic columns of treated tissue, reducing recovery time and side effects compared with non-fractionated lasers. Treating a fraction of tissue at a time, fractionated lasers leave the surrounding tissue intact to promote rapid healing while stimulating collagen remodeling. While improvements in skin tone, texture, radiance, lines and wrinkles, and acne scarring may be less substantial and require more than one session compared to an ablative procedure,1 non-ablative procedures are associated with less discomfort, less downtime, and reduced risk of side effects when compared to ablative lasers.4-6 After a non-ablative fractional laser treatment, it is common to have a sunburned sensation, erythema, swelling, tightness, itching, dryness/flaking, and the appearance of microscopic epidermal necrotic debris (MENDS), which clinically presents as a sandpapery, coffee-grounds appearance. These events typically resolve in approximately 7 to 10 days. As with ablative laser resurfacing, the condition of a patient’s skin prior to the procedure, the laser settings used, and proper care of the skin following treatment influence recovery time and outcomes.
Along with daily use of sunscreen, optimizing the condition and quality of the skin prior to laser resurfacing procedures with appropriate topical agents helps strengthen the skin barrier and its reparative mechanisms. Effective preconditioning of the skin can reduce the risk of post-procedure complications such as post-inflammatory hyperpigmentation (PIH), erythema, or scarring,7,8 and it may help diminish post-procedure discomfort by minimizing stinging, burning, and itching, thus improving the overall experience for patients.5,9,10 Integrating the use of selective topical agents that holistically address cellular and epidermal homeostasis prior to laser resurfacing helps improve recovery time, and optimizes healing post-procedure. These agents contribute to enhanced skin quality and support skin’s natural adaptive response in the immediate, intermediate, and maintenance post-
procedure phases.5,6,11-15
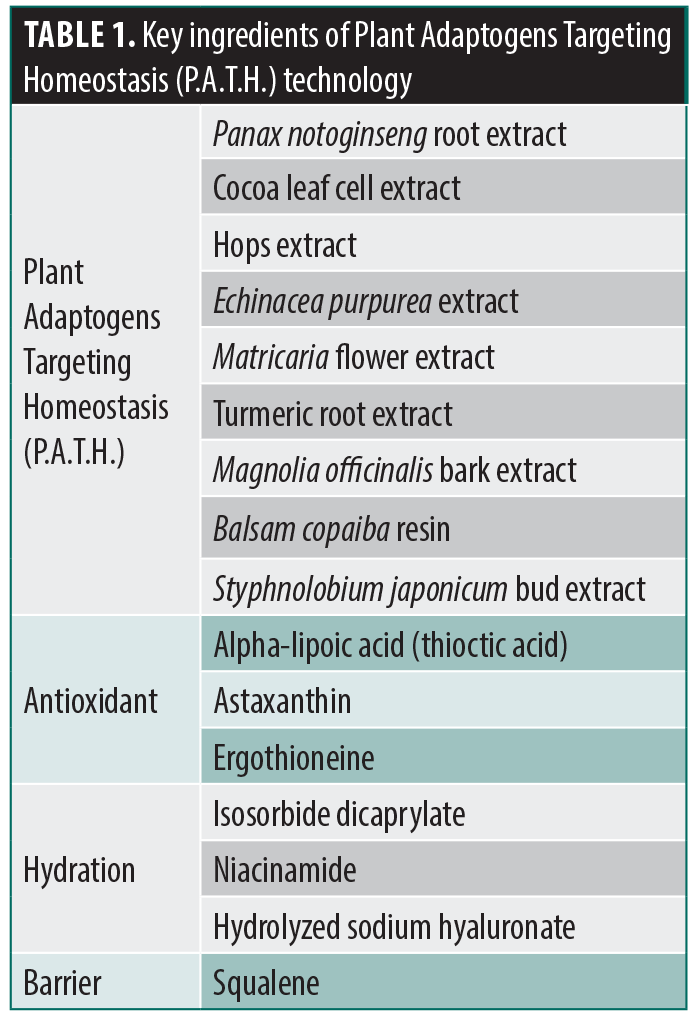
Plant-derived adaptogens act on multiple pathways and molecular targets in the skin and have an innate ability to defend against stressors.13,16-18 Possessing multifunctional properties, adaptogens have the ability to inhibit inflammation, counteract oxidative stress, reinforce skin barrier function, and support aging skin.16-18 A plant-based serum (MYS) composed of plant adaptogens targeting homeostasis (P.A.T.H.) technology leverages the innate capacity of specific botanical extracts to support the skin’s natural adaptive response to stress and achieve homeostasis (Table 1).13 A previous study conducted using an in vitro epidermal model demonstrated changes in the expression of target genes that play important roles in supporting skin’s adaptive response to stress and epidermal homeostasis.19 Mean percent improvements from baseline were demonstrated in the appearance of erythema, skin dullness, skin roughness/texture, and pore size in as early as two weeks. Additionally, a 12-week study evaluated the safety and efficacy of twice-daily application of MYS serum in fifty-three participants with mild-to-severe photodamaged skin, demonstrating significant improvements from baseline in global skin quality.13 A subsequent pilot study highlighted the benefits of applying MYS in combination with a moisturizer two weeks prior to and four weeks post-procedure following either a non-ablative laser procedure or a chemical peel.20
The study reported herein evaluated the efficacy, tolerability, and patient experience of pre- and post-procedure application of MYS serum compared to a basic moisturizing lotion (ML) in patients undergoing a single, non-ablative laser procedure.
Methods
This double-blind, randomized, controlled, split-face trial was conducted under Institutional Review Board approval (Sterling IRB, Atlanta, GA) in conjunction with current Good Clinical Practice (GCP) guidelines. A single dermatology research center recruited female participants through the site’s own efforts, and all signed written, informed consents prior to participation.
The study included female participants who were nonsmokers, aged 36 to 65 years, with Fitzpatrick Skin Types (FST) I to III with mild (3) to moderate (6) photodamaged skin based on a 10-point grading scale (0=none; 7-9=severe), presenting with at least two of the following signs of visible photoaging: erythema, prominent or enlarged pore size, dull or sallow skin tone, rough skin texture, and uneven pigmentation.
Participants with a history of cold sores were eligible if they agreed to prophylactic treatment with twice-daily valacyclovir (500mg) for 5 days beginning the day of the procedure. In addition, participants were deemed eligible for inclusion in the study following a 2-week washout period of topical skincare products containing alpha hydroxy acids (AHA), beta hydroxy acids (BHA), antioxidants (including adaptogens), vitamin C, peptides, growth factors, prescription and nonprescription retinoids, and hydroquinone. Participants who had received cosmetic treatments, such as injectable neuromodulators or dermal fillers, or who had participated in other facial cosmetic product or procedure-related studies were eligible after a 3-month washout period. Participants who had undergone microneedling, microdermabrasion, chemical peels, or similar procedures in the previous 6 months were excluded from study participation, as were patients who had undergone energy-based procedures or used isotretinoin in the previous 12 months.
Participants were randomly assigned to two groups: Group 1 applied MYS and ML to the left side of their face and ML only to the opposite side, and Group 2 applied MYS and ML to the right side of their face and ML only to the opposite side. All participants were provided with clear, simple, written instructions regarding the application of the study products and were instructed to apply the study products to their face twice daily for 2 weeks prior to their scheduled procedure. MYS was applied first and lightly massaged into the skin until completely absorbed before applying ML. Participants were instructed to wash their hands between product application to assist in maintaining the integrity of the split-face study design. Participants were also instructed to continue applying products as assigned post-procedure and were allowed up to two additional applications of ML as needed. All study products were provided in the same non-branded, white opaque bottles to ensure blinding of the investigator and participants. Participants’ pre-procedure skincare regimen also included the use of a mild gel cleanser used in the morning and evening, along with daily use of a mineral-based sunscreen (SPF 50). All participants received a single, non-ablative laser procedure (Fraxel, 1927nm, 10mJ, 30–40%, 8 passes) performed by the study investigator.
Digital photography was captured using Canfield VISIA-CR (Canfield Scientific Inc.) at baseline, Day 0/pre-procedure/skincare only, Day 0/post-procedure (prior to the study product application), and Days 1, 2, 4, 7, and 10/post-procedure. Investigator assessment of photodamage using a 10-point grading scale (0=none; 1-3= mild; 4-6=moderate; 7-9=severe) occurred at baseline, Day 0/pre-procedure/skincare only, and Day 10. Investigator assessment of skin quality parameters were based on the use of a 6-point grading scale (0=none; 5=very severe) for erythema, skin texture (visible roughness), skin dullness, pore size, and uneven pigmentation at baseline, Day 0/pre-procedure/skincare only, and Day 10. Investigator assessment of skin healing was based on the use of a 5-point grading scale (Global Assessment of Skin Healing [0=Poor to 4=Excellent]) that evaluated erythema, edema, dryness/flaking, and MENDS on Days 1, 2, 4, 7 and 10. Investigator assessment of erythema, edema, and dryness/flaking was based on the use of a 4-point grading scale (0=none; 3=severe) on Day 0/post-procedure, and Days 1, 2, 4, 7, and 10.
Participants rated severity of stinging/tingling, itching, warmth, tightness, redness, and dryness/flaking using a 4-point grading scale (0=none; 3=severe) on Day 0/immediately post-procedure (prior to application of the study products) and on Days 1, 2, 4, 7, and 10. Participant satisfaction questionnaires were completed on Day 0/pre-procedure/skincare only and on post-procedure Days 1, 2, 4, 7, and 10. Collection and evaluation of adverse events (AEs) occurred throughout the study.
Results
Fifteen participants met eligibility criteria and were randomized to either Group 1 (n=8) or Group 2 (n=7). All participants were female, with a moderate amount of photodamage and a mean age of 52 years (range: 36 to 65 years). The majority of participants were White (87%) and non-Hispanic (87%), consisting of FST I, II, and III (7%, 80%, and 13%, respectively).
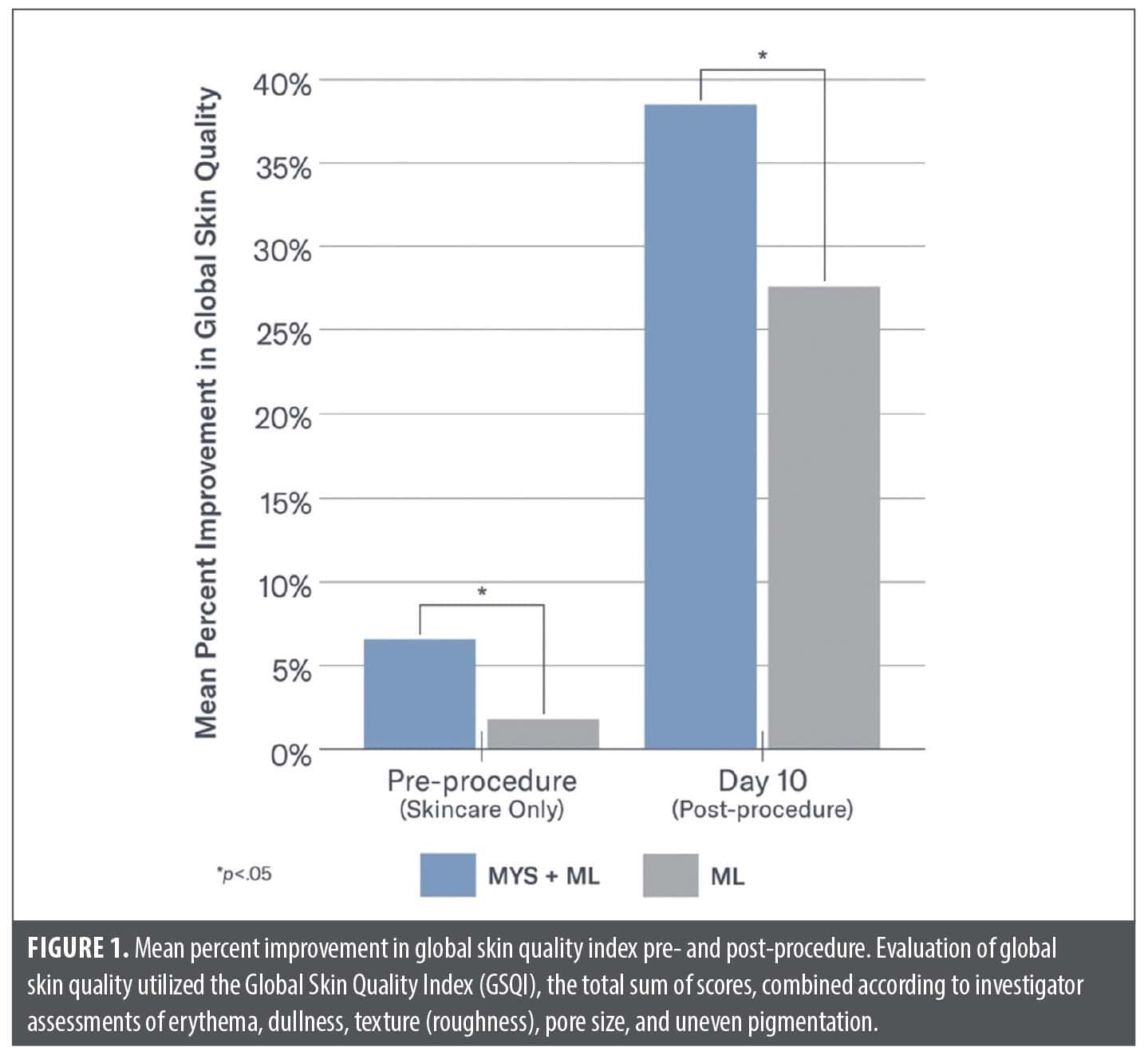
Investigator evaluations. Significant improvements in global skin quality based on the Global Skin Quality Index (GSQI) were demonstrated on the side of the face treated with MYS/ML vs. ML at Day 0 (pre-procedure/ skincare only) (7% vs. 2%; p=0.03) and Day 10 (38% vs. 27%; p=0.001) (Figure 1). Significant improvements were demonstrated on the side of the face treated with MYS/ML vs. ML on Day 10 in skin dullness (42% vs. 27%; p=0.03) and pores (32% vs. 17%; p=0.02).
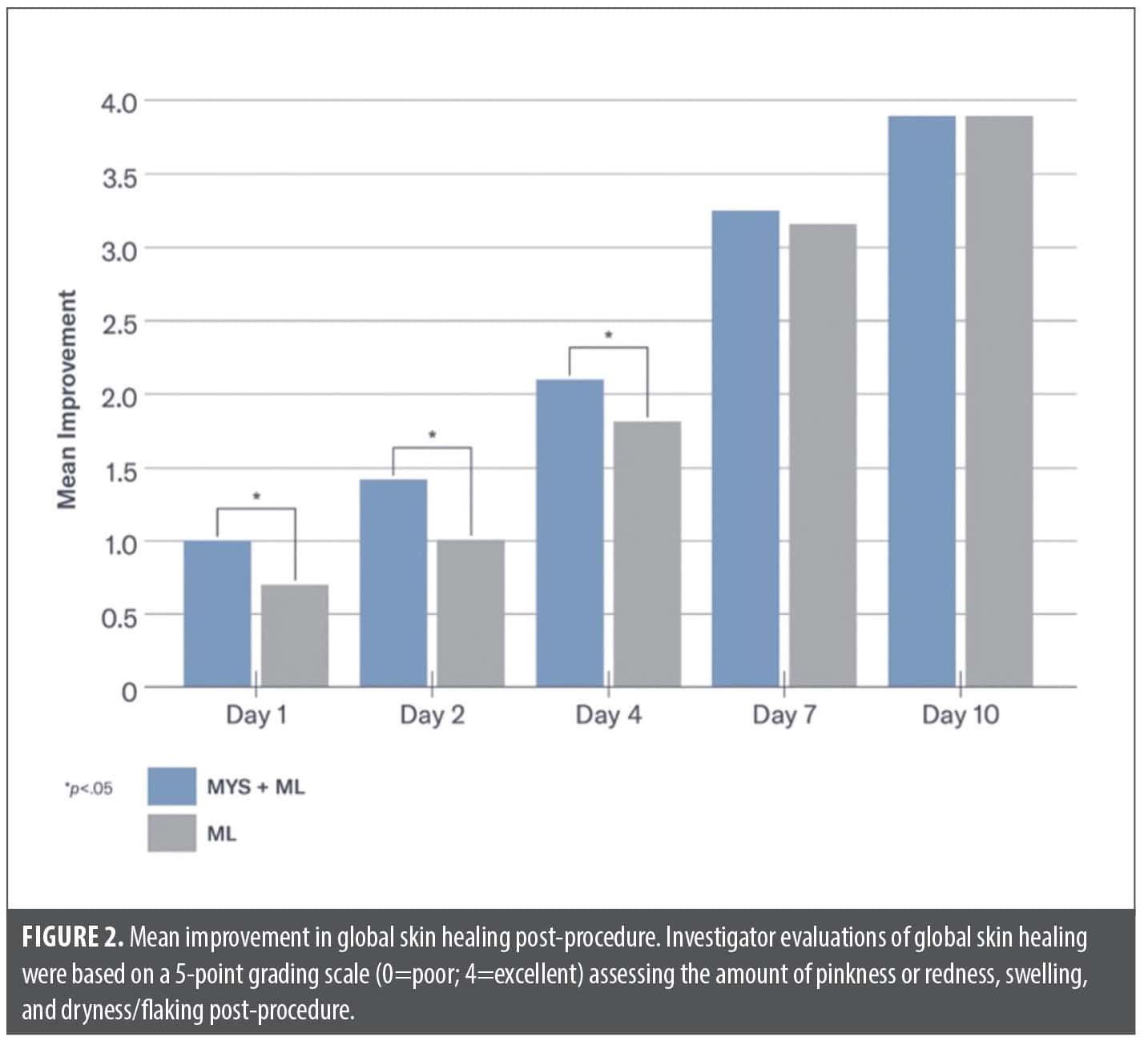
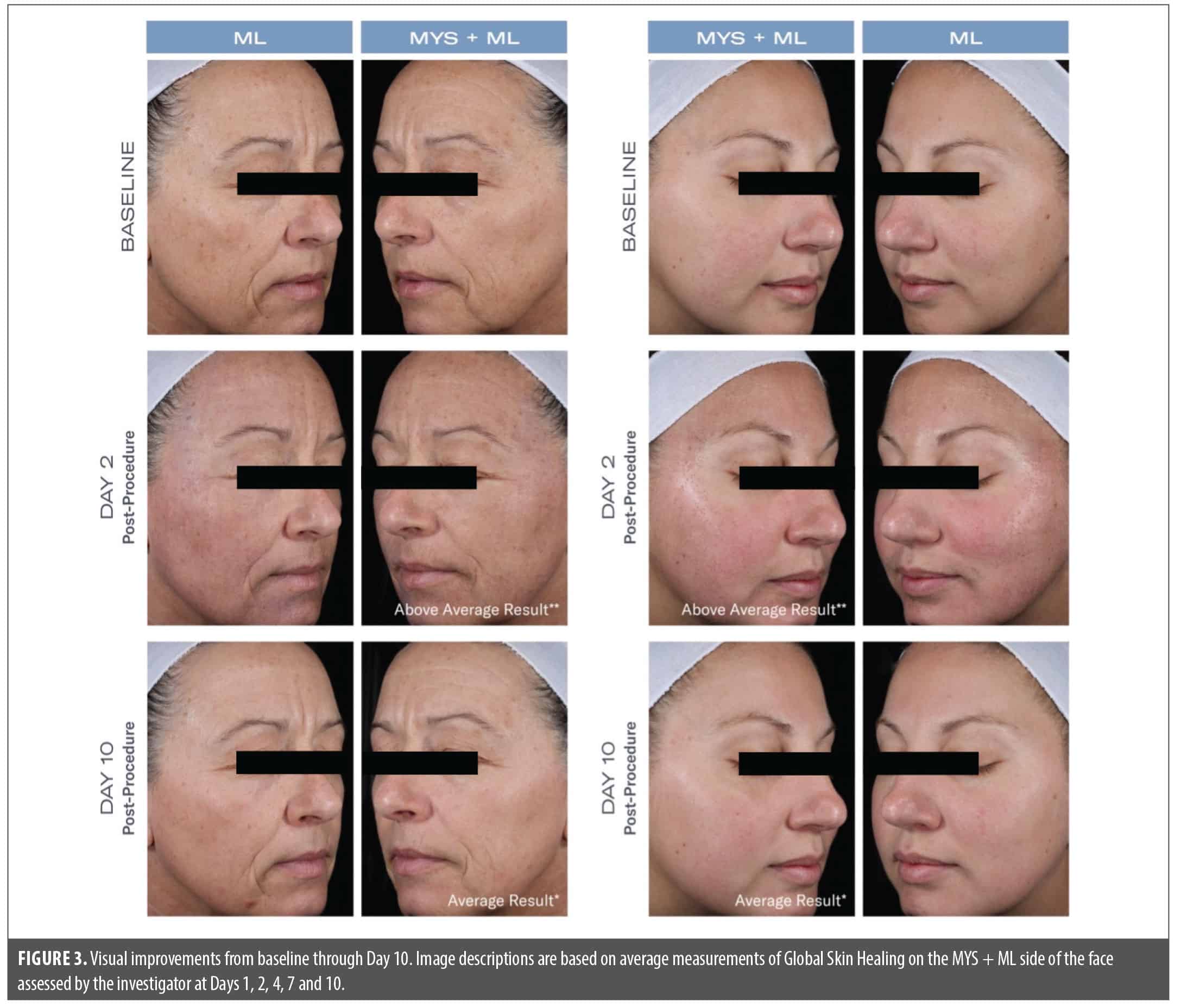
Post-procedure, significantly less edema was seen on the side of the face treated with MYS/ML vs. ML on Day 1 (–8% vs. +21%; p=0.01) and Day 2 (–22% vs. –5%; p=0.05). Significantly less dryness was demonstrated on the side of the face treated with MYS/ML vs. ML on Day 1 (1.67 vs. 2.0; p=0.05) and Day 2 (1.73 vs. 2.27; p=0.001). Earlier reductions occurred on Day 2 in erythema on the side of the face treated with MYS/ML vs. the side treated with ML (–29% and –17%, respectively). Significant improvements in global skin healing were demonstrated on the side of the face treated with MYS /ML vs. ML, post-procedure on Day 1 (p=0.04), and Days 2 and 4 (p=0.003) (Figures 2 and 3).
Patient tolerability and satisfaction. Patients reported significant reductions in dryness/flaking on the side of their face treated with MYS/ML vs. ML on Day 4 (p=0.04)
Preconditioning skin with MYS/ML for 2 weeks prior to undergoing their laser procedure led to 80% of participants reporting that the skin on the side of their face to which MYS/ML was applied was more radiant, healthy looking, and hydrated compared to the opposite side of their face in which only ML was used. Furthermore, 73% of participants reported that the overall quality of their facial skin improved more on the side of their face to which MYS/ML was applied.
Additionally, 80% of participants reported that the skin on the side of their face to which MYS/ML was applied was brighter and more luminous on Day 4 post-procedure, with less flaking and peeling experienced on Day 7. The majority of participants reported greater satisfaction with their appearance and procedurally related side effects on the side of their face treated with MYS/ML at every timepoint through Day 10. Participants also noted that MYS absorbed quickly (pre-procedure), with less stinging (post-procedure), and felt hydrating.
Four mild AEs were reported post-procedure: acne (n=2), nausea (n=1), and watery eyes (n=1). All events were deemed unrelated to the study product and resolved without sequelae.
Discussion
The popularity of minimally invasive aesthetic procedures, including non-ablative fractional laser resurfacing, can be attributed to less downtime compared to more aggressive ablative procedures. Despite reduced healing time and severity of side effects, some downtime is anticipated, as various levels of erythema, swelling, dryness, and peeling does occur depending on the condition of the skin, energy levels, and settings used during treatment, which may deter some patients from seeking treatment. Optimizing the condition of the skin prior to treatment by integrating selective topical skincare products that can reduce inflammation and oxidative stress, reinforce skin barrier function, and support the skin’s resistance to negative stressors can improve healing time and contribute to optimal outcomes.5,9 Ideally, treatment strategies should involve a streamlined, uncomplicated approach and include products that are aesthetically pleasing and safe and effective for use pre-procedure, immediately post-procedure, and long term.
Previous studies have examined the benefits of pretreating skin with petrolatum-based, silicone-based, or other occlusive agents prior to fractional laser resurfacing procedures to expedite wound healing. While these products reduce transepidermal water loss (TEWL), they are generally inelegant to use, considered to be sticky, leave a shiny film on the skin, and may cause breakouts.10,21-24
Plant-derived adaptogens have an innate ability to protect against environmental and other insults. Adaptogenic compounds act on multiple pathways and molecular targets and have multifunctional properties that support homeostasis in the skin.13,16-18 Consequently, plant-based adaptogens have been incorporated into skincare formulations for both the management of dermatologic conditions, including atopic dermatitis, psoriasis, and wound healing, as well as for support of aging skin.14
The active product used in this study (MYS serum) is a plant-based adaptogenic compound that has been previously investigated in both in vitro and in vivo studies.13,19 Using an epidermal model, the application of MYS Serum facilitated changes in the expression of target genes that are key to supporting the skin’s ability to respond to stress and achieve homeostasis.19 Results from a clinical trial reported significant improvements from baseline in the appearance of erythema, skin dullness, skin texture, pore size, and global skin quality as early as Week 2, with significant mean percent improvement in uneven pigmentation achieved by Week 8.13 In addition, a pilot study demonstrated the safety and efficacy of MYS Serum in combination with an emollient-rich moisturizing cream pre- and post-laser procedure or chemical peel, including significant improvements from baseline in global skin quality at both 2 weeks (pre-procedure/skincare only) and 4 weeks post-procedure for each treatment.20 Participants in both treatment groups reported positive benefits of the skincare regimen, noting that it improved their overall experience.
Results from the current controlled study demonstrated significant improvements in global skin quality after pretreatment with MYS/ML, leading to significantly less edema and dryness on Days 1 and 2 post-procedure on the side of the face treated with MYS/ML vs. ML. Pretreatment with MYS/ML also facilitated significant post-procedure improvements in global skin healing, and the majority of participants reported greater satisfaction with the procedure itself as well as the immediate cosmetic results compared with the use of ML alone. These findings highlight both the preconditioning and reparative benefits associated with the adaptogens in this unique serum, helping to reinforce skin barrier function while optimizing post-procedure healing. Future studies are warranted to evaluate long-term safety and efficacy outcomes in a larger and diverse population.
Conclusion
Integrating the use of a plant-based adaptogen serum before and after non-ablative fractional laser demonstrated significant mean improvements in global skin quality vs. a basic moisturizer both pre- and post-procedure. Additionally, significant improvements in global skin healing occurred on Days 1, 2, and 4 post-procedure on the side of the face treated with the serum, leading to higher levels of patient satisfaction in procedure related side effects and the overall quality of their skin.
References
- American Society of Plastic Surgeons. 2023. Accessed April 8, 2025. https://www.plasticsurgery.org/documents/news/statistics/2023/plastic-surgery-statistics-report-2023.pdf
- de Vries K, Prens EP. Laser treatment and its implications for photodamaged skin and actinic keratosis. Curr Probl Dermatol. 2015;46:129-135.
- Alexaiades-Armenakas MR, Dover JS, Arndt KA. The spectrum of laser skin resurfacing: non-ablative, fractional, and ablative laser resurfacing. J Am Acad Dermatol. 2008;58(5):719-737.
- Friedman PM, Polder KD, Sodha P, Geronemus RG. The 1440 nm and 1927 nm non-ablative fractional diode laser: current trends and future directions. J Drugs Dermatol. 2020;19(8):s3-s11.
- Farris PK, Lain T. Peri-procedural skincare: a look at the evidence. Pract Dermatol. 2022;(Sept):64-65.
- Robinson DM, Frulla AP. Randomized, split-face/décolleté comparative trial of procedure enhancement system for fractional non-ablative laser resurfacing treatment. J Drugs Dermatol. 2017;16(7):707-710.
- Hrabovsky SL. Preoperative and postoperative skin care with laser resurfacing. Semin Ophthalmol. 1998;13(3):114-122.
- Lowe NJ, Lask G, Griffin ME. Laser skin resurfacing. Pre- and posttreatment guidelines. Dermatol Surg. 1995;21(12):1017-1019.
- Amici JM, Cogrel O, Jourdan M, et al. Expert recommendations on supportive skin care for nonsurgical and surgical procedures. J Eur Acad Dermatol Venereol. 2023;37(Suppl 3):16-33.
- Angra K, Lipp MB, Sekhon S, Wu DC, Goldman MP. Review of post-laser-resurfacing topical agents for improved healing and cosmesis. J Clin Aesthet Dermatol. 2021;14(8):24-32.
- Potekaev NN, Borzykh OB, Medvedev GV, et al. The role of extracellular matrix in skin wound healing. J Clin Med. 2021;10(24):5947.
- Casas LA, Bell M, Claytor B, Ziegler ME, Widgerow AD. An analysis of patient-reported recovery outcomes of topical tripeptide/hexapeptide formulations utilized in a prospective randomized double-blind split neck and body study. Aesthet Surg J Open Forum. 2020;3(1):ojaa052.
- Draelos ZD, Grimes PE, Watchmaker J, Nelson DE. A multi-center trial evaluating a serum comprised of plant-based adaptogens targeting skin quality. J Clin Aesthet Dermatol. 2024;17(2):15-19.
- Kim J, Kim J, Lee YI, Almurayshid A, Jung JY, Lee JH. Effect of a topical antioxidant serum containing vitamin C, vitamin E, and ferulic acid after Q-switched 1064-nm Nd:YAG laser for treatment of environment-induced skin pigmentation. J Cosmet Dermatol. 2020;19(10):2576-2582.
- Ud-Din S, Bayat A. Controlling inflammation pre-emptively or at the time of cutaneous injury optimizes outcome of skin scarring. Front Immunol. 2022;13:883239.
- Panossian AG, Efferth T, Shikov AN, et al. Evolution of the adaptogenic concept from traditional use to medical systems: pharmacology of stress- and aging-related diseases. Med Res Rev. 2021;41(1):630-703.
- Pomatto LCD, Davies KJA. The role of declining adaptive homeostasis in ageing. J Physiol. 2017;595(24):7275-7309.
- Ray A, Gulati K, Anand R. Stress, adaptogens and their evaluation: an overview. J Pharma Reports. 2016;1(2):1000110.
- Draelos ZD, Grimes PE, Watchmaker J, Nelson DE. Gene expression analysis of a topical serum comprised of plant-based adaptogens developed to support homeostasis and skin quality. J Clin Aesthet Dermatol. 2024;17(2):43-46.
- Watchmaker J, Nelson DB. Compatibility of an integrated skincare regimen comprised of a serum containing plant adaptogens and a cream addressing three natural components of moisturization pre-and post-procedure. Abstract Presented at SCALE 2024. J Clin Aesthet Dermatol. 2024;17:9(Suppl1):24.
- Leight-Dunn H, Chima M, Hoss E. Wound healing treatments after ablative laser skin resurfacing: a review. J Drugs Dermatol. 2020;19(11):1050-1055.
- Mortensen JT, Bjerring P, Cramers M. Locobase repair cream following CO2 laser skin resurfacing reduces interstitial fluid oozing.J Cosmet Laser Ther. 2001;3(1):155-158.
- Murakami Y, Saya Y, Morita E, Matsunaka H. Novel petrolatum-based ointment that is highly moisturizing and has superior usability with increased adherence in patients with facial dry skin. J Cosmet Dermatol. 2020;19(10):2650-2655.
- Torres A, Rego L, Martins MS, et al. How to promote skin repair? In-depth look at pharmaceutical and cosmetic strategies. Pharmaceuticals (Basel). 2021;16(4):573.