 by Gabriele F. Muti, MD, and Matteo Basso, MD
by Gabriele F. Muti, MD, and Matteo Basso, MD
Dr. Muti is with the Department of Plastic Surgery, European Dermatological Institute, in Milan, Italy. Dr. Basso is with the Studio Dermatologico in Rome, Italy.
Funding: No funding was provided for this study.
Disclosures: Dr. Muti has been an investigator and speaker for Merz Pharmaceuticals GmbH and Merz Pharma Italy. Dr. Basso has no conflicts of interest relevant to the content of this article.
Keywords: Lateral periorbital line, crow’s feet, incobotulinumtoxinA
Abstract: Background. IncobotulinumtoxinA is a botulinum neurotoxin type A that is free from complexing proteins and is used in various therapeutic indications and aesthetic medicine. It is approved for the treatment of glabellar frown lines in the United States. In Europe, it is also approved for the treatment of lateral periorbital lines (crow’s feet) and for the combined treatment of upper facial lines, including glabellar frown lines, crow’s feet, and horizontal forehead lines.
Methods. In the present study, incobotulinumtoxinA was injected at two different dilutions to treat female subjects aged 40 to 50 years who had moderate-to-severe lateral periorbital lines at maximum contraction according to a score of 2 or 3 points on the 5-point Merz Aesthetics Scales (MAS). For Group 1 (n=20), 50U of incobotulinumtoxinA were reconstituted with 1.60mL of 0.9% NaCl, and for Group 2 (n=20), a reconstitution volume of 0.55mL was used.
Results. Merz Aesthetics Scales scores were markedly improved by at least one point in both groups at one month and three months. The mean Merz Aesthetics Scales scores at one month were 0.4 and 0.6 points for Group 1 and Group 2, respectively, corresponding to a mean improvement of 2.0 and 1.8 points compared with baseline, respectively. conclusion: No significant differences in efficacy and tolerability of incobotulinumtoxinA were seen between the two dilutions at any time point.
J Clin Aesthet Dermatol. 2017;10(9):27–29
Introduction
Botulinum neurotoxin is a product of anaerobic fermentation of the bacterium Clostridium botulinum. It inhibits acetylcholine release, causing a complete blockade of cholinergic transmission at the neuromuscular junction, resulting in temporary muscle relaxation.1 In 1989, a botulinum neurotoxin was first approved for the treatment of blepharospasm, hemifacial spasm, and strabismus.2 Neurotoxin serotype A preparations have been approved for aesthetic use and are the most popular of all cosmetic procedures worldwide.3
IncobotulinumtoxinA (Xeomin®/Xeomeen®/ Bocouture®/XEOMIN Cosmetic TM, NT 201; Merz Pharmaceuticals GmbH, Frankfurt am Main, Germany), unlike the other major botulinum toxin formulations used in aesthetics [abobotulinumtoxinA (Dysport®; Ipsen, UK/Azzalure®; Galderma, Switzerland) and onabotulinumtoxinA (Vistabel®, Vistabex®, Botox® Cosmetic; Allergan Inc., Irvine, CA)], is free from complexing proteins, thus containing only active neurotoxin, which reduces the risk of potential immunogenicity and subsequent treatment failure.4 In various clinical studies, incobotulinumtoxinA has been shown to be equally effective and safe as onabotulinumtoxinA in reducing glabellar frown lines5,6 and lateral periorbital lines.7–9 A recent study reported similar efficacy of incobotulinumtoxinA in comparison to abobotulinumtoxinA at a dose conversion ratio of 1:3 for treating periorbital lines.10 A non-interventional study conducted in Germany, France, and the United Kingdom documented very high treatment satisfaction with incobotulinumtoxinA among physicians and patients.11
In 2014, incobotulinumtoxinA was approved in Europe for the treatment of lateral periorbital lines at a recommended dose of 12U per eye area.12 The aim of the present study was to compare the efficacy of two different dilutions of incobotulinumtoxinA in treating lateral periorbital lines (i.e., crow’s feet).
Patients and Methods
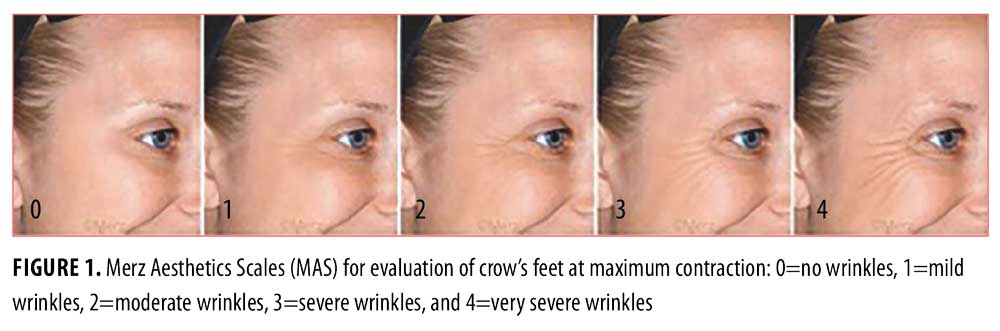
This randomized, double-blind, two-arm study was conducted at two private practices in Milan and Rome, Italy, between January 2015 and June 2015 and complied with the ethical principles of the Declaration of Helsinki. Patients gave informed consent to participate in the study as well as photoconsent to have their photos published. The study included 40 healthy female subjects aged 40 to 50 years with moderate-to-severe lateral periorbital lines at maximum contraction (2–3 points on the validated 5-point Merz Aesthetics Scales [MAS], where 0=no wrinkles, 1=mild wrinkles, 2=moderate wrinkles, 3=severe wrinkles, and 4=very severe wrinkles; Figure 1). The subjects were randomized to two groups, each with 20 subjects and a mean MAS score of 2.4 points at baseline. The periorbital lines on the right and left side of each subject’s face were quite symmetrical (2–3 points).
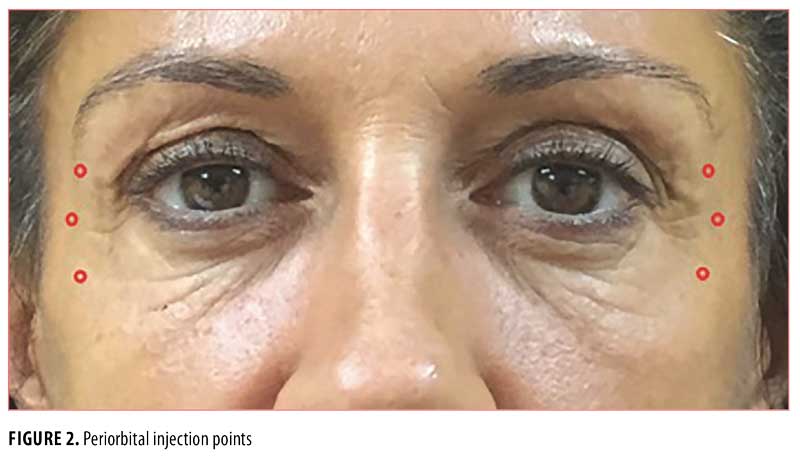
Both treatment groups were injected 1cm lateral from the bony orbital rim at three injection sites per side with 4U of incobotulinumtoxinA per injection (i.e., 12U per side; Figure 2). For Group 1, incobotulinumtoxinA (50U vial) was reconstituted with 1.60mL of 0.9% NaCl and 0.128mL (4U) injected into each injection point, while Group 2 was treated with incobotulinumtoxinA reconstituted in 0.55mL of 0.9% NaCl and injected at a volume of 0.044mL (4U) per injection point. The incobotulinumtoxinA concentrations for Group 1 and Group 2 were 31.25U/mL and 90.91U/mL, respectively.
The assessment on the 5-point MAS at maximum contraction was used for blinded cross-evaluation of the subjects by the two investigators at baseline, one month, and three months after injections. The difference between the two groups was analyzed by one-way ANOVA and statistical significance set at p<0.001 (StatPlus:Mac).
Results
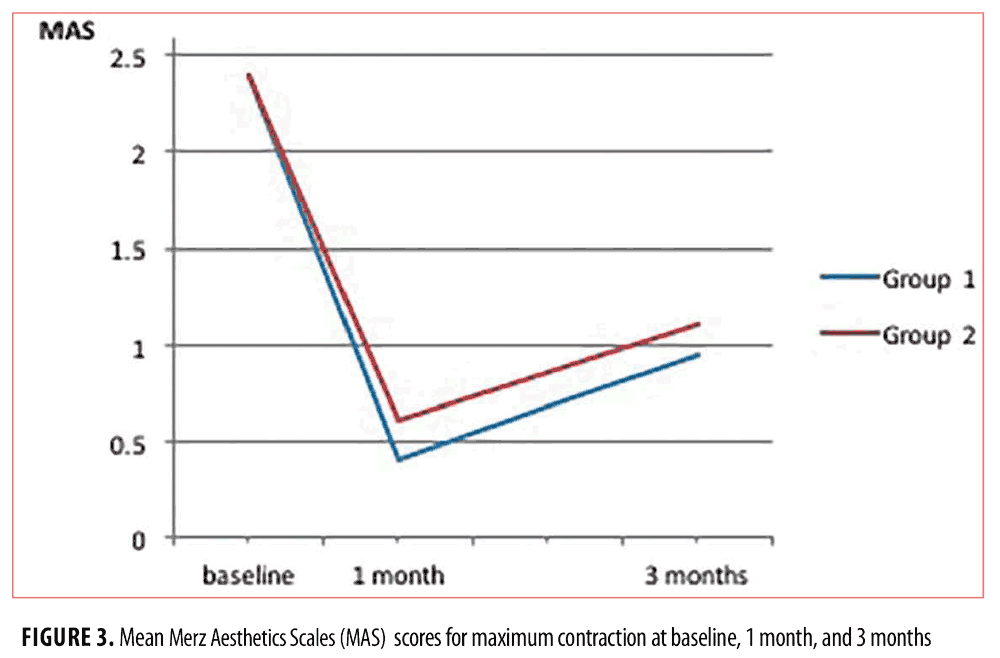
The mean severity score at maximum contraction, as assessed on the MAS at baseline, was 2.4 points for both groups (range: 2–3 points). At one month after injection, mean scores were 0.4 points in Group 1 (range: 0–1 point) and 0.6 points in Group 2 (range: 0–2 points), corresponding to an improvement in the MAS scores of 2.0 and 1.8 points, respectively. At three months, the mean MAS score at maximum contraction was 0.95 points in Group 1 (range: 0–2 points) and 1.1 points in Group 2 (range: 0–3 points). Figure 3 shows the mean scores for both groups. The mean scores of the two groups differed by 0.2 points at 1 month and by 0.15 points at 3 months. These differences were not statistically significant (p=0.00454) and can be considered as not clinically relevant.
With the exception of one case in Group 2 at three months, all individual MAS scores at one and three months had an improvement of at least one point compared with baseline.
No adverse reactions were reported in either group during the study period, and incobotulinumtoxinA was well tolerated.
Discussion
In the present study, two different dilutions of incobotulinumtoxinA were compared for their efficacy in treating lateral periorbital lines. On the one hand, less diluted incobotulinumtoxinA enables physicians to target certain muscles more precisely, and on the other hand, a higher dilution might lead to more homogeneous results due to the larger area of effect.
Both dilutions of incobotulinumtoxinA were equally effective in reducing periorbital lines as assessed by the blinded evaluator at maximum contraction. No significant differences in MAS scores were seen between the two dilution groups at any time point. Possibly the release of acetylcholine was completely blocked at both dilutions, and differences might be seen with fewer units of incobotulinumtoxinA, but not at the labeled dose of 12U per side as used in this study.
Subjects who were injected with a total of 25U incobotulinumtoxinA at five injection points for glabellar frown lines did not show any significant differences at two different dilutions (40U/mL and 25U/mL).13 Glabellar frown lines were also equally reduced by four different dilutions (100, 33.3, 20, and 10U/mL) of 30U onabotulinumtoxinA at seven injection points.14 In a study of lateral periorbital lines, single injections of 5U onabotulinumtoxinA at a concentration of 100U/mL showed no significant differences to a dilution of 20U/mL.15
Further factors affecting spreading of toxin must be considered, such as injection accuracy and depth of injection.16 In a study on different doses, dilutions, and depths of injection of abobotulinumtoxinA in patients with compensatory hyperhidrosis, the diameters of the fields of anhidrotic effects on the patients’ backs were not markedly affected by the different dilutions (250, 125, 83.3, and 62.5U/mL) or the depths of injection (1, 2, 3, and 4mm), but a double dose of abobotulinumtoxinA (10U vs. 5U) generated significantly larger diameters of anhidrotic effects.17 Taken together, the present study confirms previous findings that different concentrations of toxin solution lead to similar results, and that the key efficacy parameter is the number of units injected in the particular area.
Strengths of this study were the blinded cross-evaluation to avoid bias and the use of the validated MAS assessment scale for lateral periorbital lines. A potential limitation was the comparatively small number of subjects. Although more studies on the effects of dilution are warranted, the choice between a holistic approach to facial rejuvenation with higher dilutions or precise administration of treatment with lower dilutions is a promising prospect in the context of individualized treatment.
Conclusion
In the present study, both dilutions of incobotulinumtoxinA were equally effective in reducing lateral periorbital lines with 12 units per eye area. Both solutions were well-tolerated, and no adverse reactions were reported for either dilution. Thus, a vial of 50U incobotulinumtoxinA may be reconstituted in 1.60mL or smaller volumes, such as 0.55mL (reconstitution volume according to SmPC:1.25 mL), allowing aesthetic physicians to choose between higher dilutions aimed at smooth treatment effects or lower dilutions for precise treatment administration while obtaining consistent results at maximum contraction.
References
- Brin MF. Botulinum toxin: chemistry, pharmacology, toxicity, and immunology. Muscle Nerve Suppl. 1997;6:S146–S168.
- Frevert J. Pharmaceutical, biological, and clinical properties of botulinum neurotoxin type A products. Drugs R D. 2015;15(1):1–9.
- International Society of Aesthetic Plastic Surgeons. ISAPS International Survey on Aesthetic/Cosmetic Procedures. Performed in 2013. https://www.isaps.org/Media/Default/ global-statistics/2014%20ISAPS%20Results% 20(3).pdf. Accessed 1 Sep 2017.
- Torres S, Hamilton M, Sanches E, et al. Neutralizing antibodies to botulinum neurotoxin
- type A in aesthetic medicine: five case reports. Clin Cosmet Investig Dermatol. 2014;7:11–17.
- Sattler G, Callander MJ, Grablowitz D, et al. Noninferiority of incobotulinumtoxinA, free from complexing proteins, compared with another botulinum toxin type A in the treatment of glabellar frown lines. Dermatol Surg. 2010;36:1–9.
- Kane MAC, Gold MH, Coleman WP, et al. A randomized, double-blind trial to investigate the equivalence of incobotulinumtoxinA and onabotulinumtoxinA for glabellar frown lines. Dermatol Surg. 2015;41:1310–1319.
- Muti G, Harrington L. A prospective rater- and subject-blinded study comparing the efficacy of incobotulinumtoxina and onabotulinumtoxina to treat crow’s feet: a clinical crossover evaluation. Dermatol Surg. 2015;41:s39–s46.
- Prager W, Wissmüller E, Kollhorst B, et al. Comparison of two botulinum toxin type A preparations for treating crow’s feet: a split-face, double-blind, proof-of-concept study. Dermatol Surg. 2010;36:2155–2160.
- Lee JH, Park JH, Lee SK, et al. The efficacy and safety of incobotulinum toxin A in periocular rhytides and masseteric hypertrophy: side by side comparison with onabotulinum toxin A. J Dermatolog Treat. 2014;25(4):326–330.
- Saybel A, Artemenko A, Nikitin S, et al. A prospective, neurophysiologic comparative study to assess the efficacy and duration of effect of incobotulinumtoxinA and abobotulinumtoxinA in the treatment of crow’s feet. J Drugs Dermatol. 2015;14(11):1291–1296.
- Pavicic T, Prager W, Klöppel M, et al. IncobotulinumtoxinA use in aesthetic indications in daily practice: a European multicenter, noninterventional, retrospective study. Clin Cosmet Investig Dermatol. 2015;8:135–142.
- Bocouture [summary of product characteristics]. Merz Pharmaceuticals GmbH, Frankfurt, Germany, 2016.
- Prager W, Zschocke I, Reich C, et al. Beeinflusst die Verdünnung das kosmetische Ergebnis von BoNT/A? [Does the dilution influence the cosmetic result of BoNT/A?]. Hautarzt. 2009;60:815–820.
- Carruthers A, Carruthers J, Cohen J. Dilution volume of botulinum toxin type A for the treatment of glabellar rhytides: does it matter? Dermatol Surg. 2007;33:S97–S104.
- Carruthers A, Bogle M, Carruthers JD, et al. A randomized, evaluator-blinded, two-center study of the safety and effect of volume on the diffusion and efficacy of botulinum toxin type A in the treatment of lateral orbital rhytides. Dermatol Surg. 2007;33(5):567–571.
- Kerscher M, Yutskovskaya Y, Corcoran T. IncobotulinumtoxinA in esthetics. J Drugs Dermatol. 2013;12(6):e111–e120.
- Hexsel D, Soirefmann M, Porto MD, et al. Fields of anhidrotic effects of abobotulinumtoxinA in patients with compensatory hyperhidrosis. Dermatol Surg. 2015;41 Suppl 1:S93–S100.

