J Clin Aesthet Dermatol. 2026;19(4):26–27.
by Erik Domingues, MD
Dr. Domingues is with Modern Dermatology of Massachusetts, Fall River, Massachusetts, and the Department of Dermatology at the University of Massachusetts Chan Medical School, Worcester, Massachusetts.
FUNDING: Manuscript support was provided by Arcutis Biotherapeutics Inc.
DISCLOSURES: Dr. Domingues is a promotional speaker and/or consultant for AbbVie, Arcutis, Bristol Myers Squibb, Castle Biosciences, Galderma, Incyte, Johnson & Johnson, LEO Pharma, Pelthos, Pfizer, UCB, and Veradermics.
ABSTRACT: Periorificial dermatitis is a common inflammatory dermatosis most often presenting in adult women around the mouth with a distinctive sparing of the vermilion border. Involvement may also cluster around the eyes and nose. Steroid use is most commonly implicated in this condition. Treatments include topical and oral antibiotics, sulfur-based creams and cleansers, azelaic acid, and nonsteroidal anti-inflammatory agents. Roflumilast cream, 0.3%, was approved in 2022 for the treatment of chronic plaque psoriasis, including intertriginous areas. Here, we present the case of a 30-year-old female patient who presented with periorificial dermatitis, which cleared in 5 days with once daily roflumilast cream, 0.3%, without adverse events, tolerability concerns, or recurrence 11 months after treatment initiation. Keywords: Periorificial dermatitis, roflumilast cream, phosphodiesterase 4 inhibitor
Introduction
Perioral dermatitis (POD), also referred to as periorificial dermatitis, is a common inflammatory facial dermatosis most often observed in adult women. Small pink erythematous papules, often with a papulopustular and/or papulovesicular appearance, fine scaling, and macular erythema, commonly cluster around the mouth with a distinctive sparing of the vermilion border.1 Involvement may also cluster around the eyes and nose.2 While women aged 20 to 45 years are predominantly affected, POD can also affect children.3 A burning sensation may accompany POD in adult patients.3 The pathogenesis of POD is poorly understood; however, topical, intranasal, inhaled, and oral corticosteroids have been implicated in this condition.1 While chronic corticosteroid use may cause a more severe presentation, the exact pathophysiology is unknown.1 Other causes include cosmetic products such as makeup, sunscreen, and toothpaste; physical factors; and microorganisms such as Fusobacterium, Demodex mites, and Candida albicans.1,3 The diagnosis of POD is made clinically, rarely requiring histologic evaluation for confirmation.3 Histopathology reveals spongiosis, lymphocytic exocytosis, edema, and lymphocytic perivascular infiltration.1,3 While POD may be self-limiting and is not associated with systemic disease, many patients desire treatment due to the visible facial lesions and frequent symptomatology.3 The severity varies among patients, and, without treatment, it can become chronic and recurrent.2 An individualized treatment approach is based on severity, age, and concomitant conditions. In general, topical corticosteroids should be discontinued and potential contact allergens identified and avoided whenever possible.1,2
Multiple pharmacologic treatment options have been used for treatment of POD. Topical therapies include antibiotics, sulfur-based creams and cleansers, azelaic acid, and nonsteroidal anti-inflammatory agents such as pimecrolimus cream, often used in combination with barrier-repair moisturizers.1,3 In cases of moderate to severe POD, systemic therapy with an oral tetracycline is commonly used, with macrolides such as erythromycin used as alternative therapy.1-3 Roflumilast cream, 0.3%, is a corticosteroid-free and potent topical phosphodiesterase 4 inhibitor approved by the United States Food and Drug Administration (FDA) for the treatment of plaque psoriasis (2022); the foam formulation was approved for seborrheic dermatitis in 2023 and plaque psoriasis of the scalp and body in 2025. Additionally, roflumilast cream, 0.15%, was approved by the FDA in 2024 for the treatment of atopic dermatitis, and 0.05% strength was approved for atopic dermatitis in patients aged 2 to 5 years (2025).
This case study of a 30-year-old woman with a 1-month history of POD highlights therapeutic success after roflumilast cream, 0.3%, was selected as monotherapy to treat POD. She had complete resolution of POD after using roflumilast cream, 0.3%, once daily for 5 days.
Case Report
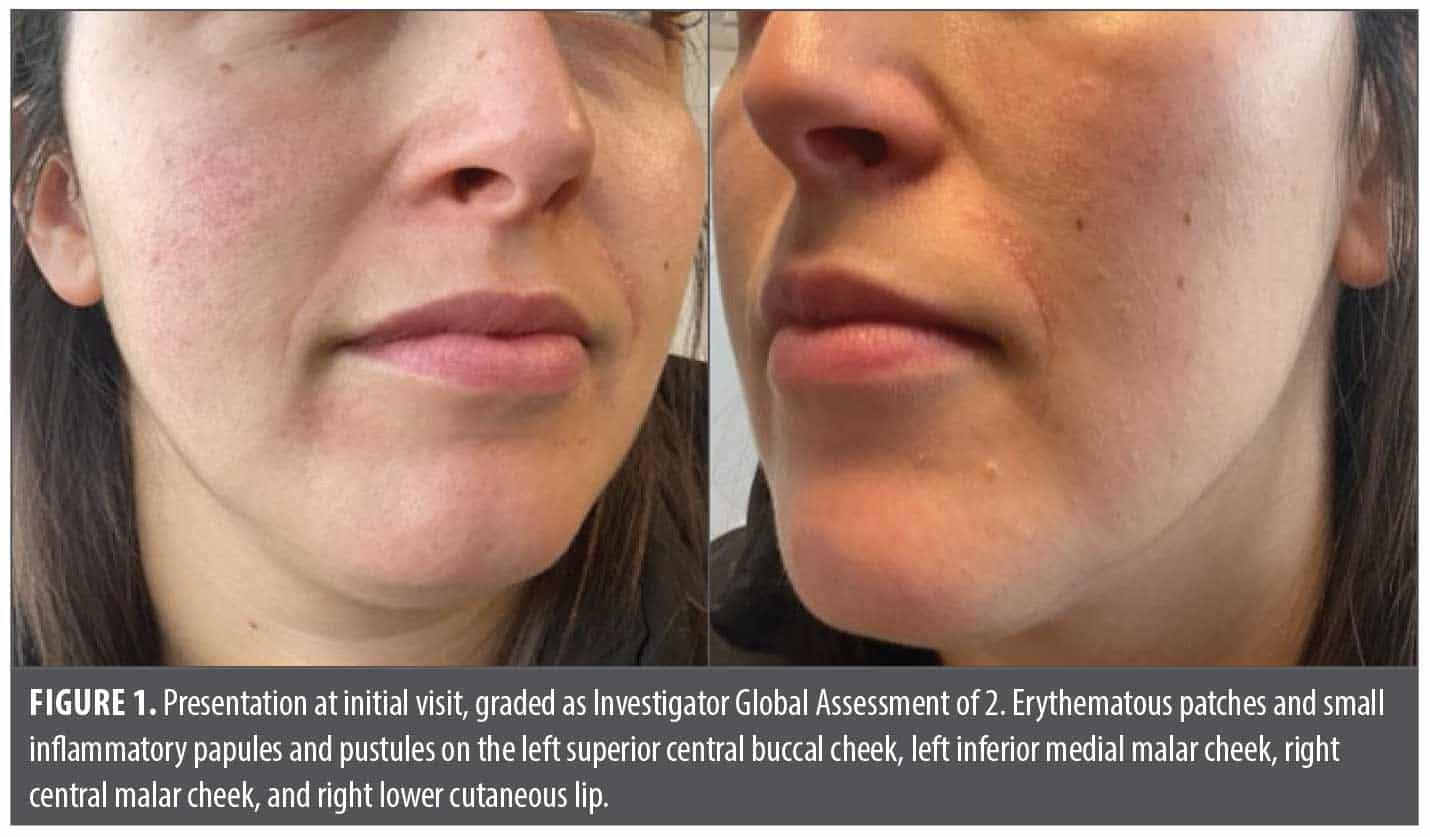
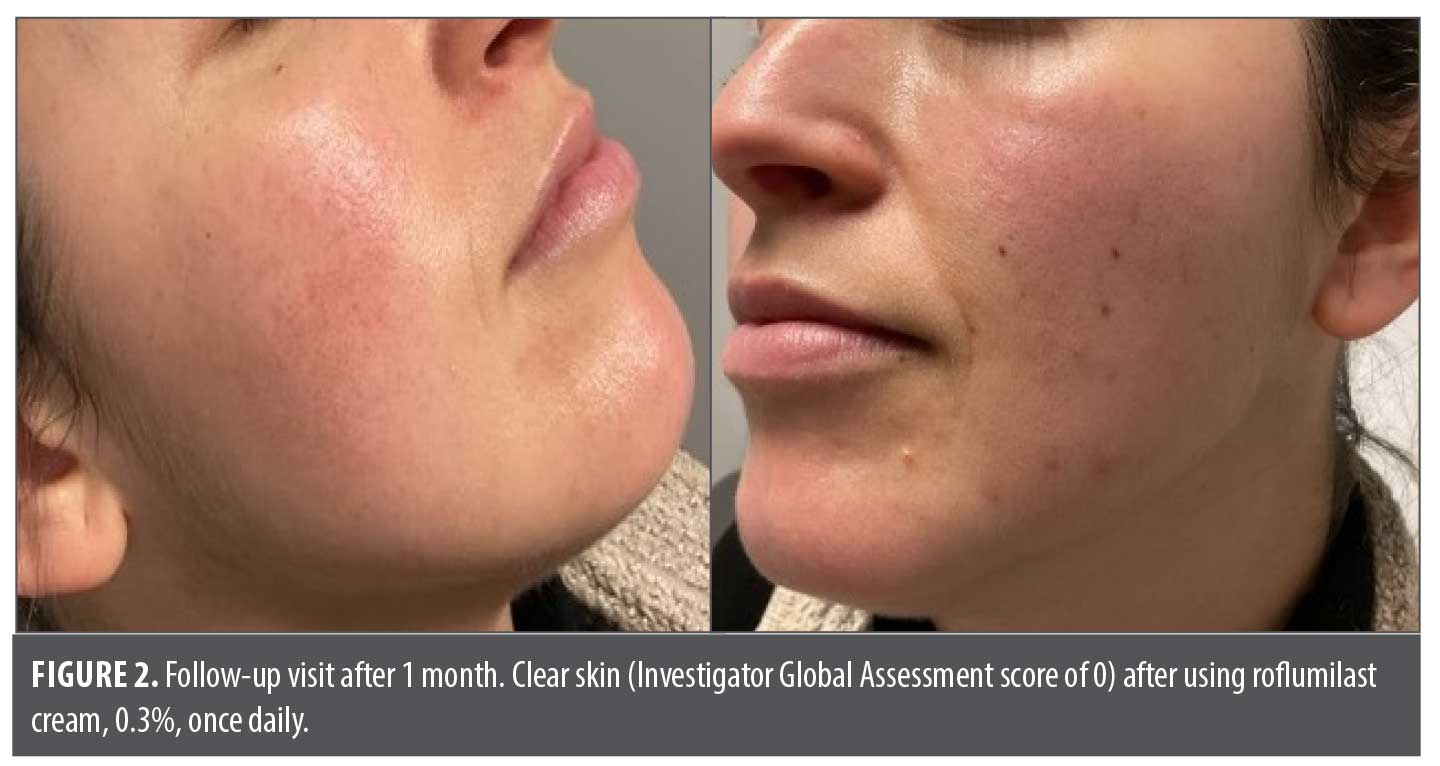
A 30-year-old woman presented with scaly, red papules of 1-month duration on the medial cheeks. Upon physical examination, there were pink, scaly, erythematous patches and small inflammatory papules on the left superior central buccal cheek, left inferior medial malar cheek, right central malar cheek, and right lower cutaneous lip (Figure 1). The patient was diagnosed with POD of moderate severity at the time of examination. Treatment options were discussed, and the patient started on topical roflumilast cream, 0.3%, once daily; this option was selected for its nonsteroidal anti-inflammatory properties and low potential for cutaneous irritation. At 1-month follow-up, the patient reported complete resolution within 5 days of treatment initiation with no further recurrences observed. She used less than 5 g of roflumilast cream, 0.3%, over the treatment duration. Further examination revealed complete skin clearance with no signs or symptoms of disease (Figure 2).
No adverse reactions were noted. In clinical trials of topical roflumilast cream, 0.3%, the most commonly reported adverse events occurring in at least 1% of patients were diarrhea, headache, insomnia, nausea, application site pain, upper respiratory tract infections, and urinary tract infections.4 The patient was instructed to use the product as needed for any recurrences of POD and to follow up if persistent or frequently recurrent. As of 11 months after initial treatment, the patient has not had recurrence of her POD.
Conclusion
This case report of a 30-year-old woman treated with roflumilast cream, 0.3%, once daily for POD resulted in clearance within 5 days of treatment initiation without recurrence of disease after 11 months. In a condition with a plethora of topical and systemic treatment options, this case highlights the utility of topical roflumilast cream, 0.3%, for POD, as it delivered rapid and sustained clearance and was well tolerated. Further evaluations including randomized and controlled studies are needed to evaluate topical roflumilast cream, 0.3%, as a potential treatment option for POD.
References
- Searle T, Ali FR, Al-Niaimi F. Perioral dermatitis: diagnosis, proposed etiologies, and management. J Cosmet Dermatol. 2021;20(12):3839–3848.
- Gray NA, Tod B, Rohwer A, Fincham L, Visser WI, McCaul M. Pharmacological interventions for periorificial (perioral) dermatitis in children and adults: a systematic review. J Eur Acad Dermatol Venereol. 2022;36(3):380–390.
- Lee GL, Zirwas MJ. Granulomatous rosacea and periorificial dermatitis: controversies and review of management and treatment. Dermatol Clin. 2015;33(3):447–455.
- Roflumilast topical cream. Prescribing information. Arcutis Biotherapeutics; July 2024. Accessed 16 June 2025. https://www.arcutis.com/wp-content/uploads/USPI-roflumilast-cream.pdf

Optimizing Surgical Tray Setup and Instrument Selection in Dermatologic Surgery