J Clin Aesthet Dermatol. 2026;19(4):20–25.
by Graham H. Litchman, DO, MS; Diego Ruiz Dasilva, MD; and James Q. Del Rosso, DO
Dr. Litchman is with with Vivida Dermatology, Las Vegas, Nevada and Touro University Nevada, Henderson, Nevada. Dr. Ruiz Dasilva is with Forefront Dermatology, Virginia Beach, Virginia and Eastern Virginia Medical School, Norfolk, Virginia. Dr. Del Rosso is with JDR Dermatology Research, Las Vegas, Nevada and Touro University Nevada, Henderson, Nevada.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Litchman: Arcutis, Castle Biosciences, Galderma, Pfizer, Sanofi & Regeneron, UCB. Dr. Ruiz Dasilva: AbbVie, Arcutis, Dermavant, Galderma, Janssen, LEO Pharma, Lilly, Pfizer, Sanofi & Regeneron, UCB, Verrica. Dr. Del Rosso: AbbVie, Almirall, Amgen, Anaptos Bio, Apogee, Arcutis, Bausch, Beiersdorf, Biofrontera, Botanix, Bristol Myers Squibb, Cage Bio, Cara, Cassiopea, Dermata, Dermavant (Organon), Ferndale, Galderma, Incyte, Johnson and Johnson, La Roche Posay, LEO Pharma, Lilly, L’Oréal, MC2, Moonlake, Nektar, Novan, Nutrafol, Pfizer, Ralexar, RBC Consultants, Regeneron, Sanofi, Sente, Sun Pharma, Takeda, UCB, Verrica, Vyne.
ABSTRACT: Nail psoriasis is a chronic inflammatory manifestation frequently associated with cutaneous psoriasis and psoriatic arthritis (PsA). Fingernails and/or toenails may be affected by psoriasis. The management of nail psoriasis can be challenging due to marked variability in clinical presentation, its chronicity, its frequent refractory nature despite a variety of therapies, and the slow rate of nail plate growth over several months. Unfortunately, the common clinical misdiagnosis as onychomycosis often leads to delay in an accurate diagnosis of nail psoriasis as well as exposure of the patient to topical and/or oral antifungal therapies that are not effective for psoriasis. Traditional treatment options for nail psoriasis often require extended courses of therapy to achieve significant results. Both of these cases were characterized by limited or absent psoriatic skin involvement, with extensive symptomatic psoriatic fingernail dystrophy and presence of PsA. This article describes the use of bimekizumab, a dual interleukin (IL) 17A and IL-17F inhibitor, for the rapid resolution of severe psoriasis in 2 patients with extensively dystrophic fingernail psoriasis and concomitant PsA, both causing marked interference with daily physical functioning and significant psychosocial impairment. Both patients rapidly achieved marked improvement of all signs and symptoms of severe psoriasis involving all affected fingernails and joints, within at least 1 to 3 months. These results add real-world evidence that further highlights the potential for accelerated improvement with specific biologic therapies effective for plaque psoriasis, with both cases supporting the rapid onset and high magnitude of efficacy for symptomatic severe psoriasis presenting with extensive dystrophy of all fingernails and PsA. Keywords: Nail psoriasis, bimekizumab, psoriatic arthritis, biologic therapy
Introduction
Nail psoriasis is a chronic inflammatory disorder of the nail unit, most commonly seen in patients with cutaneous psoriasis and psoriatic arthritis (PsA), that can affect fingernails and/or toenails. Anatomy of the nail unit consists of the nail matrix, nail bed, nail plate, hyponychium, and surrounding periungual skin including the proximal and lateral nail folds. The nail matrix is the germinative epithelium located beneath and extending distally beyond the proximal nail fold and is primarily responsible for formation of the nail plate. It is divided into proximal and distal (visible as the white lunula) portions, with nail matrix integrity being crucial for normal nail plate growth, appearance, and function. The nail bed lies beneath the nail plate, from the lunula’s distal edge to the hyponychium. It contains a basal layer and supports nail plate growth but does not produce the plate. The underlying vascular tissue of the nail bed gives the healthy nail unit its pink color, visible through the translucent plate. The nail plate forms the visible portion of the nail unit and is made of keratinized anucleate cells. It is anchored by the nail folds and bordered distally by the hyponychium. The hyponychium is the protective zone beneath the free edge of the nail plate, with the transverse onychodermal band serving to prevent the entry of external liquids, microbes, and debris, and is connected to the supporting fingertip. It is distinct from the nail bed and seals the subungual space. The periungual tissues, including the nail folds and eponychium (cuticle), protect the nail unit, with the eponychium providing a seal that prevents liquids, debris, and microbes from penetrating under the nail folds. The soft tissues of the nail unit are highly vascularized and innervated, aiding in both sensory and protective functions.1–3
The nail matrix and nail bed are the most common sites of primary psoriatic inflammation, which induces pathologic nail unit involvement with psoriasis. Inflammation in the nail matrix typically results in several potential visible manifestations. These include nail plate pitting, leukonychia (white spots within the nail plate), and/or crumbling (reflecting confluence of multiple broadening nail pits). Red spots within the lunula or diffuse erythema of the lunula (which is normally white in color) may also be observed in nail psoriasis. Nail bed involvement leads to distal onycholysis, “oil-drop” discoloration (salmon patch) reflective of focal onycholysis, subungual hyperkeratosis, and occasionally splinter hemorrhages.3,4
Epidemiologically, nail unit involvement occurs in approximately 50% to 79% of patients with skin psoriasis and up to 80% to 90% of those with PsA.3–5 Psoriatic nail changes may be the initial or sole manifestation of psoriasis and are a risk factor for the current presence or subsequent development of psoriatic arthritis.4,5 Fingernails are more commonly affected than toenails, and the severity of nail disease may or may not correlate with the extent of cutaneous involvement.
Beyond prevalence, several factors increase the likelihood for nail unit involvement, including longer disease duration, more severe cutaneous psoriasis, scalp and genital involvement, and a personal or family history of PsA.2 Trauma or injury can also precipitate psoriatic nail lesions (Koebner phenomenon). Nail psoriasis is associated with significant pain, functional impairment, and psychosocial distress, all of which consistently impair quality of life on many levels, including interpersonal interactions and work productivity.3–5
Treatment options for nail psoriasis are guided by the locations and extent of nail unit involvement, associated skin and joint disease, and the impact on a patient’s quality of life. For mild, limited nail disease (typically ≤ 3 nails, without significant skin or joint involvement), the conventional initial approach, notably published prior to the availability of biologic agents with augmented efficacy, has been high-potency topical corticosteroid application, often combined with a topical vitamin D analog, and intralesional corticosteroid injections (the most effective of these options but usually with temporary benefit); these options are used with the goal of targeting the nail matrix and bed to address pitting, onycholysis, and subungual hyperkeratosis.5,6 When nail unit involvement is more moderate to severe, or when there is concomitant skin and/or joint disease, oral agents, including methotrexate, acitretin, cyclosporine, and apremilast, have historically been suggested. However, the more recent availability of biologic therapies for psoriasis (tumor necrosis factor [TNF] α inhibitors and interleukkn [IL] 17, IL-12/23, and IL-23 inhibitors) demonstrate more optimal efficacy in improving nail psoriasis outcomes, especially in patients with PsA and with more extensive cutaneous disease.4–12 Risk factors such as longer disease duration, severe skin involvement, and PsA may necessitate escalation to systemic or biologic therapy.5–7 Although limited, in patients who are truly averse to an injectable therapy, there are data showing that oral deucravacitinib, a tyrosine kinase 2 (TYK-2) inhibitor in the Janus kinase (JAK) class, can improve psoriatic
nail disease.13
The typical duration required for psoriatic nail changes to clear is substantially longer than for cutaneous lesions. Changes over time, specifically in psoriatic fingernail disease undergoing treatment, are highly dependent on the anatomic locations and magnitude of involvement with psoriatic inflammation. Psoriatic fingernail disease often takes substantially longer to improve than for cutaneous lesions due to the slow growth rate of fingernails (3–4 mm/month). Most therapies require at least 3 to 6 months for significant improvement, with complete clearance often taking 6 to 12 months or longer. For example, biologic agents that target IL-17 have demonstrated marked improvement in nail psoriasis by 16 to 36 weeks in randomized controlled clinical trials, with sustained clearance rates up to 60% to 88% at 36 to 60 weeks. Conventional systemic agents generally may require similar or longer durations to take effect, and improvement in psoriatic nail matrix disease may lag behind isolated psoriatic nail bed disease.3,5–11
Here we present 2 cases of severe psoriasis affecting all 10 fingernails and associated manifestations of PsA treated with bimekizumab therapy to achieve complete clearance of disease and symptomatology within 3 months. Both patients provided informed consent for the use of their cases and photographs for publication and public use.
Case #1
A 56-year-old White woman with no significant past medical history other than vague symptoms of fatigue and joint pain presented with chronic fingernail changes and periungual inflammation. She had previously been treated for presumed eczematous dermatitis, onychomycosis, and chronic paronychia for several years without clinical improvement. Her prior therapies included multiple courses of oral and topical antibiotics, including oral doxycycline; mupirocin cream, 2%; topical corticosteroids (triamcinolone cream, 0.1%; mometasone cream, 0.1%); intralesional triamcinolone injections (10 mg/mL); topical ruxolitinib cream; topical efinaconazole solution, 10%; and oral antifungal agents (terbinafine, fluconazole), all with negligible or no improvement.
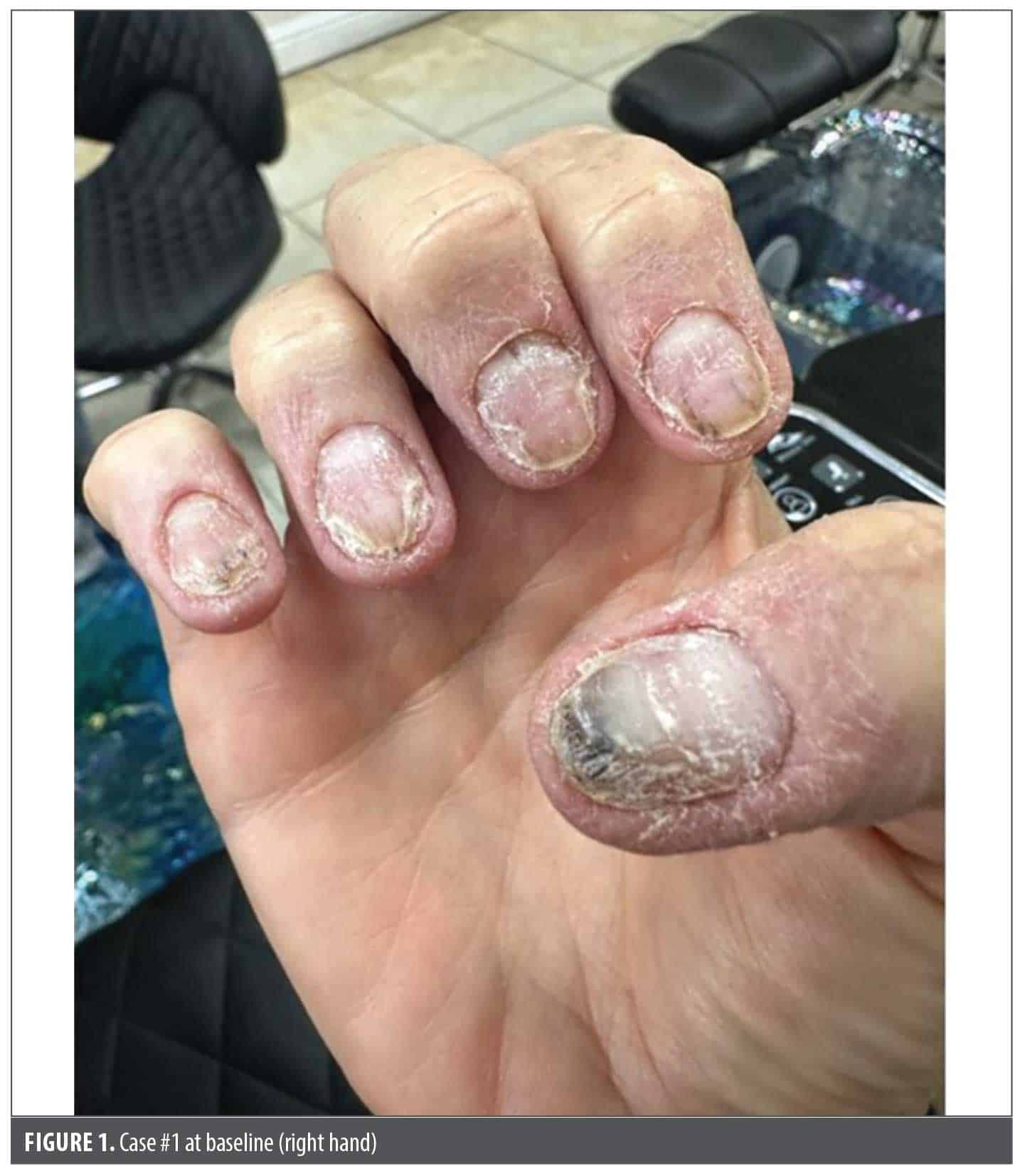
During our initial consultation, a thorough history and physical examination were performed. The patient reported being significantly distressed as she was embarrassed about the appearance of her nails. She reported that she would often hide her fingernails, avoid shaking hands, and did not visit the nail salon for years (which was an activity she frequently enjoyed). She was previously seen by a rheumatologist for what they deemed to be rheumatoid arthritis and the fingernail changes were attributed to a presumed diagnosis of onychomycosis, for which she underwent multiple rounds of oral fluconazole, oral terbinafine, and topical efinaconazole. All 10 fingernails demonstrated dystrophic changes, including nail plate pitting, leukonychia, and crumbling characteristic of nail psoriasis. Based on the concurrent presence of pain in multiple joints (fingers, wrists, lower back, and sacroiliac joint), joint stiffness that worsens with inactivity and improves with movement, and mild discomfort/tenderness upon palpation of affected finger joints, a clinical diagnosis of severe fingernail psoriasis with PsA was made (Figure 1). At this current evaluation, other than the periungual erythema, edema, and scaling involving the adjacent nail folds, there was no evidence of cutaneous psoriasis including evaluation of the scalp, palms, soles, and anogenitalia. Given the extensive dystrophic fingernail disease, despite absence of other cutaneous involvement with psoriasis, the patient was initiated on bimekizumab 320 mg administered subcutaneously once monthly, which is the Food and Drug Administration-approved dosing regimen that encompasses concurrent treatment of both plaque psoriasis and PsA.
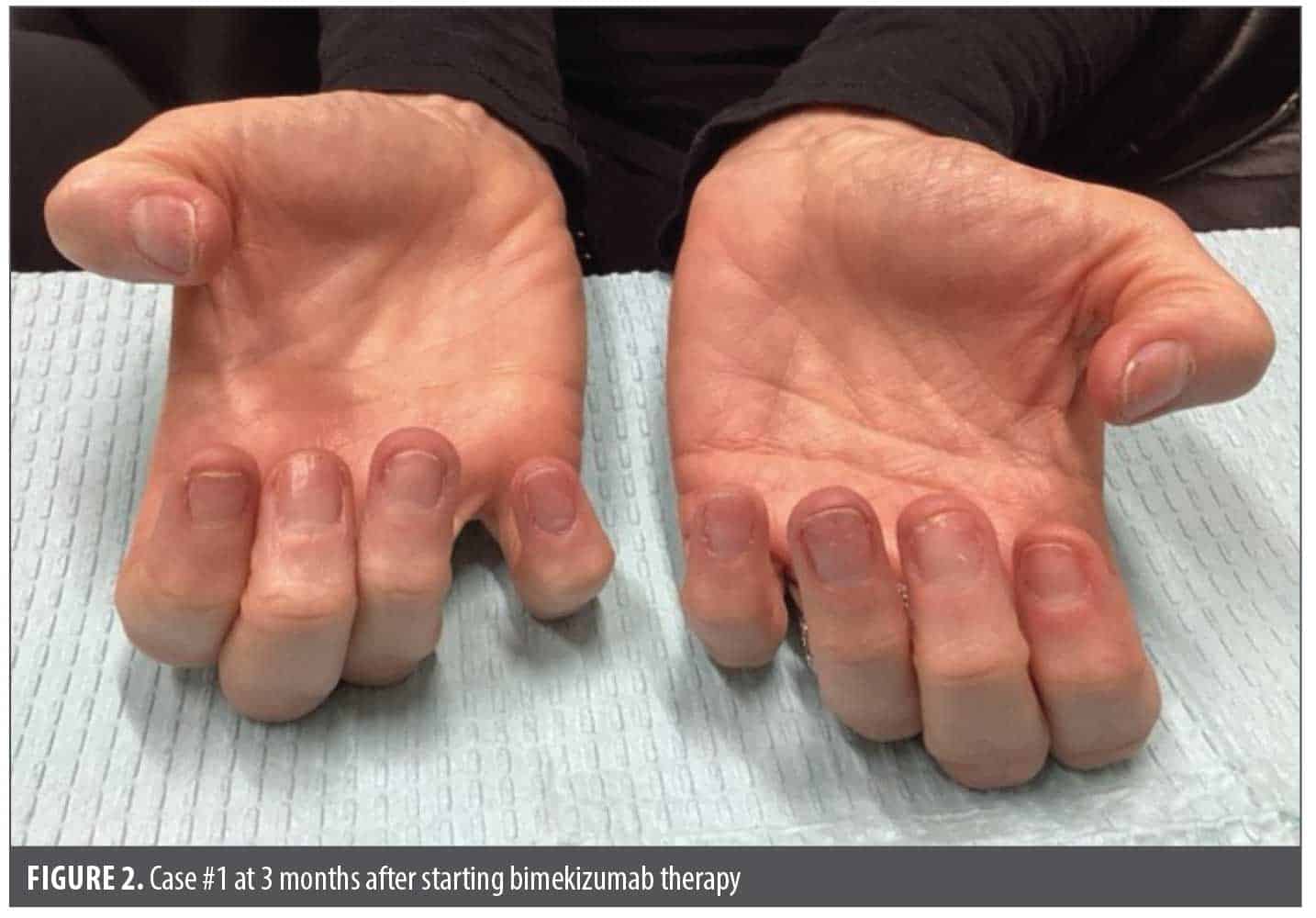
At 1-month follow-up, she demonstrated significant improvement in periungual erythema with progressive improvements in signs and symptoms noted at 2 months of follow-up. At the 3-month follow-up, all signs of fingernail psoriasis and all joint symptoms were completely resolved (Figure 2). The patient reported that her fingernails had been completely clear since the end of approximately 2 months after starting bimekizumab therapy, reflecting resolution within < 3 months of treatment.
Case #2
A 63-year-old White woman presented with a history of slowly worsening “brittle nails” affecting the fingers on both hands for over 10 years. She was otherwise healthy without any other medical disorders and did not take any medications other than those referred to below. Her toenails were not affected. Over the past year, all of her fingernails progressively changed with significant thickening distally, onset of pain, and marked loss of manual dexterity that regularly impaired her ability to function physically and caused her considerable social embarrassment, anxiety, and psychosocial distress. There had not been use of any over-the-counter treatments other than her usual personal intermittent manicuring, and she was not a consistent user of nail polish. Additionally, she had not undergone any nail procedures including at salons or spas, denies recent or remote history of trauma, and has no known family history of any nail disorders, fungal infections, or psoriasis. Her present review of systems was otherwise unremarkable including arthritis and/or joint swelling/redness. When asked if she had a history of psoriasis or other conditions affecting anywhere on her skin at present or in the past, she recalled being told she had psoriasis on her skin at 1 time over a decade ago by a general physician but recalled no other details about it, and it never persisted or recurred.
She was previously treated for a clinical diagnosis of onychomycosis with multiple courses of topical and oral antifungal agents without clinical improvement. Her prior topical therapies included mupirocin cream, 2%; efinaconazole solution, 10%; and ciclopirox nail lacquer, 8%, all without clinical improvement. Courses of oral antifungal therapy were also sequentially prescribed, including terbinafine, fluconazole, and itraconazole, all without response. During the continued course of her management, she had undergone several nail clippings sent for periodic acid-Schiff staining as well as polymerase chain reaction testing which at times revealed staphylococcal species and Malassezia spp, which were believed to be contaminants and not pathogenically related to the current nail findings.
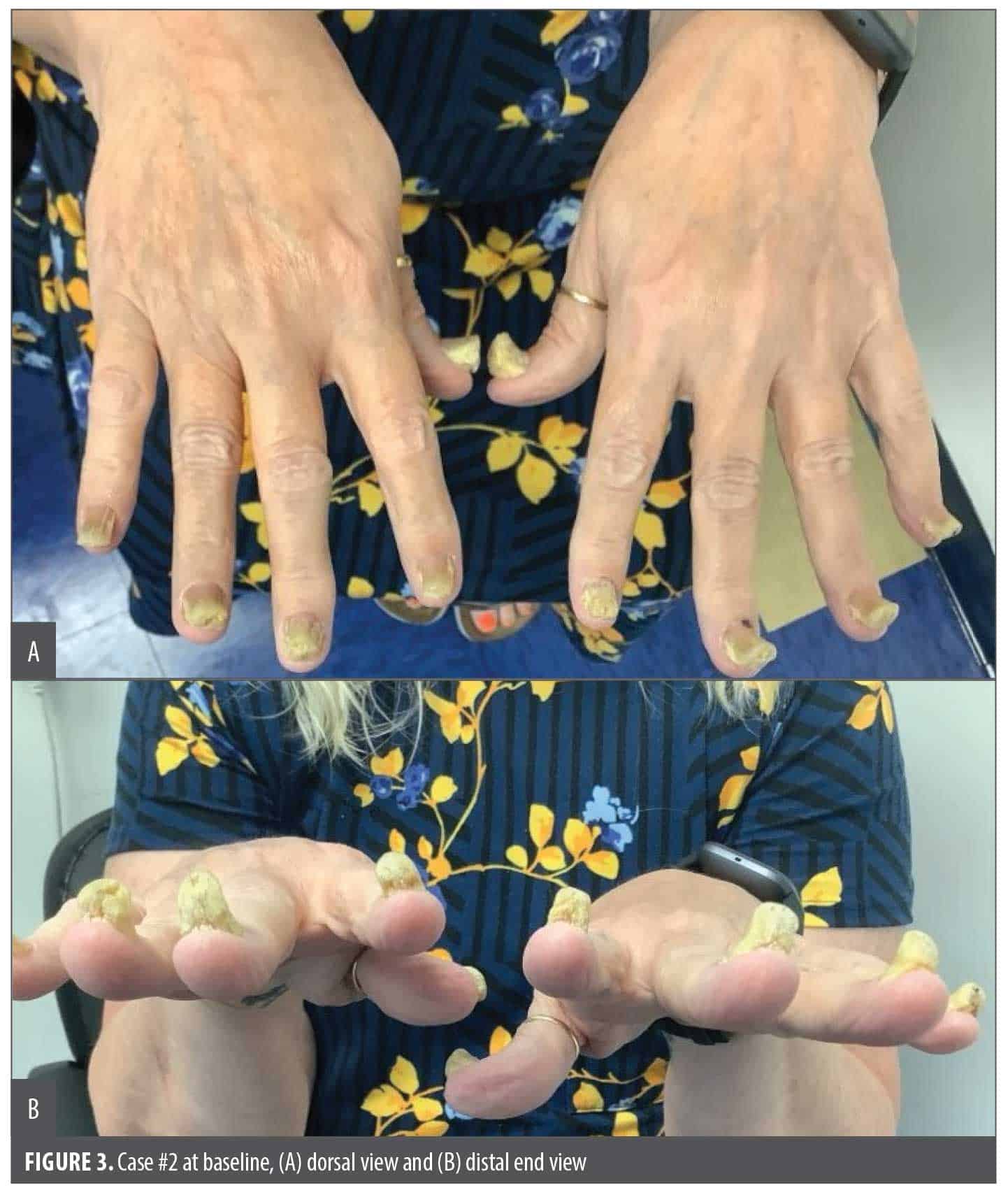
During her initial dermatology visit, all 10 fingernails demonstrated dystrophic changes, including subungual hyperkeratosis, crumbling, and onychogryphosis (Figure 3A, Figure 3B). She had severe discomfort with minimal pressure on the nail units, limiting her ability to function in even the most basic activities such as driving, opening items, carrying certain items, and typing. On skin examination, there was no evidence of cutaneous disease involving the skin, including the scalp, palms, soles, and anogenital region. Findings supporting an initial clinical diagnosis of PsA were noted presenting as mild-to-moderate symptomatic dactylitis with tenderness of multiple interphalangeal and metacarpophalangeal joints of several fingers and early morning stiffness of the first and second finger joints on both hands that resolved with movement within a few hours. The patient did not report any back stiffness and/or pain or any history of ocular symptoms or eye redness. A longitudinal nail unit biopsy, which included the distal nail matrix, nail bed, and nail plate, was performed and histologically confirmed the diagnosis of psoriasis.
Despite the absence of psoriatic skin involvement and any definitive evidence of psoriasis in the past, a severity rating of severe psoriasis affecting both hands and all 10 fingers/fingernails was documented in her record based on the following criteria: (1) progressively worsening extensive deformity of all 10 fingernails affected by biopsy-proven psoriasis, (2) significant and frequent physical discomfort (pain) interfering with hand and finger function and quality of life, (3) marked interference every day with multiple manual activities of normal daily living, (4) adverse impact on intimate relations requiring touch, (5) inability to perform normal job-related activities (ie, typing, filing, carrying items), and (6) marked psychosocial stress such as anxiety, embarrassment, and fear of touching others. The documented clinical presence of symptomatic PsA further supports an overall diagnosis and severity rating of severe psoriasis with PsA in this patient.
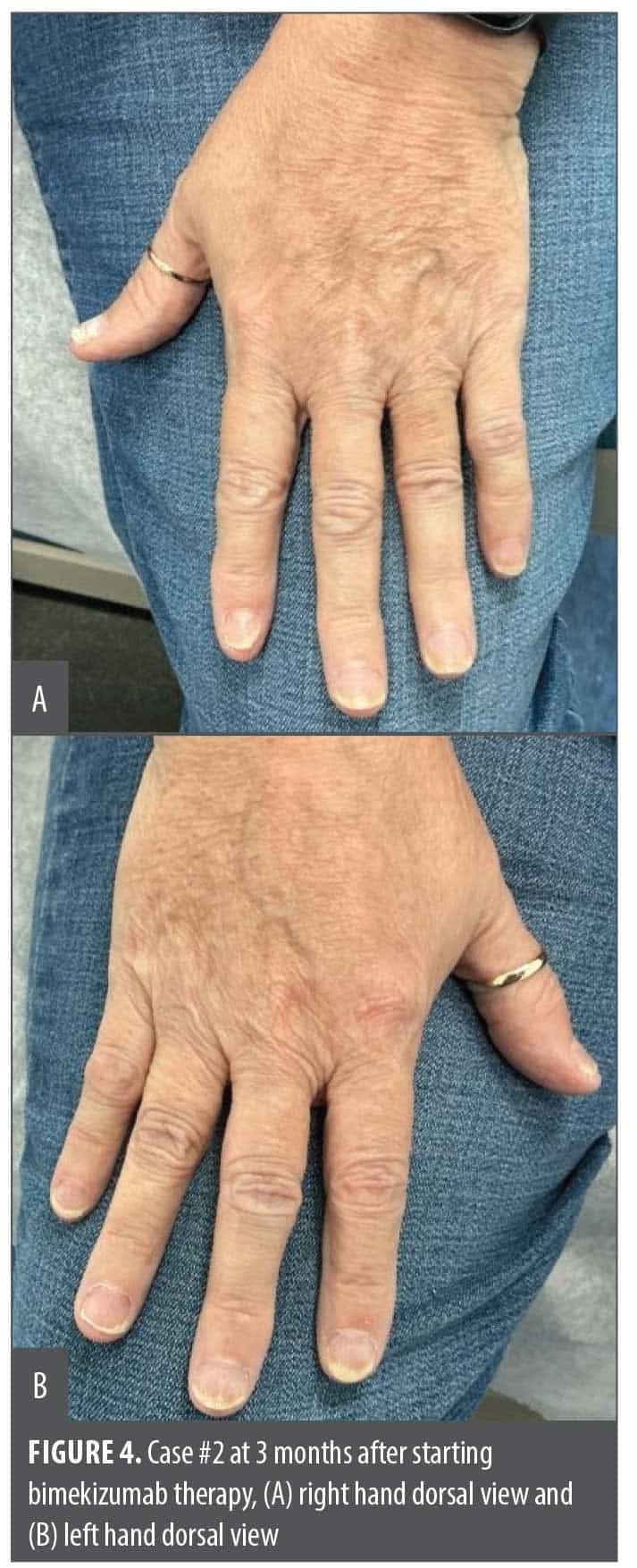
After a discussion of the diagnosis, confirmed absence of fungal infection, and the lack of anticipated response to topical therapy alone, the available systemic therapy options were reviewed. Treatment was initiated with subcutaneous bimekizumab therapy using the dosing regimen approved for moderate to severe plaque psoriasis (320 mg once monthly). This dosing regimen simultaneously addresses the presence of PsA, which is highly convenient. At the 3-month follow-up after initiating bimekizumab therapy, she demonstrated significant improvement in the dystrophic appearance of all affected fingernails with complete resolution of pain and joint stiffness and full return of all manual activities with her hands and fingers without limitations (Figure 4A, Figure 4B). The patient reported that her loss of hand and finger dexterity and tactile discomfort, the associated pain, and the joint symptoms were all at least 90% better within a month of the initial dose. She denied any adverse effects with treatment and was very pleased with the therapeutic outcome.
Discussion
These 2 cases illustrate the unusually rapid resolution of severe psoriasis involving all 10 fingernails and the associated PsA with bimekizumab therapy. Nail psoriasis remains a therapeutic challenge due to its chronicity, slow nail growth, and common association with cutaneous psoriasis and frequent association with joint disease, even in cases with limited skin involvement. Clinical improvement in nail findings typically lags behind skin and joint symptoms and is highly dependent on the anatomic location(s) and intensity of psoriatic inflammation affecting the nail unit. Most interventions involving primarily the distal portion of the fingernail unit require at least 3 to 6 months for significant visible clinical response, and up to 12 months or longer for complete clinical clearance in cases of panungual psoriatic involvement, reflecting the slow rate of nail plate growth and the need for sustained therapy.3,7–9
Biologic therapies, including TNF-α inhibitors, IL-17 inhibitors, and IL-23 inhibitors, have demonstrated superior efficacy compared to conventional systemic and topical agents in nail psoriasis, particularly in the setting of concomitant PsA.3,5–12,14 In major clinical trials, ixekizumab and secukinumab achieved Nail Psoriasis Severity Index (NAPSI) improvements of 60% to 88% by 36 to 60 weeks, with complete clearance typically not reported until later time points.6,7,11 Similarly, recent studies of bimekizumab have shown marked nail improvement by 16 weeks, with progressive clearance thereafter.4,6,8,12
Our 2 cases reflect real-world effective use of subcutaneous bimekizumab therapy for symptomatic and highly dystrophic fingernail psoriasis and PsA. The outcomes were not restricted by the inherent limitations of inclusions and exclusions that are mandated in controlled clinical trials, thus supporting the importance of published real-world evidence in case reports in addition to phase 2, 3, and 4 randomized controlled clinical trials. Both patients achieved full clinical resolution of psoriatic nail disease after < 3 months of therapy. To our knowledge, the outcomes in both cases represent a more rapid therapeutic response than has been documented overall in the literature, likely due to study methodologies and the natural variability among different study investigators when incorporating the protocol-mandated efficacy assessment criteria. The rapid clearance may reflect the high efficacy of bimekizumab in both nail psoriasis and PsA, with the potential for a greater impact on efficacy with inhibition of both IL-17A and IL-17F.14–17 The rapidity and magnitude of response is consistent with what has been reported in multiple randomized controlled short-term and long-term studies using bimekizumab for plaque psoriasis.8,12,14–16
Conclusion
Nail unit psoriasis is a common manifestation in patients affected by psoriasis and is often present concurrently with or as a predictor for PsA. The clinical presentations of nail psoriasis are highly variable, and the response to treatment is typically more prolonged than with psoriasis involving skin. These 2 cases underscore the potential of IL-17 inhibitors, and more specifically highly favorable therapeutic outcomes with bimekizumab, to achieve accelerated improvement with clearance of psoriatic nail disease and associated signs and symptoms of PsA. Importantly, these results provide encouragement that more prolonged treatment courses may not be needed to achieve clearance of psoriatic nail disease, especially in those cases presenting with dystrophic nail changes that are primarily distal in location. Larger collaborations of real-world case-based evidence and prospective clinical trials will be needed to confirm whether such rapid responses are often reproducible, to identify the patterns of psoriatic nail disease that can clinically predict the greater likelihood of early clearance, and to accurately differentiate which agents are most likely to provide rapid and effective improvement of psoriatic nail disease and of psoriatic arthritis.
References
- Del Rosso JQ, Basuk PJ, Scher RK. Ricci AR. Dermatologic diseases of the nail unit. In: Scher RK, Daniel CR, eds. Nails: Therapy, Diagnosis, Surgery. 2nd ed. WB Saunders Company; 1997:172–200.
- Haneke E. Anatomy of the nail unit and the nail biopsy. Semin Cutan Med Surg. 2015;34(2):95–100.
- Ji C, Wang H, Bao C, et al. Challenge of nail psoriasis: an update review. Clin Rev Allergy Immunol. 2021;61(3):377–402.
- Crowley JJ, Weinberg JM, Wu JJ, Robertson AD, Van Voorhees AS; National Psoriasis Foundation. Treatment of nail psoriasis: best practice recommendations from the Medical Board of the National Psoriasis Foundation. JAMA Dermatol. 2015;151(1):87–94.
- Thomas L, Azad J, Takwale A. Management of nail psoriasis. Clin Exp Dermatol. 2021;46(1):3–8.
- de Vries ACQ, Bogaards NA, Hooft L, et al. Interventions for nail psoriasis. Cochrane Database Syst Rev. 2013;2013(1):CD007633.
- Hwang JK, Ricardo JW, Lipner SR. Efficacy and safety of nail psoriasis targeted therapies: a systematic review. Am J Clin Dermatol. 2023;24(5):695–720.
- Campione E, Artosi F, Shumak RG, et al. Fast clinical response of bimekizumab in nail psoriasis: a retrospective multicenter 36-week real-life study. Pharmaceuticals (Basel). 2024;17(10):1378.
- Egeberg A, Kristensen LE, Vender R, et al. Sustained resolution of nail psoriasis through 5 years with ixekizumab: a post-hoc analysis from UNCOVER-3. Acta Derm Venereol. 2022;102:adv00787.
- Reich K, Sullivan J, Arenberger P, et al. Effect of secukinumab on the clinical activity and disease burden of nail psoriasis: 32-week results from the randomized placebo-controlled TRANSFIGURE trial. Br J Dermatol. 2019;181(5):954–966.
- Huang IH, Wu PC, Yang TH, et al. Small molecule inhibitors and biologics in treating nail psoriasis: a systematic review and network meta-analysis. J Am Acad Dermatol. 2021;85(1):135–143.
- Hagino T, Onda M, Saeki H, Fujimoto E, Kanda N. Effectiveness of bimekizumab for genital, nail, and scalp lesions with psoriasis: a 24-week real-world study. J Dermatol. 2024;51(12):1658–1664.
- Hagino T, Saeki H, Fujimoto E, Kanda N. Long-term effectiveness and safety of deucravacitinib for psoriasis: a 52-week real-world study of genital, scalp and nail lesions. Clin Exp Dermatol. 2025;50(5):952–959.
- Battista T, Scalvenzi M, Martora F, Potestio L, Megna M. Nail psoriasis: an updated review of currently available systemic treatments. Clin Cosmet Investig Dermatol. 2023;16:1899–1932.
- Merola J, Warren RB, Thaçi D, et al. Bimekizumab complete clearance of both skin and nail psoriasis: comparative efficacy in phase III/IIIb Studies. Am J Clin Dermatol. 2025;26(6):967–979.
- Ruggiero A, Potestio L, Camela E, et al. Bimekizumab for the treatment of psoriasis: a review of the current knowledge. Psoriasis (Auckl). 2022;12:127–137.
- Nie T, Shirley M. Bimekizumab: a review in psoriatic arthritis. Drug. 2024;84(5):587–598.

Optimizing Surgical Tray Setup and Instrument Selection in Dermatologic Surgery