 J Clin Aesthet Dermatol. 2021;14(7):22–25
J Clin Aesthet Dermatol. 2021;14(7):22–25
by Mohammed Al-Haddab, MD; Azhar Alali, MD; and Homaid Al-otiabi, MD
Dr. Al-Haddab is with King Saud University in Riyadh, Saudi Arabia. Dr. Alali is with the Ministry of Health Saudi Arabia in Medinah, Saudi Arabia. Dr. Al-Otiabi is with the Ministry of Health in Makkah, Saudi Arabia.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors report no conflicts of interest relevant to the content of this article.
ABSTRACT: Background. Lip hyperpigmentation is a common cosmetic problem with multiple causative factors and limited treatment options.
Objective. This study sought to assess the efficacy and safety of Q-switched (QS) double-frequency 532-nm neodymium-doped yttrium aluminum garnet (Nd:YAG) laser therapy for dark lips.
Methods. A prospective pilot study of 24 patients with lip hyperpigmenation treated with a QS double-frequency 532-nm Nd:YAG laser was performed. Photographs taken before and three months after the laser therapy sessions were evaluated for improvement and/or any complication by two blinded dermatologists using the color score chart and a visual analog scale (VAS). Patients were asked to assess their pain and satisfaction scores following the procedure using a VAS.
Results. The mean VAS scores of the photographs decreased by 10.18 percent, from a baseline of 6.09±1.53 to 5.47±1.72 (p=0.16). Also, the color score decreased by 9.38 percent, from 2.44±0.61 to 2.23±0.71 points (p=0.23) after the last treatment. Overall, the photographic assessment indicated that, of the 24 participants, 14 (58.3%) showed moderate to excellent improvement, four (16.7%) showed mild or no improvement, and six (25.0%) showed worsening/mottling of the pigmentation. The overall mean pain and satisfaction scores were 4.08±1.14 and 6.50±2.92, respectively.
Conclusions. QS Nd:YAG 532-nm laser therapy appears to be a safe and moderately effective treatment for hyperpigmentation of the lips. Further studies should evaluate a larger number of patients and undertake a longer period of follow-up.
Keywords: Q-switched 532-nm Nd:YAG laser, safety, effectiveness, patient satisfaction
Lip hyperpigmentation is a common cosmetic concern and lowers the self-esteem of some affected individuals.1 A clinical diagnosis of lip hyperpigmentation is established in individuals presenting with diffuse dark-hued islands or even dark spots interspersed on the lip surface, not involving areas of vascular supply in the mucosa of the lip, which are most often reddish in color.2 The etiology of lip hyperpigmentation might be physiologic or pathologic and due to a number of causes, such as genetics, permanent makeup, tobacco use, perioral ephelides, labial lentigines, benign melanosis, and prolonged use of some drugs (e.g., minocycline cyclophosphamide, doxorubicin, and tricyclic antidepressants).2–6 The treatment of these conditions depends on the underlying causative factors and includes avoidance of smoking and drugs, anti-inflammatory medications, and laser treatments.2,7 However, the effectiveness of these treatment approaches varies from very poor to excellent. Beyond these, some patients can present with idiopathic lip hyperpigmentation. This is mainly seen in women with darker skin types (Fitzpatrick phototypes III–V), who present for management due to cosmetic concerns or embarrassment.4,5 In Thailand, such patients tend to mask lip hyperpigmentation by tattooing with a red or pink color.8 Currently, evidence to guide the treatment of idiopathic lip hyperpigmentation is crucial but lacking, and patients are usually placated with the hope that lip darkening will resolve with time. Some previous reports have demonstrated successful laser treatments for benign lip hyperpigmentation.2,5,8 However, the effectiveness of these treatment approaches varies in terms of potential benefits and side effects.2 Recent research has shown that laser therapy might be effective in the management of hyperpigmentation of the lip and face without damaging the surrounding skin.9 This is because the absorption spectrum for melanin is broad, ranging from 351 to 1,064 nm—a property that supports the treatment of pigmented lesions with a wide variety of lasers.9 One of the commonly used laser types is the continuous wave laser, which can repeatedly deliver short but intense light projections onto the skin. For example, Q-switched (QS) neodymium-doped yttrium aluminum garnet (Nd:YAG) laser pulses are typically 3 to 7 nanoseconds in length and allow selective photothermolysis of pigmented lesions without significant damage to the surrounding skin.5,8,9 Preliminary studies using Q-switched Nd:YAG lasers have shown promising results. Recent case reports have indicated that QS Nd:YAG laser therapy was effective in the treatment of lip pigmentation, lip mucosa tattoos, and a variety of pigmentary lesions on the face.5,8–10 However, some other studies have demonstrated that the application of lasers in the management of hyperpigmentation occasionally leads to undesirable side effects, such as labial edema, pain, scale and bleb formation, and hypopigmentation.6,8,11 In Saudi Arabia, several studies have demonstrated that pigmentary disorders are among the common presentations in dermatology clinics, with one study suggesting it to be the fourth most common dermatosis.12–14 Although one case report from Saudi Arabia demonstrated that Nd:YAG (532-nm) laser therapy was effective in the treatment of a case of benign lip pigmentation,5 to the best of our knowledge, there have been no prospective studies to assess the efficacy and safety of QS double-frequency 532-nm Nd:YAG laser therapy in the treatment of lip hyperpigmentation performed in the country. Thus, in this prospective pilot study, we examined the efficacy and safety of QS double-frequency 532-nm Nd:YAG laser therapy in the treatment of benign lip hyperpigmentation among women in Saudi Arabia.
Methods
Study design. This was a prospective observational study carried out from January to June 2019 in our dermatology unit. The potential benefits and risks of the procedure were fully disclosed to each patient, and written informed consent was obtained from all participants. The study was approved by the institutional review board (E-17-2757).
Inclusion and exclusion criteria. The participants were 24 Saudi women (aged >18 years) who presented with diffuse homogeneous brown or dark brown lips, which were clinically evaluated and diagnosed to be benign lip pigmentation. Prospective participants were excluded if they were younger than 18 years of age, were pregnant or lactating, had a personal history of skin cancer or radiotherapy, had lesions having a keloid-forming tendency, were smokers, were using systemic retinoid medication, and/or had any active lesions on the lips requiring biopsy to rule out malignancy.
Treatment protocol and follow-up. The laser system used was a QS double-frequency 532-nm Nd:YAG laser (AlexTrivatage Laser system; Candela Corporation, Wayland, Massachusetts). The laser unit emits a wavelength of 532nm with the maximum fluence of 2 to 2.4 J/cm2 for a 532-nm green light, nanosecond pulse duration with a 0-Hz repeat, and a 3-mm spot size. The lips were treated with four sessions of laser therapy biweekly, according to the skin type and degree of lip pigmentation. Topical anesthesia (topical lidocaine 2.5% plus prilocaine 2.5% cream) was applied for 20 minutes under occlusion before each session. After each laser session, patients were instructed to use a mid-potency topical corticosteroid with an antibacterial cream for 2 to 3 days and to limit their sun exposure. A broad-spectrum sunscreen lotion was used by all patients for the entire study period. Patients were called by phone a day after their procedure for follow-up and to inquire if they had any history of herpes labialis—particularly those who had no clear history of previous eruption of herpes. A total of two of the 24 patients had experienced a previous episode of herpes labialis before laser treatment, and prophylactic valacyclovir 500mg twice daily for three days was given to them from the day of the laser treatment. None of the patients had active herpes after laser sessions and none required therapeutic doses of valacyclovir.
Patients were photographed before treatment and three months after their last treatment session. Photographs were evaluated for improvement or any complication by two blinded consultant dermatologists.
All participants were assessed objectively by the dermatologists (using the color score and photographic assessment) and subjectively via their own self assessments.
Objective assessments. Photographs of patients were taken in the same location with constant illumination and distance at baseline, before and after each therapy session, and at each follow-up visit using a digital still camera (VISIA Facial Imaging Device; Canfield Scientific Inc., Fairfield, New Jersey). The digital photographs obtained were independently and blindly assessed for improvement by the two dermatologists before laser therapy and at the end of the follow-up period using a visual analog scale (VAS) ranging from 0 to 10 (where 0=no pigmentation and 10=severe dark pigmentation).
The level of darkness of the lips in photographs was also evaluated by the dermatologists using the specific color score chart described by Sharquie et al.15 Based on the chart, the level of darkness of the pigmentation was graded from 0 to 4 (where 0=no pigmentation, 1=light brown, 2=brown, 3=dark brown, and 4=black).
Subjective assessments. The level of satisfaction of the participants with the treatment received with respect to their perceived improvement in lip pigmentation was graded with a VAS ranging from 0 to 10 (where 0=extreme dissatisfaction and 10=satisfaction).
All patients were also asked about whether they experienced pain during laser therapy at end of their follow-up visit. The patients were asked to grade this pain using a VAS ranging from 0 to 10 (0=no pain at all and 10=severe pain).
Statistical analysis. Data were analyzed with the Statistical Package for the Social Sciences version 23 software program (IBM Corporation, Armonk, New York). Continuous variables were summarized as mean ± standard deviation values and paired t-tests were used to assess the differences in parameters before and after treatment. Categorical variables were presented as frequency and percentages. A p-value of less than 0.05 was considered to be statistically significant.
Results
A total of 24 Saudi women aged from 20 to 58 years (mean: 42.4±14.7 years) with Fitzpatrick Skin Types III to V were included in the present study. The baseline color score was 2.44±0.61 and the baseline VAS score was 6.09±1.53. The patients received a median of four sessions (range: 3–4 points) of laser therapy at a median of two-week intervals (range: 1–3 weeks) with 532-nm Nd:YAG laser at fluences of 2 to 2.4 J/cm2.
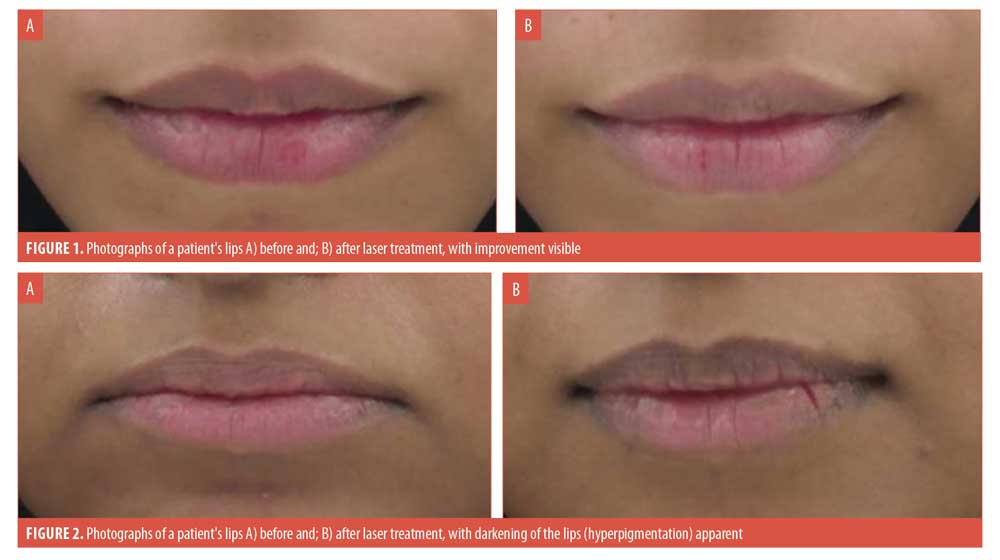
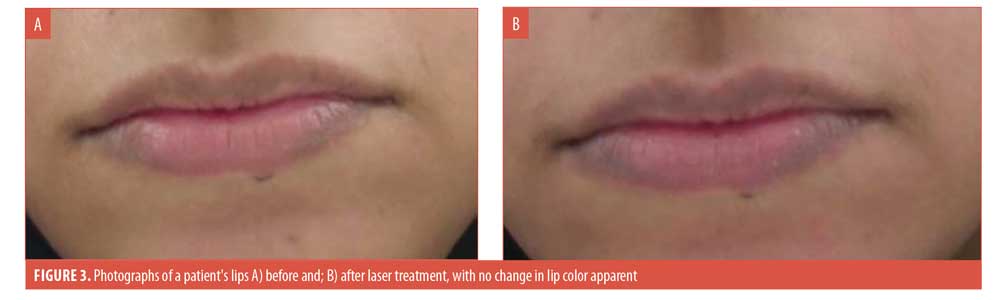
The mean VAS score decreased by 10.18 percent from a baseline of 6.09±1.53 to 5.47±1.72 (p=0.16). Also, the color score decreased by 9.38 percent from a baseline of 2.44±0.61 to 2.23±0.71 (p=0.23) after the last treatment. Overall, the photographic assessment indicated that, of the 24 study participants, 14 (58.3%) showed moderate to excellent improvement (Figure 1), four (16.7%) showed mild or no improvement (Figure 2), and six (25.0%) showed worsening/mottling of their pigmentation (Figure 3), respectively. The overall mean pain and satisfaction scores were 4.08±1.14 and 6.50±2.92, respectively.
The changes in mean pain and satisfaction scores according to the patient’s response to laser therapy are summarized in Table 1. The mean pain score was 4.67±1.63 among patients who had worsening of the pigmentation and 3.71±0.83 among those with moderate to excellent improvement following the laser therapy. However, the difference was not statistically significant (p=0.17). Also, the mean satisfaction score was 5.33±3.32 among patients who had worsening of the pigmentation and 7.43±2.17 among those with moderate to excellent improvement following the laser therapy, and this difference was similarly not statistically significant (p=0.18). Next, a correlation analysis between pain score, satisfaction score, color score, and photographic VAS score was performed, and there was a weak and insignificant negative correlation between pain and satisfaction scores (r=-0.22; p=0.31). Also, there was a was a weak and insignificant positive correlation between pain and color scores (r=0.32; p=0.13), and between pain and photographic VAS score (r=0.34; p=0.10).

Discussion
In this study, we found that the 532-nm Nd:YAG laser therapy led to a moderate to excellent improvement in lip pigmentation among Saudi women. However, we also found that two-fifths of the women had either no improvement or worsening of the lip pigmentation, with one quarter experiencing worsening of the lip pigmentation. While women who showed moderate to excellent improvements in lip pigment clearance had higher VAS, color, and satisfaction scores as well as lower pain scores, the differences did not reach statistical significance. Also, there was a weak correlation between pain and satisfaction, VAS, and color scores. About three-fifths of the women in this study showed moderate to excellent clearance of lip hyperpigmentation. Previous reports have suggested that there is a number of variations in the response of patients to 532-nm Nd:YAG laser therapy for hyperpigmentation in the lip and face.5–8 A case report from Saudi Arabia indicated that 532-nm Nd:YAG laser therapy showed excellent clearance of benign lip hyperpigmentation.5 A prospective, randomized, controlled trial that evaluated low-fluence QS 1,064-nm Nd:YAG laser therapy versus QS 532-nm Nd:YAG laser therapy in the treatment of hyperpigmented lips reported that all subjects in the 1,064-nm group demonstrated no change in scale evaluation; however, 6.7 percent experienced a one-point decline, 20 percent experienced a one-point improvement, and 73.33 percent experienced no change in the QS 532-nm group, and the differences between two laser groups were not significant (p=0.539).6 In addition, there was a case report which showed that the use of diode 940-nm laser therapy to treat lip hyperpigmentation was characterized by the formation of purpura for three days and the development of crusts, which sloughed off within four to seven days.11 In our study, we found that two-fifths of the women had either no improvement or worsening of lip pigmentation, with one-quarter experiencing worsening of lip pigmentation. This suggests that 532-nm Nd:YAG laser therapy for treating hyperpigmentation of the lip is not 100-percent effective as suggested by previous case reports.5,8 This is consistent with findings from other studies that assessed the effectiveness of 532-nm Nd:YAG laser therapy for the treatment of pigmentary lesions in the face, such as melasma, freckles, and frictional dermal melanosis.16–18 Beyond these, the use of 532-nm Nd:YAG laser therapy for hyperpigmentation of the lip has a number of undesirable side effects, such as mild to moderate lip swelling, burning sensation, itching, dryness, minimal bleeding, hypopigmentation, and purpura after the treatment.6,8,11 We strongly recommend ruling out any history of mucosal or cutaneous malignancies and examining the area of treatment before each treatment session.
Pain scores were lower and satisfaction scores were higher among patients who experienced excellent improvement in their lip hyperpigmentation following the administration of the 532-nm Nd:YAG laser. Also, these variables weakly correlated with the VAS and color scores. Although these relationships did not reach statistical significance, probably because of the small sample size of this study, this result still suggests that the greater the improvement, the more likely the patient will be satisfied and have less pain. A recent trial comparing 532-nm Nd:YAG and 1,064-nm Nd:YAG laser therapy showed that patients exposed to the 532-nm Nd:YAG laser had a significantly higher score, believed to be due to more reactions that were created during QS 532-nm treatment.6
Limitations. Importantly, our study has some limitations. First, the sample size of this study was small. This affected the findings of some of the associations assessed. Second, the patients were followed-up for a short period of three months. It is not clear whether the changes in lip pigmentation persisted beyond this period or improved subsequently. Further studies need to evaluate a larger number of patients and undertake a longer period of follow-up.
Conclusion
Our results indicate that low-fluence 532-nm Nd:YAG laser therapy is safe and has a modest effect in the treatment of hyperpigmentation of the lips. Further studies are needed to test different lasers, wavelengths, and energies in the management of dark lips, and caution is needed to avoid possible adverse events. Future studies should evaluate a larger number of patients and undertake a longer period of follow-up.
Acknowledgments
We extend special thanks to the staff nurses of the clinics of the Dermatology Department at King Khalid University Hospital for their help and support.
References
- Stephen ID, McKeegan AM. Lip colour affects perceived sex typicality and attractiveness of human faces. Perception. 2010;39(8): 1104–1110.
- Vachiramon V, McMichael AJ. Approaches to the evaluation of lip hyperpigmentation. Int J Dermatol. 2012;51(7):761–770.
- Lucena EE, Castro AC, Farias DB, et al. Prevalence of lip and perioral ephelides in beach workers. An Bras Dermatol. 2013;88(1):56–63.
- Ge Y, Jia G, Lin T. Q-switched Nd:YAG laser treatment for labial lentigines associated with Peutz-Jeghers syndrome. J Dtsch Dermatol Ges. 2015;13(6):551–555.
- Alharbi MA. Q-switched double-frequency Nd:YAG (532 nm) laser is an effective treatment for racial lip pigmentation. J Cosmet Dermatol. 2019;18(6):1672–1674.
- Limpjaroenviriyakul N, Jurairattanaporn N, Kamanamool N, et al. Low-fluence Q-switched Nd:YAG 1064-nm laser versus Q-switched Nd:YAG 532-nm laser in the treatment of hyperpigmented lips: a prospective, randomized, controlled, evaluator-blinded trial. Lasers Med Sci. 2019;35(1):165–171.
- Shi VY, Joo JS, Sharon VR. Multiple labial melanotic macules occurring after topical application of calcineurin inhibitors. Dermatol Online J. 2014;20(8):13030/qt45k792vj.
- Kunachak S, Kunachakr S, Kunachakr S, et al. An effective treatment of dark lip by frequency-doubled Q-switched Nd:YAG laser. Dermatol Surg. 2001;27(1):37–40.
- Goel A, Gatne V, Molvi M. QS Nd YAG Laser: single technology with multiple cosmetic applications. Gavin J Dermatol Res Ther. 2016;2016(1):42–45.
- Kirby W, Chen C, Desai A, Desai T. Successful treatment of cosmetic mucosal tattoos via Q-switched laser. Dermatol Surg. 2011;37(12):1767–1769.
- Vidyaa I, Farista H, Sana. Management of hyperpigmentation of lips with 940 nm diode laser: two case reports. Int Journal of Laser Dent. 2014;4(1):31–38.
- Al Shammrie F, Al Shammrie A. Pattern of skin disease in Hail region of Saudi Arabia. J Dermatol Dermatol Surg. 2017;21(2):62–65.
- Alshamrani HM, Alsolami MA, Alshehri AM, et al. Pattern of skin diseases in a university hospital in Jeddah, Saudi Arabia: age and sex distribution. Ann Saudi Med. 2019;39(1): 22–28.
- Alakloby OM. Pattern of skin diseases in Eastern Saudi Arabia. Saudi Med J. 2005;26(10):1607–1610.
- Sharquie KE, Al-Tikreety MM, Al-Mashhadani SA. Lactic acid as a new therapeutic peeling agent in melasma. Dermatol Surg. 2005;31(2):149–154.
- Kwon HH, Choi SC, Jung JY, Park GH. Combined treatment of melasma involving low-fluence Q-switched Nd:YAG laser and fractional microneedling radiofrequency. J Dermatolog Treat. 2019;30(4): 352–356.
- Ogbechie-Godec OA, Elbuluk N. Melasma: an up-to-date comprehensive review. Dermatol Ther (Heidelb). 2017;7(3):305–318.
- Al-Dhalimi MA, Maluki AH, Tauma A. Efficacy and safety of 532-nm and 1,064-nm Q-switched Nd:YAG laser treatment of frictional dermal melanosis over bony prominences (Lifa disease). Dermatol Surg. 2015;41(1):136–141.
