J Clin Aesthet Dermatol. 2026;19(2):43–46.
by Mara O’Connor, BS, and Peter Lio, MD
Ms. O’Connor and Dr. Lio are with the Northwestern University Feinberg School of Medicine in Chicago, Illinois.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no conflicts of interest relevant to the contents of this article.
ABSTRACT: The interrupted skin barrier seen in atopic dermatitis (AD) increases exposure to environmental antigens including those in food, which contributes to the increased prevalence of food allergies in the AD population. This relationship is further explored in the dual allergen exposure hypothesis, which examines the differences in immune system response between cutaneous and oral exposures. It was previously thought that avoiding early oral exposure would decrease the likelihood of developing food allergy; however, several studies have demonstrated that earlier oral exposure increases tolerance. On the other hand, exposure to food antigens through the skin activates the helper T-cell 2 pathway, which promotes development of allergy. There have also been several studies demonstrating that aggressive treatment of AD decreases the risk of developing food allergies. Based on these findings, there is concern that inclusion of food-based ingredients in personal care products, such as moisturizers that act as main components of AD treatment regimens, may increase the likelihood of developing allergies to the included foods. However, there are many commonly used food-based ingredients that have low rates of allergy, which raises the question of why some antigens are more likely to result in allergic sensitization while many others remain benign. Keywords: Atopic dermatitis, food allergy, dual allergen exposure hypothesis, food-based ingredients, personal care products
Introduction
Many patients are impacted by allergic diseases, including atopic dermatitis (AD) and food allergies, prompting extensive research into their interconnectedness.1,2 Research indicates that individuals with AD have a higher prevalence of food sensitization and food allergy compared with those without AD.3 Importantly, the risk of developing food allergies correlates with the severity and early onset of AD. Children with more severe AD are at a higher risk of developing food allergies.4,5 A compromised skin barrier in patients with AD facilitates the entry of allergens, including food proteins, leading to sensitization. This transcutaneous exposure is a key factor in the development of food allergies among these individuals.6
The dual allergen exposure hypothesis (DAEH) helps explain the well-established association between AD and food allergy. The DAEH proposes that early transcutaneous exposure to food allergens results in food allergies; however, early oral exposure to food allergens increases the chance of tolerance, protecting against allergy.7 As demonstrated in Figure 1, the hypothesized mechanism for transcutaneous exposure involves Langerhans cells presenting the food allergen to naïve T cells, leading to a helper T cell (TH) 2 response. The TH2 response is responsible for humoral immunity, including immunoglobulin E (IgE) production by B cells. In contrast, when food allergens are taken up by dendritic cells and presented to naïve T cells in the gastrointestinal tract, this prompts a Th1 and regulatory T-cell response, which helps to establish tolerance.7 Recent literature builds upon this theory, referencing conditions such as celiac disease that have both intestinal and cutaneous presentations, which suggests that sensitization through the skin may also have a direct impact on the gut.8
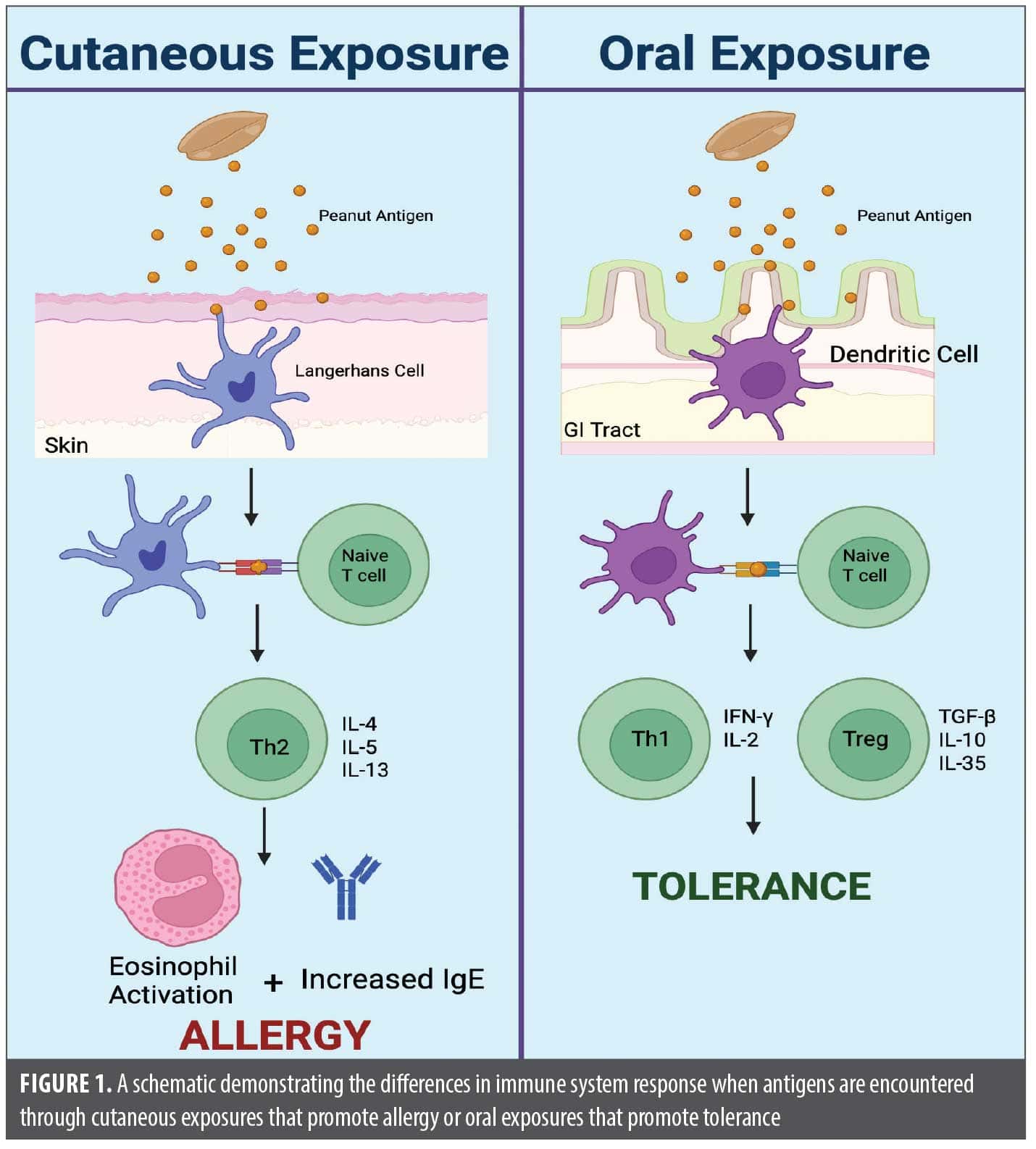
Historically, it was thought that early oral exposure to food allergens increased the likelihood of developing food allergy. Previous allergy guidelines encouraged excluding common allergenic foods for any child with a high risk of atopic disorders until the age of 3.9 However, these guidelines have been altered since being disputed by several studies. The Learning Early About Peanut Allergy study was a randomized trial that found that early introduction of peanuts into infant diets significantly reduced the likelihood of developing peanut allergy in high-risk children.10
Along with studying early food introduction, there have also been trials examining whether aggressive treatment of AD, ostensibly to improve the skin barrier and decrease cutaneous sensitization, could lead to a decrease in the development of food allergy. The two-step egg introduction for prevention of egg allergy in high-risk infants with eczema (PETIT) study investigated the stepwise introduction of egg plus aggressive treatment of patient’s atopic dermatitis.11 Remarkably, the PETIT study found that 8% of patients in the egg introduction plus aggressive AD treatment group developed egg allergy, compared to 38% of patients in the placebo group, suggesting that controlling the AD may lead to decreased risk of food allergy.11
Another study examined the development of egg allergy in patients with AD but solely compared enhanced early skin treatment of AD to conventional reactive treatment. The enhanced group received topical corticosteroids on both lesional and nonlesional skin as proactive therapy. The conventional reactive treatment group received topical corticosteroids only on lesional skin. The investigators found that enhanced treatment significantly reduced hen’s egg allergy, with 31.4% of the enhanced treatment group developing allergy, compared to 41.9% of the conventional treatment group.12 These studies support the notion that aggressive treatment of AD decreases the likelihood of developing food allergy. These findings align with DAEH and support that transcutaneous exposure is a main pathway to developing sensitization to food allergens.
Both the PETIT trial and the study examining egg allergy rates between different AD treatments suggest that treatment of AD can decrease the likelihood of developing food sensitization. One of the foundations of AD treatment is moisturization. However, given that many moisturizers contain food-related ingredients, it is important to consider the potential for sensitization, as patients will apply these to their damaged skin barrier. For example, the Avon Longitudinal Study of Parents and Children found a significant relationship with the use of skin preparations containing peanut oil and peanut allergy.4
Oat has moisturizing and anti-inflammatory properties and is one of the most common food ingredients found in skin care products, particularly those directed toward patients with eczema.13 A common oat-containing moisturizer was used in the STOP-AD trial. The STOP-AD trial investigated whether daily moisturizer use from birth to 2 months reduced the incidence of AD in high-risk infants at 12 months. Notably, the study found a lower incidence of AD in the oat-containing moisturizer group (32.8%) compared to the control group (46.4%).14
In response to the publication of this trial, Ryczaj et al15 warned of potential adverse effects of products with food-based ingredients. The oat-derived ingredients in skin care products could lead to immediate and delayed-type sensitivity reactions, with the majority of these cases occurring in patients with AD.16 Specifically, it was demonstrated that children with AD have high oat sensitization through oat atopy patch tests and skin prick tests. In this study, 32% of oat cream users had a positive atopy patch test in comparison to 0% in the nonusers group.17 There has also been one case report of anaphylaxis to oats after cutaneous sensitization through skin care products for treatment of AD.18 However, the STOP-AD trial authors responded with the results of a post-study survey, including food frequency and food diaries, of participating families at 1 year. In the survey results, no child who participated in the study, in either arm, reported to any allergic reactions to oats.19 The STOP-AD authors concluded: “Given the wide use of oat-containing products and the extremely low incidence of clinical reactions to oat protein, the real-world risk of percutaneous sensitization to oat leading to clinically significant reactions must be very low generally, and specifically much lower than to other more allergenic foods such as peanut.”19 In addition to oat, there are several other frequently used ingredients in moisturizers that have not been demonstrated to increased likelihood of food allergy. This calls into question why certain foods are more likely to elicit allergic reactions in moisturizers compared to others, despite the mechanistic understanding of sensitization through the skin.
Importantly, there are variations in the level of allergenicity between different foods. In the US, only 9 foods account for 90% of symptomatic allergic reactions from food proteins.20 Many sources report on 11 high-priority food allergens, and this list does not include oat or coconut.21,22 In fact, oat allergy is so infrequent that a multicenter food challenge study used oat for the placebo food challenge, which emphasizes the scarcity of oat allergy.23
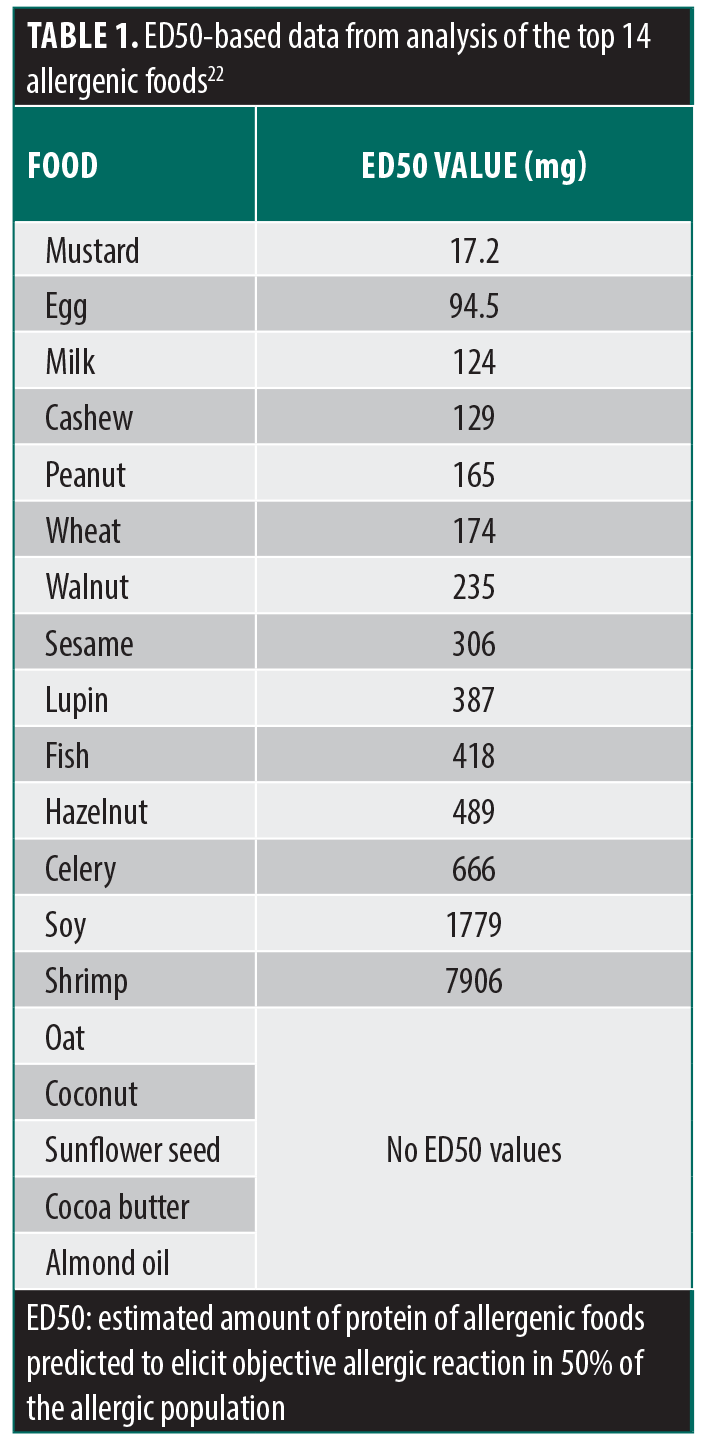
There are several studies examining the differences in allergic potency between foods by determining the Eliciting Dose values for these allergenic foods.21,22,24 The Eliciting Dose values are defined as the number of milligrams of total protein in an allergenic food predicted to elicit objective allergic reactions in 1% (ED01) of the allergic population. The discrete dose ED01 for several allergenic foods include 0.05mg of cashew, 0.2mg of peanut, 0.2mg of egg, 0.2mg of milk, 0.7mg of wheat, and 26.2mg of shrimp.24 These Eliciting Dose values allow for direct comparison of allergenic potencies between foods; cashew was found to be the most potent at the ED01 value. Table 1 shows the allergenicity of different foods relative to one another based on their eliciting dose for 50% of the allergic population (ED50). ED50 was specifically selected for this table based on the recommendation from the International Life Science Institute noting that ED50 is the best parameter for comparing the different potencies of food allergens.25 As the purpose of these studies is to establish reference doses for precautionary food labeling for the most common allergens, there are limited data regarding eliciting doses for other moisturizer-related ingredients such as oat. Further investigation is needed to understand the variability in the pathophysiology of both cutaneous and oral sensitization leading to differing ranges of food allergy risk in patients with AD and the general population.
Additionally, when patients are diagnosed with AD, food triggers are often suspected and elimination diets are considered.10 One study found that up to 71% of patients with AD had attempted some type of dietary modification to try to improve their disease.26 Given the frequency at which elimination diets are trialed by patients, the potential outcomes of the reintroduction of avoided foods are important to highlight when discussing treatment options with patients. A retrospective review of 298 patients with AD who previously tolerated foods found that when the eliminated foods were reintroduced, nearly 1 in 5 (19%) patients now demonstrated symptoms of acute IgE reactions. In other words, patients who were not allergic to foods at the start were now allergic after dietary elimination. Worse, 30% of these reactions met criteria for anaphylaxis.27 This study demonstrates the necessity of continuous oral exposure to food allergens to maintain tolerance and the potentially severe consequences of trialing elimination diets.
Overall, as the prevalence of AD and food allergy remains high, it is important to continue to investigate the relationship between these two diagnoses and the DAEH. Previous investigations have included trialing prevention strategies such as enhanced treatment of AD with topical corticosteroids and adapting allergy guidelines to begin earlier introduction of highly allergenic foods. The STOP-AD trial studied daily emollient use for the prevention of AD and maintenance of the skin barrier to block cutaneous sensitization of food allergens. Even though they used a moisturizer that contained oat ingredients, no cases of oat allergy were reported, and oat allergy remains rare in the population despite widespread addition of oat to skincare products. This study, along with the established common use of food-related ingredients in skin care products, raises the question as to why we are more susceptible to develop sensitization to certain foods.
References
- Togias A, Cooper SF, Acebal ML, et al. Addendum guidelines for the prevention of peanut allergy in the United States: report of the National Institute of Allergy and Infectious Diseases-sponsored expert panel. J Allergy Clin Immunol. 2017;139(1):29-44.
- Sicherer SH, Sampson HA. Food allergy: a review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J Allergy Clin Immunol. 2018;141(1):41-58.
- Tsakok T, Marrs T, Mohsin M, et al. Does atopic dermatitis cause food allergy? A systematic review. J Allergy Clin Immunol. 2016;137(4):1071-1078.
- Lack G, Fox D, Northstone K, Golding J; Avon Longitudinal Study of Parents and Children Study Team. Factors associated with the development of peanut allergy in childhood. N Engl J Med. 2003;348(11):977-985.
- Thompson MM, Hanifin JM. Effective therapy of childhood atopic dermatitis allays food allergy concerns. J Am Acad Dermatol. 2005;53(2 Suppl 2):S214-S219.
- Elias PM. Primary role of barrier dysfunction in the pathogenesis of atopic dermatitis. Exp Dermatol. 2018;27(8):847-851.
- Lack G. Epidemiologic risks for food allergy. J Allergy Clin Immunol. 2008;121(6):1331-1336.
- Davis KL, Claudio-Etienne E, Frischmeyer-Guerrerio PA. Atopic dermatitis and food allergy: more than sensitization. Mucosal Immunol. 2024;17(5):1128-1140.
- Lio PA. Updated guidelines on peanut allergy prevention in infants with atopic dermatitis. Cutis. 2018;101(6):398-399.
- Du Toit G, Roberts G, Sayre PH, et al; LEAP Study Team. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372(9):803-813.
- Natsume O, Kabashima S, Nakazato J, et al; PETIT Study Team. Two-step egg introduction for prevention of egg allergy in high-risk infants with eczema (PETIT): a randomised, double-blind, placebo-controlled trial. Lancet. 2017;389(10066):276-286.
- Yamamoto-Hanada K, Kobayashi T, Mikami M, et al; PACI Study Collaborators. Enhanced early skin treatment for atopic dermatitis in infants reduces food allergy. J Allergy Clin Immunol. 2023;152(1):126-135.
- Paudel D, Dhungana B, Caffe M, Krishnan P. A review of health-beneficial properties of oats. Foods. 2021;10(11):2591.
- Ní Chaoimh C, Lad D, Nico C, et al. Early initiation of short-term emollient use for the prevention of atopic dermatitis in high-risk infants-the STOP-AD randomised controlled trial. Allergy. 2023;78(4):984-994.
- Ryczaj K, Kulus M, Feleszko W. Food allergens in emollients. Allergy. 2023;78(4):1121-1122.
- Pootongkam S, Nedorost S. Oat and wheat as contact allergens in personal care products. Dermatitis. 2013;24(6):291-295.
- Boussault P, Léauté-Labrèze C, Saubusse E, et al. Oat sensitization in children with atopic dermatitis: prevalence, risks and associated factors. Allergy. 2007;62(11):1251 1256.
- Radhakrishna N, Prickett S, Phan T, Rolland JM, Puy R, O’Hehir RE. Anaphylaxis to oats after cutaneous sensitization by oatmeal in skin products used for the treatment of atopic dermatitis. J Allergy Clin Immunol Pract. 2016;4(1):152-153.
- Irvine AD, Hourihane JOB; STOP-AD Team. Reply to correspondence “Food allergens to emollients”. Allergy. 2023;78(4):1123.
- Iglesia EGA, Kwan M, Virkud YV, Iweala OI. Management of food allergies and food-related anaphylaxis. JAMA. 2024;331(6):510-521.
- Allen KJ, Remington BC, Baumert JL, et al. Allergen reference doses for precautionary labeling (VITAL 2.0): clinical implications. J Allergy Clin Immunol. 2014;133(1):156-164.
- Taylor SL, Baumert JL, Kruizinga AG, et al. Establishment of Reference Doses for residues of allergenic foods: report of the VITAL Expert Panel. Food Chem Toxicol. 2014;63:9-17.
- Purington N, Chinthrajah RS, Long A, et al. Eliciting dose and safety outcomes from a large dataset of standardized multiple food challenges. Front Immunol. 2018;9:2057.
- Remington BC, Westerhout J, Meima MY, et al. Updated population minimal eliciting dose distributions for use in risk assessment of 14 priority food allergens. Food Chem Toxicol. 2020;139:111259.
- Houben G, Burney P, Chan CH, et al. Prioritisation of allergenic foods with respect to public health relevance: report from an ILSI Europe Food Allergy Task Force Expert Group. Food Chem Toxicol. 2016;89:8-18.
- Jensen P. Use of alternative medicine by patients with atopic dermatitis and psoriasis. Acta Derm Venereol. 1990;70(5):421-424.
- Chang A, Robison R, Cai M, Singh AM. Natural history of food-triggered atopic dermatitis and development of immediate reactions in children. J Allergy Clin Immunol Pract. 2016;4(2):229-236.e1.
Feeding the Skin? Foods in Personal Care Products and the Risk of Allergy
Categories:
J Clin Aesthet Dermatol. 2026;19(2):43–46.
by Mara O’Connor, BS, and Peter Lio, MD
Ms. O’Connor and Dr. Lio are with the Northwestern University Feinberg School of Medicine in Chicago, Illinois.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no conflicts of interest relevant to the contents of this article.
ABSTRACT: The interrupted skin barrier seen in atopic dermatitis (AD) increases exposure to environmental antigens including those in food, which contributes to the increased prevalence of food allergies in the AD population. This relationship is further explored in the dual allergen exposure hypothesis, which examines the differences in immune system response between cutaneous and oral exposures. It was previously thought that avoiding early oral exposure would decrease the likelihood of developing food allergy; however, several studies have demonstrated that earlier oral exposure increases tolerance. On the other hand, exposure to food antigens through the skin activates the helper T-cell 2 pathway, which promotes development of allergy. There have also been several studies demonstrating that aggressive treatment of AD decreases the risk of developing food allergies. Based on these findings, there is concern that inclusion of food-based ingredients in personal care products, such as moisturizers that act as main components of AD treatment regimens, may increase the likelihood of developing allergies to the included foods. However, there are many commonly used food-based ingredients that have low rates of allergy, which raises the question of why some antigens are more likely to result in allergic sensitization while many others remain benign. Keywords: Atopic dermatitis, food allergy, dual allergen exposure hypothesis, food-based ingredients, personal care products
Introduction
Many patients are impacted by allergic diseases, including atopic dermatitis (AD) and food allergies, prompting extensive research into their interconnectedness.1,2 Research indicates that individuals with AD have a higher prevalence of food sensitization and food allergy compared with those without AD.3 Importantly, the risk of developing food allergies correlates with the severity and early onset of AD. Children with more severe AD are at a higher risk of developing food allergies.4,5 A compromised skin barrier in patients with AD facilitates the entry of allergens, including food proteins, leading to sensitization. This transcutaneous exposure is a key factor in the development of food allergies among these individuals.6
The dual allergen exposure hypothesis (DAEH) helps explain the well-established association between AD and food allergy. The DAEH proposes that early transcutaneous exposure to food allergens results in food allergies; however, early oral exposure to food allergens increases the chance of tolerance, protecting against allergy.7 As demonstrated in Figure 1, the hypothesized mechanism for transcutaneous exposure involves Langerhans cells presenting the food allergen to naïve T cells, leading to a helper T cell (TH) 2 response. The TH2 response is responsible for humoral immunity, including immunoglobulin E (IgE) production by B cells. In contrast, when food allergens are taken up by dendritic cells and presented to naïve T cells in the gastrointestinal tract, this prompts a Th1 and regulatory T-cell response, which helps to establish tolerance.7 Recent literature builds upon this theory, referencing conditions such as celiac disease that have both intestinal and cutaneous presentations, which suggests that sensitization through the skin may also have a direct impact on the gut.8
Historically, it was thought that early oral exposure to food allergens increased the likelihood of developing food allergy. Previous allergy guidelines encouraged excluding common allergenic foods for any child with a high risk of atopic disorders until the age of 3.9 However, these guidelines have been altered since being disputed by several studies. The Learning Early About Peanut Allergy study was a randomized trial that found that early introduction of peanuts into infant diets significantly reduced the likelihood of developing peanut allergy in high-risk children.10
Along with studying early food introduction, there have also been trials examining whether aggressive treatment of AD, ostensibly to improve the skin barrier and decrease cutaneous sensitization, could lead to a decrease in the development of food allergy. The two-step egg introduction for prevention of egg allergy in high-risk infants with eczema (PETIT) study investigated the stepwise introduction of egg plus aggressive treatment of patient’s atopic dermatitis.11 Remarkably, the PETIT study found that 8% of patients in the egg introduction plus aggressive AD treatment group developed egg allergy, compared to 38% of patients in the placebo group, suggesting that controlling the AD may lead to decreased risk of food allergy.11
Another study examined the development of egg allergy in patients with AD but solely compared enhanced early skin treatment of AD to conventional reactive treatment. The enhanced group received topical corticosteroids on both lesional and nonlesional skin as proactive therapy. The conventional reactive treatment group received topical corticosteroids only on lesional skin. The investigators found that enhanced treatment significantly reduced hen’s egg allergy, with 31.4% of the enhanced treatment group developing allergy, compared to 41.9% of the conventional treatment group.12 These studies support the notion that aggressive treatment of AD decreases the likelihood of developing food allergy. These findings align with DAEH and support that transcutaneous exposure is a main pathway to developing sensitization to food allergens.
Both the PETIT trial and the study examining egg allergy rates between different AD treatments suggest that treatment of AD can decrease the likelihood of developing food sensitization. One of the foundations of AD treatment is moisturization. However, given that many moisturizers contain food-related ingredients, it is important to consider the potential for sensitization, as patients will apply these to their damaged skin barrier. For example, the Avon Longitudinal Study of Parents and Children found a significant relationship with the use of skin preparations containing peanut oil and peanut allergy.4
Oat has moisturizing and anti-inflammatory properties and is one of the most common food ingredients found in skin care products, particularly those directed toward patients with eczema.13 A common oat-containing moisturizer was used in the STOP-AD trial. The STOP-AD trial investigated whether daily moisturizer use from birth to 2 months reduced the incidence of AD in high-risk infants at 12 months. Notably, the study found a lower incidence of AD in the oat-containing moisturizer group (32.8%) compared to the control group (46.4%).14
In response to the publication of this trial, Ryczaj et al15 warned of potential adverse effects of products with food-based ingredients. The oat-derived ingredients in skin care products could lead to immediate and delayed-type sensitivity reactions, with the majority of these cases occurring in patients with AD.16 Specifically, it was demonstrated that children with AD have high oat sensitization through oat atopy patch tests and skin prick tests. In this study, 32% of oat cream users had a positive atopy patch test in comparison to 0% in the nonusers group.17 There has also been one case report of anaphylaxis to oats after cutaneous sensitization through skin care products for treatment of AD.18 However, the STOP-AD trial authors responded with the results of a post-study survey, including food frequency and food diaries, of participating families at 1 year. In the survey results, no child who participated in the study, in either arm, reported to any allergic reactions to oats.19 The STOP-AD authors concluded: “Given the wide use of oat-containing products and the extremely low incidence of clinical reactions to oat protein, the real-world risk of percutaneous sensitization to oat leading to clinically significant reactions must be very low generally, and specifically much lower than to other more allergenic foods such as peanut.”19 In addition to oat, there are several other frequently used ingredients in moisturizers that have not been demonstrated to increased likelihood of food allergy. This calls into question why certain foods are more likely to elicit allergic reactions in moisturizers compared to others, despite the mechanistic understanding of sensitization through the skin.
Importantly, there are variations in the level of allergenicity between different foods. In the US, only 9 foods account for 90% of symptomatic allergic reactions from food proteins.20 Many sources report on 11 high-priority food allergens, and this list does not include oat or coconut.21,22 In fact, oat allergy is so infrequent that a multicenter food challenge study used oat for the placebo food challenge, which emphasizes the scarcity of oat allergy.23
There are several studies examining the differences in allergic potency between foods by determining the Eliciting Dose values for these allergenic foods.21,22,24 The Eliciting Dose values are defined as the number of milligrams of total protein in an allergenic food predicted to elicit objective allergic reactions in 1% (ED01) of the allergic population. The discrete dose ED01 for several allergenic foods include 0.05mg of cashew, 0.2mg of peanut, 0.2mg of egg, 0.2mg of milk, 0.7mg of wheat, and 26.2mg of shrimp.24 These Eliciting Dose values allow for direct comparison of allergenic potencies between foods; cashew was found to be the most potent at the ED01 value. Table 1 shows the allergenicity of different foods relative to one another based on their eliciting dose for 50% of the allergic population (ED50). ED50 was specifically selected for this table based on the recommendation from the International Life Science Institute noting that ED50 is the best parameter for comparing the different potencies of food allergens.25 As the purpose of these studies is to establish reference doses for precautionary food labeling for the most common allergens, there are limited data regarding eliciting doses for other moisturizer-related ingredients such as oat. Further investigation is needed to understand the variability in the pathophysiology of both cutaneous and oral sensitization leading to differing ranges of food allergy risk in patients with AD and the general population.
Additionally, when patients are diagnosed with AD, food triggers are often suspected and elimination diets are considered.10 One study found that up to 71% of patients with AD had attempted some type of dietary modification to try to improve their disease.26 Given the frequency at which elimination diets are trialed by patients, the potential outcomes of the reintroduction of avoided foods are important to highlight when discussing treatment options with patients. A retrospective review of 298 patients with AD who previously tolerated foods found that when the eliminated foods were reintroduced, nearly 1 in 5 (19%) patients now demonstrated symptoms of acute IgE reactions. In other words, patients who were not allergic to foods at the start were now allergic after dietary elimination. Worse, 30% of these reactions met criteria for anaphylaxis.27 This study demonstrates the necessity of continuous oral exposure to food allergens to maintain tolerance and the potentially severe consequences of trialing elimination diets.
Overall, as the prevalence of AD and food allergy remains high, it is important to continue to investigate the relationship between these two diagnoses and the DAEH. Previous investigations have included trialing prevention strategies such as enhanced treatment of AD with topical corticosteroids and adapting allergy guidelines to begin earlier introduction of highly allergenic foods. The STOP-AD trial studied daily emollient use for the prevention of AD and maintenance of the skin barrier to block cutaneous sensitization of food allergens. Even though they used a moisturizer that contained oat ingredients, no cases of oat allergy were reported, and oat allergy remains rare in the population despite widespread addition of oat to skincare products. This study, along with the established common use of food-related ingredients in skin care products, raises the question as to why we are more susceptible to develop sensitization to certain foods.
References
Share:
Recent Articles:
Challenges of Treating Recurrent Merkel Cell Carcinoma in the Rural US: A Case Report
Case Series: Successful Management of Refractory Plaque Psoriasis With Combined Laser Therapy and Topical Roflumilast in 2 Patients
Quality of Life in Young Adults With Acne
From Clinic to Conference: A Practical Guide for Dermatology PAs and NPs Preparing a Scientific Poster
Letter to the Editor: April 2026
Categories:
Recent Articles:
Plant Exosome Injection: A New Boost for Postlaser Vascular Repair
Hispanic and Latino Ethnicity and Acculturation: Influences on Skin Cancer Prevention Attitudes and Behaviors
Optimizing Surgical Tray Setup and Instrument Selection in Dermatologic Surgery
Severe Nail Psoriasis and Early Psoriatic Arthritis: Illustrative Cases Confirming That Severity of Disease Is Not Solely Dependent on the Extent of Body Surface Area
The Treatment of Perioral (Periorificial) Dermatitis With Topical Roflumilast 0.3% Cream: An Illustrative Case Study With Rapid Onset and Prolonged Remission