J Clin Aesthet Dermatol. 2026;19(4):35–37.
by Simona A. Alomary, BA; Nicole J. Baker, BS; Nicholas Schell, BA, MCIT; Temitayo Ogunleye, MD; Susan Taylor, MD; and Heather Milbar, MD
Ms. Alomary is with Rutgers New Jersey Medical School, Newark, New Jersey, and Sidney Kimmel Medical College at Thomas Jefferson University, Philadelphia, Pennsylvania. Ms. Baker is with Sidney Kimmel Medical College at Thomas Jefferson University and the University of Pennsylvania Perelman School of Medicine, Department of Dermatology, Philadelphia, Pennsylvania.
Mr. Schell and Drs. Ogunleye, Taylor, and Milbar are with the University of Pennsylvania Perelman School of Medicine, Department of Dermatology, Philadelphia, Pennsylvania.
FUNDING: No funding was provided for this article.
DISCLOSURES: Dr. Taylor has served as a consultant, advisory board member, and/or speaker for AbbVie, Arcutis, Armis Scientific, Avita, Beiersdorf, Biorez, Bristol Myers Squibb, Cara
Therapeutics, Dior, EPI Health, Evolus, Galderma, GloGetter, Hugel America, Incyte, Johnson & Johnson, Lilly, L’Oréal USA, MedScape, MJH LifeSciences, Pfizer, Piction Health, Sanofi, Scientis US, UCB, and Vichy Laboratories. She has received royalties from McGraw-Hill. She has served as an investigator for Allergan, Concert Pharmaceuticals/Sun Pharma, Croma-Pharma GmbH, Lilly, and Pfizer. Dr. Ogunleye has served as a consultant, advisory board member, and/or speaker for Beiersdorf, MJH LifeSciences, Dermatology Times, and Health Central. Dr. Milbar and Ms. Alomary, Ms. Baker, and Mr. Schell have no conflicts of interest to disclose.
ABSTRACT: Objective: To compare thyrotropin (TSH) levels across nonscarring alopecia (NSA) subtypes—alopecia areata (AA), telogen effluvium (TE), and androgenetic alopecia (AGA)—in a primary analysis of all patients, a secondary analysis of euthyroid patients stratified into low-normal (0.5–2.5 mIU/L) and high-normal (2.5–4.5 mIU/L) categories, and a tertiary analysis of patients with thyroid dysfunction. Methods: A retrospective chart review was conducted on patients diagnosed with AA, TE, or AGA at a single academic dermatology department between August 2017 and August 2024. TSH values were drawn within 3 months of the initial visit, and patient demographics were extracted from the electronic medical record. Patients with thyroid dysfunction were excluded from the secondary analysis. TSH values within the euthyroid range were stratified into low-normal and high-normal categories. A tertiary analysis of patients with frank thyroid dysfunction was later conducted. Descriptive statistics, Kruskal-Wallis tests, and Bonferroni-corrected pairwise comparisons were performed. Results: Among the 1,411 patients initially identified, most TSH values were within the euthyroid range. In a secondary analysis of 1,291 euthyroid patients, there was no significant difference in TSH distribution across NSA subtypes, including when stratified into low-normal vs high-normal categories. Among patients with thyroid dysfunction (8.5%), those with TE had the highest relative proportions of both hypothyroidism (5.0%) and hyperthyroidism (5.5%). Black or African American patients with AA and TE had lower mean TSH levels than White patients; however, this was not observed in AGA. Limitations: Limitations of this study include the retrospective design and the potential for incomplete or biased medical records. Conclusion: TSH levels among patients with NSA did not vary significantly based on NSA subtype. Racial differences in mean TSH were observed in AA and TE, but the clinical significance remains uncertain. Keywords: TSH, nonscarring alopecia, alopecia areata, telogen effluvium, androgenetic alopecia
Introduction
Thyroid hormones are known to regulate key aspects of hair growth and cycling through genomic and nongenomic pathways.1 Both hypothyroidism and hyperthyroidism have been linked to diffuse hair shedding and structural hair changes.2 In dermatologic practice, clinicians often screen patients with nonscarring alopecia (NSA) for thyroid dysfunction; however, the relevance of subtle thyrotropin (TSH) variations within the euthyroid range remains unclear. Reproductive endocrinology literature has suggested that high-normal TSH may reflect subclinical hypothyroidism, with potential associations to infertility and adverse pregnancy outcomes.3 Given the established role of thyroid hormones in hair growth and follicle cycling, these findings raise the possibility that variation in TSH levels within the euthyroid range could also be relevant to hair loss. Prior studies have documented associations between thyroid dysfunction and various forms of NSA; however, TSH variation within the euthyroid range among patients with hair loss has not been well characterized.4,5 Despite this, there remains uncertainty about how dermatologists should interpret subtle TSH variations that fall within normal laboratory ranges. Decisions about screening for thyroid dysfunction, how to interpret borderline TSH values, and whether subclinical abnormalities warrant closer monitoring or intervention vary widely in practice. Therefore, this study aims to explore whether TSH levels differ across NSA subtypes—alopecia areata (AA), telogen effluvium (TE), and androgenetic alopecia (AGA)—and to assess whether any observed variation has potential clinical relevance for screening or management.
A retrospective chart review was conducted on patients diagnosed with AA, TE, or AGA at a single academic dermatology department between August 2017 and August 2024. TSH values were drawn within 3 months of the initial visit, and patient demographics were extracted from the electronic medical record. TSH values within the euthyroid range were stratified into low-normal (0.5–2.5 mIU/L) and high-normal (2.5–4.5 mIU/L) categories. Descriptive statistics, Kruskal-Wallis tests, and Bonferroni-corrected pairwise comparisons were performed. Patients with frank thyroid dysfunction (TSH <0.5 or >4.5 mIU/L) or documented thyroid disease (via International Statistical Classification of Diseases, Tenth Revision codes) were excluded from a secondary analysis but were included in a tertiary analysis.
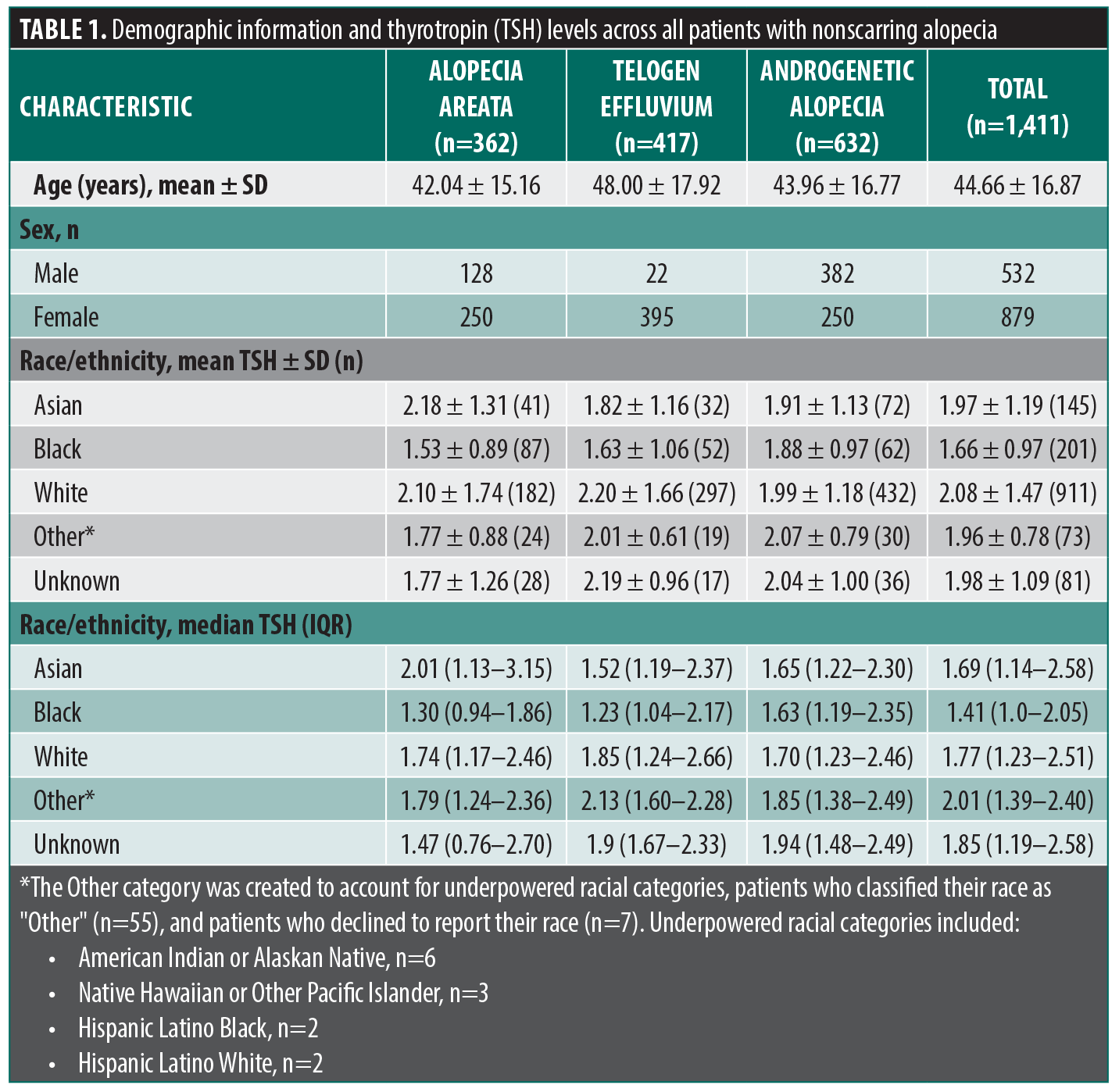
A total of 1,411 patients with NSA were identified as part of the primary analysis: 362 with AA, 417 with TE, and 632 with AGA (Table 1). Nearly all patients’ TSH levels (91.5%) were within the euthyroid range (0.5–4.5 mIU/L). Patients identifying as Black or African American had lower TSH levels than White patients in both AA (Black: 0.37–5.76 mIU/L; White: 0.02–15.4 mIU/L; P<0.001) and TE (Black: 0.15–5.22 mIU/L; White: <0.01–15.67 mIU/L; P=0.002), with no racial differences observed for AGA (Black: 0.43–4.29 mIU/L; White: <0.01–9.15 mIU/L; P=0.49). There was no overall difference in TSH levels across all 3 NSA subtypes (P=0.09).
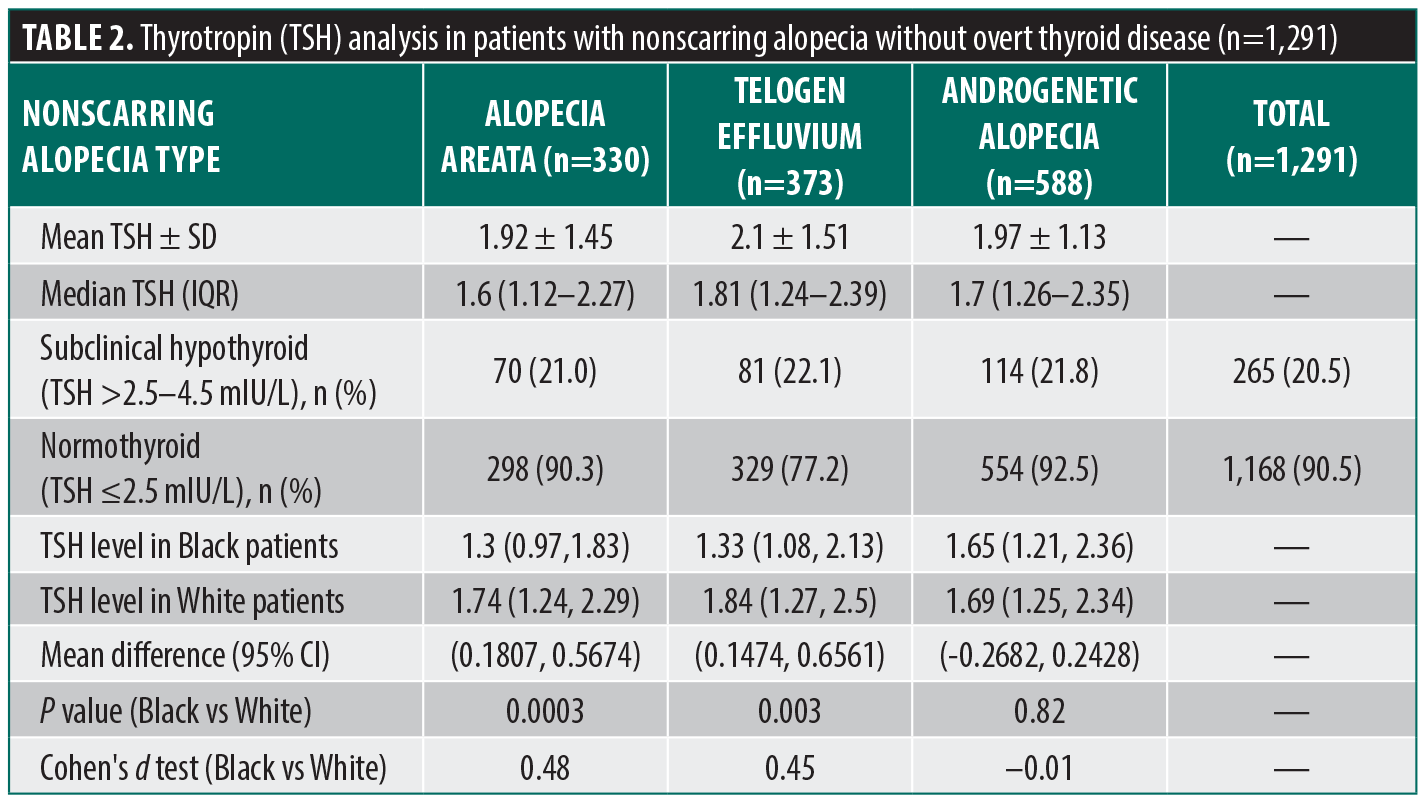
After excluding patients with overt thyroid dysfunction or documented thyroid disease, 1,291 patients remained in the secondary analysis: 330 patients with AA, 373 with TE, and 588 with AGA (Table 2). There was no significant difference in the distribution of NSA subtypes across the euthyroid range (P=0.94). Racial differences in TSH levels persisted: Black patients had lower mean TSH than White patients in AA (1.48 vs 1.85; P=0.0003) and TE (1.59 vs 1.99; P=0.003), with no difference in AGA (P=0.82). These racial differences are consistent with trends reported in prior endocrinologic studies.5,6 Exploratory analyses stratifying by gender and use of AGA as a control group showed no significant differences across subtypes.
Among the 8.5% of patients with abnormal TSH values, there were no statistically significant differences across NSA subtypes as part of a tertiary analysis. Based on patient counts (Table 3), AGA accounted for the largest number of hypothyroid patients (n=24), while TE accounted for the largest number of hyperthyroid patients (n=21). When proportions were calculated relative to each NSA subtype cohort size, patients with TE showed slightly higher rates of hyperthyroidism (5.0%) and hypothyroidism (5.5%) than AGA (3.2% and 3.8%) or AA (4.7% and 4.1%). This suggests that although AGA may be the most common NSA subtype overall, thyroid dysfunction was disproportionately represented among patients with TE. This finding is consistent with existing literature that identify TE as the NSA subtype most often associated with thyroid dysfunction.6,7
These findings suggest that variation across the euthyroid spectrum is not significantly associated with development of NSA. Notably, even in the primary analysis that included both hyperthyroid and hypothyroid patients, TSH levels in patients with TE did not differ significantly from that in patients with AA or AGA, despite TE being the hair-loss pattern most often considered in the context of thyroid dysfunction. This may reflect the timing of laboratory testing relative to the onset of TE, as TSH abnormalities may be transient. In addition, patients with AA or TE who identified as Black consistently had lower mean TSH levels than their White counterparts. The clinical significance of these differences is not fully understood; however, prior endocrinologic studies have reported similar population trends and propose several potential mechanisms, including genetic influences, variations in dietary iodine intake and deiodinase activity, and racial differences in physiologic thresholds for TSH suppression.8–10 Although variation in TSH levels within the euthyroid spectrum has been considered clinically meaningful in other areas of medicine such as reproductive endocrinology, this was not observed in patients with NSA.
Overall, these results support an individualized approach to screening for thyroid disease in patients with NSA. In clinical practice, individualized screening may involve reserving thyroid testing for patients who exhibit symptoms suggestive of thyroid dysfunction or have a personal or family history of autoimmune disease. This approach helps avoid unnecessary testing in asymptomatic patients while ensuring that thyroid testing is pursued when it is clinically likely to affect management. As universal thyroid screening is not recommended in current dermatology guidelines, the results of this study support a selective symptom-based strategy.
As a retrospective study, this analysis is limited by the potential for incomplete or biased medical records, which may hinder accurate assessment of thyroid function and its temporal relationship to hair loss onset. TSH values were obtained within 3 months of the initial visit; however, thyroid function can fluctuate over time, thus a single time-point measurement of TSH may not accurately reflect longitudinal thyroid status. Some patients may have also had laboratory testing performed outside our institution, introducing the possibility of minor interassay variability. Race and ethnicity were extracted from the medical record and may be subject to misclassification if patient-reported information was entered incorrectly. Prospective studies are warranted to determine whether treatment of subclinical thyroid disease influences clinical outcomes in patients with hair loss.
References
- Popa A, Carsote M, Cretoiu D, Dumitrascu MC, Nistor CE, Sandru F. Study of the thyroid profile of patients with alopecia. J Clin Med. 2023;12(3):1115.
- Hussein RS, Atia T, Bin Dayel S. Impact of thyroid dysfunction on hair disorders. Cureus. 2023;15(8):e43266.
- Maraka S, Singh Ospina NM, Mastorakos G, O’Keeffe DT. Subclinical hypothyroidism in women planning conception and during pregnancy: who should be treated and how? J Endocr Soc. 2018;2(6):533–546.
- Jain VK, Kataria U, Dayal S. Study of diffuse alopecia in females. Indian J Dermatol Venereol Leprol. 2000;66(2):65–68.
- Vincent M, Yogiraj K. A descriptive study of alopecia patterns and their relation to thyroid dysfunction. Int J Trichology. 2013;5(1):57–60.
- Bin Dayel S, Hussein RS, Atia T, et al. Is thyroid dysfunction a common cause of telogen effluvium?: a retrospective study. Medicine (Baltimore). 2024;103(1):e36803.
- Cheng T, Fang H, Wang Y, et al. The diagnostic value of serum ferritin for telogen effluvium: a cross-sectional comparative study. Clin Cosmet Investig Dermatol. 2021;14:137–141.
- Surks MI, Boucai L. Age- and race-based serum thyrotropin reference limits. J Clin Endocrinol Metab. 2010;95(2):496–502.
- Schectman JM, Kallenberg GA, Hirsch RP, Shumacher RJ. Report of an association between race and thyroid stimulating hormone level. Am J Public Health. 1991;81(4):505–506.
- Koch L. Thyroid gland: TSH reference limits specific for age, sex and ethnicity. Nat Rev Endocrinol. 2011;7(2):61.


Optimizing Surgical Tray Setup and Instrument Selection in Dermatologic Surgery