 by Lily I. Jiang, PhD; Peter D. Hino, MD, FAAD; Lydia Parker, MD; Thomas J. Stephens, PhD; John McCook, BSC (Pharm); and Vincent Gotz, MS (Pharm), MBA
by Lily I. Jiang, PhD; Peter D. Hino, MD, FAAD; Lydia Parker, MD; Thomas J. Stephens, PhD; John McCook, BSC (Pharm); and Vincent Gotz, MS (Pharm), MBA
Drs. Jiang, Hino, and Stephens are with Thomas J. Stephens and Associates, Inc. in Richardson, Texas. Dr. Parker is with the Parker Skin and Aesthetic Clinic in Beachwood, Ohio. Mr. McCook is with Discovery Partners LLC in Frisco, Texas. Mr. Gotz is with MDRejuvena, Inc. in Carlsbad, California.
Abstract: Objective. The objective of this study was to assess clinical safety and efficacy of a novel acne treatment regimen in adult women.
Methods. Participants in the study included an ethnically diverse group of adult women (n=24) with mild-to-moderate acne who were treated twice daily with a topical regimen (cleanser, acne cream, and rebalancing gel) for eight weeks. Following baseline assessments, subjects returned to clinic at Weeks 2, 4, and 8 for clinical assessments and self-assessment questionnaires.
Results. Twenty-one of the 24 enrolled women completed the eight-week clinical trial. Statistically significant clinical improvements were seen in both acne and aging parameters over time. The product regimen was well tolerated without adverse reactions commonly seen with topical acne products.
Conclusion. The regimen demonstrated efficacy and tolerability in adult women with acne and signs of skin aging.
Keywords: Adult female acne, acne treatment, liposomal sodium copper chlorophyllin complex, salicylic acid
J Clin Aesthet Dermatol. 2018;11(6):46–51
Introduction
Acne vulgaris, a chronic, inflammatory disease of the pilosebaceous follicles, is a pervasive skin disorder that presents regardless of sex, skin color, or ethnicity. An estimated 40 to 50 million Americans suffer from the disorder, including more than 85 percent of teenagers.1–3 However, while acne is typically perceived as a disease of adolescents, adult acne remains a significant problem. Studies have shown a worldwide prevalence of female adult acne to be as high as 41 percent, with adult acne being more prevalent in women than men.4–6 Acne is the most common reason for dermatologist visits in the United States for individuals aged 15 to 44 years.7 As seen in adolescent acne, adult acne (particularly among women) can have a negative impact on social, emotional, and psychological well-being.8–11 For these reasons, effective treatments are important.
All of the commonly employed topical medications for acne have certain limitations to treatment: topical benzoyl peroxide can cause dryness, cutaneous irritation, and staining; oral and topical antibiotics can lead to bacterial resistance; and retinoid use can result in dryness, scaling, irritation, burning, stinging, and erythema. Thus, there is need for a new topical acne treatment with good efficacy and a strong safety profile for women with mild-to-moderate acne. Additionally, antiaging effects would likely be considered added benefits to an effective topical acne treatment product.
Sodium copper chlorophyllin complex is a semisynthetic mixture of the water-soluble sodium salt of copper chlorophyllin and small molecule analogs derived from plant chlorophyll that possesses a unique combination of anti-inflammatory, antibacterial, and antioxidant activities. Sodium copper chlorophyllin complex has been used topically for many years to aid in wound healing, but the ingredient has not been used until recently as a cosmeceutical in topical products.
Topical formulations containing a liposomal dispersion of sodium copper chlorophyllin complex at concentrations of 0.025 to 0.1% have shown promise in the clinical treatment of various dermatologic conditions.12–14 A pilot study demonstrated that sodium copper chlorophyllin complex was clinically effective and well-tolerated for the treatment of mild-to-moderate photodamage and solar lentigines when used for eight weeks.12 In uncontrolled case studies, topical application of formulations containing sodium copper chlorophyllin complex demonstrated a reduction in facial redness, decreased pore size, and improved skin texture.14 Another study reported that topical copper chlorophyllin complex was clinically effective and well-tolerated for the treatment of mild-to-moderate acne and large, visible pores when used for three weeks.13 Clinical efficacy parameters (e.g., global acne assessment, facial pores, oiliness, blotchiness, and acne lesion counts) have shown statistically significant improvements from baseline (p?0.05) at the three-week visit.
This study was conducted over three weeks, much shorter than typical acne studies, which last 8 to 12 weeks. Nevertheless, the results show early effects with topical use of liposomal sodium copper chlorophyllin by itself. These factors provide background support for a study in adult female acne.
Methods
Subjects. Prior to subject enrollment, the protocol, informed consent form, and recruitment materials were reviewed and approved by an institutional review board. Following an initial telephone screening, potential women between the ages of 25 and 45 years were scheduled for eligibility screening in the clinic. Among others, exclusion criteria included use of oral or topical prescription or over-the-counter (OTC) acne treatments (and topical retinoids) within the past 30 days, other dermatologic disorders of the face, and having started hormone replacement therapy or oral contraceptives less than 90 days prior to the study. The prospective subjects were advised to take precautions not to pick at or squeeze facial acne lesions prior to screening and not to apply makeup prior to each scheduled visit.
At Visit 1 (baseline), prospective subjects read and signed an informed consent form and a photo release form after the nature of the study was explained and any study-related questions were answered. Prospective subjects were acclimated to ambient temperature and humidity conditions for at least 15 minutes prior to participating in clinic procedures. Upon acclimation, subjects were evaluated for the following eligibility criteria: Fitzpatrick skin classification Types I to VI; presence of facial acne that was clinically determined to be mild to moderate (score of 1.5 to 3.5 according to the modified Cook’s grading scale,15 where 0=none and 5=very severe); and presence of 5 to 100 noninflammatory and 5 to 50 inflammatory facial acne lesions.
Subjects who passed the screening criteria participated in the following procedures:
Investigator Global Acne Assessment (IGA): subjects had an IGA performed using the scale in Table 1, derived from the grading of acne proposed by Cook (with half-point scores allowed).

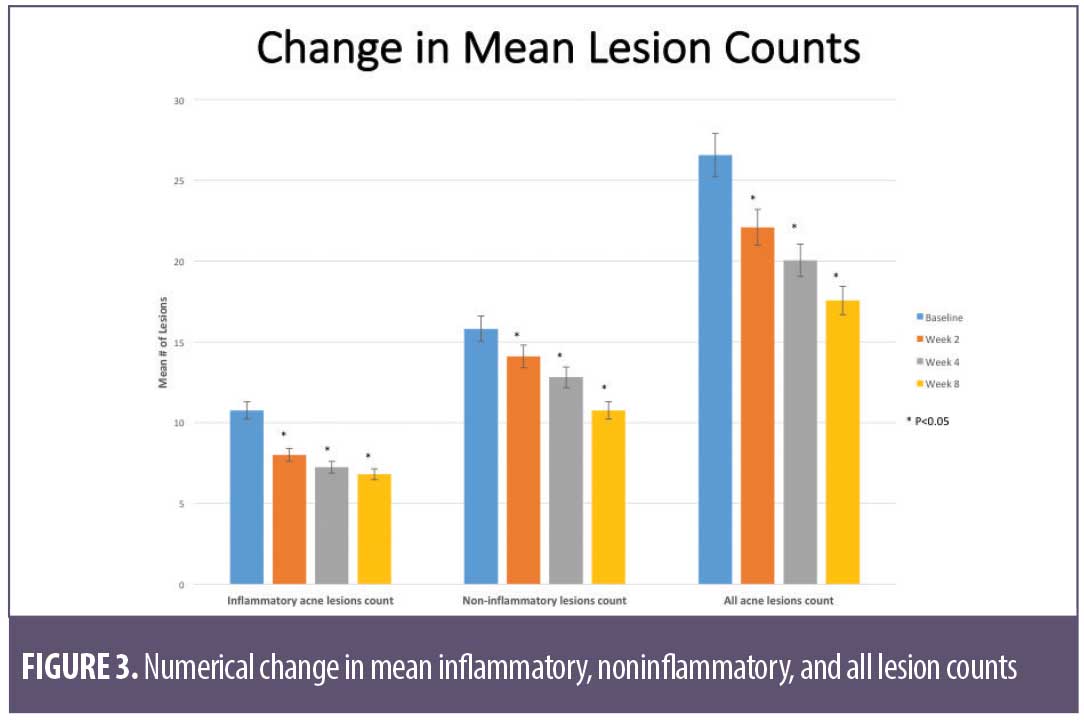
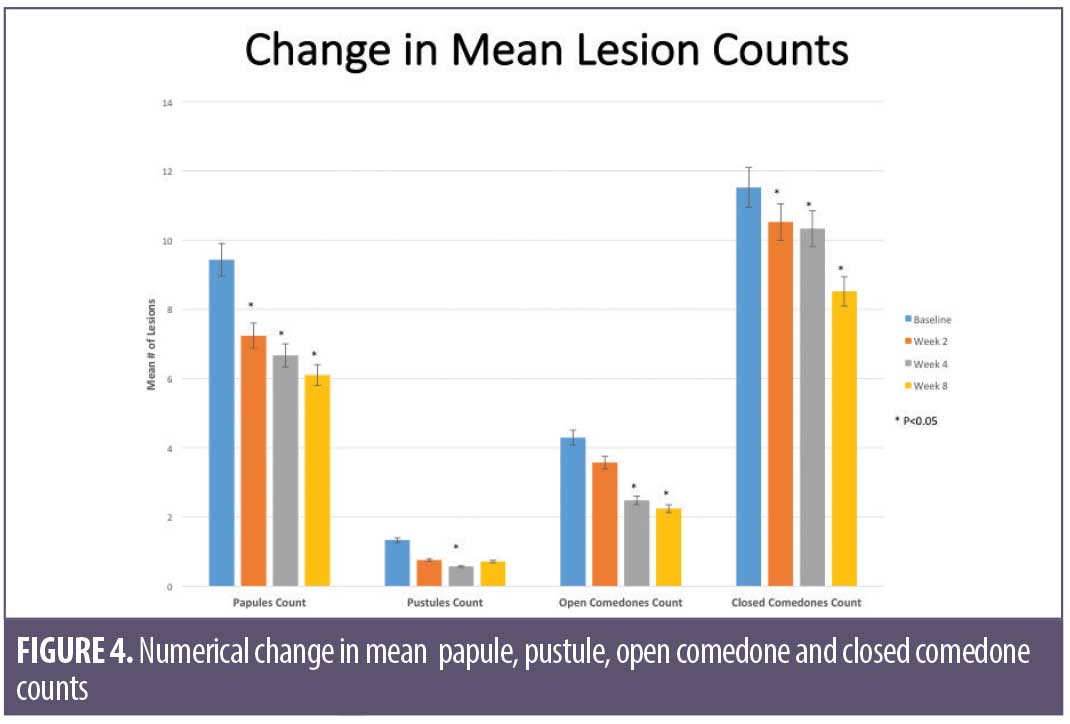
Acne lesion count: a clinical evaluator counted and recorded the number of papules, pustules, open comedones, and closed comedones separately on the following facial locations/quadrants: forehead, left cheek, chin (including the area above the upper lip), and right cheek. Papules and pustules were classified as inflammatory acne lesions, while open and closed comedones were classified as non-inflammatory lesions.
Clinical grading of efficacy parameters: subjects were clinically graded for efficacy parameters (Table 2), including overall assessment of inflammatory acne lesions, globally on the face using a modified Griffiths 10-point scale according to the following numerical definitions (with half-point scores allowed): 0=none (best possible condition); 1 to 3=mild; 4 to 6=moderate; 7 to 9=severe (worst possible condition).
Tolerability evaluations. Local cutaneous tolerability was evaluated by assessing the signs and symptoms of objective and subjective irritation globally on the right and left side of each subject’s face. Objective irritation (e.g., erythema, edema, dryness/scaling) was clinically graded by an expert evaluator using a four-point scale (0=none, 1=mild, 2=moderate, and 3=severe), whereas subjective irritation (e.g., burning, stinging, itching, dryness/tightness) was assessed by subjects on a separate four-point scale (0=none, 1=mild, 2=moderate and 3=severe)
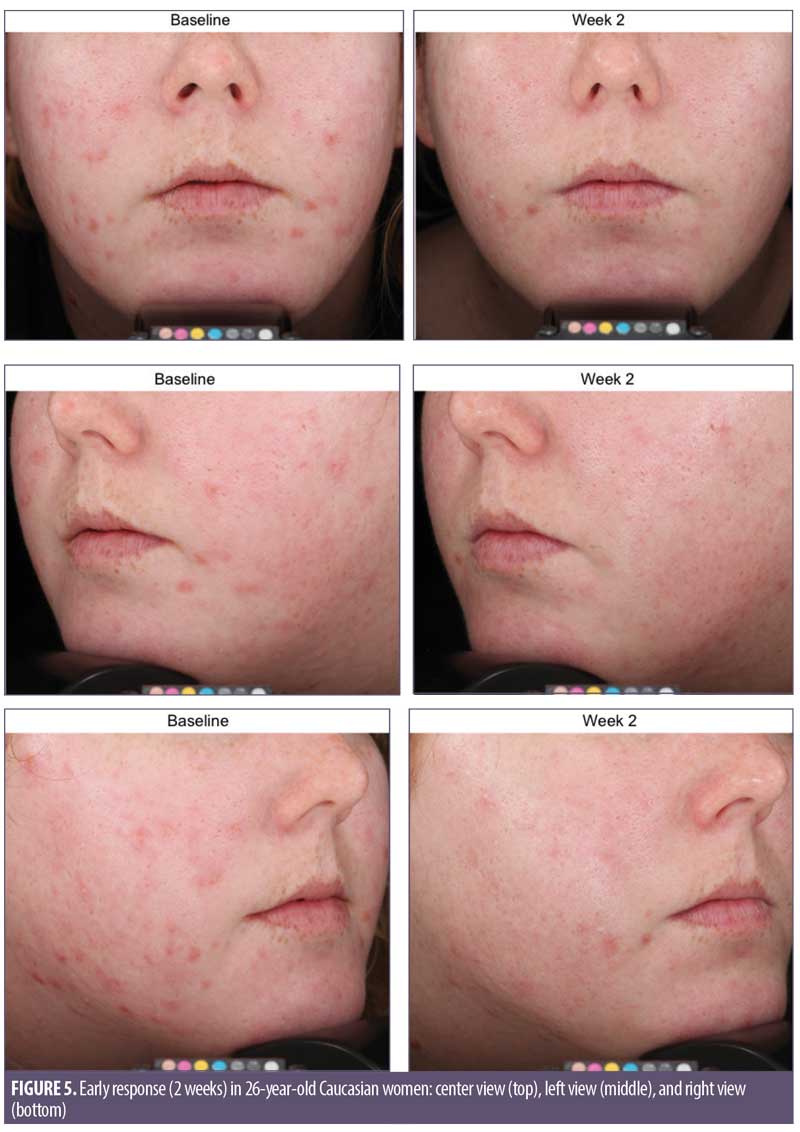
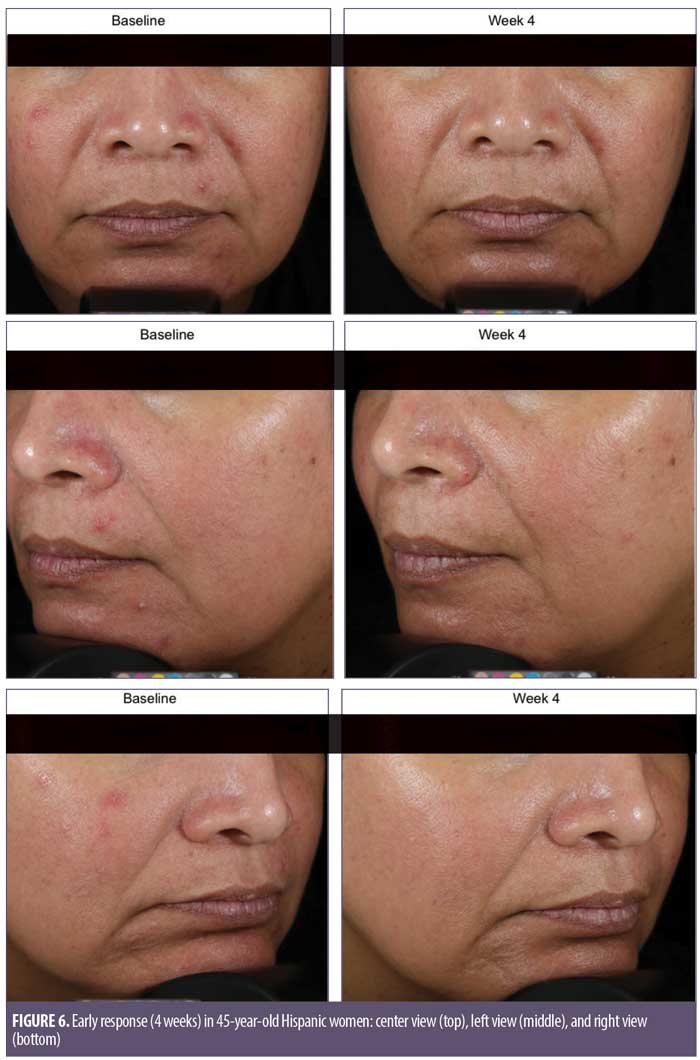
Digital imaging. Prior to photography procedures, clinic personnel ensured subjects had a clean face with no makeup and had removed any jewelry from the areas to be photographed. Full-face digital images were taken of each subject (right side, left side, and center views) using the VISIA-CR photo-station (Canfield Imaging Systems, Fairfield, New Jersey, USA) with a Canon Mark II 5D digital SLR camera (Canon Inc., Tokyo, Japan).
Subjects were distributed preweighed units of the acne treatment regimen test materials and provided with the following usage instructions:
Use the regimen twice per day, morning and evening.
Wash face with facial cleanser.
After washing, apply a pea-sized amount of over-the-counter (OTC) acne treatment cream to the fingertips and gently spread over the face.
After the product dries, apply two pumps of rebalancing gel to the fingertips and gently spread over the face.
Subjects were provided with a calendar of study visits, written usage instructions, and a daily diary to record product application times and comments. Subjects returned to the clinic for Visit 2 (Week 2), Visit 3 (Week 4), and Visit 4 (Week 8). Subjects participated in the following procedures at each visit (unless otherwise indicated):
Clinic personnel recorded concomitant medications and questioned subjects regarding changes in their health. Adverse events were recorded if applicable.
Daily diaries were returned to the clinic and reviewed for adherence. New diaries were distributed at Weeks 2 and 4.
Test material units were collected, visually inspected, and weighed for adherence. Test materials were returned to subjects or new units were distributed as needed at Weeks 2 and 4.
Subjects completed a self-assessment questionnaire.
Subjects acclimated for at least 15 minutes and then participated in the following procedures as described for baseline: digital imaging procedures; Investigator global acne assessment, acne lesion count, and clinical grading of efficacy parameters; and tolerability evaluations.
Biostatistics and Data Management
Wilcoxon signed-rank test with p?0.05 was utilized to evaluate mean changes from baseline for all clinical efficacy and tolerability parameters. The testing hypothesis was that the mean change from baseline is equal to zero. Self-assessment questionnaires completed by the subjects at Week 8 were tabulated, and a top-box analysis was performed.
Results
Twenty-one of the 24 enrolled female subjects completed the clinical study. Of the three subjects with early discontinuation, one experienced an adverse event (sickle cell crisis considered unlikely to be related to treatment regimen), one requested withdrawal, and one was lost to follow-up. Demographic characteristics of the subjects are presented in Table 3.
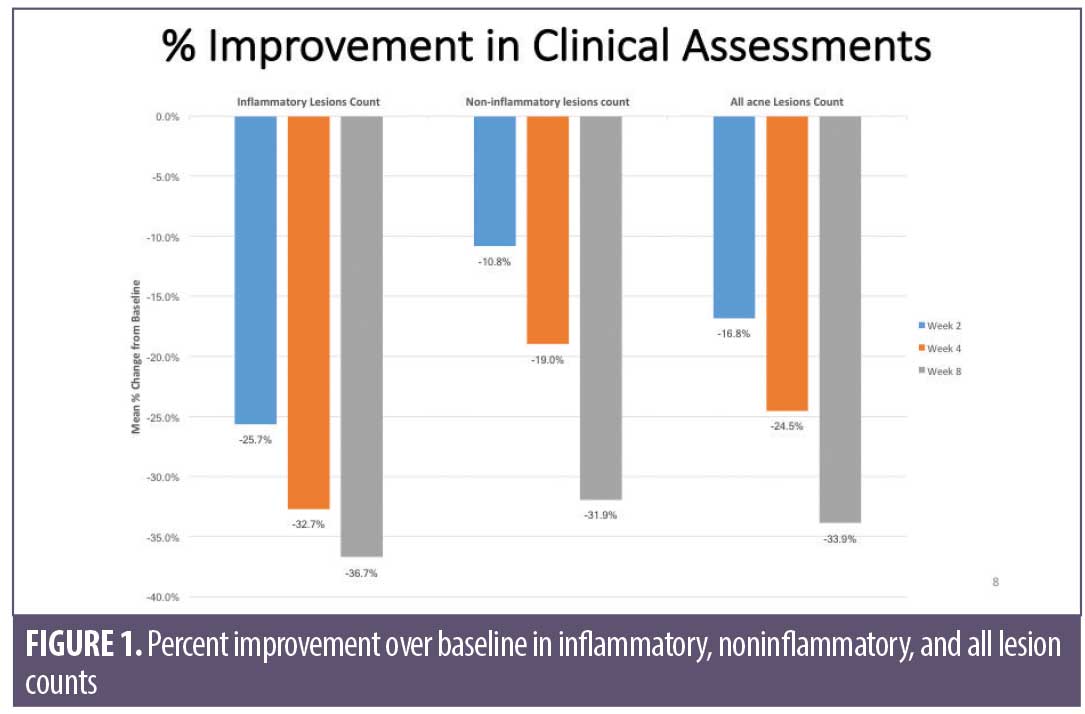
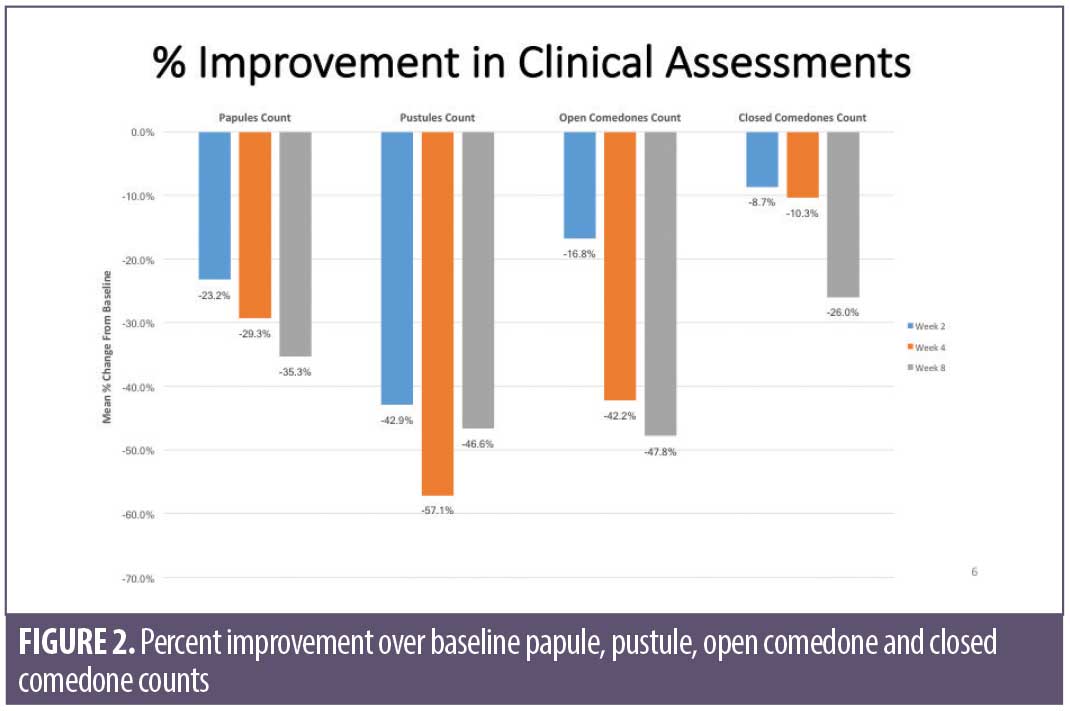
Overall results of this single-center clinical trial indicate that the acne treatment regimen (OTC acne treatment cream, rebalancing gel, and facial cleanser) was effective in improving facial acne conditions when used over the course of eight weeks by women with mild-to-moderate acne on the face. The regimen was well-tolerated and well-perceived by the subjects. Use of the regimen produced a statistically significant improvement in the following acne and aging parameters when compared to baseline: Investigator’s Global Acne Assessment scores at Weeks 2, 4, and 8; acne lesion count values for papules, closed comedones, inflammatory acne lesions, noninflammatory acne lesions, and all acne lesions (combined) on the global face at Weeks 2, 4, and 8; acne lesion count values for pustules at Week 4 and open comedones at Weeks 4 and 8; scores for skin smoothness (tactile), skin tone (color) evenness, overall redness of inflammatory lesions, and overall size of inflammatory lesions at Weeks 2, 4, and 8; and scores for pore size and clarity at Weeks 4 and 8 (Figures 1–4). Figures 5 and 6 document photographic responses to treatment. Use of the regimen produced no statistically significant changes from baseline in scores for erythema, dryness/scaling, edema, burning, stinging, itching, and dryness/tightness at any time point. Analysis of the self-assessment questionnaire indicated that a statistically significant proportion of subjects selected favorable responses as compared to unfavorable responses regarding the regimen after eight weeks of use for inquiries regarding reducing number of blemishes, improving evenness of skin tone, improving skin texture, reducing appearance of pores, making skin feel softer/smoother, and improving appearance of fine lines and wrinkles.
 Discussion
Discussion
Acne remains a major concern for many women in adulthood. Adult female acne can be a chronic condition with negative psychological, emotional, and social impacts.16,17 It is unclear whether the actual prevalence of adult female acne is increasing or whether this group is simply better informed of their options for treatment and/or less tolerant of the condition.5 A web-based survey confirmed that treatment expectations for adult female acne were similar across racial/ethnic groups, with subjects expecting a quick resolution (overnight to within 2 weeks). Expectations might impact treatment satisfaction and medication adherence.18
The regimen studied consisted of a facial cleanser, OTC acne cream containing 2% salicylic acid, and a rebalancing gel containing liposomal sodium copper chlorophyllin complex. In low concentrations, this ingredient has shown clinical benefits in addressing photodamaged skin, pore size, acne, and facial redness.12–14 In addition, sodium copper chlorophyllin complex has demonstrated, both in vitro and in vivo, the ability to inhibit hyaluronidase.19,20 The clarifying gel has moisturizing, antioxidant, anti-aging, and anti-inflammatory ingredients designed to address characteristics of aging skin, such as reduced hyaluronic acid content, loss of collagen and elastin, buildup of reactive oxygen species, and reduction in water and lipid content.21,22 Certainly, the salicylic acid product contributed to the benefits seen with use of the regimen. However, topical salicylic acid is often associated with skin dryness and irritation.18 In this study, use of the three-product regimen produced no statistically significant changes from baseline in scores for erythema, dryness/scaling, edema, burning, stinging, itching, and dryness/tightness at any time point. The test regimen appeared to have a beneficial effect on overall outcome; however, the differential anti-acne effects of salicylic acid and sodium copper chorophyllin complex cannot be quantified by the study design that was employed.
Overall, the skin ages as a result of a combination of the effects of time (intrinsic) and the environment (extrinsic). It is well-documented that clinical manifestations of aging skin include loss of tone and elasticity, uneven skin tone, coarsened texture, enlarged pores, irregular pigmentation, and lines and wrinkles.23,24 Because of previous observations of improvement in facial photodamage with sodium copper chlorophyllin complex,12 assessments (investigator and self) were made into various parameters of aging of the face. At Week 8, all of the investigator-assessed clinical parameters (i.e., pore size, skin smoothness, clarity, skin tone, redness of inflammatory lesions, and size of inflammatory lesions) showed statistically significant improvements as compared with baseline. In addition, the subject self-assessment questionnaires also showed statistical improvements in several aging parameters (Table 4).

Conclusion
An ethnically diverse group of women treated with an acne regimen showed statistically significant clinical improvements in both acne and certain aging parameters in as little as two weeks with progressive improvements in most parameters from Week 2 to Week 8. Importantly, the product regimen was very well-tolerated without the adverse reactions commonly seen with topical acne products.
References
- Burton JL, Cunliffe WJ, Stafford I, Shuster S. The prevalence of acne vulgaris in adolescence. Br J Dermatol. 1971;85(2):119–126.
- Thiboutot DM. Overview of acne and its treatment. Cutis. 2008;81(1 Suppl):3–7.
- James WD. Clinical practice: acne. N Engl J Med. 2005;352(14):1463–1472.
- Goulden V, Stables GI, Cunliffe WJ. Prevalence of facial acne in adults. J Am Acad Dermatol. 1999;41(4):577–580.
- Williams C, Layton AM. Persistent acne in women: implications for the patient and for therapy. Am J Clin Dermatol. 2006;7(5):
281–290. - Poli F, Dreno B, Verschoore M. An epidemiological study of acne in female adults: results of a survey conducted in France. J Eur Acad Dermatol Venereol. 2001;15(6):541–545.
- Landis ET, Davis SA, Taheri A, Feldman SR. Top dermatologic diagnoses by age. Dermatology Online J. 2014;20(4):22368.
- Dunn LK, O’Neill JL, Feldman SR. Acne in adolescents: quality of life, self-esteem, mood, and psychological disorders. Dermatology Online J. 2011;17(1):1.
- Lasek RJ, Chren MM. Acne vulgaris and quality of life of adult dermatology patients. Arch Dermatol. 1998;134(4):454–458.
- Mallon ME, Newton JN, Klassen A, et al. The quality of life in acne: a comparison with general medical conditions using generic questionnaires. Br J Dermatol. 1999;140(4):672–676.
- Callender VD, Alexis AF, Daniels SR, et al. Racial differences in clinical characteristics, perceptions and behaviors, and psychological impact of adult female acne. J Clin Aesthet Dermatol. 2014;7(7):19–31.
- Sigler ML, Stephens TJ. Assessment of the safety and efficacy of topical copper chlorophyllin in women with photodamaged facial skin. J Drugs Dermatol. 2015;14(4):401–404.
- Stephens TJ, McCook JP, Herndon JH. Pilot study of topical copper chlorophyllin complex in subjects with facial acne and large pores. J Drugs Dermatol. 2015;14(6):589–592.
- Vasily DB. Topical treatment with liposomal sodium copper chlorophyllin complex in subjects with facial redness and erythematotelangiectatic rosacea: case studies. J Drugs Dermatol. 2015;14(10):1157–1159.
- Cook CH, Centner RL, Michaels SE. An acne grading method using photographic standards. Arch Dermatol. 1979;115(5):
571–575. - Collier CN, Harper JC, Cantrell WC, et al. The prevalence of acne in adults 20 years and older. J Am Acad Dermatol. 2008;58(1):
56–59. - Tanghetti EA, Kawata AK, Daniels SR, et al. Understanding the burden of adult female acne. J Clin Aesthet Dermatol. 2014;7(2):
22–30. - Rendon MI, Rodriguez DA, Kawata AK, et al. Acne treatment patterns, expectations, and satisfaction among adult females of different races/ethnicities. Clin Cosmet Invest Dermatol. 2015;8:231–238.
- McCook JP, Dorogi PL, Vasily DB, Cefalo DR. In vitro inhibition of hyaluronidase by sodium copper chlorophyllin complex and chlorophyllin analogs. Clin Cosmet Investig Dermatol. 2015;8:443–448.
- McCook JP, Stephens TJ, Jiang LI, et al. Ability of sodium copper chlorophyllin complex to repair photoaged skin by stimulation of biomarkers in human extracellular matrix. Clin Cosmet Investig Dermatol. 2016;9:1–8.
- Bauman L. Skin agin and its treatment. J Pathol. 2007;211:241–251.
- Farage MA, Miller KW, Elsner P, Maibach HI. Characteristics of the aging skin. Adv Wound Care. 2013;2(1):5–10.
- Gilchrest BA. Skin aging and photoaging: an overview. J Am Acad Dermatol.1989;21(3 Pt 2):610–613.
- Lupo MP. Cosmeceutical peptides. Dermatol Surg. 2005;31(7 Pt 2):832–836.

